Abstract
In the present study, the effects of the NO donor “sodium nitroprusside” (SNP) and the ethylene inhibitor “CoCl2” combined with 2.2 μM 6-benzyladenine (BA), and the antioxidant vitamin E “α-tocopherol” in combination with 2.2 μM zeatin (ZT) and 0.2 μM α-naphthaleneacetic acid (NAA) on in vitro shoot proliferation of Sideritis raeseri Boiss. & Heldr. subsp. raeseri, one of the seven endemic mountain tea species of the Greek flora were investigated. The basal nutrient culture medium used in all experiments was the MS (Murashige, Skoog, Physiol Plant 15:473–497, 1962). Taking simultaneously into consideration all shoot proliferation parameters evaluated, the explants performance was optimum and significantly better among others when treated with 2.2 μM BA + 10 μM SNP or 2.2 μM BA + 2.5 μM CoCl2, or 2.2 μM ZT + 0.2 μM NAA + 11.6 μM α-tocopherol. In particular, 10 μM SNP gave 4.6 ± 0.6 shoots/explant and 100% shoot multiplication percentage while 20 μM SNP exhibited the greatest shoot length (17.5 ± 1.8 mm). However, SNP (10–40 μM) caused hyperhydricity symptoms to the 30–100% of explants. CoCl2 applied at 2.5 μM led to the production of 5.5 ± 0.7 shoots/explant, 11.8 ± 1.1 mm shoot length and 100% shoot multiplication percentage and concurrently decreased the percentage of vitrified explants from 33.3 to 20%. 11.6 μM α-tocopherol gave 6.7 ± 0.8 shoots/explant, 14.5 ± 0.7 mm shoot length, 100% shoot multiplication percentage, diminishing vitrification percentage from 41.6 to 8.3% (33.3% decrease) and completely inhibiting the appearance of necrotic explants, indicating its antioxidant properties as a vitamin. Shoot-tip explants responded better and exhibited better shoot proliferation results when cultured in MS medium with 2.5 μM CoCl2 for 5 weeks or 11.6 μM α-tocopherol for 9 weeks rather than with SNP for 4 weeks. In this study, three new different protocols were established for the in vitro shoot proliferation stage of S. raeseri.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among the threatened wild-growing species in Greece are those of the genus Sideritis (Lamiaceae). Taxonomic revision (Papanikolaou and Kokkini 1982) of Sideritis L. section Empedoclia, reported the following seven species of the genus Sideritis as part of the Greek flora: S. raeseri Boiss. & Heldr., S. clandestina Chaub. & Borry, S. scardica Griseb., S. euboea Heldr., S. athoa Papanikolaou & Kokkini, S. perfoliata L. and S. syriaca L. From the various species of Sideritis only S. raeseri, which is the most widespread, is systematically cultivated in Greece. In particular, S. raeseri is distributed in central, north-central and northern parts of the country, typically in the range of Pindos (Strid and Tan 1991) and it self-sows in Parnassus, Timfristos (Velouchi) and other mountains of Aetolia, Doris and Fthiotida (Goliaris 1984).
Conservation of endemic, endangered (IUCN 2001), medicinal and aromatic plants including Sideritis genus native in Greece is beyond regional scope and becomes of global significance. Sideritis species are of great economic importance which is not only related to their use as a spice. In fact, as recent studies have pointed out, Sideritis species are traditionally used in many other ways as their essential oils have antimicrobial, antifungicidal, cytotoxic, antiviral, nematicidal and antioxidant properties (Gonzalez-Burgos et al. 2011; Todorova and Trendaflova 2014; Yigıt 2014). Seeds of Sideritis species collected from native populations exhibit dormancy and have a low germination ability under laboratory and natural conditions (about 5%) (Evstatieva and Koleva 2000). Micropropagation is more complicated and sophisticated than traditional methods, requires special training, the initial costs for setting up a micropropagation unit are relatively high and special laboratory equipment is needed, as well as expensive and very pure chemicals (Cachiţă-Cosma 1987). However, micropropagation method was considered necessary for the mass vegetative propagation of S. raeseri as the nature of plants belonging to Sideritis genus is such that their asexual propagation with cuttings results in low rooting percentages and losses during acclimatization stage. Tissue culture of Sideritis species has been reported on several occasions (Sanchez-Gras and Segura 1987, 1988, 1997; Garcia-Granados et al. 1994; Faria et al. 1998; Erdağ and Yόrekli 2000; Uҫar and Turgut 2009; Papafotiou and Kalantzis 2009; Danova et al. 2013). In vitro culture of Sideritis spp. as an approach for conservation of the natural habitats and provision of herb with high and permanent quality has also been important in order to increase the accumulation of biologically active compounds and obtain herb with permanent and good quality (Gonzalez-Burgos et al. 2011; Todorova and Trendaflova 2014). Therefore, in vitro propagation is a suitable method for plant regeneration, micropropagation and long-term storage of virus-free plant material in a commercial scale, preventing the destruction of the flora, in comparison with traditional propagation systems including seed, cutting, grafting, air-layering etc. (Garcia-Gonzales et al. 2010).
