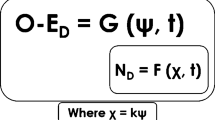Abstract
One big challenge faced by cognitive science is the development of a unified theory that integrates disparate scales of analysis of cognitive phenomena. In this paper, I offer a unified framework that provides a way to integrate neural and behavioral scales of analysis of cognitive phenomena—typically addressed by neuroscience and experimental psychology, respectively. The framework is based on the concept of resonance originated in ecological psychology and aims to be the foundation for a unified theory for radical embodiment; that is, a unified theory for that dissident part of cognitive science that shares a methodological commitment to dynamic systems theory and remains skeptical about the adequacy of mechanism and representationalism as the guiding ideas in the field. In the course of my presentation, I analyze different issues regarding the requirements and constraints unification poses to radical embodiment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Unified scientific theories are understood in two different although interrelated ways. On the one hand, a unified scientific theory is thought to be able to explain phenomena from different domains by showing that they are essentially the same phenomena or that they obey the same laws (Morrison 2000). An example of such kind of unified theory may be found in Kepler’s optics. Kepler elaborated his account of optics (Kepler 1604) while he was trying to justify the scientific reliability of optical instruments such as telescopes to study celestial bodies. In the process, Kepler equated the formation of retinal images with the production by light of any other kind of image on other surfaces (Gal and Chen-Morris 2013). By doing so, Kepler revolutionized the field of optics by turning it into the study of the formation of images by light and brought together phenomena from different domains—e.g., images projected in the lenses of telescopes and retinal images—under the same physical–mathematical formulation.
On the other hand, a unified scientific theory is taken to be able to reduce or, at least, to connect two other scientific theories, usually operating in different domains, under the same principles without taking them to be always addressing the same phenomenon. This is the case, for instance, of the classic examples of the deductive-nomological model of scientific explanation (Hempel and Oppenheim 1948; Hempel 1965) and its associated model of reduction (Nagel 1961; Gylmour 1969), such as Newton’s unification of Kepler’s celestial mechanics and Galileo’s terrestrial mechanics. Newton offered a common explanation along with a common mathematical formalization for the motions of celestial and terrestrial bodies that were previously explained by Kepler’s and Galileo’s theories respectively. Under this understanding of unification, a unified theory is so not just because it explains previously disparate phenomena, but because it theoretically and methodologically integrates previously independent theories. Of course, such an integration can lead to a unitary account of phenomena previously regarded to different domains—e.g., the motions of celestial and terrestrial bodies as essentially the same kind of motion—but such a move is not strictly required: phenomena can be explained in terms of the same principles and mathematical formulations and still be understood as essentially different or as belonging to different domains, levels of description, or scales of analysis.Footnote 1
These two senses of ‘unified scientific theory’ are relevant to cognitive science. A unified theory of cognitive science should be able to explain disparate phenomena, such as perception, motor control, decision-making, reasoning, emotions, etc., in terms of the same basic principles or by describing the same basic mechanism. For example, such is the case for the unified theory of cognition based on an integrated cognitive architecture proposed by Allen Newell (1990). At the same time, a unified theory of cognitive science should integrate the theories that operate at the different scales of neuroscience, psychology, philosophy, etc., and should coordinate them under a unitary theoretical and methodological approach. I take this second sense of unity to be more fundamental than the first one in the case of cognitive science. The use of integrated resources from various disciplines makes the explanation for the disparate phenomena we label as ‘cognitive’ easier. This is the reason why, in the following, I will focus on this second sense of unification.
In this paper, I present a unified framework that aims to integrate two typical scales of analysis of cognitive phenomena: the behavioral scale and the neural scale. These two scales of analysis are not two distinct ontological levels, but two different scopes from which the cognitive activities of the same organism can be studied. The scales refer to the spatiotemporal dimensions of the organism and, usually, experimental psychology aims to explain the behavioral scale and neuroscience aims to explain the neural scale.Footnote 2 The framework I propose is based on the concept of resonance originated in ecological psychology and aims to integrate non-computational, non-representational models and theories of perception, action, and cognition both from experimental psychology and neuroscience. In this sense, the proposed framework aims to provide the foundation of a unified theory for radical embodiment; that is, for the part of the cognitive sciences that remains skeptical of mechanism and representationalism as the main guiding forces for the sciences of the mind. In the course of the presentation of the framework, I address three issues regarding unification in radical embodiment: the kind of framework that can be considered as a unified one, the possibility of unification while maintaining some degree of independence between the two scales of analysis integrated in the unified framework, and the relations between the framework and mechanistic explanations.
In Sect. 2, I explain what it is meant by ‘radical embodiment’ and I define the scope, aims, and constraints of the unified framework proposed here. Then, in Sect. 3, I present the abstract formulation of the framework and analyze its relationship with existing dynamics-based hypotheses and theories in radical embodiment and some of their central concepts. In Sect. 4, I develop a more concrete instantiation of the unified framework that integrates behavioral dynamics (Warren 2006), neural reuse (Anderson 2014), and coordination dynamics (Kelso 1995; Kelso and Tognoli 2007). The concrete instantiation of the framework provides a more detailed characterization of the way it can integrate the neural and behavioral scales of analysis of cognitive phenomena. Finally, in Sect. 5, I address two issues open for future research: the study of the relation of constitution between the two scales of analysis integrated into the unified framework and the possibility of understanding it in terms of mechanistic explanation.
2 Radical embodiment
It is important to recall that there are different paradigms, theories, and opinions about how to best understand cognition. This is a truism both regarding different moments of the history of the sciences of the mind and regarding contemporary cognitive science. However, it does not speak against the possibility of having unified theories in cognitive science. We might not be ready to have one unified theory of cognition, but the task can be to “get some candidate theories that have a large enough scope in order to get the gains inherent in such unification” (Newell 1990, p. 17). In this sense, it is worth pursuing unified theories from different standpoints in our path to a future unified theory of cognition.
Most contemporary cognitive science is predominated by a cognitivist paradigm that favors the understanding and explanation of cognition in terms of mechanisms and representations (Bechtel 2009; Garnham 2009). According to cognitivism, a unified theory of cognition consists of a set of computational mechanisms that are in the business of building up and manipulating internal models of their environments to solve cognitive tasks. Through computational mechanisms, cognitive systems build up and manipulate internal representations of their surroundings to perceptually guide their locomotion (Kawato 1999) as well as build up and manipulate internal representations of the plausible future states affairs to make decisions, for example Engin and Vetschera (2017). A unified theory of this kind has been one of the targets of contemporary cognitive science in the last decades and has been pursued in several ways.Footnote 3 However, not all contemporary cognitive scientists agree with the cognitivist paradigm despite its dominance in the field. At least part of this group of dissident cognitive scientists work under a paradigm I will refer as radical embodiment.
Radical embodiment can be described in two different ways. On the one hand, it can be described in terms of its opposition to the dominant cognitivist paradigm. That is, radical embodiment is characterized by the rejection of the use of computational mechanisms and representations to pursue explanations in cognitive science (e.g., Gibson 1979; Varela et al. 1991; Hutto and Myin 2013).Footnote 4 On the other hand, radical embodiment can be described in terms of its positive account of explanation in cognitive science: as cognitive systems are dynamical, complex systems that engage in a set of specific interactions with their environments, perception, action, and cognition are best understood in terms of dynamical explanations (e.g., Chemero 2009; Di Paolo et al. 2017; van Gelder 1998; Walmsley 2008); where ‘dynamical explanations’ are covering-law-like explanations of the dynamics of those cognitive systems—i.e., the change of a system across time—in terms of differential equations that make no reference to notions such as representation or computation.Footnote 5 Such a commitment restricts radical embodiment to a subset of ecological psychologists and enactivists—those Chemero (2009) labels as radical embodied cognitive scientists. The application of this kind of explanation in cognitive science has increased over the few last decades and several dynamic models have been used to explain different phenomena like learning (Newell et al. 2008), locomotion (Fajen and Warren 2003), interpersonal coordination (Schmidt and Richardson 2008), speech production (Port 2003), brain–body coordination (Jirsa et al. 1998; Pillai and Jirsa 2017), behavioral variability (Nalepka et al. 2017), human–computer interactions (Nalepka et al. 2019; Lobo et al. 2019), and brain activity (Tognoli and Kelso 2014) among others.
Despite the growing success of radical embodiment, it cannot be said that there is a unified theory or a unified framework that gives coherence to the field as a whole. What unifies the different applications of dynamical explanations to cognitive phenomena is the use of the same mathematical methods based on dynamic systems theory—what Turvey (1992) named strategic reductionism—but not an underlying theory or framework for all of them. Dynamical explanations of brain states and dynamical explanations of behavior, for example, share epistemological and methodological commitments regarding the correct explanatory approach to cognition, but they do not share a theory that makes them aspects of a unitary explanation of the relevant phenomena. To be so, dynamical explanations at neural and behavioral scales of analysis should be part of a theory that connects them under the same principles and that shows how the activity at the neural scale enables and constrains the activity at the behavioral scale and vice versa. This would unite the dynamical approaches to neuroscience and to experimental psychology and, thus, would count as a unified theory for radical embodiment. In the rest of this paper, I offer a unified framework that may serve as the foundation for such a unified theory.
3 Unifying radical embodiment: a candidate framework
In order to be successful, a unified framework for radical embodiment needs to establish some constraints regarding the scales of analysis of cognitive phenomena. Dynamical explanations have been developed at several of those scales (e.g., behavioral scale, muscular–skeletal scale, neural scale) and a completely unified theory for radical embodiment should encompass all of them. For the sake of simplicity, however, the framework I present here focuses on the scales of behavior and the neural system. Connecting these two fundamental scales should be enough to have a simple, unified framework based on dynamical explanations and able to bring experimental psychology and neuroscience together. Moreover, as we shall see, the very nature of the proposed framework allows for its expansion to integrate all the other scales of analysis.
