Abstract
Ziziphora persica (Lamiaceae) has been used as infusions and decoctions in traditional medicine for various purposes such as sedative, carminative, and stomach pain. In the proliferation stage of shoot culture, MS medium was treated with different concentrations of methyl jasmonate (MJ) and salicylic acid (SA) (50, 100 and 150 µM) either individually or in combination with various concentrations (25 µM SA + 25 µM MJ, 25 µM SA + 50 µM MJ, 50 µM SA + 25 µM MJ, 50 µM SA + 50 µM MJ). In the present study, we investigated the quantitative changes in pigments, growth parameters, and volatile compounds in the shoot culture of nodal segments. Volatile compounds were extracted and subsequently analyzed using GC-mass spectrometry. The highest amounts of chlorophyll a and carotenoids were detected in SA25 + MJ25 treatment, whereas chlorophyll b was highly found in the control treatment. Besides, significant amounts of detected volatile compounds belonged to alkane hydrocarbons, whereas mono and sesquiterpenes were found in low amounts.
Key message
SA show higher toxic effect than MJ and affective than synergic use of elicitors on the organ culture of Z. persica. Elicitors can increase volatile compounds in different pattern compared to the previously in vivo production.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ziziphora persica is an herb and annual plant that belong to order of Lamiales, family of Lamiaceae, and genus of Ziziphora (Baytop 1984). It is widely distributed in Iran and is known as a valuable plant due to its various applications. Its leaves, flowers, and stems are applied as wild vegetable or additive food for their flavor. In addition, different species of this genus have been used as infusions and decoctions for various purposes such as sedative, carminative, and stomach pain in Iranian traditional medicine (Ghahreman 1995). The studies conducted on essential oils and various extracts of Z. persica have reported several antibacterial and antioxidant effects (Meral et al. 2002). The most prominent essential oils which are extracted from the aerial parts of field-grown Z. persica include the following compounds: pulegone, limonene, and piperitenone (Ozturk and Ercisli 2006), pulegone, 1,8-cineon, beta-pinene, menthone, gama-terpinene, p-myrcene, and beta-cymene (Nezhadali and Shirvan 2010), and dodecane, decane, tetradecane, ethyl palmitate, and hexadecane (Nadaf et al. 2013).
Plants’ cell, tissue, and organ culture technology have been used in producing pharmaceutical valuable compounds and other chemicals in recent years (Baque et al. 2012). Stem and root cultivation is applied in order to produce momentous pharmaceutical compounds, since the synthesis of secondary metabolites is raised in differentiated tissues. Organ cultures are relatively stable, and secondary metabolites can be produced in shorter time periods than the parent plants can (Subroto et al. 1996).
The production of secondary metabolites in plants is mainly related to the environmental stresses and their accumulation incited by elicitors (Zhao et al. 2005). The elicitors are compounds with biotic and abiotic origins (Zhao et al. 2010). Biotic elicitors include polysaccharides, proteins, glycoproteins or the cell walls parts of fungi and plants (cellulose and pectin), and microorganisms (chitin, glucagon). On the other hand, abiotic elicitors include heavy metals, UV radiation, and some chemical compounds with different mechanisms such as salicylic acid, methyl jasmonate, and nitrate (Vasconsuelo and Boland 2007; Yu et al. 2006).
Among the different elicitors, salicylic acid and methyl jasmonate (MJ) are known as effective elicitors for the induction of secondary metabolite synthesis in plant cell/organ cultures (Yu et al. 2002). The salicylic acid (SA) belongs to phenolic compounds in plants which are able to affect growth and development, photosynthesis and transpiration rates, and ion uptake and transportation. Moreover, the salicylic acid causes changes in leaf morphology and the structure of chlorophyll (Popova et al. 2003). This compound is one of the stress-signaling molecules that play an effective role in plant’s resistance to pathogens and other stress factors (Rao et al. 2000). The effect of salicylic acid on secondary metabolites especially on the enhancement of phenolic compounds was elucidated (Li et al. 2016).
Jasmonic acid (JA) and its methyl ester, namely MJ, have various biological effects. Plant treatment with mentioned compounds causes leaves’ senescence, falling, twisting, stomatal closure, and beta-carotene and ethylene synthesis as well as an inhibition in root growth (Szabo et al. 1999; Yu et al. 2002). JA and MJ have been suggested as important signaling compounds for the process that lead to an increased production of secondary metabolites and play a key role in signal transduction processes that adjust defense responses in plants (Zhao et al. 2005; Walker et al. 2002).
