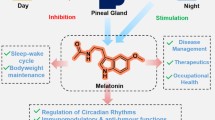
Abstract
The last four decades, we assist to an increasing scientific interest on melatonin, a circadian hormone, a metabolic regulator which influences not only plants’ metabolism and their defense against pathogens but mostly the animals and humans’ metabolic pathways, their response to circadian disruption, stress and burnout syndrome. In humans, as a hormonal regulator, produced in the pineal grand as well in mitochondria, melatonin is involved in different, complex intracellular signaling pathways, with antioxidant and immune stimulating effects, proving to act as a circadian synchronizer, as a preventive and therapeutic agent in many degenerative diseases, and especially in hormone-dependent cancers. Preclinical or clinical studies showed recently the mechanisms involved in regulating the cellular activity, its role in aging and circadian disturbances and impact on degenerative diseases. Melatonin proved to have an anti-inflammatory, antiapoptotic and powerful antioxidant effect by subtle mechanisms in mitochondrial metabolic pathways. This overview includes recent and relevant literature data related to the impact of endogenous and exogeneous melatonin on the prevention of cancer progression and treatment of various degenerative diseases. Metabolomics, an emerging new omics’ technology, based on high performance liquid chromatography coupled with mass spectrometry is presented as an encouraging technique to fingerprint and realize a precise evaluation and monitoring of the turnover of melatonin and its metabolites in different pathological circumstances.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Melatonin (N-acetyl-5-methoxytryptamine) is an indole derivative discovered in 1958, a natural neuro-transmitter acting as a circadian hormone is primarily produced at night and released by the pineal gland, which regulates the sleep–wake cycle. For decades melatonin was considered as an animal hormone involved in synchronizing the circadian rhythm, blood pressure regulation and seasonal reproduction [1, 2], but later, in the 90’s was found also in plants, being now widely recognized as a universal amphiphilic molecule, able to penetrate and influence all cells’ activity [3]. A comparative picture of the melatonin biosynthesis pathway in plants and animals in humans is presented in Fig. 1.
Tryptophan, an essential amino acid is produced de novo in plants through the specific shikimate pathway, a seven step biosynthesis which converts shikimic acid to chorismate, the precursor of some aromatic amino acids, including tryptophan [4] . In animals, tryptophan is released by protein degradation and acquired from diet. Serotonin and then melatonin are synthesized in both plants and animals, but on different mechanisms, including as intermediates tryptamine and 5-methoxy tryptamine or 5-Hydroxy Tryptophan and Acetyl serotonin, respectively, as presented in Fig. 1. These pathways are regulated by specific enzymes located in chloroplasts and mitochondria [5, 6].
Melatonin synthesized in plants proved to have direct effects on vertebrate’s plasma membranes binding to specific receptors [6]. Melatonin biosynthesis in animals and humans take place either in pineal grand, its biosynthetic pathways being activated by darkness and depressed by light, but also in mitochondria [7, 8]. The release of melatonin follows a circadian rhythm generated by the suprachiasmatic nuclei in response to daylight alterations, therefore this internal “chronobiotic” substance normalizes the biological rhythms and adjusts the timing of other biomolecules (hormones, neurotransmitters, etc.) and processes. Comparative studies recently published evaluated the key-mechanisms of melatonin roles and functions, specific to plant and animal metabolic pathways [4, 9].
2 Melatonin roles and functions in plants
Melatonin has been found in many plants, in leaves, stems, roots, fruits and seeds, in different proportions [3, 10,11,12]. Its occurrence, distribution and bioavailability, as well its potential for human health differ not only among species (medicinal or food plants), but also between varieties of the same species depending on the growing conditions [3, 5].
Although the role for melatonin as a plant hormone has not been clearly established, its involvement in processes such as growth regulation and photosynthesis is well established. Melatonin performs important roles in plants as a metabolic regulator, as well as environmental stress protector, improving plants’ defense against pathogens. Its synthesis is upregulated in plants after exposure to biological stress (e.g. viral, fungal infections) or nonbiological stress (e.g. extreme temperatures, toxins, increased soil salinity, drought) [13,14,15]. Its bactericidal capacity was proved against Mycobacterium tuberculosis, as well as multidrug-resistant bacteria under in vitro conditions. In plant–fungi interaction models, melatonin was found to play a key role in plant resistance to Botrytis cinerea, and to reduce the stress tolerance of Phytophthora infestans [16]. The exposure of plants to abiotic and biotic stresses causes the increase of endogenous melatonin levels and induces the mitogen-activated protein kinase cascade and associated defense responses.
