Abstract
Nitraria tangutorum Bobr., a typical desert halophyte, plays an important ecological role because of its superior tolerance to severe drought and high salinity. Very little is known about the physiological adaptative mechanism of this species to environmental stresses. The aim of this study was to investigate the changes of antioxidant enzyme activities and the regulatory mechanism of ascorbate peroxidase (APX) activity in the calli from Nitraria tangutorum Bobr. after treatment with different NaCl concentrations. The activities of superoxide dismutase (SOD) and catalase (CAT) significantly increased in the calli treated with NaCl, while the peroxidase activity decreased. APX activity was also elevated significantly in response to NaCl, but the increase was partly abolished by H2O2 scavenger dimethylthiourea (DMTU). Furthermore, the excitatory effect of salinity on APX could be alleviated by the addition of exogenous CAT and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase inhibitor diphenylene iodonium, indicating that the modulation of the APX activity in Nitraria tangutorum Bobr. calli might be associated with NADPH oxidase-dependent H2O2 generation. Measurement and analysis using fluorescent dye 2′,7′-dichlorodihydrofluorescein diacetate showed the increase of H2O2 content in salinity-treated calli. The investigation of NADPH-dependent O −2 production in plasma membrane (PM) vesicles isolated from Nitraria tangutorum Bobr. calli revealed that salinity treatment stimulated NADPH oxidase activity. In conclusion, these results suggest that the higher activities of antioxidant enzymes play an important role in the salt tolerance of Nitraria tangutorum Bobr. calli and that the extracellular production of H2O2, depending on the excitation of PM NADPH oxidase, is responsible for enhancing the APX activity in Nitraria tangutorum Bobr. calli under salinity stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
High salinity is a most serious environmental stress that imposes both ionic toxicity and osmotic stress, leading to the reduction of plant growth and crop production (Boyer 1982; Munns et al. 2006). One of the plant responses to environment stresses is the rapid and increased generation of reactive oxygen species (ROS), including hydrogen peroxide (H2O2), which can damage lipids, proteins and nuclei acids (Shalata and Tal 1998). However, different plants develop different protection mechanisms to eliminate ROS and prevent oxidative damage. Among them, enzymatic antioxidants, including such functionally interrelated enzymes as superoxide dismutase (SOD), catalase (CAT), peroxidase (POD) and ascorbate peroxidase (APX), provide the first line of defence against ROS (Meloni et al. 2003; Yahubyan et al. 2009). A previous study showed that salt-tolerant Plantago maritima has higher activities of antioxidant enzymes and a better protection mechanism against oxidative damage under salt stress than salt-sensitive Plantago madia (Sekmen et al. 2007). Similarly, salt stress induces increased activities of antioxidant enzymes in salt-tolerant rice (Moradi and Ismail 2007). Moreover, correlations between antioxidant capacity and salt tolerance have been suggested in a large number of true halophytes, such as Limonium bicolor (Li 2008), Thellungiella halophila (Radyukina et al. 2007) and Suaeda salsa (Pang et al. 2005; Wang et al. 2008). So, the regulation of antioxidant enzyme activities may represent an important cellular mechanism for salt resistance.
Hydrogen peroxide, which may attack and damage almost every molecule in the living cell, is one of the most reactive species in biological systems (Mishra et al. 1993). Meanwhile, H2O2, as a signal molecular, is also involved in many important plant processes associated with a variety of environmental stresses (Avsian-Kretchmer et al. 2004; Joo et al. 2005). It is well known that multiple enzymatic sources are responsible for H2O2 production in plants. For example, plasma membrane (PM) nicotinamide adenine dinucleotide phosphate (NADPH) oxidase has been described as a source of extracellular H2O2 generation (Sagi and Fluhr 2001; Qin et al. 2004) because this enzyme can transfer electrons from NADPH in the cytoplasma to molecular oxygen in the apoplast to generate superoxide anion (O −2 ), which can be subsequently converted to H2O2 (Sagi and Fluhr 2001). Recently, a few investigations have demonstrated that NADPH oxidase-dependent H2O2 plays an important role in plant responses to abiotic stresses (Avsian-Kretchmer et al. 2004; Joo et al. 2005) and the abscisic acid signal transduction pathway (Kwak et al. 2003), as well as the modulation of plant calcium (Yang et al. 2007). However, no clear relationships between the generation of NADPH oxidase-dependent H2O2 and the regulation of antioxidant enzyme activities have been reported in plants treated with salt.
