Abstract
We describe the feeding habits of 70 blue sharks (Prionace glauca) and 39 salmon sharks (Lamna ditropis) caught at 0–7 m depth at night by research drift gillnets in the transition region of the western North Pacific during April–May of 1999 and 2000. Blue sharks of 50–175 cm total length fed on a large variety of prey species, consisting of 24 species of cephalopods and 16 species of fishes. Salmon sharks of 69–157 cm total length fed on a few prey species, consisting of 10 species of cephalopods and one species of fish. Important prey for the blue sharks were large, non-active, gelatinous, meso- to bathypelagic cephalopods (e.g., Chiroteuthis calyx, Haliphron atlanticus, Histioteuthis dofleini and Belonella borealis) and small myctophid fishes. Important prey for the salmon sharks were mid-sized, active, muscular, epi- to mesopelagic squids (e.g. Gonatopsis borealis, Onychoteuthis borealijaponica and Berryteuthis anonychus). Our results suggest that blue sharks feed on cephalopods mainly during the daytime when they descend to deep water. Salmon sharks may feed opportunistically with no apparent diurnal feeding period. Blue sharks and salmon sharks have sympatric distribution in the transition region in spring; they have different feeding habits and strategies that reduce competition for food resources.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The blue shark, Prionace glauca, is distributed worldwide in tropical and subtropical pelagic waters, and is considered to be the most abundant shark in the world (Compagno 1984). It grows larger than 3 m in total length and undergoes a large scale north–south migration in the North Pacific during its life history (Nakano and Seki 2003). The salmon shark, Lamna ditropis, is a little smaller (reaching about 2.5–3 m in total length) and endemic to the subtropical to subarctic waters of the North Pacific (Sano 1960, 1962). It migrates northward during spring and summer and southward during fall and winter (Tanaka 1980).
The feeding habits of the blue shark have been reported from the eastern North Atlantic (Stevens 1973; Clarke and Stevens 1987), the central Pacific (Strasburg 1958; Seki 1993), off California (Tricas 1979; Harvey 1989), and the Gulf of Alaska (Le Brasseur 1964). These authors reported that blue sharks feed largely on fishes and cephalopods; however few studies have attempted to evaluate the qualitative and quantitative importance of prey species in the diet. The salmon shark preys on fishes, especially salmonids in northern areas (Sano 1960, 1962) and mackerels and sardine in southern areas (Tanaka 1980), but its feeding habits have not been studied in detail.
The transition region of the North Pacific occurs near 30°–45° N between subtropical and subarctic regions (Favorite et al. 1976). It is a highly productive region during spring to fall, and both subtropical and subarctic fishes and cephalopods pass through it during their feeding migrations (Pearcy 1991). During the late 1970’s–1992, large-scale pelagic drift gillnet fisheries were conducted in this region by Japanese, South Korean and Taiwanese fleets targeting mainly neon flying squid (Ommastephes bartramii), tunas and billfishes. McKinnell and Seki (1998) analyzed catch statistics of the Japanese driftnet fishery in 1990–1991 and reported that the total numeric catch comprised 67% cephalopods, 32% bony fishes, 1% elasmobranchs and 0.3% seabirds, turtles and marine mammals. The elasmobranchs included blue sharks (93.7%) and salmon sharks (5.2%). Although sharks composed only a small number of the catch, they become important components of the food web in this region when considering their size and amount of prey consumed.
During 1999–2003, the Japan Fisheries Agency conducted drift-gillnet surveys in the transition region of the North Pacific to study the feeding habits of various marine animals (Watanabe et al. 2003, 2004). The present paper describes the prey compositions of blue shark and salmon shark collected in the transition waters of the western North Pacific and evaluates the quantitative importance of their prey.