3.1 A resonance-based framework: what is resonance?
Resonance is a widely observed phenomenon in nature and has been described in several fields (e.g., acoustic resonance, mechanical resonance, orbital resonance, optical resonance, or electrical resonance). Put simply, resonance occurs when a vibrating system forces another system to vibrate at a greater amplitude at some specific frequencies—especially at those related to the latter’s natural frequency. A canonical example of resonance is the body of a violin or a guitar resonating to the vibration of one of its strings and amplifying its sound. By means of this example, it can be seen that resonance is closely related to two main concepts. On the one hand, it is related to amplification. In music, resonance is used to amplify the sound of instruments and to give specific features to that sound. For instance, a guitar sounds like a guitar due to the sound of its vibrating strings but also due to the resonance introduced by its body. On the other hand, resonance is related to coupling. In resonant phenomena, both the driving system (the string, in the example) and the driven system (the body of the instrument, in the example) are coupled at some specific frequency. They vibrate at the same frequency or, at least, at two lawfully related frequencies. This second aspect of resonance is what inspired J. J. Gibson to use the concept in ecological psychology.Footnote 6
In the very first pages of The Senses Considered as Perceptual Systems, Gibson (1966) proposes for the first time the concept of resonance as a metaphor for the role of the brain in perceptual events:
Instead of supposing that the brain constructs or computes the objective information from a kaleidoscopic inflow of sensations, we may suppose the orienting of the organs of perception is governed by the brain so that the whole system of input and output resonates to the external information. (1966, p. 5).
Resonance is presented as an alternative to information-processing as the foundation for perception. Gibson uses the concept a second time in The Ecological Approach to Visual Perception (1979) within a discussion of the perception of persistence and change:
In the case of a persisting thing, I suggest, the perceptual system simply extract the [informational] invariants from the flowing array; it resonates to the invariant structure or is attuned to it. (Gibson 1979, p. 249).
In this second instance, Gibson suggests that resonance may be understood in terms of coupling (or attunement) between perceptual systems and perceptual information available in the energy flows of the environment (e.g., light, chemicals, vibrating air, and so on): perceptual systems—constituted by the sensory organs, the nervous system, the motor system, and their joint activity—are attuned to the invariant structure of environmental energy flows (perceptual information) just as, for example, a radio is attuned to a radio station by extracting the signal of that particular radio station from the whole flowing array of frequencies. In principle, no substantive notion of information-processing that requires different kinds of internal transformations of perceptual information (e.g., combining with other sources of information, composition or de-composition, etc.) is involved in the perceptual activity as just the ability to resonate to perceptual information is needed for perception to occur.Footnote 7
3.2 A resonance-based framework: from a metaphor to the framework
Gibson’s notion of resonance has remained metaphorical during the development of ecological psychology. However, based on the Gibsonian tradition and the work of others that have explored the resonant properties of neural systems (e.g., Large 2008; Roach et al. 2018), I am going to propose a non-metaphorical, operational concept of resonance as the central notion of a unified framework for radical embodiment.Footnote 8 It is my contention that we must understand ecological resonance as the process that couples the dynamics of the behavior of cognitive systems and the dynamics of their nervous systems in terms of whatever information is relevant to the on-going perceptual task.Footnote 9 There are two aspects of this claim that need to be unpacked.
First, if an organism is a cognitive system, its dynamics at the behavioral scale of analysis are the dynamics of the organism–environment interaction (O–ED) constrained by the information available at that scale (ψ), that is, the information available to the organism due to its interactions with the environment (for further characterization of this information, see Bingham 1988; Lee 2009; Turvey et al. 1981). On the other hand, granting that the organism has a neural system, the dynamics at the neural scale of analysis are the dynamics of the organism’s neural system (ND) and have their own constraints (χ). The phenomenon of resonance occurs when both dynamics, O–ED and ND, are informationally coupled. That is, when both dynamics are jointly constrained by the same information, χ = kψ, where k is a coupling coefficient.
Second, the relevant notion of informational coupling in ecological resonance follows Warren’s account (2006).Footnote 10 Unlike Beer (1995) or Kelso (1995), for example, who claim that two systems are coupled when some parameters of one system are functions of the state variables of the other system, Warren proposes a notion of coupling that is not solely parametric but can also be based on the coupling of state variables themselves through informational constraints, for instance (see Warren 2006, p. 369). Moreover, the idea that relevant information generated at the scale of the dynamics of the organism–environment interactions constrains the dynamics of the neural system is not just a speculative proposal. On the contrary, there are empirical results that describe events in which that kind of information or a similar one is actually constraining neural dynamics of different systems—e.g., Aguilera et al. (2013), Kelso and Tognoli (2007), Large (2008), and van der Weel and van der Meer (2009).
This simple, abstract framework based on resonance is a starting point for the development of a unified theory for radical embodiment because of two features. First, the framework describes the approaches usually regarded by neuroscience and experimental psychology as two complementary approaches to the same cognitive system. The neural scale of analysis is not opposed to or separated from the behavioral scale of analysis in any strong sense. On the contrary, the neural scale is nested within the behavioral scale. Thus, if two explanations of the same cognitive phenomenon are respectively developed at each of the scales, the two explanations are not in conflict. Even more, it is not clear that there is any need for the reduction of one to the other. They can just be taken as two explanations of the same cognitive phenomenon with regard to the same cognitive system. And both explanations are equally relevant because they reveal features of the cognitive phenomenon in different spatiotemporal dimensions.Footnote 11 In this sense, the cognitive phenomena that neuroscience and experimental psychology try to explain are not disparate, but the same cognitive phenomena approached from different scales. The question is not whether one scale can be reduced to the other, but how can we understand the relationship between the scales in a coherent manner. The resonance-based framework offers a clear way to tackle this question.
And second, the dynamics at the behavioral and neural scales of analysis are connected by task-relevant information in a fully operative fashion. The information available in the environment of cognitive systems has been thoroughly studied in radical embodiment, especially in ecological psychology (e.g., Lee 2009; Segundo-Ortín et al. 2019), and resonance is a well-known physical process. Given that, the proposed framework fits well with one important tenet of radical embodiment: that neural systems are best understood in terms of the way their dynamics are constrained by the information generated in organism–environment interactions. In this sense, a resonance-based framework provides a way to generate and test hypotheses regarding the connection of the two relevant scales of analysis and, thus, is a candidate unified framework for radical embodiment.
3.3 Resonance, dynamics, and informational coupling
As the resonance-based framework shares the usual dynamics-based methodology of radical embodiment, it is inspired and influenced by proposals such as the dynamical hypothesis (van Gelder 1998), the dynamical approach to autonomous agents (Beer 1995, 2003), or the behavioral dynamics model (aka information-based control; Warren 2006), among others. However, these proposals do not specifically address the fundamental issue targeted by the resonance-based framework: the unification of perception–action (experimental psychology) and neural activity (neuroscience) as two different but integrated scales of cognitive events (for the relevance of the issue, see Krakauer et al. 2017; Pillai and Jirsa 2017).
The dynamical hypothesis elaborated by van Gelder (1998) consists of taking cognitive systems to be dynamical systems (p. 615) and of promoting a single-scale explanatory strategy based on the use of dynamical models to understand the cognitive performance of the system at its highest relevant level of causal organization (p. 622). In this sense, as the resonance-based framework also describes cognitive systems as dynamical systems, the influence of van Gelder’s hypothesis is undeniable. However, while van Gelder’s explanatory strategy is focused on just one scale (the one with the highest level of causal organization),Footnote 12 the resonance-based framework acknowledges the possibility of accounting for cognitive events at different scales. This strategy acknowledges current practices in cognitive science—experimental psychology and neuroscience are targeting cognitive phenomena at different scales—and permits the development of a unified account of those scales through the integration of theories operating at them.
The multi-scale character inherent to the resonance-based framework is also present in the behavioral dynamics approach (Warren 2006) and the dynamical approach to autonomous agents (Beer 1995, 2003). Both of them acknowledge the necessity of understanding cognitive phenomena at different scales. In the case of Warren’s proposal, the dynamics of behavioral activities can be described at the scale of the organism (in the form of control laws and information; see Warren 2006, p. 368) and at the scale of the organism–environment system (in the form of behavioral dynamics; see Warren 2006, p. 366). In the case of Beer’s dynamical approach, behavior can be characterized at the scale of the organism–environment system (Beer 2003, pp. 223–225), at the scale of the interactions of organism and environment taken as two different systems (Beer 2003, pp. 226–230), and at the scale of organism’s neural dynamics (Beer 2003, pp. 231–235). The influence of Warren’s and Beer’s proposals in radical embodiment and, in particular, in the resonance-based framework can hardly be overemphasized. However, both differ from the resonance-based framework in terms of unification: Warren’s approach, rooted in the ecological tradition, makes no reference to the study of behavior at the neural scale; and Beer’s approach includes caveats against interpreting it as modeling real-life behaviors or nervous systems (Beer 1995, p. 190) and caveats that promote its usage in exploratory theoretical refinement (Beer 2003, p. 210). In this sense, the resonance-based framework, although influenced and inspired by these proposals, is a novel approach insofar as it promotes a unified understanding of the behavioral and neural scales of cognitive phenomena and the integration of the neuroscientific and psychological theories that separately account for them in radical embodiment.
A further issue regarding the place of the resonance-based framework within radical embodiment has to do with the differences between its fundamental concept (resonance) and the notion of coupling, commonly used to refer to the relationship of two interacting and co-constrained dynamical systems. There is a basic similarity between resonance and coupling: resonance is a special kind of coupling. When a body of a guitar resonates to one of its strings, for example, body and string are coupled at some frequency. In this sense, at the abstract level, resonance and coupling are equivalent concepts and resonance can be studied in terms of coupled variables. However, as a special kind of coupling, resonance has some particularities that cannot be attributed to other forms of coupling and that are relevant to evaluate cognitive systems at different scales.
First, models of networks composed by nonlinear neural oscillators—i.e., models of the neural scale—exhibit a feature known as nonlinear resonance that accounts for some network properties as stimuli filtering, neural entrainment, or stimuli anticipation (Large 2008; Sanches de Oliveira et al. 2019). These features are not common to all coupled oscillators, but only to those that have specific resonant properties (e.g., coupling to frequency harmonics). And second, while coupling remains an abstract property of the interaction of dynamical systems, resonance may be found in the physiology of neurons, neural networks, and brains in different forms. In this sense, resonance goes beyond the abstraction of coupling and opens the possibility to connect neural dynamics and physiology (more on this in the next section). These particular properties of resonance are the ones that make it, instead of mere coupling, an interesting guiding concept for a model that aims to unify theories at different scales of cognitive systems.