Sodium nitroprusside (SNP) releases nitric oxide (NO), a highly reactive gas and a ubiquitous bioactive molecule which plays a central role in signal transduction in plant stress response (Arasimowicz and Wieczorek 2007). The effects of NO on different types of cells indicate that NO is a potent oxidant or an effective antioxidant (Qiao and Fan 2008). SNP releases NO in a pH dependent manner that promotes plant growth and development and reduces senescence (Kolberz et al. 2008). NO produced by SNP has currently been considered a new member of phytohormones (Leterrier et al. 2012) and may scavenge other reactive intermediaries like reactive oxygen species (ROS) (Laspina et al. 2005). NO synthesis was induced by cytokinins in tobacco, parsley and Arabidopsis cell cultures (Tun et al. 2001).
Accumulation of ethylene in culture vessels may induce growth abnormalities in plants generated in vitro including inhibition of growth, leaf epinasty, leaf senescence and diminution of foliar area (Steinitz et al. 2010). However, the influence of ethylene in plant cells and tissues grown in vitro is diverse and often controversial depending on plant species and even the cultivar (Jha et al. 2007). The addition of ethylene inhibitors such as cobalt chloride (CoCl2) to culture media have been demonstrated to improve regeneration and growth performance in both dicot and monocot plant tissue cultures (Sandra and Maira 2013). Cobalt is an inorganic micronutrient used in most plant tissue culture media based on level established by Murashige and Skoog (1962) for tobacco tissue culture. The low level of cobalt ion promotes growth factors such as plant height, number of leaves per plant as well as fresh and dry weight of leaves and roots (Hilmy and Gad 2002). Cobalt increases the growth of seedlings and alleviates the senescence of aged tissues, inhibiting the activities of the enzyme 1-aminocyclopropane-1-carboxylic acid (ACC) oxidase and thus reducing ethylene production (Li et al. 2005). The promoting effect of cobalt can be ascribed to its role in several physiological activities like growth, photosynthesis and respiration (El-Sheekh et al. 2003; Aziz Eman et al. 2007).
Tocopherols (α-, β-, γ-, and δ-tocopherol) represent a group of lipophilic antioxidants that are synthesized only by photosynthetic organisms. It is widely believed that the main functions of tocopherols are protection of pigments and proteins of photosystem and polyunsaturated fatty acids from oxidative damage caused by reactive oxygen species (Woo et al. 2014). α-Tocopherol (vitamin E) is a common term used for tocopherols and tocotrienols. It is an important antioxidant that is directly involved in scavenging oxygen free radicals and quenching lipid peroxidation chain reactions that occur during oxidation reactions with polyunsaturated fatty acids (Sattler et al. 2004). α-Tocopherol reactions result in the formation of tocopheroxyl radicals that react with other antioxidants to regenerate the active molecule (Liebler 1993; Carelli et al. 2005). Regarding its role in plant tissue culture systems, vitamin E improved in vitro recovery of cryopreserved blackberry shoot tips (Uchendu and Reed 2010). Using sunflower cell cultures, a suitable in vitro production system of natural α-tocopherol was established (Caretto et al. 2004). In addition, there was a substantial improvement in somatic embryogenesis of Loblolly pine (Pinus taeda L.) in the early-stage of somatic embryo growth by supplementing the medium with vitamin E (Pullman et al. 2006). According to Woo et al. (2014) the expression of tobacco tocopherol cyclase in rice regulated antioxidative defense and drought tolerance.
The objectives of this study were to develop and establish three optimal protocols for in vitro clonal shoot multiplication of S. raeseri, an endemic to Greek flora species. There is no experimental evidence available that directly shows the effect of SNP, CoCl2 and α-tocopherol on in vitro shoot proliferation of Sideritis spp. Therefore, this research study focused on investigating the exogenous effect of these three agents on shoot proliferation of S. raeseri under in vitro conditions.
Materials and methods
For the initial establishment of the plant material in vitro, shoot tips of S. raeseri, 1.5-2 cm long were cut and removed from mother plants maintained in a peat:perlite (1:1) substrate in pots under unheated-greenhouse conditions. For the disinfection of the experimental plant material, shoot tip explants were soaked in 70% ethanol for 1 min followed by 2% NaOCl solution for 15 min under continuous stirring and then washed into sterile distilled water for 4–5 times. The successfully established explants (pathogen-free) were sub-cultured every 4 weeks until a sufficient amount of plant material was obtained. The nutrient culture medium used for all experiments was the MS (Murashige and Skoog 1962).
Three experiments were conducted aiming to enhance in vitro shoot proliferation of S. raeseri shoot tip explants. In the first experiment, SNP (Fluka Biochemika) was exogenously applied at 4 concentrations (0, 10, 20, 40 μM) in combination with 2.2 μM ΒΑ (Sigma–Aldrich). In the second experiment, CoCl2 6H2O (Sigma–Aldrich) was tested at 9 concentrations (0.1-contol, 0.5, 1, 2.5, 5, 10, 25, 50, 100 μM) in combination with 2.2 μM ΒΑ. The normal concentration of CoCl2 6H2O, which is an essential microelement of MS culture medium is 0.1 μM (=0.025 mg/l) (Murashige and Skoog 1962), thus, this concentration was considered the control treatment of this experiment. In the third experiment, α-tocopherol (Sigma–Aldrich) was added to culture medium at five concentrations (0, 2.3, 5.8, 11.6, 23.2 μM) in combination with 2.2 μM zeatin (ZT; Sigma–Aldrich) and 0.2 μM α-naphthaleneacetic acid (NAA; Sigma–Aldrich). For all three experiments, the MS culture medium was also supplemented with 30 g/l sucrose (Duchefa Biochemie). In the first and second experiments with SNP and CoCl2, respectively, 3 g/l of Phytagel (Sigma–Aldrich) was used as gelling agent while 3 g/l of Gelrite (Duchefa Biochemie), another type of gelling agent was applied for the third experiment with α-tocopherol. The pH of the culture medium in all three experiments was adjusted to 5.8 before adding the gelling agent and afterwards the medium was sterilized at 121 °C for 20 min. Shoot-tip explants were transferred for the first and second experiments with SNP and CoCl2, respectively into flat-base glass test tubes of 25 × 100 mm containing 10 ml of MS medium and for the third experiment with α-tocopherol into Magenta vessels containing 35 ml of MS medium. All cultures were maintained in a growth chamber. The chamber was programmed to maintain 16-h light duration (40 μmol/m2/s) supplied by cool white fluorescent lamps and a constant temperature of 22 ± 2 °C. Αfter 4 weeks of culture for the first experiment with SNP, 5 weeks for the second experiment with CoCl2 and 9 weeks for the third experiment with α-tocopherol, measurements were taken regarding shoot proliferation characteristics including shoot number/explant, shoot length (mm), shoot multiplication percentage (i.e. number of initial explants able to proliferate new vegetative masses/total number of initial explants × 100%), callus formation, vitrification and necrosis percentages (%).