Materials and methods
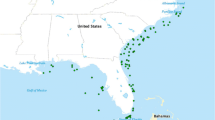
Sharks were sampled with research drift gillnets during research cruises conducted by the National Research Institute of Far Seas Fisheries (RIFSF) during 4–26 May 1999 and, 27 April–27 May 2000. The fishing grounds extended from 35° N to 42° 30’ N and from 155° E to 178° W (Fig.1). The research drift gillnets consisted of 10 different mesh sizes (48, 55, 63, 72, 82, 93, 106, 121, 138 and 157 mm) that each formed three units of the net (one unit measured 50-m long and 7-m deep) and an additional 20 units of 115-mm mesh. At each sampling station, gillnets was deployed at dusk and retrieved at dawn on the following day. A total of 20 fishing operations were conducted in 1999, and 41 were conducted in 2000. Conductivity Temperature Depth profiler casts to 500 m depth were conducted at each station and between stations to observe vertical oceanographic profiles.
Sharks were measured for total length and their stomachs were removed on board. The stomachs were tied off at both ends, cut from the body and then frozen for later analyses in the laboratory. We collected 35 blue shark stomachs and six salmon shark stomachs in 1999, and 35 and 33 in 2000, respectively.
In the laboratory, each stomach was thawed and weighed before its contents were removed. The contents were placed in a plastic pan, and the inner wall was rinsed carefully to collect all prey remnants. Each empty stomach was weighed, and this weight was subtracted from the full stomach weight to determine the weight of the stomach contents. Undigested and partially digested fishes and cephalopods were sorted and identified to the lowest taxonomic level based on external morphology. Where possible, the body length, standard or total length (ST, TL) for fishes, the dorsal mantle length (DML) for cephalopods, and the wet weight were measured. From half-digested items, such as fish skulls and cephalopod buccal masses, sagittal otoliths and beaks were extracted for identification. Finally fish sagittal otoliths, cephalopod beaks, fragments of zooplankton and debris were sorted from the remaining stomach contents.
Pairs of otoliths from fishes were sorted into left and right otoliths. Typically the left ones were identified to the lowest possible taxonomic level based on Ohizumi et al. (2001) and their otolith collection at RIFSF. When the numbers of left and right otoliths in a stomach differed, the larger number was used to estimate the number of prey fish. The maximum otolith diameter (MDO) was measured to the nearest 0.05 mm with a dissecting microscope and micrometer. Cephalopod beaks were separated into upper and lower beaks, and the lower beaks were identified to the lowest possible taxonomic level based on Clarke (1986), Kubodera and Furuhashi (1987), Kubodera and Ohizumi (2001), and the beak reference collection at the National Science Museum, Tokyo. The number of lower beaks was used to estimate the number of prey cephalopods. The rostral length of the lower beak (LRL) was measured to the nearest 0.1 mm with vernier calipers. The MDO and LRL were used to estimate the intact size and weight of prey items using regression formulae hitherto reported (Kubodera 1982; Clarke 1986; Kubodera and Furuhashi 1987; Kubodera and Shimazaki 1989; Ohizumi et al. 2001; Kubodera and Ohizumi 2001). For the prey species for which regression formulae are unavailable, appropriate body weights reported in the literature (Hart 1973; Amaoka et al. 1983) and/or deduced from our data are given.
Using the frequency of occurrence (F) of each prey, the proportion that each prey formed of the total number of prey (Cn), and the proportion that each prey formed of the total wet weight of the stomach contents (WW), an index of relative importance (IRI) proposed by Pinkas et al. (1971) was calculated using the equation:
The index is represented by the size of the rectangle resolved by plotting the three values on a three-way graph.
Results
Distribution and size of sharks
Most of the blue sharks were collected in the transition zone south of the subarctic boundary (Fig.1), while the salmon sharks occurred in both the transition zone and transitional domain (Fig. 2).
Among the 70 blue sharks from which stomachs were removed, 44 individuals were measured for total length (TL) on board. The TLs ranged from 50 to 175 cm with a mode at about 80–110 cm (Fig. 3). Among the 39 salmon sharks from which stomachs were removed, 26 individuals were measured for total length on board. The TLs ranged from 69 to 157 cm with dominant size classes of 90–100, 100–110, and 130–140 cm (Fig. 4).