Another fundamental aspect of the relationship between resonance and coupling is that the proposed framework based on ecological resonance characterizes the former in terms of the informational coupling of the dynamics of different scales of nested systems. First, such nesting follows from the observation that the dynamics at the neural scale are just a subpart of the dynamics of the organism–environment interaction. In this sense, ecological resonance is not framed in terms of the classic picture in which internal states are opposed to external states. On the contrary, neural states are nested within and constrained by organism–environment states. And second, the nature of the constraint that the dynamics of the organism–environment interaction exert on the neural dynamics is of informational coupling, meaning that the dynamics at both scales enter into a relationship of common change and variance in terms of the relevant information. Thus, perceivers are sensitive to relevant perceptual information insofar as their dynamics, concretely the dynamics of the neural system, are affected by that information in terms of coupling.Footnote 13
Informational coupling is at the core of ecological resonance, but its general description might still be seen as too vague. On the one hand, the fact that the dynamics of the nervous system are affected by the dynamics of the organism–environment interactions simpliciter is trivial. Any contemporary account of the dynamics of the nervous system acknowledges that they change depending on the events at the scale of the dynamics of the organism–environment system. On the other hand, the concept of coupling is very broad in itself and, therefore, an abstract description of what it takes for coupling to be informational may not be enough to grasp the notion. For this reason, a concrete example of informational coupling may shed some light in the discussion.
The proposed framework based on ecological resonance remains theoretical for the time being and, therefore, it is not possible to find empirical examples that fully implement it. However, it is possible to find studies that speak to the plausibility of the framework and to a concrete understanding of informational coupling. Imagine, for example, a situation in which a perceiver must avoid an approaching object. The relevant perceptual information in this kind of situation—at least for radical embodiment—is a well-known perceptual variable, tau (τ) (Lee 2009), that allows perceivers to know the time it will take for an approaching object to hit their visual system. In this sense, if the perceiver is sensitive to tau, she is sensitive to the relevant perceptual information to accomplish object avoidance. In such a situation, the way tau constrains perceiver’s neural system dynamics can be labeled as an event of ecological resonance. A situation similar to the one in this example was explored by van der Weel and van der Meer (2009).
In their study, 10 babies were looking to a screen in which looming figures (aka “approaching objects”) were projected. The value of tau (τ) was manipulated for different looming rates and figures. Then, van der Weel and van der Meer analyzed the theta-rhythm oscillatory activity of babies’ visual cortex—related to attentional processes (Orekhova et al. 1999)—during these looming situations. They found that the theta-rhythm oscillatory activity was tau-coupled: the tau-value of the change in neural rhythm’s temporal structure was linearly correlated with the tau-value generated by the perceiver–environment interaction in the experimental setting. Concretely, van der Weel and van der Meer ran a recursive linear regression to determine the strength of the tau-coupling of the dynamics at the neural scale (i.e., the dynamics of the nervous system or ND) and the dynamics of the looming figures at the organism–environment scale (i.e., the dynamics of the organism–environment interaction or O–ED) given the coupling equation τND = KτO − ED, where K is the constant of coupling. They found that the percentage of tau-coupling was 79.4% with over a 95% correlation between both scales (van der Weel and van der Meer 2009, p. 1389).Footnote 14
The study developed by van der Weel and van der Meer (2009) shows what I take to be a clear instance of informational coupling in the sense of ecological resonance. Relevant perceptual information (i.e., tau) is available in the environment of perceivers due to their dynamical interaction with it (i.e., due to the organism–environment interaction). Then, the dynamics of the nervous system are informationally constrained by tau (τ) and, consequently, coupled to the dynamics of the organism–environment interaction. The informational coupling itself is defined in terms of a coupling equation that includes the informational variable (τ) and a coupling parameter (K). In this sense, babies are sensitive to the relevant perceptual information by a process of informational coupling that can be explained in terms of the informational constraints between the scales of the nested system. Crucially for radical embodiment, this explanation avoids any appeal to robust notions of encoding or internal model.Footnote 15
3.4 Resonance and physiology
So far, ecological resonance has been defined as the informational coupling of the dynamics of the organism–environment interactions and the dynamics of the nervous system. Also, the way ecological resonance may be an interesting resource for a framework that intends to account for the relationship between brain and behavior in radical embodiment has been pointed out. However, there are two fundamental and interrelated questions regarding the biophysical and physiological characteristics of ecological resonance that must be addressed before the theoretical framework based on it can be considered minimally completed. First, the general phenomenon of resonance seems to imply the coupling of two oscillatory systems, but not all variables of relevant information are oscillatory in character. For example, the relevant perceptual information in van der Weel and van der Meer’s study (2009), that depends on continuous looming, is not oscillatory. Also, in general, the perception of visual scenes and events is not oscillatory either. Thus, the question is: how do nervous systems resonate to non-oscillatory scenes and events? In other words, how do nervous systems resonate to non-oscillatory perceptual information? And second, ecological resonance operates at the scale of neural and behavioral dynamics, but this fact does not entail a process of resonance at the physiological scale. It is possible for neural systems to be computational at the physiological scale while acting as resonators at the scale of neural dynamics, for example. In this sense, radical embodiment might be still resting on computational principles without acknowledging it. Therefore, the question is: why is resonance the principle that accounts for informational coupling and not computation or feature detection, for example? In other words, can we talk about resonance in the physiological sense?
The most plausible answer to the question about how neural systems resonate to non-oscillatory information is to consider that biology capitalizes on the oscillatory features needed for the process of resonance. Thinking about flutes helps to unpack this claim. An interesting aspect of flutes is that they get non-oscillatory, continuous air flows when flutists blow through their embouchure holes even though sound actually requires oscillatory motions or air flows. How can flutes sound, then? Indeed, when the continuous air flows get into the flute, the joint activity of these flows and the components of the body of the flute (breathe hole, flue, ramp, and so on) produce new oscillatory flows and some of them are radiated as sounds. In other words, flutes produce oscillatory patterns of flows out of non-oscillatory ones due to the physical properties of their material constitution.
Similarly, it is possible for the biophysical properties of nervous systems to capitalize the transformation of non-oscillatory perceptual information into oscillatory perceptual information they can resonate with. This possibility is compatible with the intrinsically oscillatory nature of sensory receptors that, at the end of the day, are in charge of registering the environmental energy flows from where perceptual information can be extracted. In this sense, sensory receptors may play in neural systems the same role material components (e.g., flue, breathe hole, or ramp) play in flutes.
Such a way of answering the question of the detection of non-oscillatory perceptual information speaks to the question of resonance at the physiological scale as well. If the biophysical properties of nervous systems transform any energy input into an oscillatory one, it is possible that the physiological nature of single neurons and neural networks is also resonant. Indeed, processes of physiological resonance that could account for informational coupling have already been described. Lau and Zochowski (2011), for example, have shown that the ubiquitous variations of subthreshold oscillations in the brain lead single-neurons to have flexible resonance profiles (aka resonance frequency shifts) that, consequently, lead to complex patterns of coupling between neural networks and their input flows.Footnote 16 In this sense, and taking that relevant perceptual information is present in these input flows, processes of resonance frequency shift might be good candidates to be the physiological principle for the informational coupling entailed by ecological resonance. Moreover, other processes of resonance described in the literature of neurophysiology and neural networks—single-neuron resonance (Hutcheon and Yarom 2000; Kasevich and LaBerge 2011), coherence resonance (Yu et al. 2018), network resonance (Helfrich et al. 2019), or stochastic resonance (Ikemoto et al. 2018)— along with the nonlinear resonance of neural oscillators that allows them to filter, to be entrained with, and to anticipate the arriving stimulation (Large 2008; Sanches de Oliveira et al. 2019), constitute further resources to connect ecological resonance to the biophysical and physiological underpinnings of nervous systems without postulating encoding or computational principles.
4 Developing the unified framework: resonance in context
The claim that a resonance-based framework may be a good candidate unified framework for radical embodiment will be best understood by showing the way it can be applied to the integration of disparate dynamical explanations both at the behavioral and neural scales of analysis. To do so, I evaluate the way resonance is able to unify one very successful account of behavior that operates at the behavioral scale, behavioral dynamics (Warren 2006), with a theory for the functional organization of the brain, neural reuse (Anderson 2014), and a way to understand its neural dynamics, coordination dynamics (Kelso and Tognoli 2007).
4.1 The behavioral scale: behavioral dynamics
As we have seen, radical embodiment promotes the use of the tools provided by dynamic systems theory to explain cognitive phenomena. These tools consist of differential equations that capture the change of some variables over time in terms of some control parameters. Those variables refer to features of systems that change over time and capture their quantitative and qualitative evolution (e.g., the relative change of the population predators and prey in a given environment). Control parameters refer to other features of those systems that determine the change of the variables. These are the main components of typical dynamical explanations.
One example of a successful dynamical explanation is behavioral dynamics (Warren 2006). Also known as information-based control, behavioral dynamics is a model for the prospective control of behavior based on the information generated at the scale of organism–environment interactions. In behavioral dynamics, organism and environment are taken to be two dynamical systems that constrain each other. On the one hand, the organism mechanically constrains the environment by the forces exerted on it (e.g., movements). On the other hand, the environment informationally constrains the organism through the structures of her sensory fields (e.g., the structure of light in the visual field or the structure of sound in the auditory field). Through these constraints, the two dynamical systems are coupled and that coupling can be studied, according to Warren, from a broader scale: the dynamics of the organism–environment system as a unit or behavioral dynamics.