Nitraria tangutorum Bobr., a shrub belonging to the Nitraria genus in Zygophyllaceae, is a typical plant in the desert with the strong ability to resist salinity and alkalinity, drought, wind and dust, and it can efficiently fix moving sands and decrease wind speed (Yang and Furukawa 2006). Additionally, this species owns high potential economic values as a source of edible and medicinal compounds. For instance, the fruit of Nitraria tangutorum Bobr. can better decrease blood lipid and have an antioxidative effect (Suo et al. 2004). It can prevent and cure atherosclerosis as well. Nitraria tangutorum Bobr. usually distributes in those desert areas in the north-west of China. A previous study mainly focused on a relationship between anatomical features and adaptability to desert environment in this species (Yang and Furukawa 2006). There are few researches on the physiological adaptative responses of Nitraria tangutorum Bobr. to salt environment. Salinity tolerance is a complicated whole-plant phenomenon, and it is necessary to understand the integration and expression of salinity-tolerant mechanisms from the plant-cell level to the whole-plant level (Dracup 1991). A comprehensive study of the physiological adaptative mechanism at the cellular level would be of great help in understanding the processes controlling plant growth and survival in a hostile environment. Therefore, in the present study, we used the calli from Nitraria tangutorum Bobr. to investigate the responses of antioxidative enzyme and the relationship between the regulation of APX activity and the generation of H2O2 after salinity treatment.
Materials and methods
Plant material and growth condition
Seeds of Nitraria tangutorum Bobr. were obtained from Minqin desert botanical garden of Gansu province. After having been surface-sterilised for 12 s with 75% (v/v) ethanol and then in 0.1% HgCl2 for 10 min, the seeds were rinsed six times and then soaked in sterile distilled water for 2 days. The embryos were extracted and incubated on 30 ml of growth regulator-free Murashige and Skoog (MS) solid medium. After subculture for about 25 days, the cotyledons from aseptic seedlings were cut to about 0.3 cm and subsequently placed on MS solid medium supplemented with 0.3 mg l−1 6-benzyladenine (6-BA) and 1 mg l−1 α-naphthaleneacetic acid (NAA) for inducing callus. Initial calli were incubated for about 18 days and then subculture was performed every 18–20 days on the same medium.
MS medium in all experiments was supplemented with 0.3 mg l−1 6-BA and 1 mg l−1 NAA. NaCl (50, 100 and 200 mM) was added in MS medium for salt stress. H2O2, dimethylthiourea (DMTU), CAT and diphenylene iodonium (DPI) (prepared with sterilised water) were added on the surface of MS medium after filter sterilisation. Control calli were cultured on MS medium without NaCl. All calli were maintained at 24 ± 1.5°C in the dark for 3, 6 or 9 days, and then evaluated and washed by distilled water. Excess water was blotted with filter paper.
Antioxidant enzyme activity measurement
After 3, 6 and 9 days of culture on MS medium containing 0.3 mg l−1 6-BA, 1 mg l−1 NAA and different NaCl concentrations (0, 50, 100 or 200 mM), calli were collected for analysing enzyme activities. The overall procedure was carried out at 4°C. One gram of plant material was ground with 1 ml of chilled NaH2PO4/Na2HPO4 buffer (phosphate-buffered saline [PBS], 50 mM, pH 7.8) containing 0.1 mM ethylenediaminetetraacetic acid (EDTA) and 1% polyvinylpyrrolidone (PVP). After centrifugation for 30 min at 15,000g, the supernatant (enzyme extraction) was collected for the measurement of SOD, POD and CAT activities.
SOD (EC 1.15.1.1) activity was estimated based on the method described by Dhindsa and Matow (1981). A quantity of 50 μl of the enzyme extraction was added to 3 ml of a reaction mixture consisting of 50 mM PBS (pH 7.6), 13 mM methionine, 75 μM nitroblue tetrazolium (NBT) and 0.1 mM EDTA-Na2. The reaction was started by the addition of 2 μM lactochrome. After illumination for 30 min at 25°C using a non-illumination as the reference, the absorbance was recorded at 560 nm. The complete reaction medium without enzyme incubated in the dark was used as the dark control. One unit of enzyme activity (U) was defined as the quantity of SOD required to produce a 50% inhibition of the reduction of NBT and the results were given as units of the SOD activity per gram of fresh weight (U g−1 FW).
A modification of the method of Aebi (1974) was used to assay CAT (EC 1.11.1.6) activity. Briefly, 100 μl of the enzyme extraction was added to 3 ml of 50 mM PBS buffer (pH 7.0). After 5 min pre-incubation at 25°C, 15 mM H2O2 was added to start the reaction and the absorbance changes were recorded at 240 nm for 3 min. An absorbance change of 0.01 unit min−1 was defined as 1 unit of CAT activity, and the CAT activity was expressed as U g−1 FW.