Several studies have reported the effects of MJ and SA on the production of secondary metabolites such as enhanced diterpenoid (carnosic acid and carnosol) content in shoot culture of Salvia officinalis by MJ (Grzegorczyk-Karolak and Wysokińska 2009), induced high bacoside A yield in shoot culture of B. monnieri by MJ (Sharma et al. 2013), and increased anthocyanin biosynthesis in callus culture of Daucus carota by SA (Sudha and Ravishankar 2003).
So far, there is no report on the in vitro production of volatile compounds in Z. persica after implementing MJ and SA treatment on tissue culture. Therefore, in the present investigation, various concentrations of the elicitors (MJ and SA) are applied on shoot culture of Z. persica, and then some morphological traits and biochemical aspects such as photosynthetic pigments and chemical composition of the volatile compounds are examined.
Materials and methods
Plant material
The mature seeds of Z. persica were obtained from the Botanical Garden of Agriculture in Tabriz, Iran. The seeds were sterilized using 70% ethanol and 20% sodium hypochlorite for 3 and 15 min, respectively. Then, they were washed three times using sterile distilled water and planted in basal solid MS medium (Murashige and Skoog 1962). Planted seeds in MS medium were maintained under cool-white light illumination at 40 µmol m−2 s−1 with the daily photoperiod of 16 h at 25 ± 2 °C. After 1 month, the developed shoots with 2–3 nodes were cut and transferred to the fresh solid MS medium containing 1000 µM BA and 200 µM NAA for shoot multiplication. Furthermore, the liquid culture of developed shoots was performed. To this end, the developed shoots were transferred to test tubes containing liquid MS medium and then subjected to various concentrations of elicitors under continuous shaking at 120 rpm at 26 ± 2 °C. The MS medium without elicitor was treated as a control in this experiment. After 2 weeks of treatment, growth parameters consisting of the shoot, root, and leaf numbers, shoot and root length (cm), and fresh and dry weight (DW) of regenerated shoots per explants were determined.
Provision and treatment of elicitors
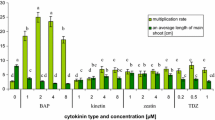
In this study, the two elicitors (MJ and SA) were purchased from Sigma. They were dissolved in 96% ethanol and sterile water to provide stock solutions and were sterilely filtered using 0.22 µM membrane filters. Elicitors with different concentrations (50, 100 and 150 µM) were added to MS liquid medium containing NAA (200 µM) and BA (1000 µM) and then were sterilized and distributed in the test tubes. The shoots transferred to the mentioned medium and were maintained under light to grow in a shaker at 120 rpm at 26 ± 2 °C. The synergism effects of the elicitors were studied as follows: 25 µM SA + 25 µM MJ, 25 µM SA + 50 µM MJ, 50 µM SA + 25 µM MJ and 50 µM SA + 50 µM MJ. The untreated shoot was used as a control culture. The experiments were performed with three replications.
Extraction of photosynthetic pigments
Photosynthetic pigments content of tissues was determined using Wellburn’s method (1994). For spectral determination of the concentration of chlorophyll a and b as well as the total carotenoids, 0.06 g of in vitro produced shoots were crushed in 2 mL of dimethyl sulfoxide. The extracts with a total volume of 2 mL were centrifuged (5900×g for 10 min). Using supernatant, the pigment content of in vitro produced shoots was measured spectrophotometrically through determining the absorbance on 665, 649, and 480 nm and using the following formula. The results were expressed as mg of pigments per g of fresh weight (FW).
Extraction of volatile products
For volatile compound analysis, 0.3 g of in vitro produced shoots of each sample was twice crushed with n-hexane (2 and 5 mL) for 3 min (Pourebad et al. 2015).
Gas chromatography-mass spectrometry (GC–MS)
Volatile compounds were analyzed and identified via GC–MS using an Agilent 6890 gas chromatograph equipped with a fused-silica capillary HP-5 column (30 × 0.25 × 0.25 µm film thickness). Injection start temperature was 150 °C, and the start temperature was 70 °C. It was kept at this temperature for 3 min, and then with the speed of 10 °C per min temperature, it was changed to 120 °C for 2 min. Then, with speed of 10 °C per min, it reached to the temperature of 150 °C. Then, after staying for 2 min at this temperature and changing with 7 °C per min, it changed to the temperature of 240 °C and remained at this temperature for 5 min. The identification of the compounds was performed comparing their retention indices and mass spectra with those reported in the articles (Adams 1995).