In general terms, behavioral dynamics model the environment as a dynamical system e. Its changes, ė, are a function of e’s previous states and the forces produced by the organism, F. The organism is modeled as another dynamical system o. Its changes, ȯ, are a function of o’s previous states and the information, i, provided by the environment. Thus, the coupling of these two dynamical systems is facilitated by F and i. Given this, there is a broader scale that captures the dynamics of the organism–environment interactions as a unitary event. This scale is named behavioral dynamics and captures behavior (x) and behavioral changes (ẋ and ẍ) of an organism during its ongoing activity in its environment. Behavioral dynamics emerge from the dynamics at the lower scale and, at the same time, capture that lower scale in an abstract fashion (see Fig. 1).
Schema of behavioral dynamics. The way the environment changes, ė, depends on the previous states of the environment, e, and the forces exerted on it by the agent, F. The way the organism changes, ȯ, depends on the previous states of the organism, o, and the informational constraints posed by the environment, i. From these interactions, a regularity, x, emerges at the scale of the organism–environment interactions, and its change ẋ and/or ẍ are described as behavioral dynamics
An example of a concrete model of behavioral dynamics is Fajen and Warren’s (2003) steering model for navigation in sparsely crowded environments. This model predicts the trajectories followed by agents given the constraints posited by the elements of their environment (obstacles and goal). In Fajen and Warren’s model (2003), the goal is understood as an attractor for the steering of an agent (see Fig. 2). Given an arbitrary reference axis, the steering of an agent, ϕ, and the position of the goal, ψg, give rise to the angle βg. To guide her own steering towards the goal, the agent must close that angle βg. Such closing is defined in terms of the dynamical equation of a damped mass-spring that defines the two control parameters b (damping) and k (stiffness). Otherwise, obstacles are defined in the same terms, but the agent must open the angle βo to avoid hitting them. In this sense, the steering of an agent (ϕ) is attracted by the goal and repelled by the obstacles. In both cases, the changes in the steering of the agent are modeled in terms of “stretching” or “compressing” the mass-spring that connects (in the model) her current steering and the position of the goal or the obstacles.

(from Warren 2006, p. 374, figure 7)
Graphical depiction of the relation between an agent and a goal in Fajen and Warren’s steering model (2003). Given an arbitrary reference axis, the steering of an agent, ϕ, and the position of the goal, ψg, form an angle βg. The goal acts as an attractor for the steering of the agent and such attraction is modelled using the equation of a damped mass-spring and its two typical parameters for damping (b) and stiffness (k)
When the dynamics of navigation in a sparsely crowded environment are defined in the above way, they can be captured with the following differential equation:
where \(\ddot{\phi }\) captures the acceleration in the change of the steering of the agent, − bg (− \(\dot{\phi }\)) is the damping term and − kg βg and + ko βo are the attraction and repulsion terms, respectively. The remaining terms modulate the decay of attraction towards the goal or repulsion from the obstacles as a function of distance—to the goal (dg) or to the obstacles (do)—and given the control parameters CN. Concretely, C1 determines the decay rate of attraction towards the goal, C2 determines the minimum value of attraction towards goals, and C3 and C4 determine the decay rate of repulsion from the obstacles (including the influence of stiffness).
The damping term, − bg (– \(\dot{\phi }\)), is included in the model to prevent oscillations in steering. When navigating an environment, agents do not oscillate around the new heading direction after a change in steering, but just smoothly get to the new direction. This is the feature of steering behavior captured by the damping term. Otherwise, the attraction and repulsion terms, − kg βg and + ko βo, are modulated by the spring stiffness parameter k to capture the strength of angular acceleration: the strength of attraction towards the goal or the strength of repulsion from the obstacles. The stiffness of a spring is its resistance to deformation—i.e., its rigidity, its resistance to being compressed or stretched. Thus, the higher the value of stiffness (k), the more the attraction or the repulsion, respectively. Put simply, damping and stiffness modulate the force needed to stretch or compress the mass-spring that connects (in the model) the current steering of the agent and the position of the goal or the obstacles.
Once navigation in a sparsely crowded environment is described in terms of Fajen and Warren’s model and is formalized in Eq. (1), the regularities (trajectories) at the organism–environment scale can be predicted. In other words, the regularities at the behavioral scales of analysis can be captured. The robustness of the model has been supported by several studies on perception and action (Bruggeman and Warren 2005; Fajen and Warren 2003, 2005; Warren and Fajen 2004; Lobo et al. 2019).
The last noteworthy aspect of Fajen and Warren’s model is the role of relevant perceptual information in navigation. Warren’s proposal (2006) is that behavioral dynamics both capture and emerge from the organism–environment interactions when constrained by the relevant perceptual information. In the case of organisms, behavioral changes are described as ȯ = ψ (o, i), where ȯ is the change of the state of organism while dynamically coupled to their environment. This change is defined as a function ψ of organisms’ previous states, o, and of relevant perceptual information, i (see Fig. 1). In Fajen and Warren’s model, the relevant perceptual information is provided by the angles βg and βo, that are defined as the difference between the current steering of the agent and the positions of the goal (βg = ψg − ϕ) and the obstacles (βo = ψo − ϕ), respectively. It is possible to define these angles in terms of variables “specified by optic flow and the proprioceptive locomotor axis [egocentric direction], whereas the direction of a goal [or an obstacle] … is given by its visual direction” (Warren and Fajen 2004, p. 332; see also Warren et al. 2001; Wilkie and Wann 2003). In the case of βg,Footnote 17 the relevant perceptual information can be formulated as:
where βg is the difference between the steering of the agent and the location of the goal, ϕloco − ψg determines the egocentric direction with respect to the goal, and ϕflow − ψg determines the visual angle between the goal and the steering of the agent in the optic flow. The structure of this flow is co-determined by the layout of the environment, w, and the velocity of the agent, v. In this sense, task-relevant information is defined, precisely, at the behavioral scale of analysis; namely, in terms of the organism–environment interactions.
Behavioral dynamics provides a dynamical explanation of behavior—of the control of behavior for navigating an environment in the case of Fajen and Warren’s steering model (2003)—and, thus, it is an example of an explanatory framework for radical embodiment at the scale usually addressed by experimental psychology. However, as might be inferred from the previous discussion, the viability of behavioral dynamics is based on one aspect of the relationship of organisms and information: organisms must be sensitive to the relevant information and must use it in terms of their own intrinsic dynamics (see Fig. 1). Behavioral dynamics, though, does not explain how organisms carry out such a process.
4.2 The neural scale: neural reuse and coordination dynamics
Although behavioral dynamics is an instance of an explanatory framework based on dynamical explanations in radical embodiment, it remains silent about the way organisms are able to integrate relevant perceptual information in their own intrinsic dynamics to make their contribution to the overall dynamics of the organism–environment interaction. Such an integration is probably carried out by the nervous system to an important extent—at least in those organisms with a nervous system—and, thus, a unified theory for radical embodiment needs to connect the dynamics at the behavioral scale of analysis with those at the neural scale of analysis to provide a substantive unification. To do so, first, radical embodiment needs a description of the neural scale compatible with its main tenets.
As already noted, radical embodiment rejects computation and representations as explanatory tools. Given these constraints, the idea of nervous systems composed of more or less computational mechanisms that realize cognitive phenomena is a non-starter. A framework or a theory that explains cognitive phenomena in terms of a computational mechanism at the scale of the nervous system—or any other scale, actually—might be unifying, but it would not be a unified theory for radical embodiment.Footnote 18 For this reason, the neural system must be also addressed in terms of dynamical explanations. That is, the explanation at the neural scale of analysis (i) must take the neural system to be best understood in terms of its dynamics and (ii) must offer methods to explain and predict those dynamics.
In order to understand the neural system in terms of its dynamics, the theory of neural reuse offers a promising framework (Anderson 2014; Raja and Anderson 2019). Neural reuse’s main tenet is that neural regions, although having their own propensities to participate in specific tasks (e.g., visual perception, decision making, or motor control), are used and re-used in many different cognitive phenomena. Thus, neural reuse predicts that, for each cognitive phenomenon, we will find groups of dynamically coordinated neural regions participating in them (Anderson et al. 2013). These groups of neural regions are named TALoNS (Transiently Assembled Local Neural Subsystems) and consist of temporary coalitions of different neural networks which support different cognitive phenomena. In this sense, the dynamics of the neural system can be understood as the change from TALoNS to TALoNS as cognitive tasks change as well.
Neural reuse and its related concept of TALoNS provide a way to understand neural systems in terms of their dynamics. In this context, at the neural scale of analysis of cognitive systems, the computational capacities of each neural region are not the relevant explananda—as is proposed by (the dominant) computational neuroscience. The relevant explananda are the dynamics of the different coalitions of neural regions (TALoNS) that support different cognitive phenomena. However, in order to have a complete explanation of the neural scale of cognitive systems, these very dynamics must be modeled and coordination dynamics (see Kelso 1995; Kelso and Tognoli 2007) provides the conceptual and empirical tools to do so.
In coordination dynamics, the neural system is taken to be composed by non-linearly coupled non-linear oscillators (Tognoli and Kelso 2009, p. 33) in which coupling is captured by a specific form of the HKB model.Footnote 19 Given this, the temporal dynamics of the synchronized states of different neural regions are modeled in terms of their relative phase of oscillation. Based on these patterns of synchronization between neural regions, proponents of coordination dynamics identify three different states (or schemes) of coordination: uncoupled, phase-coupled, and metastable (Tognoli and Kelso 2014, p. 36; see Fig. 3). Uncoupled brain regions are usually so because they do not interact with each other. Uncoupling is a feature found between regions but not within them—i.e., intrinsic dynamics of brain regions always hold some form of coupling as they hold local interactions. Otherwise, phase-coupled brain regions are usually so because, despite having their own intrinsic dynamics, their level of interaction is strong enough as to overcome their own “personalities” and to make them engage in a coordinated activity. These two schemes of coordination have been thoroughly studied in recent years (e.g., Bressler and Kelso 2001; Bressler and Tognoli 2006; Fries 2005; Singer 2005; Uhlhaas et al. 2009; Wang 2010), but in terms of biological or cognitive systems, they are idealizations. It is very rare if not impossible to find stable patterns of coordination lasting through time. On the contrary, what it is normally found in biological and cognitive systems—like the brain—are alternated moments of different patterns of stability and instability. This phenomenon is captured by the third schema of coordination mentioned above, the metastable one.