POD (EC 1.11.1.7) activity was measured following a modification of the method of Rao et al. (1996). A quantity of 5 μl of the enzyme extraction was mixed with 3 ml of reaction mixture containing 50 mM PBS (pH 6.5) and 20 mM guaiacol. After pre-incubation at 25°C for 5 min, 40 μl of 0.05% H2O2 was added to initiate the reaction. The changes in the absorbance at 470 nm within 3 min were recorded for calculating the POD activity; one unit of POD activity was defined as an absorbance change of 0.1 unit min−1 and the POD activity was expressed as U g−1 FW.
APX activity measurement
The calli cultured for 3, 6 and 9 days on MS medium containing 0.3 mg l−1 6-BA, 1 mg l−1 NAA and different NaCl concentrations (0, 50, 100 or 200 mM) were collected for our analysis of the APX activity. One gram of plant material was ground with 1 ml of chilled 50 mM PBS buffer (pH 7.0) containing 1 mM EDTA and 1 mM ASA. After centrifugation for 30 min at 15,000g, the supernatant (enzyme extraction) was collected for the measurement of APX (EC 1.11.1.11) activity.
The assay of APX activity was performed as described by Nakano and Asada (1981), with some modifications. The assay was carried out in a reaction mixture consisting of 50 mM PBS (pH 7.0), 0.5 mM ascorbate (ASA), 0.1 mM H2O2 and 100 μl of the enzyme extraction. The changes in the absorbance at 290 nm were recorded at 25°C for 1 min after the addition of H2O2. One unit of APX activity was defined as an absorbance change of 0.1 unit min−1 and the APX activity was expressed as U g−1 FW.
H2O2 content measurement
After 3 days of culture on MS medium containing 0.3 mg l−1 6-BA, 1 mg l−1 NAA and various NaCl concentrations (0, 50, 100 and 200 mM), calli were collected for the measurement of H2O2 level according to the method of Sergiev et al. (1997). One gram of fresh calli was ground in an ice bath with 2 ml of 0.1% trichloroacetic acid. The homogenate was centrifuged at 12,000g for 20 min and 0.7 ml of the supernatant was mixed with 0.7 ml 10 mM PBS buffer (pH 7.0) and 0.7 ml 1 M KI. The H2O2 content was estimated by measuring the absorbance at 390 nm, and a standard curve was made using H2O2 in the same way.
After treatment with 0 and 50 mM NaCl for 3 days, calli were collected and the fluorescence intensity of H2O2 was visualised using a fluorescent dye 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA) as described by Pei et al. (2000). The calli were immersed in 50 μM H2DCFDA in 10 mM tris-(hydroxymethyl) amino methane (Tris)–HCl buffer (pH 7.2) containing 50 mM KCl for 20 min at 37°C and were washed three times in Tris–HCl buffer (10 min each) to remove excess dye. Then, the fixed callus cells were treated with 1% pectinase (in MS medium without agar) for 20 min. Examinations of H2DCFDA fluorescence were observed using a laser scanning confocal microscope (LSCM, LSM510 Meta, Zeiss) with the following settings: excitation = 488 nm, emission = 525 nm. To quantify fluorescence, images were processed and analysed with Zeiss LSM 510 software.
Plasma membrane purification
The calli collected from the subculture medium were transferred to MS medium supplemented with different NaCl concentrations (0 and 50 mM), 0.3 mg l−1 6-BA and 1 mg l−1 NAA. After 3 or 6 days of treatment, PM vesicles were isolated to determine the NADPH oxidase activity. The PM-rich vesicles were fractionated by two-phase partitioning using the method of Buckhout et al. (1989), with some modifications. All steps were carried out at 4°C. The calli were immediately ground in the isolation medium (1:2 w/v) containing 250 mM sucrose, 25 mM Tris–HCl, pH 7.5, 5 mM EDTA, 1 mM ethylene glycol tetraacetic acid, 1 mM 1,4-dithiothreitol (DTT), 2 g PVP/100 g cell and 1 mM phenylmethylsulfonyl fluoride (PMSF). The homogenate was filtered through four layers of cheesecloth and the resulting filtrate was centrifuged at 15,000g for 20 min. The supernatant was then centrifuged for 30 min at 100,000g and the crude microsomes were resuspended in 0.049% PBS buffer (pH 7.8). The PM fraction was isolated by adding the microsomal suspension to a two-phase partition system consisting of 6.3% polyethylene glycol 3350, 6.3% Dextran T-500, 8.7% sucrose, 0.014% KCl and 0.049% PBS buffer (pH 7.8). The resulting upper phases obtained after three successive rounds of partitioning were pelleted by centrifugation at 100,000g for 30 min. The pellets were washed with a resuspension buffer (containing 50 mM Tris–HCl, pH 7.4, 250 mM sucrose, 1 mM EDTA, 1 mM DTT and 1 mM PMSF) and then resuspended in the resuspension buffer.