Stomach contents weights
Several stomachs were sliced during removal and/or their ends came untied, so stomach contents weight was accurately measured in 42 of the 70 blue shark stomachs, and 25 of the 39 salmon shark stomachs. Stomach contents weights ranged from 0 to 1,705 g for blue sharks and 0 to 1,465 g for salmon sharks. Stomach contents weights tended to increase with increasing size, but this was not always the case (Figs. 5 and 6). Stomachs that had a large volume were usually filled with digestive fluid and/or seawater.
Prey species composition in number and frequency of occurrence
Blue sharks
Of the 70 stomachs examined, 57 contained prey items. A total of 430 prey items were collected, including 220 (51.2%) cephalopods and 210 (48.8%) fishes. The average number of prey per stomachs with contents was 6.9 in 1999 and 8.2 in 2000 (overall mean = 7.5). The frequency of occurrence was 74.5% for cephalopods and 66.7% for fishes (Table 1).
Blue sharks fed on a large variety of prey species, including 24 species of cephalopods from 15 families, 16 species of fishes from 8 families and an unidentified shark. In addition, several amphipods, euphausiids, copepods and decapods were found in the stomachs, but they may have come from the stomachs of prey so have been eliminated from analyses.
The most numerous cephalopod prey over the 2 years was the squid, Chiroteuthis calyx, which accounted for 7.0% of the total prey (TP) and 28.1% by frequency of occurrence (FO). Other cephalopod prey included the pelagic octopods, Haliphron atlanticus (6.5% TP, 17.5% FO), Belonella borealis (6.3% TP, 12.3% FO), Japetella diaphana (4.0% TP, 19.3% FO), the squid, Gonatus pyros (3.0% TP, 10.5% FO), Gonatus onyx (2.6% TP, 8.8% FO), Octopoteuthis sp. (2.3% TP, 10.5% FO), Histioteuthis dofleini (2.1% TP, 14.0% FO), H. inermis (1.9% TP, 7.0% FO), Ancistrocheirus lesueuri (1.6% TP, 8.8% FO), Gonatus berryi (1.6% TP, 5.3% FO) and Gonatus sp. (1.6% TP, 8.8% FO). Each of the remaining 12 cephalopod species accounted for less than 1.5% of the total prey and less than 6% by frequency of occurrence.
The most numerous fish prey was Maurolicus imperatorius which accounted for 10.2% of the total prey, but occurred in only one stomach. Other fish prey included four myctophids, Electrona risso (7.9% TP, 31.6% FO), Symbolophorus californiensis (4.4% TP, 14.0% FO), Ceratoscopelus warmingi (2.3% TP, 12.3% FO) and Lampanyctus alatus (1.4% TP, 3.5% FO), and Pacific pomfret, Brama japonica (1.2% TP, 3.5% FO). Each of the remaining 11 fish species accounted for less than 1.5% of the total prey and 3.5% of frequency of occurrence.
Salmon sharks
Of the 39 stomachs examined, 31 contained prey items. A total of 105 prey items were collected, including 83 (79%) cephalopods and 22 (21%) fishes. The average number of prey per stomachs with contents was 8.7 in 1999 and 2.8 in 2000 (overall mean = 3.4). The frequency of occurrence was 100% for cephalopods and 48.4% for fishes (Table 2). Several amphipods, euphausiids, and copepods were also found in the stomachs, but were not included in our analyses.
The most numerous cephalopod prey over the 2 years was the squid, Gonatopsis borealis, accounting for 31.4% of the total prey with a frequency of occurrence of 38.7%. Other cephalopod prey included the squid, Onychoteuthis borealijaponica (11.4% TP, 25.8% FO), Berryteuthis anonychus (8.6% TP, 19.4% FO), Gonatus sp. (6.7% TP, 12.9% FO), Gonatus kiddendorffi (2.9% TP, 9.7% FO), and Gonatus berryi (2.9% TP, 9.7% FO). Each of the four remaining species accounted for less than 2% of the total prey and 6.5% by frequency of occurrence.