(from Tognoli and Kelso 2014, p. 37; figure 1). (Color figure online)
Three schemes of coordination (phase-coupled, metastable, and uncoupled) at four different scales (model, behavior, brain microscale, and brain macroscale). All charts (A–L) show the relative phase (ϕ; y-axis) between two oscillators through time (x-axis) given the three different schemes of coordination and the four scales of observation. In general terms, the more horizontal the lines are, the more coordination is achieved. Thus, we can see how (green) lines are mostly horizontal all the time in the phase-coupled charts (A, D, G, J) and (purple) lines are mostly diagonal—non-coupled—all the time in the uncoupled charts (C, F, I, L). However, the (colored) lines in the metastable charts (B, E, H, K) combine both moment of phase-coupling and uncoupling through time without ever stay in any of them
Coordination that exhibits metastability or metastable regimes (Kelso 2012; Tognoli and Kelso 2014) combines moments of synchronization (or integration), named dwells, and moments of non-synchronization (or segregation), named escapes. Metastability allows brain regions to be non-linearly coupled (or quasi-synchronized) with other brain regions that hold different intrinsic dynamics without the necessity of a strong phase-coupling. Moreover, the moments of coupling give rise to moments of non-coupling without the need for a different kind of mechanism. The transitions are part of the dynamics of the system. Thus, metastable regimes provide a way to model TALoNS as the temporary alliances of neural regions to solve cognitive tasks.
The combination of neural reuse and coordination dynamics offers a framework to develop dynamical explanations of the dynamics at the neural scale of analysis of cognitive systems. Radical embodiment can find—and indeed has found—in it a way to explain cognitive phenomena at the scale typically addressed by neuroscience. However, the explanation of the activity of cognitive systems at their neural scale of analysis in terms of neural reuse and coordination dynamics do not offer a way to relate the findings at that scale with the parallel findings at the behavioral scale of analysis—the one addressed by behavioral dynamics, for example. As we have seen, a unified theory or framework for radical embodiment must connect the two scales in an integrated fashion. This is, precisely, what resonance helps to do.
4.3 Resonance: a unified framework
As I have described in Sect. 3, resonance is the process by which the dynamics at the scale of the neural system are constrained by the same informational variable that constrains the dynamics at the scale of behavior. Given this and the particular theoretical context developed in the present section, how can behavioral dynamics and neural reuse/coordination dynamics be integrated by a process of resonance?
The most natural way to integrate behavioral dynamics and neural reuse/coordination dynamics can be found in coupling the dynamics at the neural scale of analysis with the dynamics at the behavioral scale of analysis (see Fig. 4). As noted before, resonance is a special kind of coupling and, therefore, the coupling of behavioral and neural dynamics in the framework is an instance of the particularities of resonance. In a concrete sense, unlike other notions related to coupling, ecological resonance allows for non-parametric, informational coupling. In this sense, ecological resonance is theoretically understood as a dynamical coupling in which relevant perceptual information, i, constrains the dynamics at the neural scale, ṅ, given a function of resonance of the form ṅ = ρ(n, i), where n stands for the states of the nervous system. Importantly, notice that the relevant information does not necessarily work as a parameter for the dynamics of the nervous system but is modeled as a state variable of the function of resonance. In other words, the relevant perceptual information constrains the composition of both neural and behavioral dynamics, not just the specific form those dynamics take. An example of this role of information in constraining both scales in terms of informational coupling can be found, again, in van der Weel and van der Meer’s (2009) study discussed above. In that study, the relevant perceptual information (tau for time-to-contact; see Lee 2009) is a component of both dynamics: both dynamics are coupled through their respective tau values, but tau itself is not parametrizing the coupling.Footnote 20 Thus, one signature of the ecological notion of resonance is the presence of ecological information in both dynamics (more on this below).
Concrete unified framework based on resonance (notice it is an expansion of Fig. 1). Behavioral dynamics and neural reuse/coordination dynamics are integrated in a unified framework based on resonance. As it does with the dynamics at the scale of organism–environment interactions, task-relevant information, i, also constrains the dynamics at the neural scale given a function ṅ = ρ (n, i). These dynamics constitute a proper part ∂o of the overall dynamics of the organism ȯ (yellow arrow). (Color figure online)
Importantly, the dynamics at the neural scale that result from the resonant process constitute a proper part ∂o of the overall dynamics of the organism (ȯ). In other words, the neural scale is nested in the organism. Such a nesting makes the resonant coupling between behavioral and neural dynamics not only a relation of mutual constraint but also a relation of constitution—neural dynamics are a part (∂o) of the overall dynamics of the organism (ȯ). Most of the time such a distinction between constraint and constitution will not entail practical differences in terms of modeling or experimental research, but the analysis of the kind of nesting itself is an interesting topic for future research (see next section).
The relation between behavioral dynamics and neural reuse/coordination dynamics in the proposed integrative model may be further detailed. TALoNS—the temporal synchronization of neural regions to support cognitive phenomena—can be understood as the neural states defined in the resonance function (n). In this sense, the dynamics of the neural states, ṅ, are understood as the changes occurred to temporally stable assemblies of neural regions due to the specific constraints posited by the dynamics of the organism–environment interactions. When TALoNS are understood in such a way, they are constrained by relevant perceptual information in terms of the resonance function ṅ = ρ(n, i) and participate in the overall dynamics of the organism–environment system in terms of the function ∂o = μ(ṅ). Two main consequences follow from this conceptualization of TALoNS.
The first consequence is that TALoNS must be explained in terms of sensorimotor regularities framed within behavioral dynamics. On the one hand, TALoNS are constrained by relevant perceptual information. In this sense, they are important for perception. On the other hand, TALoNS constrain the forces the organism exerts to her environment. Namely, they are also important for action. Thus, TALoNS participate in the overall perception and action dynamics. Importantly, a single TALoNS can participate in these dynamics. In other words, it is not needed to define a TALoNS in terms of perception and a subsequent one in terms of action. The one and the same TALoNS can participate in the whole sensorimotor regularity. Moreover, this fact is compatible with TALoNS being groups of coupled neural regions that have their own activation propensities. Under this understanding of TALoNS, it is fair to expect that regions of the visual and motor cortex, for example, are jointly activated given some specific perception–action dynamics.
The second consequence is that this understanding of TALoNS sets a structured framework for the study of the relations between the dynamics of the organism–environment system and the dynamics of the nervous system. As behavioral dynamics provide dynamical explanations of behavior at the scale of the dynamics of the organism–environment interactions and the relevant perceptual information that constrains that scale also constrains the dynamics of the nervous system, putting together the two scales is a matter of finding dynamic patterns that correspond to TALoNS at the scale of the dynamics of the nervous system and modeling their coupling with the dynamics of the organism–environment interactions or vice versa. The concrete details of the relation between specific TALoNS and behaviors is an empirical question, although some research is already pointing in that direction. As a hypothesis, the expectation is that the biophysical and physiological details of the integration of relevant perceptual information into the dynamics of the nervous system, described by the resonance function ṅ = ρ(n, i), will be carried out by some form of resonant process at different network and physiological scales—e.g., resonant oscillators (Gökaydin et al.’s 2016), nonlinear resonance (Large 2008), resonance frequency shifts (Lau and Zochowski 2011; Roach et al. 2018), or stochastic resonance (Ikemoto et al. 2018). As anticipated in the previous section, the physiological resonant properties of single neurons and neural networks to achieve different degrees of synchronization both among them and with external information could account for the informational coupling between behavioral and neural dynamics described in the ecological notion of resonance. For example, the notion of resonance frequency drift could account for the way different regions of the brain are sensitive to some specific informational variables and, crucially, the way in which they can switch from one variable to another one as the organism–environment interactions change in time (e.g., in perceptual learning; see Raja 2019), as has been already described in some learning tasks (Roach et al. 2018). Importantly, the way the ecological notion of resonance as a process of informational coupling and neural resonance in the form, say, of resonance frequency shifts relate to each other can be seen as a field of future research, but also as a way to explore signatures of resonance. For example, concurrent occurrence of informational coupling and resonance frequency shifts—we have ways to measure both (e.g., van der Weel and van der Meer 2009; Roach et al. 2018)—could be a signature of a resonant coupling as opposed to other kinds of coupling.
5 Open questions: scalar relations and mechanisms
The resonance-based framework presented so far may be a promising candidate unified framework for radical embodiment. It integrates the two scales of analysis usually considered most relevant in cognitive science into the same unified system while keeping their individual importance.Footnote 21 Moreover, it integrates different theories of radical embodiment into a unified framework. As just noted, the viability of the resonance-based framework is a matter of empirical research. Different empirical results support its plausibility and some other results may be taken as proofs of concept of the framework (Aguilera et al. 2013). However, as it could not be otherwise and without prejudice of other further theoretical developments, only future empirical research will assess its failure or success.
The twofold integration entailed by the resonance-based framework, however, leaves two open questions that go beyond empirical considerations. First, there is a question regarding how the details of the relation between the behavioral and the neural scales are best analyzed. It follows from the framework that cognitive phenomena are explained in a multi-scale fashion; that is, behavioral and neural scales are needed and used to account for cognitive phenomena. Moreover, the framework holds a particularity: the ontological and temporal relationship between the scales involved in the explanation is a relation of nesting: the neural scale accounts for a fast-dynamics sub-system of the system accounted by the behavioral scale. Such an approach is not typical of dominant cognitive science, where behavior is taken to be a product or a result of neural activity and, in this sense, the latter is not nested into the former but serially linked to it—allowing for some degrees of complexity in that linking, e.g., feedback loops. For this reason, the way to address the scalar relations in the resonance-based framework is worthy of being studied.