Statistical analysis
All three experiments were repeated twice and the reported data are the means of the two comparisons, separately for each experiment. The first experiment with SNP included 4 treatments with 20 replicates/treatment in total (10 explants were used the first time and other 10 explants were used the second time in the repetition of the experiment) and one shoot-tip explant placed in each test tube. The second experiment with CoCl2 included 9 treatments with 20 replicates/treatment in total (10 explants the first time and other 10 explants the second time in the repetition of the experiment) and one shoot-tip explant placed in each test tube. The third experiment with α-tocopherol included 5 treatments with 24 replicates/treatment in total (12 explants the first time and other 12 explants the second time in the repetition of the experiment) and 4 shoot-tip explants placed in each Magenta vessel. Each experiment was completely randomized and analyzed with ANOVA (Analysis of Variance) using the statistical program SPSS 17.0 (SPSS Inc., Illinois, New York, USA) at P ≤ 0.05, according to Duncan’s multiple range test ± SE in order significant differences among the treatments to be established.
Results
Experiment 1: effect of SNP on in vitro shoot proliferation of S. raeseri
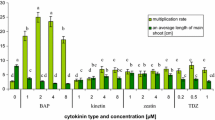
The combined effect of 10 μM SNP + 2.2 μM BA positively affected shoot proliferation in terms of shoot number/explant (4.6 ± 0.6) and shoot multiplication percentage (100%) in relation to the individual effect of 2.2 μM BA (3.2 ± 0.6 shoots/explant and 70% shoot multiplication percentage) (Table 1). However, SNP (10–40 μM) in combination with 2.2 μM BA gave similar shoot length results (14.7 ± 0.9–17.5 ± 1.8 mm) (Fig. 1b–d) to the control (16.7 ± 1.6 mm) (Fig. 1a). The highest applied SNP concentration of 40 μM was toxic and resulted in a significant reduction in shoot number and shoot multiplication percentage, caused extensive hyperhydricity to the 100% of explants and completely inhibited callus formation at the base of the explants. Vitrification percentage was increased with an increase in SNP concentration. No vitrified explants were observed in the control treatment (2.2 μM BA, SNP-free).
Experiment 2: effect of CoCl2 on in vitro shoot proliferation of S. raeseri
2.5 μM and 10–50 μM CοCl2 had a positive effect on shoot number/explant, 5.5 ± 0.7 and 5.3–5.4 respectively, in relation to the control (2.6 ± 0.6 shoots/explant) (Table 2). However, 0.5–100 μM CοCl2 + 2.2 μM BA did not promote nor inhibited the elongation of microshoots (10.6 ± 0.9–16.0 ± 1.5 mm) (Fig. 2b–i) significantly, compared with the control (0.1 μM CοCl2 + 2.2 μM BA/14.2 ± 0.8 mm) (Fig. 2a). Shoot multiplication percentage was significantly raised (87.5–100%) when explants were exposed to 0.5–100 μM CoCl2 with respect to the control (55.5%). CoCl2 (0.5–1 μM and 5–100 μM) intensified the problem of vitrified explants (40–100%) compared to the control (33.3%), whereas 2.5 μM CoCl2 diminished hyperhydricity percentage to 20%. Callus induction percentage was substantially reduced from 66.6% (control) to 20% when 100 μM CoCl2 was added to the culture medium.