NADPH oxidase activity measurement
The determination of NADPH-dependent O −2 -generation activity in the isolated PM vesicles was carried out as described by Van Gestelen et al. (1997). The assay reaction mixture consisted of 30–400 μg PM proteins, 250 mM sucrose, 0.1 mM NBT and 0.02% polyoxyethylene (20) cetyl ether in 50 mM Tris–HCl buffer (pH 7.4). After 1 min pre-incubation, the reaction was initiated with the addition of 0.1 mM NADPH and the absorbance changes at 530 nm were followed for 5 min. Rates of O −2 generation were calculated using an extinction coefficient of 12.8 mM−1 cm−1. NBT reduction by O −2 was determined from the differences of NBT reduction rates in the presence and absence of 50 units of SOD.
Soluble protein content determination
The amount of proteins was estimated according to the method proposed by Bradford (1976) and the standard curve was made using bovine serum albumin.
Statistical analysis
All values are represented by an average of at least three replicate measurements ± standard error (SE) and the significance of differences between the control and treatments was statistically evaluated by Student’s t-test methods.
Results
Effects of NaCl on the growth of the calli and antioxidant enzyme activities
The growth response of control calli is shown in Fig. 1a after culture for 18 days on MS medium supplemented with 0.3 mg l−1 6-BA and 1 mg l−1 NAA without NaCl. The calli exposed to 50 and 100 mM NaCl for 18 days exhibited good growth (Fig. 1b, c), whereas a significant reduction in growth was observed when the concentration of NaCl was increased to 200 mM (Fig. 1d).
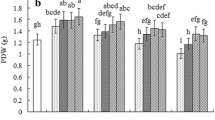
The effect of NaCl on the total SOD activity in Nitraria tangutorum Bobr. calli is shown in Fig. 2a. In comparison with untreated calli, the calli treated with 50 mM NaCl for 3 and 6 days showed about 23 and 14% increase in SOD activity, respectively, but there was an exception to this where the enzyme activity was lower at 9 days. According to our experiments, 100 and 200 mM NaCl induced a significant increase in SOD activity throughout the experimental period (3, 6 and 9 days), but in contrast to 100 mM, NaCl at 200 mM had a greater effect on the SOD activity during the same period of stress (Fig. 2a).
Effect of NaCl on antioxidant enzyme activities in Nitraria tangutorum Bobr. calli. a Superoxide dismutase (SOD). b Catalase (CAT). c Peroxidase (POD). d Ascorbate peroxidase (APX). Average values and standard errors obtained from at least three independent measurements are shown. The * indicate significantly different means with P < 0.05 when compared with the control value
Similarly, CAT activity increased significantly in Nitraria tangutorum Bobr. calli under NaCl treatment (Fig. 2b). During the entire experimental period, 50 mM NaCl induced a significant elevation in CAT activity in a durative-dependent manner. Additionally, the CAT activity increased to 119.54, 141.16 and 119.16% of the control value in the calli after treatment with 100 mM NaCl for 3, 6 and 9 days, respectively. However, compared with control calli, increased rates of CAT activity remained constant (about 40% enhancement) for 9 days under 200 mM NaCl treatment.
In contrast, NaCl treatment resulted in a significant decrease in POD activity in Nitraria tangutorum Bobr. calli (Fig. 2c). In comparison with control calli, POD activity continued to decrease in treated calli with 50 mM NaCl with the stress period increasing. Under 100 mM NaCl treatment, the enzyme activity was 89, 76 and 78% of the control value in calli cultured for 3, 6 and 9 days, respectively. Apart from these, a slight but significant decrease (about 12%) in POD activity was detected after treatment with 200 mM NaCl for 3 and 6 days, whereas in the calli treated for 9 days, the POD activity remained almost unchanged in comparison with untreated calli (Fig. 2c).