Melatonin-related growth and defense responses of plants are complex and involve specific signaling pathways and molecules, the most important being the amphiphilic nitric oxide (NO) that crosses cell membranes, induces a variety of rapid physiological reactions, e.g. NO-induced S-nitrosylation involved as a defense response, plant growth regulation and oxidative homeostasis. The exogenous melatonin induces increase of endogenous NO and up-regulate the defense-related transcription factors, resulting in enhanced stress resistance, as well promotes sugar and glycerol production, leading to increased levels of salicylic acid and NO. Melatonin and NO in plants can function cooperatively to promote lateral root growth, delay aging, and ameliorate iron deficiency [17]. Different molecular tools to study melatonin pathways and actions were reviewed, many of its effects being mediated by the melatonin receptors, but also by its antioxidant capacity [16, 18,19,20]. Therefore, melatonin is an ecofriendly compound and an economical alternative to be used to protect plants against pathogens.
A synthetic presentation of melatonin functions (as growth regulator and protective molecule against biotic and abiotic stress) and its effects in plants is presented in Fig. 2.
Many foods contain melatonin, beside other neuroactive compounds, from gamma-aminobutyric acid serotonin, to kynurenine, kynurenic acid, dopamine, norepinephrine, histamine, tryptamine, tyramine. Non-processed and fermented foods (e.g human milk) contain some of these compounds showing also antioxidant capacity [21, 22], which can ameliorate human health. Such neuroactive agents from foods are also formed by human gut microbiota, a very attractive topic which may demonstrate the interaction between microbiota and mental health as well the effects of diet on the formation of microbial metabolites, including neuroactive compounds [23]. Recently, a systematic review on the ability of food sources of melatonin to improve sleep quality was published, based on a literature search since 2005 [24].
3 Melatonin as a metabolic regulator in animals and humans
Different physiological functions have been attributed to melatonin secreted during the night, acting as a circadian rhythm synchronizer, as a regulator of the animal circadian and seasonal reproduction [7, 25,26,27], a regulator of oxidative stress either by direct detoxification or indirectly by inhibition of prooxidative enzymes and stimulation of antioxidant enzymes [19, 20, 28, 29].
Comparatively, the melatonin functions in animals and humans (as metabolic regulator and protective molecule against oxidative stress) is presented in Fig. 2, identifying some common roles and functions in both plant and animal kingdoms.
Once produced, melatonin is quickly released from the pineal gland into the bloodstream and into the cerebrospinal fluid (CSF), where its level rises during the night. This circadian “message” is essential for organisms, the disruption of its cyclic signal contributing to its pathophysiology.
Melatonin has two functional groups responsible for its amphiphilicity, the 5-methoxy group and the N-acetyl side chain, facilitating its entrance in all cells, influencing the functionality of many organs and tissues. More recent studies showed that mitochondria are also sites of melatonin synthesis [30].
The mechanisms of melatonin action are related either to its chemical properties, as scavenger of free radicals (1) or hormonal regulator of circadian rhythm (2). or on its capacity to bind protein targets including the immune-competent ones (3)
-
1.
Melatonin was first reported as a potent antioxidant and a free radical scavenger [18] against oxygen and nitrogen reactive species in vitro [28] being more effective than vitamin E [29]. Due to its electron-rich aromatic indole ring, melatonin is a potent electron donor that can reduce significantly the oxidative stress and acts as a direct free radical scavenger within mitochondria, or indirectly, by promoting the expression of antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, glutathione reductase and catalase [8, 31]. The deficiency in melatonin due to light exposure after darkness onset may increase the amount of oxidative stress, since many diseases are related to elevated oxidative damage of mitochondria [32, 33]. Melatonin occurs at high concentrations within mitochondria, exceeding its plasma concentration [20, 30, 34] and is metabolized in the liver by a cytochrome P450 enzyme to 6-hydroxymelatonin, its metabolites being conjugated with sulfuric acid or glucuronic acid for urine excretion. Only 5% of melatonin is excreted in the urine as the unchanged drug [1, 35]. Both animal and human studies have shown that melatonin may act as a radioprotector against ionizing radiation-induced cellular damage by scavenging reactive oxygen species generated during exposure [36,37,38,39].
-
2.