Effect of the concentration of the ethylene inhibitor CoCl2 combined with 2.2 μM BA on in vitro shoot proliferation of Sideritis raeseri Boiss. & Heldr. subsp. raeseri: (a) control (2.2 μM BA + 0.1 μM CoCl2), (b) 2.2 μM BA + 0.5 μM CoCl2, (c) 2.2 μM BA + 1 μM CoCl2, (d) 2.2 μM BA + 2.5 μM CoCl2, (e) 2.2 μM BA + 5 μM CoCl2, (f) 2.2 μM BA + 10 μM CoCl2, (g) 2.2 μM BA + 25 μM CoCl2, (h) 2.2 μM BA + 50 μM CoCl2, (i) 2.2 μM BA + 100 μM CoCl2
Experiment 3: effect of α-tocopherol (vitamin E) on in vitro shoot proliferation of S. raeseri
Best results in terms of shoot number/explant (6.7 ± 0.8) and shoot multiplication percentage (100%) were recorded in the combination treatment 2.2 μM ΖΤ + 0.2 μM NAA + 11.6 μM α-tocopherol (Fig. 3d), compared with the control (3.4 ± 0.5 shoots/explant and 91.6% shoot multiplication percentage) (Table 3; Fig. 3a). However, shoot length was decreased by 0.8 cm, from 22.6 ± 2.5 mm (control) to 14.5 ± 0.7 mm when 11.6 μM α-tocopherol was added to the culture medium. α-Tocopherol concentrations lower (2.3 or 5.8 μM) and higher (23.2 μM) than 11.6 μM did not promote or suppress shoot elongation of explants (Fig. 3b, c, e) substantially. According to Table 4, no change in callus induction percentage (100%) was observed with respect to the control due to α-tocopherol application. The percentage of vitrified explants was diminished from 41.6% (control 2.2 μM ΖΤ + 0.2 μM NAA) to 8.3% (33.3% decrease) by supplementing the culture medium with 11.6 μM α-tocopherol. The incorporation of 11.6 μM α-tocopherol into the culture medium led simultaneously to the production of microshoots with no apparent symptoms of necrosis (Table 4).
Effect of the concentration of α-tocopherol (vitamin E) combined with 2.2 μM ZT and 0.2 μM NAA on in vitro shoot proliferation of Sideritis raeseri Boiss. & Heldr. subsp. raeseri: (a) Control (2.2 μM ZT + 0.2 μM NAA, α-tocopherol-free), (b) 2.2 μM ZT + 0.2 μM NAA + 2.3 μM α-tocopherol, (c) 2.2 μM ZT + 0.2 μM NAA + 5.8 μM α-tocopherol, (d) 2.2 μM ZT + 0.2 μM NAA + 11.6 μM α-tocopherol, (e) 2.2 μM ZT + 0.2 μM NAA + 23.2 μM α-tocopherol
Discussion
Prior to this research work, there was available only one published protocol for in vitro shoot proliferation of S. raeseri conducted by Sarropoulou and Maloupa (2015) indicating the usefulness of MS medium supplemented with 2.2 μM BA, 0.05 μM indole-3-butyric acid (ΙΒΑ), 0.05 μM ΝΑΑ, 0.15 μM gibberellic acid (GA3) and 740 μM glycine-betaine in combination with 25 μM dikegulac on multiple shoot regeneration. Therefore, in the present study, three new protocols have been developed for in vitro shoot proliferation of S. raeseri.
In the first experiment of the present study, the combined effect of 10 μM SNP + 2.2 μM BA stimulated in vitro shoot proliferation of S. raeseri explants in terms of shoot number and shoot multiplication percentage in comparison to the effect of BA solely, showing synergistic action between the NO donor SNP and the cytokinin BA. Our results are in agreement with those presented in the Gisela 6 (Prunus cerasus × Prunus canescens) cherry rootstock by Sarropoulou et al. (2015). Cytokinins induce a burst of NO in plant cell cultures, suggesting that NO is involved in cytokinin signal transduction (Tun et al. 2001). Our records are also in line with those reported by Han et al. (2009), who found that the multiplication of Malus hupehensis Rehd. var. pinyiensis Jiang plantlets was significantly promoted by applying 20 μM SNP combined with 2 μM BA and 1 μM ZT to MS medium. Carimi et al. (2005) found that the cytokinin BA induced NO accumulation in suspension cell cultures. The positive effect of SNP on in vitro shoot proliferation has also been reported for other plant species including Albizzia lebbeck L. (Kalra and Babbar 2012), Linum usitatissimum L. (Kalra and Babbar 2010), Dioscorea opposite Thunb (Xu et al. 2009), Triticum aestivum L. (Tian and Lei 2007), Kosteletzkya virginica L. (Guo et al. 2009) and Brassica napus L. (Jhanji et al. 2012). These results indicate that NO may interact with auxin and cytokinin, linking the regulation of cell division to differentiation during de-differentiation and re-differentiation of plant cells (Otvos et al. 2005). Therefore, the supplementation of the MS culture medium with the NO-donor SNP improved the in vitro shoot proliferation potential and efficiency of S. raeseri. In the CAB-6P (Prunus cerasus L.) cherry rootstock, on the contrary, the combined effect of 10–50 μM SNP + 4.4 μM BA suppressed shoot proliferation of shoot-tip explants cultured in vitro regarding shoot number and multiplication percentage (Sarropoulou et al. 2015).
In the current study, SNP did not enhance nor suppress shoot elongation of S. raeseri explants. Similarly, in CAB-6P and Gisela 6 cherry rootstocks, the combined effect of BA + SNP did not differentiate shoot length significantly compared to the individual effect of BA (Sarropoulou et al. 2015). In contrast, shoot length of purslane (Portulaca oleracea L.) seedlings treated with 100 μM NO was significantly decreased under non-stress conditions (Fendereski et al. 2015). NO (precursor of SNP) has been reported to influence several plant developmental events in which gibberellins (GAs) play crucial roles such as seed germination, hypocotyl elongation, acquisition of photomorphogenic traits and others (Beligni and Lamattina 2000). However, so far, the actual interaction between NO and GAs has been described for only a limited number of these physiological events. In fact, the already known mechanisms underlying the interplay between GAs and NO are restricted to the inhibition of hypocotyl elongation during seedling de-etiolation (Leon and Lozano-Juste 2011). NO has been described to act upstream of GAs (Bethke et al. 2007) regulating both biosynthesis and perception/transduction of GAs (Leon and Lozano-Juste 2011).