NaCl-induced H2O2 generation account for the regulation of APX activity
Figure 2d showed that different NaCl concentrations induced the increase of APX activity in a durative-dependent manner in Nitraria tangutorum Bobr. calli. Compared with control calli, the APX activity was increased by 18 and 25%, respectively, in the calli stressed with 50 mM NaCl for 3 and 6 days, and the increase rate reached its maximum value (30%) in NaCl 9-day-treated calli. In addition, NaCl treatment induced a remarkable increase in APX activity according to stress intensity (Fig. 2d). When calli were exposed to 50, 100 and 200 mM NaCl for 9 days, there was about 30, 60 and 86% increase in APX activity in comparison with control calli, respectively.
The effect of exogenous H2O2 on APX activity was studied in the calli from Nitraria tangutorum Bobr. and the results are shown in Fig. 3. At concentrations of 5 μM or above, H2O2 significantly inhibited APX activity, whereas low H2O2 concentrations (0.5 and 1 μM) resulted in a significant enhancement in APX activity.
The application of DMTU, an H2O2 scavenger, could alleviate the salinity-induced elevation of APX activity in the calli of Nitraria tangutorum Bobr. (Fig. 4), indicating that the excitatory effect of NaCl might be due to H2O2 generation caused by NaCl. Exogenous CAT is a cell-impermeable scavenger of H2O2 (Karlsson et al. 2000). When 150 U l−1 CAT was applied together with 50 mM NaCl, the NaCl-induced increase of APX activity was abolished (Fig. 4), suggesting that NaCl might induce the excitation of APX via a pathway associated with extracellular H2O2 generation. Furthermore, the excitatory effect of NaCl could be reversed by adding 20 μM DPI (Fig. 4), a chemical inhibitor of NADPH oxidase (Auh and Murphy 1995). These data suggested that the increased APX activity might be due to PM NADPH oxidase-dependent production of extracellular H2O2 in response to salinity stress.
Effects of DMTU (5 mM), CAT (150 U l−1) and DPI (20 μM) on the excitation of ascorbate peroxidase (APX) induced by 50 mM NaCl stress for 3 days in the calli from Nitraria tangutorum Bobr. The control activity of APX (100%) corresponded to 15.47 U g−1 FW. Values represent the means of at least five independent experiments and the bars indicate the standard error. The * and + indicate significant statistical differences (P < 0.05) when compared with the columns ‘Control’ and ‘NaCl’, respectively
Effect of NaCl on hydrogen peroxide levels
The results of the measurement of H2O2 level in NaCl-treated calli and control calli are presented in Fig. 5. Compared with the control, H2O2 generation clearly increased in the calli of Nitraria tangutorum Bobr. stressed with NaCl (50, 100 and 200 mM) for 3 days (Fig. 5a). Additionally, the level of H2O2 in callus cells was determined by using LSCM and fluorophore H2DCF-DA (a H2O2-sensitive fluorescent dye). H2DCFDA fluorescence was detected in untreated cells, demonstrating that a basal level of H2O2 production occurred in the callus cells from Nitraria tangutorum Bobr. After treatment with 50 mM NaCl for 3 days, the endogenous H2O2 level increased by approximately 29% in comparison with the untreated cells (observed as green fluorescence, Fig. 5 b, c).
a Changes of H2O2 level in the calli of Nitraria tangutorum Bobr. after NaCl treatment for 3 days. Values are the averages based on at least three replicates ± standard error. The * indicate significant statistical differences (with P < 0.05) when compared with the control value. b Fluorescence intensity of H2O2 visualised using a fluorescent dye 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA) in callus cells of Nitraria tangutorum Bobr. c Mean relative H2DCFDA fluorescence densities for callus cells corresponding to Fig. 5b. Values represent the averages of at least five cells for each treatment ± standard error. Significant difference between NaCl 3-day-stressed calli and control calli is indicated with * (P < 0.05)
Effects of NaCl on PM NADPH oxidase activity
Further study was to isolate PM vesicles from Nitraria tangutorum Bobr. calli to determine the NADPH oxidase activity. The O −2 production was 9.14 nmol mg−1 protein min−1 in control PM vesicles. The treatment of calli with 50 mM NaCl for 3 and 6 days resulted in about 38 and 49% increase, respectively, in the NADPH-dependent O −2 generation as compared with that of the control PM vesicles (Fig. 6), indicating that salinity treatment stimulated PM NADPH oxidase in Nitraria tangutorum Bobr. calli.