Statistical analysis
All experiments were carried out using a completely randomized design with three replications. The data were interpreted through Analysis of Variance (ANOVA) using SPSS statistics version 19. To compare the means, the Duncan’s multiple range tests and standard error were used at P ≤ 0.05 to show the significant differences.
Results and discussion
The effects of MJ and SA concentrations on growth parameters
The shoot induction was observed in case of all media after 4–5 days of culture. According to Table 1, shoots production significantly decreased in all treatments in comparison with the one occurred in the control treatment. After the control treatment (With shoot number of 5.592 ± 0.308a), 100 µM of MJ treatment has highest shoots number (4.388 ± 0.547b) in comparison with those of other treatments. The highest shoot length belonged to the treatment with 50 µM of MJ (2.833 ± 0.110a) and was significant in comparison with that of the control. In comparison with the control, all other treatments (except 50 and 150 µM of MJ) significantly decreased the root production. Based on these results, it appears that the exerted SA, in comparison with MJ, causes a decrease in number and length of shoot and root, and these reductions were remarkable in root (Fig. 1). Manth et al. (1992) observed a remarkable reduction in root growth and toxicity due to the treatment with higher concentrations (3.5 mM) of SA on the roots of Viygica faba. Khalili et al. (2009) reported that the application of SA on root hairs of Silybum marianum reduced the root weight.
The fresh and dry weights of Z. persica decreased in accordance with the increasing concentrations of MJ and SA that is in line with the finding of Lee et al. (2015). According to the results, the exertion of SA caused a significant reduction in fresh weight of the plants, which was not followed by MJ effects. JA in higher concentrations significantly prevented biomass production in shoot culture of H. hirsutum and H. maculatum (Coste et al. 2011). The efficiency of elicitors depends on its concentration, exposure time, genotypes, and type of culture examples (suspension culture, shoot and root cultures or whole plant cultures) (Zhao et al. 2010).
Quantification of photosynthetic pigments in the produced shoots
According to the content of photosynthetic pigments, there were significant differences between all treatments and the control (Table 2). MJ and SA reduced photosynthetic pigments. This decrease reflects the role of these two elicitors in 1-aminocyclopropane- 1-carboxylic acid (ACC) synthase activity, ACC oxidase, and ethylene biosynthesis (Roustan et al. 1989; Rudell and Mattheis 2002).
Besides, MJ causes a decrease in chlorophyll amounts in Arabidopsis thaliana (Jung 2004) which is in accordance with the finding of the present study. In contrast to our findings, in terms of the influence of SA on cucumber plant, previous studies showed that SA increased chlorophyll content (Singh et al. 2010).
Synergistic effects of MJ and SA
The synergistic effects of the two mentioned elicitors at different concentrations in shoot culture of Z. persica were examined (Table 1). The synergistic effects of the elicitors, except the length of shoot, significantly reduced the number and length of roots and the shoots number. Among the four different concentrations, the highest quantity of fresh and dry weights are related to the 50 µM SA + 25 µM MJ treatment. Furthermore, among the four different concentrations and in comparison with the control, the treatment with 25 µM SA + 25 µM MJ increased (non-significant) total phenols and flavonoids content (data is not shown). Sahu et al. (2013) observed an increased production of rosmarinic acid (two fold) in the whole plant culture of Solenostemon scutellarioides using the combination usage of MJ, SA, and yeast extract. In another other study on the Valeriana amurensis plant, the synergistic effects of these two elicitors caused an increased production of valtrate (Cui et al. 2012). Based on our results, as compared to the control, the combination of 25 µM of SA + 25 µM of MJ has the highest significant effect on chlorophyll a and carotenoid synthesis.