In the present study employing S. raeseri, callus induction percentage was high (90–100%) in all SNP treatments including control except for 40 μM SNP where only the 10% of the explants induced callus. According to a previous study conducted by Sarropoulou et al. (2015) in CAB-6P and Gisela 6 cherry rootstocks, callus induction was evident only when SNP (0–50 μM) was applied with NAA and not when applied alone or in combination with BA. Dioscorea opposite Thunb tuber explants cultured in vitro, when treated with 40 μM SNP gave 87% callus induction percentage (Χu et al. 2009). Different results were exhibited by Han et al. (2009) who did not observe callus formation in Malus hupehensis explants treated with SNP.
CoCl2 (0.5–100 μM) applied simultaneously with 2.2 μM BA enhanced in vitro shoot proliferation of S. raeseri explants increasing shoot number, shoot length and shoot multiplication percentage. According to Sarropoulou et al. (2016), CoCl2 led to the production of multiple shoots in Gisela 6 cherry rootstock to a limiting degree, while it had absolutely no effect in the CAB-6P cherry rootstock. In consistency with our findings, the exogenous administration of 30 µM CoCl2 to the culture medium resulted in the optimum response of explants regarding shoot number, shoot length and shoot multiplication percentage in several other plant species including pepper (Capsicum frutescens Mill.) (Sharma et al. 2008), cucumber (Cucumber sativus L.) (Vasudevan et al. 2006) and Gisela 6 cherry rootstock (P. cerasus × P. canescens) (Sarropoulou et al. 2016). The ethylene inhibitor CoCl2 has been shown to be effective for shoot proliferation by inhibiting ethylene production in cucumber (Mhatre et al. 1998) and muskmelon (Yadav et al. 1996). According to Chae and Park (2012), 4.2 μM CoCl2 promoted shoot proliferation of Echinacea angustifolia whereas higher CoCl2 concentrations (21–84 μM) had an inhibitory effect. There is evidence which clearly shows that Co interacts with other microelements of nutrient culture medium to form complexes with plant growth regulators, improving the metabolic activities that lead to shoot initiation and development (Palit et al. 1994). Another explanation for the beneficial effect of Co on plant growth could be ascribed according to Rathsooriya and Nagarajah (2003) to an increase in leaf water potential compared with Co-untreated plants, thus enhancing the process of photosynthesis.
In the present study, shoot length of S. raeseri explants was considerably enhanced due to CoCl2 + BA application. In CAB-6P and Gisela 6 cherry rootstocks cultured in vitro, on the contrary, shoot length of explants was significantly diminished when 10–50 μM CoCl2 was exogenously applied to the MS medium (BA-free) (Sarropoulou et al. 2016). Our results are also not in line with those presented for chickpea (Cicer arietinum cv. T-3), where CoCl2 negatively affected shoot length (Khan and Khan 2010). Similar results to our study were reported for broccoli (Brassica oleracea var. italica), where the addition of different Co levels to the growth media significantly increased plant height (Gad and Abd El-Moez 2011). In greengram (Vigna radiata L. Wilczek), on the other hand, 210 μM CoCl2 significantly increased shoot length whereas higher CoCl2 concentrations resulted in a considerable decrease of shoot length (Abdul Jaleel et al. 2009). Co at high levels may inhibit shoot growth directly by inhibiting either cell division or cell elongation, or combination of both concurrently, resulting in limited exploration of culture medium volume for uptake and translocation of nutrients and water from plant tissues, and thus inducing mineral deficiency (Hemantaranjan et al. 2000).
In S. raeseri, 11.6 μM α-tocopherol combined with 2.2 μM ZT and 0.2 μΜ NAA promoted explants’ in vitro shoot proliferation in terms of shoot number and shoot multiplication percentage. Our results are in agreement with those recorded by Gupta and Datta (2003) who noted that 100 μM α-tocopherol stimulated shoot organogenesis of Gladiolus hybridus Hort in vitro culture. According to Lee and Read (1988), in Petunia hybrida “Champagne” in vitro culture, the formation of adventitious shoots from leaf segments was accelerated due to the application of α-tocopherol (1.1–4.6 μM) + BA (0.8 μM) while the combined effect of α-tocopherol + NAA did not have any effect. In contrast, 11.6 μM α-tocopherol adversely affected shoot elongation of S. raeseri explants whereas lower (2.3–5.8 μM) and higher (23.2 μM) α-tocopherol concentrations did not differentiate shoot length considerably. However, shoot height of in vitro regenerated P. hybrida “Champagne” explants was not altered significantly while increasing α-tocopherol concentration for each NAA level (Lee and Read 1988).
In S. raeseri, the percentage of vitrified explants was remarkably decreased and the phenomenon of microshoots with symptoms of browning and subsequent necrosis was completely diminished (total disappearance) under the influence of 11.6 μM α-tocopherol, showing that α-tocopherol has ameliorating effects because of its antioxidant properties, thus, enhancing shoot proliferation. According to Uchendu and Reed (2010), shoot tips of 2 blackberry cultivars with moderate (40–60%) regrowth after standard PVS2 vitrification when treated with vitamin E (11–15 mM) had improved in vitro shoot regrowth after cryopreservation (80%) compared to the controls (50%). Sattler et al. (2004) showed that tocopherols were vital in preventing lipid peroxidation in seeds of Arabidopsis thaliana L. during germination. It is evident that α-tocopherol acts as an antioxidant in S. raeseri plants defense system under in vitro conditions and stimulates the positive effect of ZT + NAA on adventitious shoot formation pointing out a possible synergism between α-tocopherol and cytokinin + auxin combination.