Changes of NADPH oxidase activity in the calli from Nitraria tangutorum Bobr. under 50 mM NaCl treatment. Values represent the means of at least three different experiments and the bars indicate the standard error. The * indicate significant statistical difference (P < 0.05) when compared with the control
Discussion
The enhancement of antioxidant enzyme activities provides protection against oxidative damage and develops plant tolerance to salinity stresses (Sekmen et al. 2007). Halophytes are known for their ability to withstand and quench toxic ROS, since these plants are equipped with a powerful antioxidant system including enzymatic and non-enzymatic components (Ksouri et al. 2008). Bor et al. (2003) and Koca et al. (2007) reported that salinity induced higher activities of antioxidative enzymes in salt-tolerant plants than in salt-sensitive cultivated plants. Moreover, it has been suggested that the upregulation of CAT and APX is one component of the tolerant adaptation of halophytes to high salinity (Pang et al. 2005). Even though much supporting evidence on the role of antioxidant enzymes in salt tolerance is available, there is little information on the antioxidant enzymes of halophyte Nitraria tangutorum Bobr. In the present study, different NaCl concentrations resulted in the remarkable increases in the activities of SOD, CAT and APX, as well as the decrease in POD activity in Nitraria tangutorum Bobr. calli (Fig. 2). SOD is usually considered as the first line of defence against oxidative stress (Wang et al. 2008), which catalyses the dismutation of two molecules of O −2 to H2O2 and O2 (Grosicka-Maciąg et al. 2008). CAT, POD and APX, which decompose H2O2 to H2O at different cellular locations, constitute the main H2O2 scavenging system in cells (Mittler 2002; Zhu et al. 2004). The overall effect of antioxidant systems depends on the intracellular balance between antioxidant enzymes rather than a single component (Amstad et al. 1994). Therefore, our results suggested that Nitraria tangutorum Bobr. calli had a stronger ability to eliminate ROS, which protected the cells from oxidative damage under salinity stress. It was also suggested that CAT and APX might be more important than POD for the scavenging of H2O2 in Nitraria tangutorum Bobr. calli exposed to salinity.
Environmental stresses are shown to stimulate H2O2 generation in plants (Avsian-Kretchmer et al. 2004). Even though the increased activities of CAT and APX may be responsible for the elimination of H2O2 in this study, the notable increase of H2O2 content was observed in Nitraria tangutorum Bobr. calli with the increase of NaCl concentration (Figs. 5). Thus, we can suggest that maintaining a low level of H2O2 is complex and depends on different cellular mechanisms in plant tissues in response to salinity stress. In the calli of Nitraria tangutorum Bobr., we found that the application of H2O2 scavenger DMTU could reverse the NaCl-induced increase in APX activity (Fig. 4). Similar to our result, Morita et al. (1999) observed that the decrease of cellular H2O2 level using diethyldithiocarbamate, an SOD inhibitor, lessened the paraguat induction of cytosolic APX. In our study, the strong inhibition in APX activity was observed in the calli exposed to exogenous H2O2 at higher concentration, whereas 0.5 or 1 μM H2O2 resulted in the excitation of APX in Nitraria tangutorum Bobr. calli (Fig. 3). These results suggest that H2O2 may play a role in the regulation of APX activity in plants. There are multiple enzymatic sources of H2O2 in plant tissues, including extracellular and intracellular enzymes (Joo et al. 2005). The investigation of exogenous CAT effect on the NaCl-induced increase of APX activity suggested that NaCl resulted in the excitation of APX activity via a pathway involving the generation of extracellular H2O2 in Nitraria tangutorum Bobr. calli (Fig. 4). Having taken into consideration the fact that PM-bound NADPH oxidase complex in plants is an important source of extracellular H2O2 generation (Pei et al. 2000), we further measured the generation of O −2 in the PM vesicles and found significant elevation in the NADPH oxidase activity in the salinity-treated calli (Fig. 6). Furthermore, 20 μM DPI remarkably reversed the excitatory effect of salinity on APX activity induced by NaCl (Fig. 4). However, the application of the NOS inhibitor Nω-nitro-l-arginine or NO scavenger 2-(4-carboxyphenyl)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide had no effect on the NaCl-induced elevation of APX activity (data not shown). Therefore, all these results indicated that NaCl induced the excitation of APX and this excitation was due to PM NADPH oxidase-dependent H2O2 production.
In conclusion, the higher activities of SOD, CAT and APX in response to salinity stress may play an important role in salt tolerance in the calli from Nitraria tangutorum Bobr. Our results also suggest that salt stress results in the excitation of NADPH oxidase associated with elevation in the extracellular H2O2 generation, which is involved in the regulation of APX activity in Nitraria tangutorum Bobr. calli under salt stress.