A single eroded otolith from an unidentified Paralepididae fish occurred in each of six stomachs, accounting for 5.7% of the total prey with a frequency of occurrence of 19.4%.
Prey species composition by weight
Blue sharks
Over the 2 years, cephalopods accounted for nearly 90% of total prey weight and 51% of the total prey number (Table 3). This difference was mainly due to the smaller number of large-sized cephalopods and the large number of small-sized myctophid fishes in the diet.
The soft parts of all cephalopod prey were digested completely; all that remained of the prey were their beaks. Chiroteuthis calyx was the most abundant, accounting for 17.6% of the total prey biomass with an individual average weight of 275 g, followed by Histioteuthis dofleini (15.4%, 805 g), Haliphron atlanticus (11.9%, 200 g), Belonella borealis (7.8%, 135 g), Octopoteuthis sp. (6.2%, 294 g), Octopoteuthis deletron (3.0%, 278 g), Japetella diaphana (1.8%, 50 g), Histioteuthis inermis (1.7%, 100 g), Gonatus berryi (1.3%, 85 g) and Gonatus onyx (1.1%, 45 g); the remaining 11 species each composed less than 1% of the total prey biomass. Four large beaks of cirrate octopuses (two from Stauroteuthis sp. and two from Cirrothauma sp.) measuring 9.4–11.3 mm LRL were collected but no regression formulae were available. Due to their huge size, we estimated the body weight of each to be 1000 g; they formed 4.3% of the total prey biomass.
Among the fish prey, Brama cf. japonica, Thunnus cf. alalunga, Alepisaurus ferox, shark unid. and Onchorhynchus sp. occurred as half-digested intact bodies or chopped flesh, and actual wet-weight measurements were made. The total prey biomass included two pieces of a chopped body from a salmonid fish (2.8% of total prey biomass), two pieces of chopped shark flesh (1.1%), two individuals of half-digested A. ferox and B. japonica (0.8% and 0.7%, respectively) and two pieces of chopped caudal fin of T. cf. alalunga (0.6%). Each of the myctophids and gonostomatids contributed less than 0.5% of the total prey biomass.
Salmon sharks
Cephalopods were the dominant prey over the 2 years, accounting for nearly 96% of the total prey biomass (Table 4). Gonatopsis borealis was the most abundant, accounting for 60.2% of the total prey biomass with an average weight of 271 g, followed by Onychoteuthis borealijaponica (19.1%, 236 g), and Berryteuthis anonychus (1.2%, 20 g). A large beak of the pelagic octopus, Ocythoe tuberculata, measuring 4.2 mm LRL was found, but could not be used to estimate the intact weight. Females of O. tuberculata grow to 30 cm mantle length and we estimated the intact individual weight to be 200 g which formed 1.3% of the total prey biomass. Each of the remaining six species contributed less than 1%.
Six otoliths of unidentified Paralepididae fishes and 16 pieces of fish flesh were found. Paralepids are small fishes that grow to about 15 cm TL, and we estimated the intact weight of each to be 15 g; they contributed 0.6% of the total prey biomass. The 16 pieces of fish flesh formed 3.1% of the total prey mass.
Relative importance of prey
For blue sharks, the IRI was highest for Chiroteuthis calyx (689), followed by Haliphron atlanticus (323), Electrona risso (262), Histioteuthis dofleini (245), Belonella borealis (172) and Japetella diaphana (111). For salmon sharks, the IRI was by far the highest for Gonatopsis borealis (3546), followed by Onychoteuthis borealijaponica (787), Berryteuthis anonychus (189), lancet fishes (122) and Gonatus spp. (92). There were no overlaps in the prey species with high IRI values between the two sharks (Fig. 7).
Discussion
Sharks in the present material
The blue sharks in the present study had a size range of 50–175 cm in total length with a mode at about 80–110 cm. According to Nakano (1994) and Nakano and Nagasawa (1996), the sharks we collected in the present study were mainly young and sub-adults, which were presumably spending their nursery period in the study area. According to Sano (1960, 1962), salmon sharks are distributed widely in subarctic waters, including the Bering Sea, and in the transition waters in the western North Pacific from April to August. The salmon sharks in the present study had a size range of 69–157 cm body length and are considered to be mainly young and sub-adults. They presumably were also using the study area as a nursery ground.