Conclusions
In conclusion, 10 μM SNP or 10–50 μM CoCl2, each applied separately intensified the stimulatory effect of the cytokinin BA (2.2 μM) on shoot proliferation in terms of shoot number and shoot multiplication percentage. α-Tocopherol at 11.6 μM acted synergistically with 2.2 μM ZT and 0.2 μM NAA in promoting shoot proliferation. Therefore, CoCl2 or α-tocopherol to a greater extent, but SNP to a lesser one due to mild hyperhydricity symptoms that causes to explants, each substance applied separately in combination with plant growth regulators (cytokinins alone and with auxins) can potentially constitute promising shoot proliferation agents in in vitro culture of several Sideritis species, as it was proved for S. raeseri studied in this research.
References
Abdul Jaleel C, Jayakumar K, Chang-Xing Z, Azooz MM (2009) Antioxidant potentials protect Vigna radiata (L.) Wilczek plants from soil cobalt stress and improve growth and pigment composition. Plant Omics J 2:120–126
Arasimowicz M, Wieczorek JF (2007) Nitric oxide as a bioactive signaling molecule in plant stress responses. Plant Sci 172:876–887
Aziz Eman E, Gad N, Badran NM (2007) Effect of cobalt and nickel on plant growth, yield and flavonoids content of Hibiscus sabdariffa L. Aust J Basic Appl Sci 1:73–78
Beligni MV, Lamattina L (2000) Nitric oxide stimulates seed germination and de-etiolation and inhibits hypocotyl elongation, three light inducible responses in plants. Planta 210:215–221
Bethke PC, Libourel IGL, Aoyama N, Chung YY, Still DW, Jones RL (2007) The Arabidopsis aleurone layer responds to nitric oxide, gibberellin, and abscisic acid and is sufficient and necessary for seed dormancy. Plant Physiol 143:1173–1188
Cachiţă-Cosma D (1987) Metode in vitro la plantele de cultură. Editura Ceres, Bucureşti
Carelli AA, Franco CI, Crapiste HG (2005) Effectiveness of added natural antioxidants in sunflower oil. Grasas Aceites 56:303–310
Caretto S, Speth EB, Fachechi C, Gala R, Zacheo G, Giovinazzo G (2004) Enhancement of vitamin E production in sunflower cell cultures. Plant Cell Rep 23(3):174–179
Carimi F, Zottini M, Costa A, Cattelan I, De Michele R, Terzi M, Lo Schiavo F (2005) NO signalling in cytokinin-induced programmed cell death. Plant Cell Environ 28:1171–1178
Chae SC, Park SU (2012) Improved shoot organogenesis of Echinacea angustifolia DC treated with ethylene inhibitors. Life Sci J 9:1725–1728
Danova K, Evstatieva L, Todorova M, Trendafilova A, Wolfram E (2013) Optimization of in vitro culture system for biomass and polyphenolics production in Inula britannica and Sideritis scardica Sofia 2 cultivar. Planta Med. doi:10.1055/s-0033-1352365
El-Sheekh MM, El-Naggar AH, Osman MEH, El-Mazaly E (2003) Effect of cobalt on growth, pigments and the photosynthetic electron transport in Monoraphidium minutum and Nitzchia perminuta. Braz. J Plant Physiol 15:159–166
Erdağ BB, Yόrekli AK (2000) Callus induction and shoot regeneration of Sideritis sipylea Boiss. (Lamiaceae). Bull Pure Appl Sci 19B(1):41–46
Evstatieva L, Koleva I (2000) Cultivation of Sideritis scardica Griseb. In: Proceeding of the first conference on medicinal and aromatic plants of South Eastern European Countries, Arandolovac, Yugoslavia, pp 189–195
Faria JLC, Kostenyuk I, Segura J (1998) Isolation, culture and plant regeneration from protoplasts of Sideritis angustifolia. J Plant Physiol 153:251–254
Fendereski F, Ghorbanli M, Sateei A (2015) Effects of copper heavy metal and interaction with nitric oxide on growth parameters, photosynthetic pigment, soluble carbohydrate content and antioxidant enzymes in Portulaca oleracea L. Iranian. J Plant Physiol 5(3):1411–1423
Gad N, Abd El-Moez MR (2011) Broccoli growth, yield quantity and quality as affected by cobalt nutrition. Agric Biol J N Am 2:226–231
Garcia-Gonzales R, Quiroz K, Carrasco B, Caligari P (2010) Plant tissue culture: Current status, opportunities and challenges. Cien Inv Agr 37(3):5–30
Garcia-Granados A, Martinez A, Onorato ME, Parra A, Recondo MB, Rivas F, Arrebola ML, Socorro O (1994) Products with biological activity obtained from in vitro micropropagated Sideritis foetens. Phytochem 35:645–650
Goliaris A (1984) Cultivation of the mountain tea. Ministry Agric Agritica 16:29–31
Gonzalez-Burgos E, Carretero ME, Gomez-Serranillos MP (2011) Sideritis spp.: uses, chemical composition and pharmacological activities—a review. J Ethnopharmacol 135(2):209–225
Guo Y, Tian Z, Yan D, Zhang J, Qin P (2009) Effects of nitric oxide on salt stress tolerance in Kosteletzkya virginica. Life Sci J 6:67–75
Gupta SD, Datta S (2003) Antioxidant enzyme activities during in vitro morphogenesis of gladiolus and the effect of application of antioxidants on plant regeneration. Biol Plant 47(2):179–183