Abbreviations
- APX:
-
Ascorbate peroxidase
- ASA:
-
Ascorbate
- CAT:
-
Catalase
- DMTU:
-
Dimethylthiourea
- DPI:
-
Diphenylene iodonium
- DTT:
-
1,4-dithiothreitol
- EDTA:
-
Ethylenediaminetetraacetic acid
- FW:
-
Fresh weight
- H2DCF-DA:
-
2′,7′-dichlorodihydrofluorescein diacetate
- H2O2 :
-
Hydrogen peroxide
- LSCM:
-
Laser scanning confocal microscope
- MS:
-
Murashige and Skoog
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NBT:
-
Nitroblue tetrazolium
- PBS:
-
Phosphate-buffered saline
- PM:
-
Plasma membrane
- PMSF:
-
Phenylmethanesulfonyl fluoride
- POD:
-
Peroxidase
- PVP:
-
Polyvinylpyrrolidone
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- Tris:
-
Tris-(hydroxymethyl) amino methane
References
Aebi H (1974) Catalase. In: Bergmeyer HU (ed) In methods of enzymatic analysis. Academic Press, New York, pp 673–677
Amstad P, Moret R, Cerutti P (1994) Glutathione peroxidase compensates for the hypersensitivity of Cu,Zn-superoxide dismutase overproducers to oxidant stress. J Biol Chem 269:1606–1609
Auh CK, Murphy TM (1995) Plasma membrane redox enzyme is involved in the synthesis of O −2 and H2O2 by phytophthora elicitor-stimulated rose cells. Plant Physiol 107:1241–1247
Avsian-Kretchmer O, Gueta-Dahan Y, Lev-Yadun S, Gollop R, Ben-Hayyim G (2004) The salt-stress signal transduction pathway that activates the gpx1 promoter is mediated by intracellular H2O2, different from the pathway induced by extracellular H2O2. Plant Physiol 135:1685–1696
Bor M, Özdemir F, Türkan I (2003) The effect of salt stress on lipid peroxidation and antioxidants in leaves of sugar beet Beta vulgaris L. and wild beet Beta maritima L. Plant Sci 164:77–84
Boyer JS (1982) Plant productivity and environment. Science 218:443–448
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buckhout TJ, Bell PF, Luster DG, Chaney RL (1989) Iron-stress induced redox activity in tomato (Lycopersicum esculentum Mill.) is localized on the plasma membrane. Plant Physiol 90:151–156
Dhindsa RS, Matowe W (1981) Drought tolerance in two mosses: correlated with enzymatic defence against lipid peroxidation. J Exp Bot 32:79–91
Dracup M (1991) Increasing salt tolerance of plants through cell culture requires greater understanding of tolerance mechanisms. Aust J Plant Physiol 18:1–15
Grosicka-Maciąg E, Kurpios D, Czeczot H, Szumiło M, Skrzycki M, Suchocki P, Rahden-Staroń I (2008) Changes in antioxidant defense systems induced by thiram in V79 Chinese hamster fibroblasts. Tox in Vitro 22:28–35
Joo JH, Wang SY, Chen JG, Jones AM, Fedoroff NV (2005) Different signaling and cell death roles of heterotrimeric G protein alpha and beta subunits in the Arabidopsis oxidative stress response to ozone. Plant Cell 17:957–970
Karlsson A, Nixon JB, McPhail LC (2000) Phorbol myristate acetate induces neutrophil NADPH-oxidase activity by two separate signal transduction pathways: dependent or independent of phosphatidylinositol 3-kinase. J Leuk Biol 67:396–404
Koca H, Bor M, Özdemir F, Türkan I (2007) The effect of salt stress on lipid peroxidation, antioxidative enzymes and proline content of sesame cultivars. Environ Exp Bot 60:344–351
Ksouri R, Megdiche W, Falleh H, Trabelsi N, Boulaaba M, Smaoui A, Abdelly C (2008) Influence of biological, environmental and technical factors on phenolic content and antioxidant activities of Tunisian halophytes. Compt Rend Biol 331:865–873
Kwak JM, Mori IC, Pei ZM, Leonhardt N, Torres MA, Dangl JL, Bloom RE, Bodde S, Jones JDG, Schroeder JI (2003) NADPH oxidase AtrbohD and AtrbohF genes function in ROS-dependent ABA signaling in Arabidopsis. EMBO J 22:2623–2633
Li Y (2008) Kinetics of the antioxidant response to salinity in the halophyte Limonium bicolor. Plant Soil Environ 54:493–497
Meloni DA, Oliva MA, Martinez CA, Cambraia J (2003) Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ Exp Bot 49:69–76
Mishra NP, Mishra RK, Singhal GS (1993) Changes in the activities of anti-oxidant enzymes during exposure of intact wheat leaves to strong visible light at different temperatures in the presence of protein synthesis inhibitors. Plant Physiol 102:903–910
