Abstract
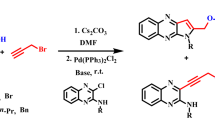
An efficient and regioselective O-alkylation of amides with a variety of electrophiles in the presence of silver nanoparticles is reported as part of our recent research on building blocks for synthesis of natural products. The nano-silver catalyst initiates O-alkylation of the amides by heteroalkyl halides. Reaction of equimolar 3-acetyl-6-chloro-4-phenylquinolin-2(1H)-one and 2-chloro-3-(chloromethyl)quinolines in the presence of silver nanoparticles in DMSO solution under reflux condition leads to the formation of 1-{1-[2-chloroquinolin-3-yl)methoxy]-6-chloro-4-phenylquinolin-3-yl}ethanones.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Major attention has been devoted to the development of regioselective reactions [1–4]. Amides are reported to undergo O-alkylation in the presence of metal salt in a suitable solvent. In continuation of the regioselective N-alkylation reaction [3], we report here regioselective O-alkylation of aromatic amides.
Quinoline derivatives are known to have interesting biological properties ranging from microbial activity to cytotoxicity [5]. Recent studies have, moreover, identified quinolones which inhibit mammalian topoisomerase II [6] as potential lead compounds in the development of anticancer drugs. Quinolines have also been used as substituents to impart the desired pharmacological and pharmacokinetic properties to quinolone antibiotics [7].
Use of metal salts is of substantial importance in organic synthesis because of high selectivity, enhanced reaction rates, and simple workup. In alkylation reactions, alkali metal salts (K, Na, Li) promote N-alkylation whereas silver salts promote O-alkylation [8, 9]. In continuation of our work on catalysis and heterocyclics [3, 5, 10–32], we have investigated an O-alkylation of 3-acetyl-6-chloro-4-phenylquinolin-2(1H)-one with 2-chloro-3-(chloromethyl)quinolines in the presence of silver nanoparticles (Scheme 1).
Results and discussion
The results from synthesis of 1-{1-[2-chloroquinolin-3-yl)methoxy]-6-chloro-4-phenylquinolin-3-yl}ethanones are summarized in Tables 1 and 2. Interestingly, the O-alkylation reaction took less time (20–45 min) for completion (Tables 1, 2). The effect of solvent and the scope of the reaction have also been investigated (Table 1). When 1-{1-[2-chloroquinolin-3-yl)methoxy]-6-chloro-4-phenylquinolin-3-yl}ethanones 1 were treated with silver nanoparticles and DMSO with conventional heating in the presence of 2-chloro-3-(chloromethyl)quinoline, 2a, the product 3a was obtained in quantitative yield (entry 14, Table 1).

The same reaction carried out with different catalysts in the absence of solvent did not proceed (Table 1, entries 1, 3, 5, 7, 9, 11 and 13). These experimental results clearly suggest that the reaction involves a heterogeneous process and the silver nanoparticles act as an efficient catalyst. The same reaction when carried out with bentonite in DMSO gave two products (Table 1, entry 2), whereas in the presence of silver nanoparticles in DMSO (Table 2, entry 14) only the O-alkylated regioselective product 3a was formed. The results suggest that the reaction proceeds well under the optimized conditions (Table 1, Entry 14). When the O-alkylation was carried out in a variety of solvents of different dielectric constant a high yield was obtained in DMSO (high dielectric constant) whereas the N-alkylated product was formed in THF (low dielectric constant).
Thus optimization of the reaction suggested that silver nanoparticles with DMSO is the appropriate combination for good regioselective product yield (Table 1). To investigate the scope of this reaction, a variety of halides were investigated (Table 2, entries 1–7). Substituted 2-chloro-3-(chloromethyl)quinoline 2 afforded the corresponding product 3 in good yields. The proposed mechanism of the reaction is depicted in Fig. 1.
The reaction of silver nanoparticles with alkyl halides may proceeds via carbonium ion formation by electrophilic attack by silver on the halogen through an SN1 mechanism and further reaction of this carbonium ion with amide will take place through covalent bond formation to the most electronegative atom of the ambident anion possessing two different reactive positions. In this reaction, the acetyl group oxygen atom and the amide group oxygen atom share the negative charge whereas the nitrogen atom of the amide group is essentially neutral and hence facilitates the regioselective O-alkylation.
In the course of this work, a few compounds with different substitution of the aromatic ring of the quinoline skeleton have been synthesized. Electron-withdrawing groups present in the 3-position of the quinoline ring 1 facilitated the reactions. This method was found to be completely regioselective. The spectral and analytical data of products 3 were in accordance with the assigned structures. Similarly, in all cases the disappearance of the –NH peak confirmed the formation of products 3b–f.