Composition of volatiles
GC–MS analysis of the examined treatments in terms of in vitro induced shoots of Z. persica revealed the presence of 20 compounds, as listed in Table 3. According to the results, alkan hydrocarbons contained the majority of these volatile compounds in the control group as well as ten various concentrations of SA and MJ treatments. All treatments were poor in terpene compounds; therefore, only one monoterpene, one sesquiterpene, and two diterpenes were detected. A large number of identified components were obtained in case of the treatments with 100 µM of SA (9 components), while a smaller number of these components were detected in the control (3 components) and 50 µM SA + 50 µM MJ (3 components) treatments. Tridecanoic acid was identified in the control treatment. Among the essential oils reported for Z. persica in a farm land, there was no trace of this fatty acid (Nadaf et al. 2013). Six volatile compounds were detected for 50 µM of SA treatment among which alkane hydrocarbons depicted a higher quantity of these compounds, and the highest percentage of compounds belonged to the α-Tetradecene. For 100 µM of SA treatment, nine compounds were identified among which alkane hydrocarbons showed the highest amount. Moreover, durene monoterpene and ethyl palmitate (a fatty acid) were found in this treatment. In this treatment, the highest percentage of compounds belonged to Tridecane. In two other studies on essential oils of Z. persica on the farms (Nadaf et al. 2013) and the Salvia nemorosa (Nadaf et al. 2012), the amount of of Tridecane was lower than the one found in this study.
According to the results, four volatile compounds were reported for the treatment of 150 µM SA which belonged to alkane hydrocarbons, except for Eicosane that was a diterpene. In 50 µM of MJ treatment, four volatile compounds were detected, and 1-Chlorooctadecane component illustrated the highest amount in this treatment. The only detected alcohol (2-Butanol) was observed in the 100 µM of MJ treatment. In 150 µM of MJ, four volatile compounds were identified. Farnesane was the only sesquiterpene detected in the 25 µM of SA + 25 µM of MJ treatment. Four volatile compounds were detected in 25 µM of SA + 50 µM of MJ treatment, in which both Phytane and Eicosane were diterpene. The amount of Phytane in the field study of Salvia nemorosa was lower than the obtained quantity in these studies (Nadaf et al. 2012). Eight volatile compounds were detected in the treatment of 50 µM of SA + 25 µM of MJ. The main component was Dodecane in this treatment which was the highest amount other treatments. Only three volatile compounds were detected in 50 µM of SA + 50 µM of MJ treatment in which Methyl palmitate (a fatty acid) was observed. Based on the results, the amount of Tridecane and Tetradecane compounds increased in all treatments in comparison with those in the control, while 100 µM of SA and 50 µM of SA + 25 µM of MJ treatments resulted in higher increases. Foliar treatment of salicylic acid (0, 200 and 400 µM) on a medicinal plant, namely Salvia macrosiphon, causes an increase in the amount of essential oils, in which the highest amount of linalool was obtained at the concentration of 400 µM (Rowshan et al. 2010).
The obtained results showed that the main medicinal compounds detected in Z. persica plant include Dodecane, Tetradecane, Methyl palmitate, α-Tetradecene, Tridecane, Eicosane, and Ethyl palmitate. Except for Methyl palmitate and α-Tetradecene, the rest of the volatile compounds correspond with those reported by Nadef et al. (2013) in case of field plants.
Based on the previous report on the cell suspension culture of Gardenia jasminoides, it was shown that the use of elicitors (MJ and SA) cause higher antioxidant capacity than its natural fruits (Liu et al. 2018); our result also confirm changes in the pattern of volatile compounds in in vitro and in vivo culture. Different volatile compounds production pattern between in vivo and in vitro culture in the presence of elicitors (MJ and SA) in this research may lead us to discuss in switching metabolites pathways; based on the investigation of Lu (2009) about the effect of salicylic acid on the metabolites pathways, it was appeared the multiple signal transduction capacity in plants. Also, Li et al. (2016) illustrated the effect of SA on up-regulation of genes involved in phenolic compounds biosynthetic pathway. Therefore, SA and MJ capacity for switching the terpenoids biosynthesis pathway to the other pathways may cause poor terpenoid compounds in the presented results.
Conclusions
The present study showed that SA had a higher toxic effect on the organ culture of Z. persica than MJ and made the most negative effects on the roots. As compared to the control condition, the elicitors (SA and MJ) increased the volatile compounds. SA with the concentration of 100 µM was found to be the best treatment for the production of volatile compounds. According to the results, the volatile compounds produced through in vitro shoot- culture of Z. persica were different from the produced volatile compounds in field grown plants (in vivo) shown in previous studies.
