Abstract
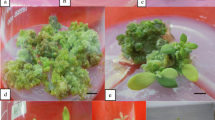
Using in vitro-grown needles of Sequoia sempervirens (D. Don.) Endl., direct shoot organogenesis was induced. The effects of three genotypes and two cytokinins, N6-benzyladenine (BA) and N-benzyl-9 (2-tetrahydropyranl) adenine (BPA), in combination with 2,4-D were investigated. Among tested cytokinins, BPA produced the highest frequency of shoot organogenesis from all three genotypes tested. Adventitious shoots were induced directly from explants without intervening callus within 5 weeks following incubation. Shoots were elongated on a 1/2 Wolter and Skoog (WS) medium supplemented with activated charcoal but without growth regulators. Later, elongated shoots were transferred to a 1/4 WS medium, but without activated charcoal and free of plant growth regulators to promote continued shoot growth. These shoots rooted spontaneously.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BA:
-
N6-benzyladenine
- BPA:
-
N-benzyl-9 (2-tetrahydropyranl) adenine
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- WS:
-
Wolter and Skoog (1966) medium
References
SM Attree LC Fowke (1993) ArticleTitleEmbryogeny of gymnosperms: advances in synthetic seed technology of conifers Plant Cell Tiss. Org. Cult. 35 1–35
SM Attree MK Pomeroy LC Fowke (1995) ArticleTitleDevelopment of white spruce [Picea glauca (Monech.) Voss] somatic embryos during culture with abscisic acid and osmoticum, and their tolerance to drying and frozen storage J. Exp. Bot. 46 433–439
EA Ball (1987) Tissue Culture Multiplication of SequoiaCell and Tissue Culture in Forestry; Case Histories: Gymnosperm Angiosperms and Palms Martinus Nijhoff Dordrecht 146–158
JM Bonga P Aderkas ParticleVon (1992) In Vitro Culture of Trees Kluwer Acad Publ Dordrecht
CH Bornman E Jansson (1980) ArticleTitleOrganogenesis in cultured Pinus sylvestris tissue Z. Pflanzenphysiol 96 1–6
Boulay (1987) In vitro propagation of tree species CE Green DA Somers WP Hackett DD Biesboer (Eds) Plant Tiss Cell Cult Alan R. Liss Inc. NY 367–382
F Bourgkard JM Favre (1988) ArticleTitleSomatic embryos from callus of Sequoia sempervirens Plant Cell Rep. 7 445–448
DJ Carrier JE Cunningham DC Taylor DI Dunstan (1997) ArticleTitleSucrose requirements and lipid utilization during germination of interior spruce (Picea glauca engelmannii complex) somatic embryos Plant Cell Rep. 16 550–554
Fouret Y, Larrieu C & Arnaud Y (1988) Rajeunissement in vitro chez la Sequoia sempervirens (endl.). (pp. 55–82). Ann Rech Sylv, AFOCEL
HJ Gladfelter GC Phillips (1987) ArticleTitleDe novo shoot organogenesis of Pinus eldarica Medw in vitro: I. Reproducible regeneration from long-term callus cultures Plant Cell Rep. 6 163–166
PK Gupta G Pullman R Timmins M Kreitinger WC Carlson J Grob E Welty (1993) ArticleTitleForestry in the 21st century; the biotechnology of somatic embryogenesis Bio/Tech 11 454–459
HR Owen AR Miller (1996) ArticleTitleHaploid plant regeneration from anther cultures of three North American cultivars of strawberry (Fragaria × ananassa Duch.) Plant Cell Rep. 15 905–909
YS Park SE Pond JM Bonga (1993) ArticleTitleInitiation of somatic embryogenesis in white spruce (Picea glauca): genetic control, cultural treatment effects, and implications for tree breeding Theor. Appl. Genet. 86 427–436
GS Pullman Y Zhang BH Phan (2003) ArticleTitleBrassinolide improves embryogenic tissue initiation in conifers and rice Plant Cell Rep. 22 96–104
InstitutionalAuthorNameSAS Institute (1985) SAS User’s Guide: Statistics EditionNumber5 SAS Institute Inc., Cary, NC
S Sato T Toya R Kawahara RF Whittier H Fukuda A Komamine (1995) ArticleTitleIsolation of a carrot gene expressed specifically during early-stage somatic embryogenesis Plant Mol. Biol. 28 39–46
EJ Staba (1969) ArticleTitlePlant tissue culture as a technique for the phytochemist Recent Adv. Phytochem. 2 75–102
C Stasolla EC Yeung (2003) ArticleTitleRecent advances in conifer somatic embryogenesis: improving somatic embryo quality Plant Cell Tiss. Org. Cult. 74 15–35
I-W Sul SS Korban (1994) ArticleTitleEffect of different cytokinins on axillary shoot proliferation and elongation of several genotypes of Sequoia sempervirens In Vitro Cell Dev. Biol. 30P 131–135
I-W Sul SS Korban (1998) ArticleTitleEffects of media, carbon sources and cytokinins on shoot organogenesis in the Christmas tree Scots pines (Pinus sylvestris L.) J Hort. Sci. Biotech. 73 822–827
B Sutton (2002) ArticleTitleCommercial delivery of genetic improvement to conifer plantations using somatic embryogenesis Ann. Forest Sci. 59 657–661
W Tang Z Guo (2001) ArticleTitleIn vitro propagation of loblolly pine via direct somatic organogenesis from mature cotyledons and hypocotyls Plant Grow Regul. 33 25–31
TA Thorpe (1988) Physiology of bud induction in conifers in vitro JW Hanover DE Kethley (Eds) Genetic Manipulation of Woody Plants Plenum Press New York 167–184
TA Thorpe IS Harry PP Kumar (1991) Application of micropropagation to forestry PC Debergeh RH Zimmerman (Eds) Micropropagation: Technology and Application Kluwer Acad Publ Dordrecht 331–336
S Arnold Particlevon T Eriksson (1979a) ArticleTitleBud induction on isolated needles of Norway spruce (Picea abies L., Karst.) grown in vitro Plant Sci. Lett. 15 363–372
S Arnold Particlevon T Eriksson (1979b) ArticleTitleInduction of adventitious buds on Norway spruce (Picea abies) grown in vitro Physiol. Plant 45 29–34
KE Wolter F Skoog (1966) ArticleTitleNutritional requirements of Fraxinus callus cultures Am. J. Bot. 53 263–269
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sul, IW., Korban, S.S. Direct shoot organogenesis from needles of three genotypes of Sequoia sempervirens. Plant Cell Tiss Organ Cult 80, 353–358 (2005). https://doi.org/10.1007/s11240-004-1365-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11240-004-1365-1