Experimental
Solvents and reagents were commercially sourced and used without further purification. Melting points were taken on Elchem microprocessor-based DT apparatus in open capillary tubes and are corrected with reference to benzoic acid. IR spectra were obtained on a Nucon infrared spectrophotometer using KBr pellets. The NMR spectra were recorded on a Bruker Avance 400 MHz spectrometer using TMS as internal standard (chemical shifts δ in ppm). LC–MS was performed with a Flash 1112 analyser in EI mode; the electron energy was 70 eV. Data are reported in the form of m/z intensity relative to the base peak. Silver nanoparticles were prepared by use of a reported method [33]. Silver sulfate (1.0 mM, 10 mL) was added dropwise to 30 mL 2.0 mM sodium borohydride solution that had been chilled in an ice bath. The reaction mixture was stirred vigorously on a magnetic stir plate. The solution turned light yellow after addition of 2 mL Ag2SO4, then a brighter yellow, after which the stirring was stopped and the stir bar removed. The clear yellow colloidal solution was then subjected to centrifugation at 5000 rpm for 30 min, after which the pellet were separated and characterized by TEM by use of an Hitachi 800, using an accelerating voltage of 120 kV; a fairly uniform particle size of 25 ± 0.5 nm was evident (Fig. 2).
General procedure for regioselective O-alkylation (3a–f)
In a typical procedure, heteroalkylhalide 2 (1 mmol) and silver nanoparticles (10 mol%) were added to a solution of the cyclic amide 1 (1 mmol) in DMSO (5 mL) and the mixture was heated under reflux at 110 °C. The reaction was complete within 20–45 min. The reaction mixture was then filtered and the supernatant liquid was added dropwise on to crushed ice. The solution was neutralized with dilute HCl. The precipitate was isolated by filtration and recrystallized from ethanol.
Synthesis of 1-{1-[2-chloroquinolin-3-yl)methoxy]-6-chloro-4-phenylquinolin-3-yl} ethanones (3a)
To a mixture of 3-acetyl-6-chloro-4-phenylquinolin-2(1H)-one 1 (1 mmol) and silver nanoparticles (10 mol%) in 5 mL DMSO, 2-chloro-3-chloromethylquinoline 2a (1 mmol) was added dropwise at 110 °C. Completion of the reaction was monitored by TLC. The reaction mixture was poured into 5% HCl to give 1-{1-[2-chloroquinolin-3-yl)methoxy]-6-chloro-4-phenylquinolin-3-yl}ethanones 3a as pale yellow solids which were recrystallized from ethanol. Product identity was confirmed by IR, NMR, and MS analysis. Data for new compounds 3a–f, which have not been reported elsewhere, are given below.
3a: Pale yellow solid, M.p. 167–169 °C, IR (KBr): 1710 cm−1, 1H NMR (400 MHz, CDCl3): δ H = 2.22 (3H, s, –CH3), 5.84 (2H, s, –CH2), 7.34 (2H, t), 7.48 (1H, d), 7.52 (3H, t), 7.57–7.67 (2H, m), 7.73 (1H, t), 7.87 (2H, q), 8.03 (1H, d), 8.33 (1H, s) ppm, 13C NMR (125 MHz, CDCl3): δ C = 32.1 (–CH3), 64.7 (–CH2), 125.5, 126.7, 4 × 127.0, 2 × 128.1, 2 × 129.2, 2 × 129.3, 2 × 129.4, 2 × 130.7, 131.2, 134.1, 136.2, 138.3, 2 × 144.6, 2 × 146.3, 201.4 ppm, LC–MS: m/z 473 [M + 1]+ for C27H18Cl2N2O2 requires 472.
3b: White solid, M.p. 162–164 °C, IR (KBr): 1708 cm−1, 1H NMR (400 MHz, CDCl3): δ H = 2.27 (3H, s, –CH3), 2.78 (3H, s, –CH3), 5.82 (2H, s, –CH2), 7.23–7.39 (3H, m), 7.44 (1H, d, J 7.6 Hz), 7.47–7.61 (5H, m), 7.69 (1H, d, J 8 Hz), 7.87 (1H, d, J 9.2 Hz), 8.28 (1H, s) ppm, 13C NMR (125 MHz, CDCl3): δ C = 17.8 (–CH3), 32.0 (–CH3), 64.9 (–CH2), 125.5, 126.8, 4 × 127.1, 2 × 128.2, 2 × 129.0, 3 × 129.4, 2 × 129.6, 2 × 130.7, 131.0, 134.0, 136.4, 138.3, 2 × 144.5, 2 × 146.5, 201.8 ppm, LC–MS: m/z 487 [M + 1]+ for C28H20Cl2N2O2 requires 486.