Han XJ, Yang HQ, Duan KX, Zhang XR, Zhao HZ, You SZ, Jiang QQ (2009) Sodium nitroprusside promotes multiplication and regeneration of Malus hupehensis in vitro plantlets. Plant Cell Tiss Org Cult 96:29–34
Hemantaranjan A, Trivedi AK, Ram M (2000) Effect of foliar applied boron and soil applied iron and sulphur on growth and yield of soybean (Glycine max L. Merr.). Indian. J Plant Physiol 5:142–144
Hilmy LM, Gad N (2002) Effect of cobalt fertilization on the yield, quality and the essential oil composition of parsley leaves. Arab Univ J Agric Sci Ain Shams Univ, Cairo, Egypt 10:803–829
IUCN (2001) IUCN red list categories: version 3.1, prepared By IUCN species survival commission, Gland, Switzerland and Cambridge, UK
Jha AK, Dahleen LS, Suttle JC (2007) Ethylene influences green plant regeneration from barley callus. Plant Cell Rep 26:285–290
Jhanji S, Setia RC, Kaur N, Kaur P, Setia N (2012) Role of nitric oxide in cadmium-induced stress on growth, photosynthetic components and yield of Brassica napus L. J Environ Biol 33:1027–1032
Kalra C, Babbar SB (2010) Nitric oxide promotes in vitro organogenesis in Linum usitatissimum L. Plant Cell Tiss Org Cult 103:353–359
Kalra C, Babbar SB (2012) Stimulatory and period-specific effect of nitric oxide on in vitro caulogenesis in Albizzia lebbeck (L.) Benth. Acta Physiol Plant 34:387–392
Khan MR, Khan MM (2010) Effect of varying concentration of nickel and cobalt on the plant growth and yield of chickpea. Aust J Basic Appl Sci 4:1036–1046
Kolberz Z, Bartha B, Erdei L (2008) Exogenous auxin-induced NO synthesis is nitrate reductase-associated in Arabidopsis thaliana root primordial. J Plant Physiol 165:967–975
Laspina NV, Groppa MD, Tomaro ML, Benavides MP (2005) Nitric oxide protects sunflower leaves against Cd-induced oxidative stress. Plant Sci 169:323–330
Lee SW, Read PE (1988) Effect of α-tocopherol on organogenesis of Petunia “Champagne” cultured in vitro. Korea Agric Sci Dig Library 9:152–160
Leon J, Lozano-Juste J (2011) Nitric oxide regulates DELLA content and PIF expression to promote photomorphogenesis in Arabidopsis. Plant Physiol 156:1410–1423
Leterrier M, Valderrama R, Chaki M, Airaki M, Palma JM, Barroso JB, Corpas FJ (2012) Function of nitric oxide under environmental stress conditions. In: Khan NA, Nazar R, Iqbal N, Anjum NA (eds) Phytohormones and abiotic stress tolerance in plants. Springer, Berlin, pp 99–113
Li CZ, Wang D, Wang GX (2005) The protective effects of cobalt on potato seedling leaves during osmotic stress. Bot Bull Acad Sin 46:119–125
Liebler DC (1993) The role of metabolism in the antioxidant function of vitamin E. Crit Rev Toxicol 23:147–169
Mhatre M, Mirza M, Rao PS (1998) Stimulatory effect of ethylene inhibitors and orthocoumaric acid in tissue cultures of cucumber, Cucumis sativus L. Ind J Exp Biol 36:104–107
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Otvos K, Pasternak TP, Miskolczi P, Domoki M, Dorjgotov D, Szucs A, Bottka S, Dudits D, Feher A (2005) Nitric oxide is required for, and promotes auxin-mediated activation of cell division and embryogenic cell formation but does not influence cell cycle progression in alfafa cell cultures. Plant J 43:849–860
Palit S, Sharma A, Talukder G (1994) Effects of cobalt on plants. Bot Rev 60:149–181
Papafotiou M, Kalantzis A (2009) Seed germination and in vitro propagation of Sideritis athoa. Acta Hortic. VI Int. Symp on new floricultural crops, Funchal, Madeira, Portugal 813:471–476
Papanikolaou K, Kokkini S (1982) A taxonomic revision of Sideritis L. section Empedoclia (Rafin.) Bentham (Labiatae) in Greece. In: Margaris N, Koedam A, Vokou D (eds) Aromatic plants: basic and applied aspects, Martinus Nijhoff Publishers, The Hague, The Netherlands, pp 101–128. Proc Int Symp on Aromatic Plants, 14–19 September 1981, Kallithea (Chalkidiki), Greece
Pullman GS, Chopra R, Chase KM (2006) Loblolly pine (Pinus taeda L.) somatic embryogenesis: Improvements in embryogenic tissue initiation by supplementation of medium with organic acids, Vitamins B12 and E. Plant Sci 170(3):648–658
Qiao W, Fan LM (2008) Nitric oxide signaling in plant responses to abiotic stresses. J Integr Plant Biol 50:1238–1246
Rathsooriya GB, Nagarajah S (2003) Studies effect of salinity with cobalt on tomato plants. Plant Soil 44:158–158
Sanchez-Gras MC, Segura J (1987) In vitro propagation of Sideritis angustifolia Lag. J Plant Physiol 130:93–99
Sanchez-Gras MC, Segura J (1988) Morphogenesis in vitro of Sideritis angustifolia: effects of auxins, benzyladenine and spermidine. Plant Sci 57:151–158
Sanchez-Gras MC, Segura J (1997) Micropropagation of Sideritis species. In: Bajaj YPS (ed) High-tech and micropropagation VI—biotechnology in agriculture and forestry, vol 40. Springer-Verlag, Berlin Heidelberg, pp 343–359