Recently, a methodological approach to the scalar relations of complex, nested systems has been developed: fractal analysis. The main rationale for this kind of analysis is the prediction that complex, nested systems exhibit fractal structures at fast-dynamics scales (Bak 1990; see also Aks 2005; Van Orden et al. 2003; Holden et al. 2013). Due to the nested scalar relations in the resonance-based framework, it seems to be amenable to fractal analysis and a testable hypothesis can be developed: the dynamics at the neural scale of analysis should exhibit a fractal structure as they are constrained by the broader dynamics at the behavioral scale of analysis. Concretely, a fractal structure is expected to be found in the dynamics of TALoNS that are constrained by the same relevant perceptual information that constrains behavior in a given cognitive task. Therefore, and following the literature in fractal analysis, the variability in the dynamics of TALoNS taken in the form of time series must show a fractal structure.
Second, it might be claimed that the resonance-based framework for radical embodiment is mechanistic: resonance might be the mechanism that relates the contributions of the neural system to cognitive phenomena and the organism–environment interactions. As we have seen, resonance is defined as the increase in the amplitude of an oscillator when a force feeds it near to its proper frequency, and so the dynamics at the neural scale of analysis may be understood as the result of the mechanism of resonance. In that picture, the dynamics at the behavioral scale of analysis and the task-relevant information generated at that scale would be a component of the mechanism. The other component of the mechanism would be the oscillatory dynamics at the neural scale of analysis. And resonance would causally constrain the interaction between the two components: one component of the mechanism (the dynamics and information at the behavioral scale) affects the amplitude of the oscillatory dynamics of the other component (the dynamics at the neural scale) by making the latter resonate—i.e., to synchronize/couple to the dynamics at the behavioral scale.Footnote 22
However, the understanding of resonance as a mechanism faces some issues at least for some forms of mechanism. One issue is the two components of the mechanism are dynamic patterns and, therefore, change over time. In other words, the dynamics of the behavioral scale of analysis are not the same ones for any cognitive phenomena and the same applies to the dynamics of the neural scale of analysis. Even more, dynamic patterns are difficult to localize and decompose: they are different ways of interaction between different or the same components of a system. Thus, it is not clear that the sense in which dynamic patterns of interaction are components of the mechanism of resonance and whether they are acceptable as components for all the mechanistic theories. Another issue is that, although resonance can be understood in the mechanistic sense just described, what drives the resonant states of both scales is not just a physical event but the information of that physical event (e.g., the relevant perceptual information for locomotion in the optic flow generated by the organism–environment interaction). Again, it is not clear whether this kind of causal constraint is acceptable for many mechanistic proposals in cognitive science. Finally, there is an issue pertaining to the relation of constraint between the different scales entailed by resonance. In the proposed model, some components (e.g., the dynamics of organism–environment systems) of the resonant mechanism are of a higher spatiotemporal scale than the mechanism itself. This is problematic for some notions of mechanism (e.g., Craver 2008) which assume that the constituting components of a mechanism are always of a lower spatiotemporal scale than the mechanism itself. Also, the relation between the mechanism of resonance and some of its possible components does not seem to be one of constitution but one of constraint. Concretely, the relationship between behavior and brain activity in terms of resonance may be seen as a one of enabling constraint: behavior at the same time enables and is enabled by brain activity [Anderson 2015; Raja and Anderson in press; see also the Gillett’s (2016) notion machresis that aims to capture cases where wholes or higher levels constrain or determine the lower level parts that compose them]. As Anderson (2015) suggests, this kind of relationship may be hard to capture by some notions of mechanism (e.g., Carver 2008) although some other notions that make room for a more substantive notion of constraint may not suffer from that shortcoming (e.g., Winning and Bechtel 2018).
These issues are by no means exhaustive regarding the compatibility or incompatibility between the notion of resonance defended here and some notions of mechanism and mechanistic explanation (see, e.g., Golonka and Wilson 2018). They are, nevertheless, issues that must be tackled if their relationship is to be clarified. That seems like a worthy enterprise for a unified theory of cognitive science that reconciles the dominant approaches with radical embodiment.
Notes
This fact is salient regarding some contemporary theories that aim to explain sets of very different phenomena. For example, self-organized criticality (Bak 1990) aims to account for the behavior of complex systems in terms of their self-organization around stable states (critical states) and the transitions between these states (catastrophes). The theory has been applied to disparate phenomena such as earthquakes or the intentionality of cognitive systems (Juarrero 1999). However, there is no commitment to the idea that the phenomena under the scope of the theory are essentially the same or of the same kind beyond the fact that there are some common theories and methods to capture them. Earthquakes and intentions, for example, are characterized in terms of self-organized criticality, but they still are qualitatively different phenomena in many regards.
For example, the activity within the nervous system, measured in milliseconds or fractions of milliseconds, belongs to the neural scale and is studied by neuroscience while the displacement of the hips in a walking task, measured in seconds or fractions of seconds (the usual cut-off for that measurement is 5 Hz), belongs to the behavioral scales and is studied by experimental psychology. Of course, the distinction between these two spatiotemporal scales may be blurry in some specific situations—as it is sometimes between biology, physiology, or psychology, for example—but they usually are easily distinguished.
Allen Newell’s (1990) own proposal is a clear example of it, but it is not the only one. Other examples are the ACT-R cognitive architecture (Anderson 1983, 2007), the Semantic Pointer Architecture developed by Chris Eliasmith (2013), or the Adaptive Resonance Theory (Grossberg 2013); as well as more recent proposals based on Bayesian models of cognitive systems (Clark 2015; Friston 2010).
Reasons for such rejection are diverse and there is no space in this paper to go through all of them. Some paradigmatic examples have to do with intrinsic problems of computation, like the frame problem (McCarthy and Hayes 1968); others have to do with the intrinsic knowledge a system based on computation needs for perceiving and acting, like the problem of the loans of intelligence (Dennett 1978; Kugler and Turvey 1987); and others with the coordination of all the effectors of a system to generate the desired behavior, as the issue labeled as “the Charles V problem” in the literature on motor control (Meijer 2001).
All the aspects of this description must be met. Notice, for example, that assuming a strategy based on dynamical explanations or using the tools of dynamical systems theory as a methodology do not per se make an approach an instance of radical embodiment. It is possible to use dynamical systems theory to describe a cognitive system while holding a computational or representational understanding of the system—e.g., some cognitive architectures that offer dynamical descriptions of neural systems and still hold computational assumptions, as in the case of Izhikevich (2007), or representational assumptions, an in the case of dynamic field theory (Schöner et al. 2016). In both cases, we would not be talking about radical embodiment.
The concept of resonance has been used in different contexts in cognitive science, but always in relation to coupling—e.g., single-neuron activity (Kasevich and LaBerge 2011), motor resonance and mirror neurons (Leonetti et al. 2015), adaptive resonance theory (Grossberg 2013), resonant processes for sequential effects in psychology (Gökaydin et al. 2016).
Notice that Gibson explicitly rejects the idea that perceptual information can be characterized as a kind of Shannon information (Gibson 1979, pp. 62–63, 242–244) and the idea that perceptual systems are channels of information (Gibson 1966, pp. 1–6, 47–58). In this sense, even the most basic notions of information-processing (e.g., the transformation of some signal in a channel) might not apply to the ecological notion of resonance. For a different example of a system able to detect information without internal processing, see the pole planimeter (Runeson 1977).
See Raja (2018) for a schematic description of the framework and a justification of its plausibility.
For the rest of the paper, I will use the formula ‘relevant information’ to refer to this information. In general, I take ecological information (Gibson 1966, 1979) to be the best characterization of information for radical embodiment, but I do not want to preclude the use of other available notions of information (e.g., O’Regan and Noë 2001; Oyama 2000). Ecological information is revealed at the scale of behavior and is not described in terms of semantic content (Gibson 1979, p. 55 and ff.; Turvey et al. 1981; Turvey and Kugler 1984; Segundo-Ortín et al. 2019) as it is not related to the state of affairs of the environment in terms of truth–values. For example, we generate optic flow by moving through the environment and that optic flow is informative of our movement (e.g., centrifugal flow specifies forward locomotion). But crucially, specific patterns of optic flow are neither true nor false of our movements, but lawfully related to them—in the ecological jargon, it is said that patterns of the optic flow are specific of movements. Due to the lack of a truth–value relationship with the environment, ecological information is said to be non-semantic, but specificational. These are the features to expect from any form of ‘relevant information’ used in the proposed model, in particular, and in radical embodiment, in general.
This feature is common to the explanation of all complex systems. For example, if one wants to explain the performance of a soccer team in a given game, she will minimally need to account for the technical skills of the individual players and for the tactical scheme of the whole team. These are two different scales of analysis (individual and collective), but they are strongly interrelated: the success of the tactical scheme depends on the interpretation of it by the individual players that concurrently depends on their technical skills; and the success of technical skills depends on the position of the player within the tactical scheme. Both scales are relevant to the explanation and irreducible to each other.
Gibson entertained an idea akin to informational coupling: “If the invariants of this [environmental] structure [i.e., ecological information] can be registered by a perceptual system, the constants of neural input will correspond to the constants of stimulus energy, although the one will not copy the other. But then meaningful information can be said to exist inside the nervous system as well as outside” (1966, p. 267).
For other examples of the plausibility of the proposed model of ecological resonance and the notion of informational coupling, see Raja (2018).
I remain agnostic regarding whether a cognitivist/representationalist account of the phenomenon of informational coupling and, more generally, of the phenomenon of ecological resonance is possible. Some scholars in the ecological tradition have argued that ecological information plays the functional role of representations in the theory and that the wording used might not be crucial in this case (Golonka and Wilson 2019). Otherwise, maybe the contemporary notion of structural representation (Rescorla 2009; Shea 2014; Ramsey 2016; Gładziejewski and Miłkowski 2017) could accommodate the notion of resonance as informational coupling. However, I think it is fair to claim that a representational account of resonance is not necessary and further argumentation should be given in order to justify such understanding.
The same applies βo.
For example, in the case of Fajen and Warren’s model (2003), although the organism’s integration of information occurs at the neural scale, the cognitive phenomenon itself (control of locomotion) remains distributed through the organism–environment system; that is, the cognitive phenomenon remains constituted (at least partially) by the behavioral scale: “[C]ontrol is distributed over the agent–environment system. I interpret this statement to imply that biology capitalizes on the regularities of the entire system as a means of ordering behavior” (Warren 2006, p. 358).