3c: White solid, M.p. 176–178 °C, IR (KBr): 1704 cm−1, 1H NMR (400 MHz, CDCl3): δ H = 2.22 (3H, s, –CH3), 2.56 (3H, s, –CH3), 5.81 (2H, s, –CH2), 7.33 (2H, s), 7.34 (2H, t), 7.40 (1H, d, J 8 Hz), 7.47 (1H, d), 7.52 (2H, m), 7.59 (1H, d, J 8 Hz), 7.75 (1H, d, J 8 Hz), 7.87 (1H, d, J 8.8 Hz), 8.26 (1H, s) ppm, LC–MS: m/z 487 [M + 1]+ for C28H20Cl2N2O2 requires 486.
3d: Pale yellow solid, M.p. 165–167 °C, IR (KBr): 1706 cm−1, 1H NMR (400 MHz, CDCl3): δ H = 2.21 (3H, s, –CH3), 2.78 (3H, s, –CH3), 5.84 (2H, s, –CH2), 7.34 (2H, t), 7.48 (1H, d), 7.52 (2H, t), 7.57–7.67 (2H, m), 7.73 (1H, t), 7.87 (2H, two d), 8.02 (1H, d), 8.31 (1H, s) ppm, LC–MS: m/z 487 [M + 1]+ for C28H20Cl2N2O2 requires 486.
3e: White solid, M.p. 154–156 °C, IR (KBr): 1706 cm−1, 1H NMR (400 MHz, CDCl3): δ H = 2.24 (3H, s, –CH3), 3.92 (3H, s, –OCH3), 5.82 (2H, s, –CH2), 7.33 (2H, d, J 7.6 Hz), 7.41 (2H, d, J 8.8 Hz), 7.47–7.51 (2H, m), 7.60 (2H, d, J 9.6 Hz), 7.87 (1H, t, J 9.6 Hz), 7.90–7.97 (2H, m), 8.73 (1H, s) ppm, 13C NMR (125 MHz, CDCl3): δ C = 17.8 (–OCH3), 32.0 (–CH3), 64.9 (–CH2), 125.5, 126.8, 4 × 127.1, 2 × 128.2, 2 × 129.0, 3 × 129.4, 2 × 129.6, 2 × 130.7, 131.0, 134.0, 136.4, 138.3, 2 × 144.5, 2 × 146.5, 201.8 ppm, LC–MS: m/z 503 [M + 1]+ for C28H20Cl2N2O3 requires 502.
3f: Pale yellow solid, M.p. 148–150 °C. IR (KBr): 1708 cm−1. 1H NMR (400 MHz, CDCl3): δ H = 2.27 (3H, s, –CH3), 4.05 (3H, s, –OCH3), 5.82 (2H, s, –CH2), 6.98 (2H, d, J 7.6 Hz), 7.21–7.31 (4H, m), 7.42–7.50 (4H, m), 7.59 (1H, d, J 8.4 Hz), 8.73 (1H, s) ppm. LC–MS: m/z 503 [M + 1]+ for C28H20Cl2N2O3 requires 502.
Conclusion
In the work discussed in this report we developed a simple and effective method for facile O-alkylation using silver nanoparticles. The method has very attractive features, for example reduced reaction times, higher yields, and economic viability of the catalyst. The operational simplicity of the procedure is also attractive.