The role of endogenous melatonin in circadian rhythm disturbances and sleep disorders is well established, and has a close association with the endogenous circadian component of the sleep rhythm. Therefore, melatonin is an internal sleep ‘facilitator’ in humans, useful in the treatment of insomnia in elderly and depressive individuals for a readjustment of circadian rhythms. Pinealocytes export melatonin to blood stream and in brain, but can be found also in retina, gastrointestinal tract, bone marrow, skin and other tissues [26, 40].
-
3.
More than 15 proteins, including receptors, enzymes, pores and transporters or other proteins (calmodulin, tubulin, calreticulin), have been suggested to interact with melatonin at sub-millimolar concentrations. The proteins that transport melatonin across membranes include mainly the glucose transporters (GLUT1) as well proton-driven oligopeptides PEPT1/2 [41]. In humans, melatonin is a full agonist of Gprotein-coupled receptors: melatonin receptor 1 (MT1, with picomolar binding affinity) and melatonin receptor 2 (MT2, with nanomolar binding affinity) [41, 42]. Recently, extensive information was reviewed on proposed melatonin targets, with guidelines concerning future experimental methodology, physiological relevance, and independent replication [40, 42, 43]. During the night, the activation of the beta-1 adrenergic receptor by norepinephrine in pinealocytes determine the elevation of the intracellular cAMP concentration and activates the cAMP-dependent protein kinase A which stimulates the melatonin synthesis. By exposure to (day)light, the noradrenergic stimulation stops, the production of melatonin being re-started in the evening (dim-light onset).
Melatonin interacts with the immune system, recently an extensive review demonstrating is role in inflammation [42]. An anti-inflammatory effect seems to be the most relevant, being related to receptors MT1 and MT2 expressed in immunocompetent cells and enhancement of cytokine production, for fighting viral and bacterial infections [44,45,46].
4 Melatonin protection against aging and circadian disorders
Sleep and aging processes are remarkably parallel, with a metabolic similarity and interaction, as shown by behavioral studies. Changes in sleep behavior affect neurocognitive phenotypes important in aging, such as learning and memory, although the underlying connections are largely unknown. The importance of melatonin in aging and in age-related diseases is underlined by the reduced production of this potent antioxidant in the pineal gland [47].
The circadian rhythm disorders have been classically associated with disorders of abnormal timing of the sleep-wake cycle. The day/night melatonin rhythm provides a “cellular clock” information, as it was observed since three decades ago [47, 48]. Blue light, at around 460–480 nm, suppresses melatonin biosynthesis, proportional to light intensity and length of exposure while wavelengths higher than 530 nm do not suppress melatonin in bright-light conditions. It was shown that wearing glasses that block blue light before bedtime may decrease melatonin loss and promotes sleepiness [49].
A mutual relationship between circadian oscillators and melatonin secretion, the readjustment of rhythms by melatonin and its synthetic analogs, the consequences for circadian dependent disorders and limits of treatment were previously reviewed [50, 51]. However, circadian dysfunction can play a role in a wider range of pathology, ranging from the increased risk for cardiometabolic diseases to malignancy in shift workers, supporting the concept of “circadian medicine” [52].
The literature data about the benefits of melatonin for circadian disruptions are still unclear. In Europe it is used for short-term treatment of insomnia in people who are over 55, while the Agency for Healthcare Research and Quality stated in 2015 that evidence of benefit in the general population is not yet established [53]. Recent reviews reported modest effects of melatonin until onset of sleep and delayed sleep phase syndrome, without significant difference in total sleep time [54,55,56].
Melatonin appears to have limited use against the sleep problems of people who have shift work, suggesting that it increases the length of sleeping time [57] causes very few side effects as tested in the short term (up to 3 months, at low doses) with or without adverse effects in several clinical trials and comparative trials [58, 59]. Although not recommended for long term use, low-dose melatonin was generally safer, and a better alternative to other sleeping medication which can’t be used for an extended period of time [59].
Supplements containing melatonin might improve sleep in people with autism, especially children, improving the sleep duration, sleep onset latency, and night-time awakening [60, 61]. Recent studies suggest that melatonin may be an efficient and safe treatment for insomnia and depression of people and children with attention deficiency and hyperactivity disorders, but larger and longer studies are needed to establish long-term safety and optimal dosage [62, 63]. Emerging metabolic phenotyping technology, can help finding responsible markers which reflect the effects of melatonin on sleep, aging and progression of diseases [50, 64].