Abbreviations
- SA:
-
Salicylic acid
- MJ:
-
Methyl jasmonate
References
Adams RP (1995) Identification of essential oils components by gas chromatography/mass spectroscopy. Allured Publishing Corporation, Illinois, pp 1–69
Baque MA, Moh SH, Lee EJ, Zhong JJ, Paek KY (2012) Production of biomass and useful compounds from adventitious roots of high-value added medicinal plants using bioreactor. Biotechnol Adv 30(6):1255–1267
Baytop T. (1984) Türkiye’de Bitkiler ile Tedavi. IU Yayinlari No. 3255, Eczacilik Fak. No. 40, 444 s. CSA b2
Coste A, Vlase L, Halmagyi A, Deliu C, Coldea G (2011) Effects of plant growth regulators and elicitors on production of secondary metabolites in shoot cultures of Hypericum hirsutum and Hypericum maculatum. Plant Cell Tissue Organ Cult (PCTOC) 106:279–288
Cui L, Wang ZY, Zhou XH (2012) Optimization of elicitors and precursors to enhance valtrate production in adventitious roots of Valeriana amurensis. ex Kom. Plant Cell Tissue Organ Cult 108:411
Ghahreman A (ed) (1995) Flora of Iran. Research institute of forest and rangelands. Tehran University Press, Tehran
Grzegorczyk-Karolak I, Wysokińska H (2009) The effect of methyl jasmonate on production of antioxidant compounds in shoot cultures of Salvia officinalis L. Herba Pol 55(3):238–243
Jung S (2004) Effect of chlorophyll reduction in Arabidopsis thaliana by methyl jasmonate or norflurazon on antioxidant systems. Plant Physiol Biochem 42:225–231
Khalili M, Hasanloo T, Tabar SKK, Rahnama H (2009) Influence of exogenous salicylic acid on flavonolignans and lipoxygenase activity in the hairy root cultures of Silybum marianum. Cell Biol Int 33:988–994
Lee EJ, Park SY, Paek KY (2015) Enhancement strategies of bioactive compound production in adventitious root cultures of Eleutherococcus koreanum Nakai subjected to methyl jasmonate and salicylic acid elicitation through airlift bioreactors. Plant Cell Tissue Organ Cult (PCTOC) 120:1–10
Li X, Guo H, Qi Y, Liu H, Zhang X, Ma P, Liang Z, Liang Z, Dong J (2016) Salicylic acid-induced cytosolic acidification increases the accumulation of phenolic acids in Salvia miltiorrhiza cells. Plant Cell Tissue Organ Cult 126:333–341
Liu ZB, Chen J-G, Yin Z-P, Shangguan X-C, Peng D, Lu T, Lin P (2018) Methyl jasmonate and salicylic acid elicitation increase content and yield of chlorogenic acid and its derivatives in Gardenia jasminoides cell suspension cultures. Plant Cell Tissue Organ Cult 134:79–93
Lu H (2009) Dissection of salicylic acid-mediated defense signaling networks. Plant Signal Behav 4(8):713–717
Manthe B, Schulz M, Schnabl H (1992) Effects of salicylic acid on growth and stomatal movements of Vicia faba L.: evidence for salicylic acid metabolization. J Chem Ecol 18:1525–1539
Meral GE, Konyalioglu S, Ozturk B (2002) Essential oil composition and antioxidant activity of endemic Ziziphora taurica subsp. Cleonioides. Int J Environ Sci Dev 73:716–718
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497
Nadaf M, Nasrabadi M, Halimi M (2012) GC-MS analysis of n-hexane extract from aerial parts of Salvia nemorosa. Middle-East J Sci Res 11:1127–1130
Nadaf M, Halimi M, Nasrabadi M (2013) Identification of compounds nonpolar extract Ziziphora persica growing in Iran by GC-MS. Middle-East J Sci Res 13:187–190
Nezhadali A, Shirvan BZ (2010) Separation, identification and determination of volatile compounds of Ziziphora persica bunge using HS-SPME/GC-MS. Int J Environ Sci Dev 1:115
Ozturk S, Ercisli S (2006) The chemical composition of essential oil and in vitro antibacterial activities of essential oil and methanol extract of Ziziphora persica Bunge. J Ethnopharmacol 106(3):372–376
Popova L, Ananieva E, Hristova V, Christov K, Georgieva K, Alexieva V, Stoinova Z (2003) Salicylic acid-and methyl jasmonate-induced protection on photosynthesis to paraquat oxidative stress. Bulg J Plant Physiol 133:152