References
R. Islam, N. Ashida, T. Nagamatsu, Tetrahedron 64, 9885 (2008)
R. Lucas, K. Teste, R. Zerrouki, Y. Champavier, M. Guilloton, Carbohydr. Res. 345, 199 (2010)
S.M. Roopan, F.N. Khan, B.K. Mandal, Tetrahedron Lett. 51, 2309 (2010)
F.N. Khan, S.M. Roopan, V.R. Hathwar, M. Akkurt, Acta Cryst E66, o972 (2010)
S.M. Roopan, F.N. Khan, Synthesis, antioxidant, hemolytic and cytotoxicity activity of AB ring core of mappicine. ARKIVOC xiii, 161 (2009)
M.P. Wentland, G.Y. Lesher, M. Reumanm, M.D. Gruett, B. Sing, S.C. Aldous, P.H. Dorff, J.B. Rake, S.A. Coughlin, J. Med. Chem. 36, 2801 (1993)
H. Tawada, H. Natsugari, E. Ishikawa, Y. Sugiyama, H. Ikeda, K. Meguro, Chem. Pharm. Bull. 43, 616 (1995)
G. Vavilina, A. Zicmanis, P. Mekss, M. Klavins, Chem. Heter. Comp. 44, 549 (2008)
M.A. Morel, E.L. Larghi, M.M. Selvero, Synlett 18, 2755 (2005)
S.M. Roopan, F.N. Khan, A.K. Kushwaha, V.R. Hathwar, M. Akkurt, Acta Cryst. E66, o960 (2010)
S.M. Roopan, F.N. Khan, M. Vijetha, V.R. Hathwar, S.W. Ng, Acta Cryst. E65, o2982 (2009)
Y. Isogai, Menggenbateer, F.N. Khan, N. Asao, Tetrahedron 65, 9575 (2009)
T. Maiyalagan, F.N. Khan, Catal. Commun. 10, 433 (2009)
P. Manivel, F. Nawaz Khan, J. Chil. Chem. Soc. 54, 180 (2009)
P. Manivel, S. Mohana Roopan, R. Sathish kumar, F. Nawaz Khan, J. Chil. Chem. Soc. 54, 183 (2009)
P. Manivel, F. Nawaz Khan, Ind. J. Chem. 48B, 825 (2009)
F.N. Khan, P. Manivel, K. Prabakaran, V.R. Hathwar, Mehmet Akkurt, Acta Cryst. E66, o1081 (2010)
F.N. Khan, P. Manivel, K. Prabakaran, V.R. Hathwar, M. Akkurt, Acta Cryst. E66, o1094 (2010)
N.T. Patil, F.N. Khan, Y. Yamamoto, Tetrahedron Lett. 45, 8497 (2004)
P. Manivel, S. Mohana Roopan, F. Nawaz Khan, Ind. J. Heter. Chem. 18, 21 (2008)
P. Manivel, S. Mohana Roopan, F. Nawaz Khan, J. Chil. Chem Soc. 53, 1609 (2008)
K. Prabakaran, P. Manivel, F.N. Khan, Tetrahedron Lett. 51, 4340 (2010)
F.N. Khan, P. Manivel, K. Prabakaran, V.R. Hathwar, Ng. Seik Weng, Acta Cryst. E66, o370 (2010)
P. Manivel, F. Nawaz khan, V.R. Hathwar, Phosphorus, Sulphur, Silicon Relat. Elem. 185, 1932 (2010)
F.N. Khan, P. Manivel, K. Prabakaran, V.R. Hathwar, Ng. Seik Weng, Acta Cryst. E65, o2732 (2009)
F.N. Khan, P. Manivel, K. Prabakaran, V.R. Hathwar, Ng. Seik Weng, Acta Cryst. E66, o488 (2010)
P. Manivel, F. Nawaz Khan, Phosphorus, Sulfur, Silicon, Relat. Elem. 184, 2910 (2009)
S. Mohana Roopan, T.R. Sakthi Sri, B. Ramakrishna Reddy, F. Nawaz Khan, Ind. J. Heter. Chem. 19, 77 (2009)
S.S. Tajudeen, F.N. Khan, Synth. Commun. 37, 3649 (2007)
S.M. Roopan, T. Maiyalagan, F.N. Khan, Can. J. Chem. 86, 1019 (2008)
V.R. Hathwar, P. Manivel, F. Nawaz Khan, T.N.G. Ro, Acta Cryst E 63, o3707 (2007)
V.R. Hathwar, P. Manivel, F. Nawaz Khan, T.N.G. Row, Acta Cryst. E 63, o3708 (2007)
S.D. Solomon, M. Bahadory, A.V. Jeyarajaslingam, S.A. Rutkowsky, C. Borito, J. Chem. Educat. 84, 322 (2007)
Acknowledgments
This study was supported by Department of Science and Technology Government of India (grant no. SR/FTP/CS-99/2006). We also acknowledge IISc, Bangalore, for providing the NMR facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gund, M., Mohana Roopan, S., Nawaz Khan, FR. et al. Regioselective O-alkylation: synthesis of 1-{2-[(2-chloroquinolin-3-yl)methoxy]-6-chloro-4-phenylquinolin-3-yl}ethanones. Res Chem Intermed 38, 1111–1118 (2012). https://doi.org/10.1007/s11164-011-0447-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-011-0447-z