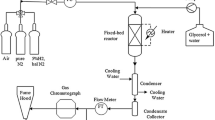
Different global kinetics for the oxidation of NO on platinum are compared in this work. The general form of the equation for the reaction rate is \(r = k \cdot c_{NO}^\alpha \cdot c_{O_2 }^\beta \cdot c_{NO_2 }^\gamma \cdot TD^\delta\) with \(TD = \left( {1 - c_{NO_2 } \cdot c_{NO}^{ - 1} \cdot c_{O_2 }^{ - 0.50} \cdot K^{ - 1} } \right)\). Furthermore steady-state and temperature-programmed experiments were performed. The best results of simulation coupled with parameter estimation were obtained using α = 0.28, β = 0.49, γ = 0 and δ = 1, along with an activation energy of 47.5 kJ mol−1.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
L. Olsson B. Westerberg H. Persson E. Fridell M. Skoglundh B. Andersson (1999) J. Phys. Chem. B 103 10433 Occurrence Handle10.1021/jp9918757 Occurrence Handle1:CAS:528:DyaK1MXntFGlsr0%3D
M. Crocoll, PhD Thesis, Universität Karlsruhe (2003)
J.M.A. Harmsen J.H.B.J. Hoebink J.C. Schouten (2001) Catal. Lett. 71 81 Occurrence Handle10.1023/A:1016660509636 Occurrence Handle1:CAS:528:DC%2BD3MXjtFynsb4%3D
L. Olsson H. Persson E. Fridell M. Skoglundh B. Andersson (2001) J. Phys. Chem. B 105 6895 Occurrence Handle10.1021/jp010324p Occurrence Handle1:CAS:528:DC%2BD3MXks1ymu7s%3D
X. Li M. Meng P. Lin Y. Fu T. Hu Y. Xie J. Zhang (2003) Top. Catal. 22 111 Occurrence Handle10.1023/A:1021480115825
H. Mahzoul J.F. Brilhac P. Gilot (1999) Appl. Catal. B 20 47 Occurrence Handle10.1016/S0926-3373(98)00093-9 Occurrence Handle1:CAS:528:DyaK1cXnvFaksL0%3D
R. Marques P. Darcy P. Costa ParticleDa H. Mellottee JM. Trichard G. Djega-Mariadassou (2004) J. Mol. Catal. A: Chem. 221 127 Occurrence Handle10.1016/j.molcata.2004.06.023 Occurrence Handle1:CAS:528:DC%2BD2cXns1Ontb8%3D
L. Olsson R.J. Blint E. Fridell (2005) Ind. Eng. Chem. Res. 44 3021 Occurrence Handle10.1021/ie0494059 Occurrence Handle1:CAS:528:DC%2BD2MXitlSjtrk%3D
S.S. Mulla N. Chen W.N. Delgass W.S. Epling F.H. Ribeiro (2005) Catal. Lett. 100 267 Occurrence Handle10.1007/s10562-004-3466-1 Occurrence Handle1:CAS:528:DC%2BD2MXivVOnsLk%3D
S. Salomons R. Hayes M. Votsmeier A. Drochner H. Vogel S. Malmberg J. Gieshoff (2007) Appl. Catal. B 70 305 Occurrence Handle10.1016/j.apcatb.2006.01.022 Occurrence Handle1:CAS:528:DC%2BD2sXivVSi
L. Mukadi R. Hayes (2002) Comp. Chem. Eng. 26 IssueID3 439 Occurrence Handle10.1016/S0098-1354(01)00763-3 Occurrence Handle1:CAS:528:DC%2BD38XhvVSmtbs%3D
J. Després, PhD Thesis, Diss. ETH No. 15006, ETH Zürich (2003)
L. Olsson E. Fridell (2002) J. Catal. 210 340 Occurrence Handle10.1006/jcat.2002.3698 Occurrence Handle1:CAS:528:DC%2BD38XmsFWnurY%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hauptmann, W., Drochner, A., Vogel, H. et al. Global kinetic models for the oxidation of NO on platinum under lean conditions. Top Catal 42, 157–160 (2007). https://doi.org/10.1007/s11244-007-0170-6
Issue Date:
DOI: https://doi.org/10.1007/s11244-007-0170-6