Pourebad N, Motafakkerazad R, Kosari-Nasab M, Farsad Akhtar N, Movafeghi A (2015) The influence of TDZ concentrations on in vitro growth and production of secondary metabolites by the shoot and callus culture of Lallemantia iberica. Plant Cell Tissue Organ Cult (PCTOC) 122(2):331–339
Rao MV, Lee HI, Creelman RA, Mullet JE, Davis KR (2000) Jasmonic acid signaling modulates ozone-induced hypersensitive cell death. Plant Cell 12(9):1633–1646
Roustan JP, Lotche A, Fallot J (1989) Stimulation of Daucus carota somatic embryogenesis by inhibitors of ethylene synthesis cobalt and nickel. Plant Cell Rep 8:182–185
Rowshan V, Khoi MK, Javidnia K (2010) Effects of salicylic acid on quality and quantity of essential oil components in Salvia macrosiphon. J Biol Environ Sci 11:77–82
Rudell DR, Mattheis JP (2002) Methyl jasmonate enhances anthocyanin accumulation and modifies production of phenolics and pigments in Fuji Apples. J Am Soc Hortic Sci 127(3):435–441
Sahu R, Gangopadhyay M, Dewanjee S (2013) Elicitor-induced rosmarinic acid accumulation and secondary metabolism enzyme activities in Solenostemon scutellarioides. Acta Physiol Plant 35:1473–1481
Sharma P, Yadav S, Srivastava A, Shrivastava N (2013) Methyl jasmonate mediates upregulation of bacoside A production in shoot cultures of Bacopa monnieri. Biotechnol Lett 35(7):1121–1125
Singh PK, Chaturvedi VK, Bose B (2010) Effect of salicylic acid on seeding growth and nitrogen metabolism in cucumber (Cucumber Sativus L.). J Stress Physiol Biochem 6(3):102–113
Subroto MA, Kwok KH, Hamill JD, Doran PM (1996) Coculture of genetically transformed roots and shoots for synthesis, translocation, and biotransformation of secondary metabolites. Biotechnol Bioeng 49(5):481–494
Sudha G, Ravishankar G (2003) Elicitation of anthocyanin production in callus cultures of Daucus carota and involvement of calcium channel modulators. Curr Sci 84(6):775–779
Szabo E, Thelen A, Peterson M (1999) Fungal elicitor preparations and methyl jasmonate enhance rosmarinic acid accumulation in suspension cultures of Coleus blumei. Plant Cell Rep 1:485–489
Vasconsuelo A, Boland R (2007) Molecular aspects of the early stages of elicitation of secondary metabolites in plants. Plant Sci 172:861–875
Walker TS, Pal Bais H, Vivanco JM (2002) Jasmonic acid-induced hypericin production in cell suspension cultures of Hypericum perforatum L. (St. John’s wort). Phytochemistry 60(3):289–293
Wellburn AR (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 30:313–317
Yu KW, Gao W, Hahn EJ, Paek KY (2002) Jasmonic acid improves ginsenoside accumulation in adventitious root culture of Panax ginseng CA Meyer. Biochem Eng J 11(2):211–215
Yu Z-Z, Fu C-X, Han Y-S, Li Y-X, Zhao D-X (2006) Salicylic acid enhances jaceosidin and syringin production in cell cultures of Saussurea medusa. Biotechnol Lett 28:1027–1031
Zhao J, Davis LC, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23:283–333
Zhao JL, Zhou LG, Wu JY (2010) Effects of biotic and abiotic elicitors on cell growth and tanshinone accumulation in Salvia miltiorrhiza cell cultures. Appl Microbiol Biotechnol 87:137–144
Acknowledgements
This study was supported by University of Tabriz, Iran.
Author information
Authors and Affiliations
Contributions
R. M. and M. K. N. conceived and designed the study; E. Z. H., R. M., J. R. and M. K. N. performed research. All authors contributed to data analysis and discussion and have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Silvia Moreno.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zare-Hassani, E., Motafakkerazad, R., Razeghi, J. et al. The effects of methyl jasmonate and salicylic acid on the production of secondary metabolites in organ culture of Ziziphora persica. Plant Cell Tiss Organ Cult 138, 437–444 (2019). https://doi.org/10.1007/s11240-019-01639-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01639-x