The HKB model was first proposed by Haken et al. (1985) as a model for phase transitions in human hand movements, but it was rapidly generalized to capture phase transitions in many other kinds of systems (e.g., Jirsa et al. 1998; Mechsner et al. 2001; Pellecchia et al. 2005; Temprado et al. 2002). In general, the HKB model is able to predict the change of the relative phase (ϕ) between two oscillators (e.g., fingers, legs, metronomes) over time; namely, how the behavior of the two oscillators is stable or not over time regarding different regimes (e.g., in-phase regime, anti-phase regime, and so on).
Remember that the tau-coupling function τND = KτO − ED by K to reflect the strength of the coupling itself. However, the informational variable, tau (τ), is a component of both dynamics (behavioral and neural).
The framework is probably not unified enough for Nagel’s standards as it entails no reduction between different explanatory scales, but it counts for sure as a unified framework in the sense promoted by Newell (1990).
Importantly, the phenomenon of resonance allows for a more complex scenario in which the driven systems also affects the dynamics of the driving system when they are resonating to each other. In this sense, the mechanism of resonance should also account for the other direction: the dynamics at the neural scale being the driving component and the dynamics at the behavioral scale being the driven one.
References
Aguilera, M., Bedia, M. G., Santos, B. A., & Barandiaran, X. E. (2013). The situated HKB model: How sensorimotor spatial coupling can alter oscillatory brain dynamics. Frontiers in Computational Neuroscience, 7, Article 117.
Aks, D. J. (2005). 1/f dynamic in complex visual search: Evidence for self-organized criticality in human perception. In M. A. Riley & G. C. Van Orden (Eds.), Tutorials in contemporary nonlinear methods for the behavioral sciences (pp. 319–352). Alexandria: National Science Foundation. Retrieved from http://www.nsf.gov/sbe/bcs/pac/nmbs/nmbs.jsp.
Anderson, J. R. (1983). The architecture of cognition. Cambridge: Harvard University Press.
Anderson, J. R. (2007). How can the human mind occur in the physical universe?. Oxford: Oxford University Press.
Anderson, M. L. (2014). After phrenology: Neural reuse and the interactive brain. Cambridge: MIT Press.
Anderson, M. L. (2015). Beyond Componential Constitution in the Brain - Starburst Amacrine Cells and Enabling Constraints. In T. Metzinger & J. M. Windt (Eds). Open MIND: 1(T). Frankfurt am Main: MIND Group. https://doi.org/10.15502/9783958570429.
Anderson, M. L., Kinnison, J., & Pessoa, L. (2013). Describing functional diversity of brain regions and brain networks. NeuroImage, 73, 50–58.
Bak, P. (1990). Self-organized criticality. Physica A, 163, 403–409.
Bechtel, W. (2009). Constructing a philosophy of science of cognitive science. Topics in Cognitive Science, 1, 548–569.
Beer, R. D. (1995). A dynamical systems perspective on agent–environment interaction. Artificial Intelligence, 72, 173–215.
Beer, R. D. (2003). The dynamics of active categorical perception in an evolved model agent. Adaptive Behavior, 11(4), 209–243.
Bingham, G. P. (1988) Task-specific devices and the perceptual bottleneck. Human Movement Science, 7, 225–264.
Bingham, G. P. (2004). A perceptually driven dynamical model of bimanual rhythmic movement (and phase perception). Ecological Psychology, 16(1), 45–53.
Bressler, S. L., & Kelso, J. A. S. (2001). Cortical coordination dynamics and cognition. Trends in Cognitive Sciences, 5(1), 26–36.
Bressler, S. L., & Tognoli, E. (2006). Operational principles of neurocognitive networks. International Journal of Psychophysiology, 60, 139–148.
Bruggeman, H., & Warren, W. H. (2005). Integrating target interception and obstacle avoidance. Journal of Vision, 5, 311.
Chemero, A. (2009). Radical embodied cognitive science. Cambridge: MIT Press.
Clark, A. (2015). Surfing uncertainty. London: Oxford University Press.
Craver, C. F. (2008). Explaining the brain: Mechanisms and the mosaic unity of neuroscience. Oxford: Oxford University Press.
Dennett, D. I. (1978). Brainstorms: Philosophical essays on mind and psychology. Montgomery: Bradford Books.
Di Paolo, E. A., Buhrmann, T., & Barandiaran, X. E. (2017). Sensorimotor life: An enactive proposal. Oxford: Oxford University Press.
Eliasmith, C. (2013). How to build a brain. New York: Oxford University Press.
Engin, A., & Vetschera, R. (2017). Information representation in decision making: The impact of cognitive style and depletion effects. Decision Support Systems, 103, 94–103.
Fajen, B. R., & Warren, W. H. (2003). Behavioral dynamics of steering, obstacle avoidance, and route selection. Journal of Experimental Psychology: Human Perception and Performance, 29, 343–362.
Fajen, B. R., & Warren, W. H. (2005). Behavioral dynamics of intercepting a moving target. Experimental Brain Research, 180, 303–319.
Fries, P. (2005). A mechanism for cognitive dynamics: Neuronal communication through neuronal coherence. Trends in Cognitive Science, 9, 474–480.
Friston, K. (2010). The free-energy principle: A unified brain theory? Nature Reviews Neuroscience, 11(2), 127–138.
Gal, O., & Chen-Morris, R. (2013). Baroque science. Chicago: University of Chicago Press.
Garnham, A. (2009). Cognitivism. In J. Symons & P. Calvo (Eds.), The Routledge companion to philosophy of psychology (pp. 99–110). New York: Routledge.
Gibson, J. J. (1966). The senses considered as perceptual systems. Boston: Houghton Miffin.
Gibson, J. J. (1979). The ecological approach to visual perception. Boston: Houghton Miffin.
Gillett, C. (2016). Reduction and emergence in science and philosophy. Cambridge: Cambridge University Press.
Gładziejewski, P., & Miłkowski, M. (2017). Structural representations: Causally relevant and different from detectors. Biology and Philosophy, 32, 337–355.
Gökaydin, D., Navarro, D. J., Ma-Wyatt, A., & Perfors, A. (2016). The structure of sequential effects. Journal of Experimental Psychology: General, 145(1), 110–123.
Golonka, S., & Wilson, A. (2018). Ecological mechanisms in cognitive science. https://psyarxiv.com/83ztv/.
Golonka, S., & Wilson, A. (2019). Ecological representations. Ecological Psychology, 31(3), 235–253.
Grossberg, S. (2013). Adaptive resonance theory: How a brain learns to consciously attend, learn, and recognize a changing world. Neural Networks, 37, 1–47.
Gylmour, C. (1969). On some patterns of reduction. Philosophy of Science, 37(3), 340–353.
Haken, H., Kelso, J. A. S., & Bunz, H. (1985). A theoretical model of phase transitions in human hand movements. Biological Cybernetics, 51, 347–356.
Helfrich, R., Breska, A., & Knight, R. T. (2019). Neural entrainment and network resonance in support of top-down guided attention. Current Opinion in Psychology, 29, 82–89.
Hempel, C. (1965). Aspects of scientific explanation and other essays in the philosophy of science. New York: Free Press.
Hempel, C., & Oppenheim, P. (1948). Studies in the logic of explanation. Philosophy of Science, 15(2), 135–175.
Holden, J., Riley, M., Gao, J., & Torre, K. (2013). Fractal analyses: Statistical and methodological innovations and best practices. Frontiers Research Topic. Retrieved from https://www.frontiersin.org/research-topics/555/fractal-analyses-statistical-and-methodological-innovations-and-best-practices.
Hutcheon, B., & Yarom, Y. (2000). Resonance, oscillation and the intrinsic frequency preferences of neurons. Trends in Neurosciences, 23, 216–222.
Hutto, D., & Myin, E. (2013). Radicalizing enactivism: Basic minds without content. Cambridge: MIT Press.
Ikemoto, S., DallaLibera, F., & Hosoda, K. (2018). Noise-modulated neural networks as an application of stochastic resonance. Neurocomputing, 277, 29–37.
Izhikevich, E. M. (2007). Dynamical systems in neuroscience: The geometry of excitability and bursting. Cambridge: The MIT Press.
Jirsa, V. K., Fuchs, A., & Kelso, J. A. S. (1998). Connecting cortical and behavioral dynamics: Bimanual coordination. Neural Computation, 10, 2019–2045.
Juarrero, A. (1999). Dynamics in action: Intentional behavior as a complex system. Cambridge: The MIT Press.
Kasevich, R. S., & LaBerge, D. (2011). Theory of electric resonance in the neocortical apical dendrite. PLoS ONE, 6(8), e23412.
Kawato, M. (1999). Internal models for motor control and trajectory planning. Current Opinion in Neurobiology, 9, 718–727.
Kelso, J. A. S. (1995). Dynamic patterns. Cambridge: MIT Press.
Kelso, J. A. S. (2012). Multistability and metastability: Understanding dynamic coordination in the brain. Philosophical Transactions of the Royal Society of London B, 367, 906–918.
Kelso, J. A. S., & Tognoli, E. (2007). Toward a complementary neuroscience: Metastable coordination dynamics of the brain. In L. I. Perlovsky & R. Kozma (Eds.), Neurodynamics of cognition and consciousness (pp. 39–59). Berlin: Springer.
Kepler, J. (1604). Optics: Paralipomena to Witelo and the optical part of astronomy [W. H. Donahue, Trans. (2000)]. Santa Fe: Green Lion Press.
Krakauer, J. W., Ghazanfar, A. A., Gomez-Marin, A., MacIver, M. A., & Poeppel, D. (2017). Neuroscience needs behavior: Corrected a reductionist bias. Neuron, 93, 480–490.
Kugler, P. N., & Turvey, M. T. (1987). Information, natural law, and the self-assembly of rhythmic movement. Hillsdale: Lawrence Erlbaum Associates.
Large, E. W. (2008). Resonating to musical rhythm: Theory and experiment. In S. Grondin (Ed.), The psychology of time. West Yorkshire: Emerald.