Volume of stomach contents
The maximum stomach-content volume recorded for a blue shark was 1,705 g in an individual measuring 141 cm in body length. The estimated body weight of this individual based on the body weight–length equation for females proposed by Nakano (1994) is 28.1 kg, giving a stomach content index (SCI) of 6.1%. The maximum stomach-content volume of a salmon shark measuring 130 cm in body length was 1,465 g. The estimated body weight of this individual based on the equation in Tanaka (1980) is 47.9 kg, so the SCI was 3.1%. Stomachs having large volume were usually filled with digestive fluid and/or seawater. These sharks may have swallowed seawater when they were entangled in gillnets; consequently the volume of fluid in the stomach may not reflect the volume of food intake.
Feeding habits of blue sharks
Blue sharks fed on a high diversity of prey species. Cephalopods and fishes each composed nearly half of the total number of prey, but cephalopods contributed over 90% of the total prey biomass. There are differences in the speed of digestion of fish and cephalopods tissues as well as in the residual periods that otoliths and beaks remain in the stomachs; fish otoliths are generally much smaller and disappear more rapidly from the stomach contents than cephalopod beaks, which may have resulted in an underestimation of the importance of fish prey.
All that remained of the cephalopod prey in the stomachs were their beaks, indicating that they were fed on long before the sharks were entangled in gillnets at night. Cephalopod prey with large IRI values were relatively large, semi-gelatinous, inactive meso- to bathypelagic inhabitants (Nesis 1982). In large trawl surveys conducted off Sanriku, northern Japan, Chiroteuthis calyx was reported from 1,000 m depth, Belonella borealis (listed as Taonius pavo) occurred at 1,000–1,500 m depth andHistioteuthis dofleini occurred at 400–1,500 m depth (Kubodera 1996; Watanabe et al. 2006). It is noteworthy that several fairly large beaks identified to the cirrate octopus, Stauroteuthis sp. and Cirrothauma sp., were found in the stomachs. Roper and Brundage (1972) reported that all cirrate octopods, except the Opisthoteuthidae, are considered to be benthopelagic having gelatinous bodies that are neutrally buoyant, which allow them to “float” above the bottom at depths below 2,300 to 2,500 m. They also pointed out that while the sizes of cirrate octopods that have been reported in the literature is rather small, underwater photographs show that individuals larger than 1 m in total length are relatively common. This suggests that blue sharks might be able to swim to depths over several thousand meters to forage.
For fish prey, small mesopelagic myctophids and gonostomatids were numerically important and all of them occurred as otoliths, except for individuals of Lampanyctus alatus, which were half digested. On the other hand, the large pelagic fishes occurred as chunks of flesh, except for two individuals of Brama cf. japonica and Alepisaurus ferox, which were intact. Thunnus cf. alalunga was identified based on its caudal fin; a shark was identified based on its skin, and Oncorhynchus sp. was identified based on its scales and the abdominal portion of body. This indicates that these pelagic fishes were fed on just before the sharks were entangled in gillnets at night, while the small myctophids and gonostomatids were fed on long enough before capture to allow them to be digested, leaving only the otoliths. The large pelagic fishes may have been bitten and cut into chunks of flesh by the sharks when they were entangled in gillnets.
Carey and Scharold (1990) reported that the swimming depth of blue sharks observed by acoustic telemetry in the Atlantic Ocean was between the surface and the thermocline (about 80–150 m depth) during the night, and that the sharks made several rapid dives to depths of 300–600 m depth during the day. Nakano and Seki (2003) mentioned that the pattern of vertical movement of blue sharks was consistent with a visual predator that might detect the silhouette of prey against light from the surface. This could be true for small mesopelagic prey but sharks would need different tactics for detecting large semi-gelatinous cephalopods in the deep sea.