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Moradi F, Ismail AM (2007) Responses of photosynthesis, chlorophyll fluorescence and ROS-scavenging systems to salt stress during seedling and reproductive stages in rice. Ann Bot 99:1161–1173
Morita S, Kaminaka H, Masumura T, Tanaka K (1999) Induction of rice cytosolic ascorbate peroxidase mRNA by oxidative stress; the involvement of hydrogen peroxide in oxidative stress signalling. Plant Cell Physiol 40:417–422
Munns R, James RJ, Läuchli A (2006) Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot 57:1025–1043
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Pang CH, Zhang SJ, Gong ZZ, Wang BS (2005) NaCl treatment markedly enhances H2O2-scavenging system in leaves of halophyte Suaeda salsa. Physiol Plant 125:490–499
Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406:731–734
Qin WM, Lan WZ, Yang XI (2004) Involvement of NADPH oxidase in hydrogen peroxide accumulation by Aspergillus niger elicitor-induced Taxus chinensis cell cultures. J Plant Physiol 161:355–361
Radyukina NL, Kartashov AV, Ivanov YV, Shevyakova NI, Kuznetsov VV (2007) Functioning of defense systems in halophytes and glycophytes under progressing salinity. Rus J Plant Physiol 54:806–815
Rao MV, Paliyath C, Ormrod DP (1996) Ultraviolet-B- and ozone-induced biochemical changes in antioxidant enzymes of Arabidopsis thaliana. Plant Physiol 110:125–136
Sagi M, Fluhr R (2001) Superoxide production by plant homologues of the gp91phox NADPH oxidase. Modulation of activity by calcium and by tobacco mosaic virus infection. Plant Physiol 126:1281–1290
Sekmen AH, Türkan I, Takio S (2007) Differential responses of antioxidative enzymes and lipid peroxidation to salt stress in salt-tolerant Plantago maritima and salt-sensitive Plantago media. Physiol Plant 131:399–411
Sergiev I, Alexieva V, Karanov E (1997) Effect of spermine, atrazine and combination between them on some endogenous protective systems and stress markers in plants. Compt Rend Acad Bulg Sci 51:121–124
Shalata A, Tal M (1998) The effect of salt stress on lipid peroxidation and antioxidants in the leaf of the cultivated tomato and its wild salt-tolerant relative Lycopersicon pennellii. Physiol Plant 104:169–174
Suo YR, Wang HL, Wang HQ (2004) Research on decreasing blood lipid and anti-oxidative effect of fruit of Nitraria tangutorum Bobr. from Qaidam Basin. Nat Prod Res Dev 16:54–58
Van Gestelen P, Asard H, Caubergs RJ (1997) Solubilization and separation of a plant plasma membrane NADPH-O −2 synthase from other NAD(P)H oxidoreductases. Plant Physiol 115:543–550
Wang CQ, Xu C, Wei JG, Wang HB, Wang SH (2008) Enhanced tonoplast H+-ATPase activity and superoxide dismutase activity in the halophyte Suaeda salsa containing high level of betacyanin. J Plant Growth Regul 27:58–67
Yahubyan G, Gozmanova M, Denev I, Toneva V, Minkov I (2009) Prompt response of superoxide dismutase and peroxidase to dehydration and rehydration of the resurrection plant Haberlea rhodopensis. Plant Growth Regul 57:49–56
Yang SM, Furukawa I (2006) Anatomical adaptations of three species of Chinese xerophytes (Zygophyllaceae). J For Res 17:247–251
Yang YL, Xu SJ, An LZ, Chen NL (2007) NADPH oxidase-dependent hydrogen peroxide production, induced by salinity stress, may be involved in the regulation of total calcium in roots of wheat. J Plant Physiol 164:1429–1435
Zhu Z, Wei G, Li J, Qian Q, Yu J (2004) Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.). Plant Sci 167:527–533
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (grant no. 30960064) and the National Science Foundation for Distinguished Young Scholars of China (grant no. 30625008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Y., Shi, R., Wei, X. et al. Effect of salinity on antioxidant enzymes in calli of the halophyte Nitraria tangutorum Bobr.. Plant Cell Tiss Organ Cult 102, 387–395 (2010). https://doi.org/10.1007/s11240-010-9745-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-010-9745-1