Sandra AT, Maira O (2013) Effect of culture medium consistence and silver nitrate on micropropagation of two potato (Solanum tuberosum) cultivars. Rev Colomb Biotecnol 15(2):55–62
Sarropoulou V, Maloupa E (2015) Effect of exogenous dikegulac on in vitro shoot proliferation ofSideritis raeseri L.—Greek mountain tea species. Agric For 61(4):153–159
Sarropoulou V, Dimassi-Theriou K, Therios I (2015) Effect of sodium nitroprusside on micropropagation and biochemical parameters of CAB-6P and Gisela 6 cherry rootstocks. Turk J Biol 39:595–610
Sarropoulou V, Dimassi-Theriou K, Therios I (2016) Effect of the ethylene inhibitors silver nitrate, silver sulphate and cobalt chloride on micropropagation and biochemical parameters in the cherry rootstocks CAB-6P and Gisela 6. Turk J Biol 40(3):670–683
Sattler SE, Gilliland LU, Magallanes-Lundback M, Pollard M, DellaPenna D (2004) Vitamin E is essential for seed longevity and for preventing lipid peroxidation during germination. Plant Cell 16:1419–1432
Sharma A, Kumar V, Giridhar P, Ravishankar GA (2008) Induction of in vitro flowering in Capsicum frutescens under the influence of silver nitrate and cobalt chloride and pollen transformation. Electronic J Biotechnol 11:1–6
Steinitz B, Barr N, Tabib Y, Vaknin Y, Bernstein N (2010) Control of in vitro rooting and plant development in Corymbia maculate by silver nitrate, silver thiosulfate and thiosulfate ion. Plant Cell Rep 29(11):1315–1323
Strid A, Tan T (1991) Mountain flora of Greece, vol 2. Edinburgh University Press, Edinburgh, p 974
Tian XR, Lei YB (2007) Physiological responses of wheat seedlings to drought and UV-B radiation. Effect of exogenous sodium nitroprusside application. Russ J Plant Physiol 54:676–682
Todorova M, Trendafilova A (2014) Sideritis scardica Griseb., an endemic species of Balkan peninsula: traditional uses, cultivation, chemical composition, biological activity. J Ethnopharmacol 152(2):256–265
Tun NN, Holk A, Scherer FE (2001) Rapid increase of NO release in plant cell cultures induced by cytokinin. FEBS Lett 509:174–176
Uçar E, Turgut K (2009) In vitro propagation of some mountain tea (Sideritis) species. Ziraat Fakόltesi Dergisi. Akdeniz universitesi 22:51–57
Uchendu EE, Reed BM (2010) Vitamins C and E improve in vitro recovery of cryopreserved blackberry shoot tips. CryoLetters 31(2):169–197
Vasudevan A, Selvaraj N, Ganapathi A, Chang WC, Manickavasagam M, Kasthurirengan S (2006) Effect of ethylene inhibitors on in vitro shoot multiplication and their impact on ethylene production in cucumber (Cucumber sativus L.). Res J Bot 1:110–117
Woo HJ, Sohn SI, Shin KS, Kim JK, Kim BG, Lim MH (2014) Expression of tobacco tocopherol cyclase in rice regulates antioxidative defense and drought tolerance. Plant Cell Tiss Organ Cult 119:257–267
Xu J, Yin H, Wang W, Mi Q, Liu X (2009) Effects of sodium nitroprusside on callus induction and shoot regeneration in micropropagated Dioscorea opposita. Plant Growth Regul 59:279–285
Yadav RC, Saleh MT, Grumet R (1996) High frequency shoot regeneration from leaf explants of muskmelon. Plant Cell Tiss Organ Cult 45:207–214
Yigit D (2014) Composition and antimicrobial activity of essential oil of Sideritis cypria species from TRNC. Near East University, Faculty of Pharmacy, Department of Pharmaceutical Botany. Master Thesis
Acknowledgements
This research project was funded under the Action “Research & Technology Development Innovation projects (AgroETAK)”, MIS 453350, in the framework of the Operational Program “Human Resources Development”. It was co-funded by the European Social Fund and by National Resources through the National Strategic Reference Framework 2007–2013 (NSRF 2007–2013) coordinated by the Hellenic Agricultural Organisation “DEMETER” (Institute of Genetic Breeding and Natural Resources/Scientific supervisor: Dr Eleni Maloupa).
Author contributions
VS performed all the experimental work, designed and managed the experiments, did all the statistics and wrote the first draft of the manuscript. EM contributed by scientific advices, checked the manuscript and gave her approval prior to its submission for potential publication. All the authors have read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sarropoulou, V., Maloupa, E. Effect of the NO donor “sodium nitroprusside” (SNP), the ethylene inhibitor “cobalt chloride” (CoCl2) and the antioxidant vitamin E “α-tocopherol” on in vitro shoot proliferation of Sideritis raeseri Boiss. & Heldr. subsp. raeseri . Plant Cell Tiss Organ Cult 128, 619–629 (2017). https://doi.org/10.1007/s11240-016-1139-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-016-1139-6