Our data suggest that blue sharks, especially young and sub-adults that migrate to the transition zone in April and May feed mainly on large, semi-gelatinous, neutrally buoyant, inactive meso- to bathypelagic cephalopods presumably during the day when they dive to the deep. Deep, oceanic, large gelatinous squids (cranchiids and histioteuthids) are also preyed by blue sharks in the Bay of Biscay (Clarke and Stevens 1987).
Feeding habits of salmon sharks
Salmon sharks fed on a relatively low diversity of prey species. For the same reasons mentioned for blue sharks, the importance of fish in the prey might be underestimated.
The salmon sharks fed extensively on Gonatopsis borealis, followed by Onychoteuthis borealijaponica; both are epi- to mesopelagic, muscular, medium-sized squids that are abundant in the transitional waters in the North Pacific (Murata et al. 1976; Naito et al. 1977; Kubodera et al. 1983). The third most important prey item was Berryteuthis anonychus, a small epi- to mesopelagic gonatid squid distributed in the subarctic and transition waters of the North Pacific (Okutani et al. 1988; Nesis 1997; Bower et al. 2002).
All the cephalopods were identified based on their beaks, though the degree of digestion varied among the specimens. The stomach of a 130 cm TL shark contained seven lower beaks of Gonatopsis borealis and one lower beak of Onychoteuthis borealijaponica in the same state of digestion. The total estimated wet weight of all squid prey was 3,004 g (Table 5). The estimated body weight of a 130 cm BL shark is 47.9 kg (Tanaka 1980), so a salmon shark can ingest prey of 6.3% of its weight in a short feeding period.
Sano (1960) mentioned that salmon sharks are opportunistic feeders and stressed that large and mature sharks found near the Aleutians from May through July feed extensively on salmonid fishes (Oncorhyncus nerka, O. keta, O. gorbuscha) of commercial size (40–50 cm FL). Later, Sano (1962) recognized that the dominant prey of salmon sharks varies temporally and geographically; sockeye salmon dominated in 1959, while in 1960, squids dominated from May to August near the Aleutians Islands.
Our study suggests that salmon sharks, especially young and sub-adults that migrate to the transition zone in April and May, feed largely on epi- to mesopelagic, active, muscular, medium sized squids which might be very abundant and suitable prey for salmon sharks in this region during spring.
Conclusion
Although both blue sharks and salmon sharks have sympatric distributions in the transition zone of the western North Pacific in spring, they have different feeding habits and strategies that reduce competition for food resources. Judging from the potential biomass of blue sharks in the study area (Nakano 1994) and our results, there should be a huge biomass of meso- and bathypelagic large, semi-gelatinous cephalopods occurring in this region. Blue sharks in this region have adopted a feeding strategy similar to that of sperm whales, diving deeply during the day for hunting non-active sizable cephalopods in the deep (Clarke 1980; Okutani and Satake 1978; Amano and Yoshioka 2003).