Lau, T., & Zochowsky, M. (2011). The resonance frequency shift, pattern formation, and dynamical network reorganization via sub-threshold input. PLoS ONE, 6(4), e18983.
Lee, D. N. (2009). General Tau theory: Evolution to date. Special issue: Landmarks in perception. Perception, 38, 837–858.
Leonetti, A., Puglisi, G., Siugzdaite, R., Ferrari, C., Cerri, G., & Borroni, P. (2015). What you see is what you get: Motor resonance in peripheral vision. Experimental Brain Research, 233, 3013–3022.
Lobo, L., Nordbeck, P. C., Raja, V., Chemero, A., Riley, M., Travieso, D., et al. (2019). Route selection and obstacle avoidance with a short-range haptic sensory substitution device. International Journal of Human–Computer Studies, 132, 25–33.
McCarthy, J., & Hayes, P. (1968). Some philosophical problems from the standpoint of artificial intelligence. Stanford: Stanford University.
Mechsner, F., Kerzel, D., Knoblich, G., & Prinz, W. (2001). Perceptual basis of bimanual coordination. Nature, 414, 69–73.
Meijer, O. G. (2001). Making things happen: An introduction to the history of movement science. In M. L. Latash & V. M. Zatsiorsky (Eds.), Classics in movement science (pp. 1–57). Champaign: Human Kinetics.
Morrison, M. (2000). Unifying scientific theories: Physical concepts and mathematical structures. Cambridge: Cambridge University Press.
Nagel, E. (1961). The structure of science: Problems in the logic of scientific explanation. New York: Harcourt, Brace and World.
Nalepka, P., Kallen, R. W., Chemero, A., Saltzman, E., & Richardson, M. J. (2017). Herd those sheep: Emergent multiagent coordination and behavioral-mode switching. Psychological Science, 28(5), 630–650.
Nalepka, P., Lamb, M., Kallen, R. W., Shockley, K., Chemero, A., Saltzman, E., et al. (2019). Human social motor solutions for human–machine interaction in dynamical task contexts. PNAS, 116(4), 1437–1446.
Newell, A. (1990). Unified theories of cognition. Cambridge: Harvard University Press.
Newell, K., Liu, Y.-T., & Mayer-Kress, G. (2008). Landscapes beyond the HKB model. In A. Fuchs & V. K. Jirsa (Eds.), Coordination: Neural, behavioral and social dynamics (pp. 27–44). Berlin: Springer.
O’Regan, J. K., & Noë, A. (2001). A sensorimotor account of vision and visual consciousness. Behavioral and Brain Sciences, 24, 939–1031.
Orekhova, E. V., Stroganova, T. A., & Posikera, I. N. (1999). Theta synchronization during sustained anticipatory attention in infants over the second half of the first year of life. International Journal of Psychophysiology, 32, 151–172.
Oyama, S. (2000). The ontogeny of information. Durham: Duke University Press.
Pellecchia, G., Shockley, K., & Turvey, M. T. (2005). Concurrent cognitive task modulates coordination dynamics. Cognitive Science, 29, 531–557.
Pillai, A. S., & Jirsa, V. K. (2017). Symmetry breaking in space-time hierarchies shapes brain dynamics and behavior. Neuron, 94, 1010–1026.
Port, R. (2003). Meter and speech. Journal of Phonetics, 31, 599–611.
Raja, V. (2018). A theory of resonance: Towards an ecological cognitive architecture. Minds and Machines, 28(1), 29–51.
Raja, V. (2019). From metaphor to theory: The role of resonance in perceptual learning. Adaptive Behavior, 27(6), 405–421.
Raja, V., & Anderson, M. L. (2019). Radical embodied cognitive neuroscience. Ecological Psychology, 31(2), 166–181.
Raja, V., & Anderson, M. L. (in press). Behavior considered as an enabling constraint. In F. Calzavarini & M. Viola (Eds.), Neural mechanisms: New challenges in the philosophy of neuroscience. New York: Springer.
Ramsey, W. M. (2016). Untangling two questions about mental representation. New Ideas in Psychology, 40, 3–12.
Rescorla, M. (2009). Cognitive maps and the language of thought. The British Journal for the Philosophy of Science, 60, 377–407.
Richardson, M. J., Shockley, K., Fajen, B. R., Riley, M. A., & Turvey, M. T. (2008). Ecological psychology: Six principles for an embodied–embedded approach to behavior. In P. Calvo & T. Gomila (Eds.), Handbook of cognitive science: An embodied approach (pp. 161–188). San Diego: Elsevier.
Roach, J. P., Pidde, A., Katz, E., Wu, J., Ognjanovski, N., Aton, S. J., et al. (2018). Resonance with subthreshold oscillatory drive organized activity and optimizes learning in neural networks. PNAS, 115(13), E3017–E3025.
Runeson, S. (1977). On the possibility of “smart” perceptual mechanisms. Scandinavian Journal of Psychology, 18, 172–179.
Sanches de Oliveira, G., Raja, V., & Chemero, A. (2019). Radical embodied cognitive science and ‘real cognition’. Synthese. https://doi.org/10.1007/s11229-019-02475-4.
Schmidt, R., & Richardson, M. (2008). Dynamics of interpersonal coordination. In A. Fuchs & V. K. Jirsa (Eds.), Coordination: Neural, behavioral, and social dynamics (pp. 282–308). Berlin: Springer.
Schöner, G., Spence, J. P., & The DFT Research Group. (2016). Dynamic thinking: A primer on dynamic field theory. Oxford: Oxford University Press.
Segundo-Ortín, M., Heras-Escribano, M., & Raja, V. (2019). Ecological psychology is radical enough: A reply to radical enactivists. Philosophical Psychology, 32(7), 1001–1023.
Shea, N. (2014). Exploitable isomorphism and structural representation. Proceedings of the Aristotelian Society, CXIV, 77–92.
Shtrahman, E., & Zochowski, M. (2015). Pattern segmentation which activity dependent natural frequency shift and sub-threshold resonance. Nature Scientific Reports, 5, 8851. https://doi.org/10.1038/srep08851.
Singer, W. (2005). The brain—An orchestra without a conductor. Max Planck Research, 3, 15–18.
Temprado, J. J., Monno, A., Zanone, P. G., & Kelso, J. A. S. (2002). Attentional demands reflect learning-induced alterations of bimanual coordination dynamics. European Journal of Neuroscience, 16, 1390–1394.
Tognoli, E., & Kelso, J. A. S. (2009). Brain coordination dynamics: True and false faces of phase synchrony and metastability. Progress in Neurobiology, 87, 31–40.
Tognoli, E., & Kelso, J. A. S. (2014). The metastable brain. Neuron, 81, 35–48.
Turvey, M. T. (1992). Ecological foundations of cognition: Invariants of perception and action. In H. L. Herbert, P. W. van der Broek, & D. C. Knill (Eds.), Cognition: Conceptual and methodological issues (pp. 85–117). Washington, DC: American Psychological Association.
Turvey, M. T., & Kugler, P. N. (1984). A comment on equating information with symbol strings. American Journal of Physiology, 246(6.2), 925–927.
Turvey, M. T., Shaw, R. E., Reed, E. S., & Mace, W. M. (1981). Ecological laws of perceiving and acting: In reply to Fodor and Pylyshyn (1981). Cognition, 9(3), 237–304.
Uhlhaas, P. J., Pipa, G., Lima, B., Melloni, L., Neuenschwander, S., Nikolić, D., et al. (2009). Neural synchrony in cortical networks: History, concept and current status. Frontiers in Integrative Neuroscience, 3, 17.
van der Weel, F. R., & van der Meer, A. L. H. (2009). Seeing it coming: Infants’ brain responses to looming danger. Naturwissenschaften, 96, 1385–1391.
van Gelder, T. (1998). The dynamical hypothesis in cognitive science. Behavioral and Brain Sciences, 21, 615–665.
Van Orden, G. C., Holden, J. G., & Turvey, M. T. (2003). Self-organization of cognitive performance. Journal of Experimental Psychology: General, 132(3), 331–350.
Varela, F., Thompson, E., & Rosch, E. (1991). The embodied mind: Cognitive science and human experience. Cambridge: MIT Press.
Walmsley, J. (2008). Explanation in dynamical cognitive science. Minds and Machines, 18, 331–348.
Wang, X. J. (2010). Neurophysiological and computational principles of cortical rhythms in cognition. Physiological Reviews, 90, 1195–1268.
Warren, W. H. (2006). The dynamics of perception and action. Psychological Review, 113(2), 358–389.
Warren, W. H., & Fajen, B. R. (2004). From optic flow to laws of control. In V. F. Hendricks & J. Symons (Eds.), Optic flow and beyond (pp. 307–338). Berlin: Springer.
Warren, W. H., Kay, B. A., Zosh, W. D., Duchon, A. P., & Sahuc, S. (2001). Optic flow is used to control human walking. Nature Neuroscience, 4(2), 213–216.
Wilkie, R., & Wann, J. (2003). Controlling steering and judging heading: Retinal flow, visual direction, and extraretinal information. Journal of Experimental Psychology: Human Perception and Performance, 29(2), 363–378.
Wilson, A., & Bingham, G. P. (2008). Identifying the information for the visual perception of relative phase. Perception & Psychophysics, 70, 465–476.
Winning, J., & Bechtel, W. (2018). Rethinking Causality in Biological and Neural Mechanisms: Constraints and Control. Minds & Machines, 28, 287–310.
Yu, H., Zhang, L., Guo, X., Wang, J., Cao, Y., & Liu, J. (2018). Effect of inhibitory firing pattern on coherence resonance in random neural networks. Physica A, 490, 1201–1210.
Acknowledgements
I am grateful to Michael Anderson, Jonathan Bowen, Tony Chemero, Valerie Hardcastle, Tom Polger, and the audience of the meeting ‘Reconceiving Cognition’ organized by the Centre for Philosophical Psychology at the University of Antwerpen (Belgium) for their helpful comments and suggestions at different stages of the development of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raja, V. Resonance and radical embodiment. Synthese 199 (Suppl 1), 113–141 (2021). https://doi.org/10.1007/s11229-020-02610-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11229-020-02610-6