References
Amaoka K, Nakaya K, Araya H, Yasui T (eds) (1983) Fishes from the north-eastern Sea of Japan and the Okhotsku Sea off Hokkaido. Japan Fisheries Resource Conservation Associaiton, Tokyo, 371 pp
Amano M, Yoshioka M (2003) Diving behavior of a sperm whale revealed by the suction cup attached TDR tag. Mar Ecol Prog Ser 258:291–295
Bower JR, Murphy J, Sato Y (2002) Latitudinal gradients in body size and maturation of Berryteuthis anonychus (Cephalopoda: Gonatidae) in the northeast Pacific. Veliger 45:309–315
Carey FG, Scharold JV (1990) Movements of blue shark (Prionace glauca) in depth and course. Mar Biol 106:329–342
Clarke MR (1980) Cephalopoda in the diet of sperm whales of the southern hemisphere and their bearing on sperm whale biology. Disc Rep XXXVII:324
Clarke MR (1986) A handbook for the identification of cephalopod beaks. Clarendon Press, Oxford, pp 273
Clarke MR, Stevens JD (1987) Cephalopods, blue shark and migration. J Mar Biol Assoc UK 54:949–957
Compagno LJV (1984) FAO species catalog Vol.4: sharks of the world; Part 2-Carcharhiniformes. Fisheries Synopsis No.125., FAO, Rome, Italy, 655 pp
Favorite F, Dodimead AJ, Nasu K (1976) Oceanography of the subarctic Pacific region, 1960–71. Bull Int Natl N Pac Fish Comm 33:1–187
Hart JL (1973) Pacific fishes of Canada. Fish Res Board Canada Bull 180:740
Harvey JT (1989) Food habits, seasonal abundance, size, and sex of the blue shark, Prionace glauca, in Monterey Bay, California. Calif Fish Game 75(1):33–44
Kubodera T (1982) Ecological studies of pelagic squids in the subarctic Pacific region. PhD thesis, Hokkaido University, Hakodate, 255 pp (in Japanese)
Kubodera T (1996) Cephalopod fauna off Sanriku and Joban districts, northeastern Japan. Mem Natn Sci Mus Tokyo 29:187–207
Kubodera T, Ohizumi H (2001) Manual for the identification of cephalopod beaks in the western North Pacific National Science Museum, Tokyo Available from the Internet URL http://www.research.kahaku.go.jp/zoology/Beak-E/index.htm
Kubodera T, Furuhashi M (1987) A manual for identification of myctophid fishes and squids in the stomach contents. Report on the development of ecosystem modeling in the northern North Pacific, supplement. Japanese Fisheries Agency, Tokyo, 65 pp (in Japanese)
Kubodera T, Shimazaki K (1989) Cephalopods from the stomach contents of the pomfret (Brama japonica Hilgendorf) caught in surface gillnets in the northern North Pacific. J Cephalopod Biol 1:71–83
Kubodera T, Pearcy WG, Murakami K, Kobayashi T, Nakata J, Mishima S (1983) Distribution and abundance of squids caught in surface gillnets in the subarctic Pacific, 1977–1981. Mem Fac Fish Hokkaido Univ 30:1–49
LeBrasseur RJ (1964) Stomach contents of blue shark (Prionace glauca L.) taken in the Gulf of Alaska. J Fish Res Bd Can 21(4):861–862
McKinnell S, Seki M (1998) Shark bycatch in Japanese high seas squid driftnet fishery in the North Pacific Ocean. Fish Res 39:127–138
Murata M, Ishii M, Araya H (1976) The distribution of the oceanic squids, Ommastrephes bartrami (Lesueur), Onychoteuthis borealijaponicus Okada, Gonatopsis borealis Sasaki and Todarodes pacificus Steenstrup in the Pacific Ocean off north eastern Japan. Bull Hokkaido Reg Fish Res Lab 41:1–29 (in Japanese with English summary)
Naito M, Murakami K, Kobayashi T, Nakayama N, Ogasawara J (1977) Distribution and migration of oceanic squids (Ommastrephes bartramii, Onychoteuthis borealijaponica, Berryteuthis magister and Gonatopusis borealis) in the western subarctic Pacific region. Spec Vol Res Inst North Pac Fish Hokkaido Univ Fac Fish 321–337(in Japanese with English abstract)
Nakano H (1994) Age, reproduction and migration of blue shark in the North Pacific Ocean. Bull Nat Res Inst Far Seas Fish 31:141–219 (in Japanese with English abstract and summary)
Nakano N, Nagasawa K (1996) Distribution of pelagic elasmobranches caught by salmon research gillnets in the North Pacific. Fish Sci 62(6):860–865
Nakano H Seki MP (2003) Synopsis of biological data on the blue shark, Prionace glauca Linnaeus. Bull Fish Res Agen 6:18–55
Nesis K (1982) Cephalopods of the World. In: Burgess LA (eds) Translated from Russian by LevitovBS. T.F.H. Publications Inc. 351 pp
Nesis K (1997) Gonatid squids in the subarctic North Pacific: ecology, biogeography, niche diversity and role in the ecosystem. Adv Mar Biol 32:243–324
Ohizumi H, Watanabe H, Moku M, Kawahara S (2001) Species identification for otoliths of myctophid fishes in the western North Pacific. Aquabiology 23(6):626–637 (in Japanese with English abstract)
Okutani T, Kubodera T, Jefferts K (1988) Diversity, distribution and ecology of gonatid squids in thesubarctic Paicifc: a review. Bull Ocean Res Inst Univ of Tokyo 26(Part I):159–192
Okutani T, Satake Y (1978) Squids in the diet of 38 sperm whales caught in the Pacific waters off northeastern Honshu, Japan, February 1977. Bull Tokai Reg Fish Res Lab 93:13–27
Pearcy WG (1991) Biology of the transition region. NOAA Tech Rep 105:39–55
Pinkas L, Oliphant MS, Iverson ILK (1971) Food habits of albacore, bluefin tuna, and bonito in California waters. Calif Fish Game 152:1–105
Roper CFE, Brundage WL (1972) Cirrate octopods with associated deep-sea organisms: new biological data based on deep benthic photographs (Cephalopoda). Smith Contr Zool 121:1–46
Sano O (1960) The investigation of salmon shark as a predator on salmon in the North Pacific, 1959. Bull Hokkaido Reg Fish Res Lab 22:68–82 (in Japanese with English summary)
Sano O (1962) The investigation of salmon shark as a predator on salmon in the North Pacific, 1960. Bull Hokkaido Reg Fish Res Lab 24:148–162 (in Japanese with English summary)
Seki MP (1993) The role of the neon flying Squid, Ommastrephes bartrami, in the North Pacific Pelagic food web. Bull INPFC 53(2):207–215
Stevens JD (1973) Stomach contents of the blue shark(Prionace Glauca L.) off south-west England. J Mar Biol Assoc UK 53:357–361
Strasburg DW (1958) Distribution, abundance, and habits of pelagic sharks in the Central Pacific Ocean. Fish Bull 138:335–361
Tanaka A (1980) Biological research on salmon sharks, Lamna ditropis, in the northwestern North Pacific. In: Report on development of sharks: new fisheries resources (North Pacific) in 1979. Japan Fisheries Resource Conservation Associaiton, Tokyo, pp 59–84 (In Japanese with English abstract)
Tricas TC (1979) Relationship of the blue shark,Prionace Glauca, and its prey species near Santa Catalina Island. Calif Fish Bull 77(1):175–182
Watanabe H, Kubodera T, Ichii T, Kawahara S (2004) Feeding habits of neon flying squid Ommastraphes bartramii in the transitional region of the central North Pacific. Mar Ecol Prog Ser 266:173–184
Watanabe H, Kubodera T, Kawahara S (2003) Feeding habits of Pacific pomfret Brama japonica in the transition zone of the central North Pacific. Fish Sci 69:269–276
Watanabe H, Kubodera T, Moku M, Kawaguchi K (2006) Diel vertical migration of squid in the warm core ring and cold water masses in the transition region of the western North Pacific. Mar Ecol Prog Ser 315:187–197
Acknowledgments
We are grateful to the captains, officers, crew, and researchers of the research vessels Kaiun-Maru of the Aomori Prefectural Fisheries Experiment Station and Wakatori-Maru of the Sakai Fisheries Senior High School in Tottori Prefecture for their help sampling under severe sea conditions. We also thank Mr. S. Kawahara and N. Baba of the Japanese Fisheries Agency (JFA) for allowing us to examine stomach contents of sharks. Dr. H. Nakano, National Research Institute of the JFA provided information on blue sharks in the North Pacific. We also thank Dr. J. Bower, Hokkaido University, for his proof reading and comments on a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kubodera, T., Watanabe, H. & Ichii, T. Feeding habits of the blue shark, Prionace glauca, and salmon shark, Lamna ditropis, in the transition region of the Western North Pacific. Rev Fish Biol Fisheries 17, 111–124 (2007). https://doi.org/10.1007/s11160-006-9020-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11160-006-9020-z