Recently, the process by which light inhibits the melatonin production and disturbs circadian rhythms, at chronic night shift workers, associated with cancer risk was reviewed [65]. An association between the excessive light exposure at night and the prevalence of degenerative diseases, including cancer, especially female breast and male prostate cancer was reported. There were identified at least two obvious physiological consequences of nighttime light exposure, namely a reduction in circulating melatonin levels and the chrono disruption, both perturbations aggravating the tumor growth in experimental animals. While an increased tumor frequency has been proved to be a consequence of chrono disruption, cancer does not represent the only pathophysiological change associated with melatonin offset and circadian disruptions.
5 Melatonin and degenerative diseases
5.1 Mechanisms and effects
Degenerative diseases are a consequence of a continuous process of cells dysfunction, affecting tissues or organs, which deteriorate increasingly over time. Such diseases are the neurodegenerative ones (e.g. Parkinson and Alzheimer diseases), cardiovascular diseases, metabolic syndromes (e.g. diabetes type 2) and neoplastic pathology (e.g. cancers). Melatonin secretion by the pineal gland progressively declines with age, in circadian disorders but especially in stress-related pathologies, inducing degenerative diseases.
Figure 3 shows the diagrammatic representation of main mechanisms of melatonin in cells, at cytoplasmic, mitochondria and nuclear level. Such mechanisms can explain the key role of melatonin as a master, pleiotropic regulator in relation to apoptosis, normal/cancer cell metabolism, immune defense and inflammation, autophagy, etc..
Diagrammatic representation of main mechanisms of melatonin in cells, at cytoplasmic, mitochondrial and nuclear level. Details about signaling steps are presented in the text. Abbreviations: MT1, MT2- melatonin specific plasma membrane receptors; MT3 – quinine reductase acting as cytoplasmic; GLUT1, PEPT1/2 – transport carriers crossing cell membrane; RZR/ROR- nuclear receptors. REV-ERB - Per1,2 – periodicity-related proteins; PLS- Phospholipase C; DAG Diacylglycerol; IP3 – Inositoltriphosphate; AC- Adenylyl cyclase; GC- Guanylyl cyclase; cAMP- cyclic AMP, cGMP – cyclic GMP; AcCoA-Acetyl-Coenzyme A; Pyr- pyruvate; PDC- pyruvate dehydrogenase complex; PDK-Pyruvate dehydrogenase kinase; BMALI/CLOCK –heterodimers which regulate SIRT(sirtuin) expression; c-Myc, cyclin D, CDK4/6 - protooncogenes involved in cell cycle and proliferation
The molecular events related to Light-at-Night whereby melatonin counteracts cancer progression are vast and complex and include different mechanisms, inducing instability at genomic level via Clock genes [66], (1), specific intracellular targets via transmembrane MT1/MT2 receptors or cytosolic MT3 or nuclear RZR/ROR receptors with impact on cell proliferation (2) [42], changes in tumor glucose metabolism (3) [67,68,69] or indirectly via T cell signaling (4) [70].
-
1.
Circadian rhythms control most biological processes and their disruption or an aberrant function in the expression of clock genes are associated with a number of cancers including some hormone-dependent and independent cancers. The melatonin production and secretion oscillates through the light:dark cycle and is related to the circadian machinery genes via proteasome inhibition and sirtuins to alter indirectly the clock genes in cancer. Concomitantly, melatonin exerts pro-apoptotic, anti-proliferative and pro-oxidative effects, metabolic shifting, reduction in neovasculogenesis and inflammation, and restores chemosensitivity of cancer cells. Finally, melatonin improves the life quality of patients. The heterodimeric clock-proteins system BMAL1/CLOCK, responsible for the upregulation of SIRT expression which is highly active in cancer cell proliferation is affected by melatonin which inhibits the nuclear receptor ROR expression of this and repress the cell proliferation via c-myc, Cyclin D and CDK4/6 transcription factors arrest the cell cycle in G0 phase and stimulates DNA repair and apoptosis. Meanwhile melatonin inactivates the proteasome complex, by complex mechanisms, well described recently [66]. A recent review [66, 71] focuses on the main functions of melatonin on clock genes and circadian-controlled genes, the ways of melatonin to dysregulate the expression of such genes in many cancer types such as breast, prostate, liver, and colon cancers, leukemia and melanoma.
-
2.
Pineal melatonin is highly permeable to cells, its transport across plasma membrane being mediated by GLUT1 or PEPT1/2 carriers or by specific receptors MT1 and MT2, as well is targeting the cytoplasmic MT3 receptor (a quinone reductase) and nuclear receptors RZR-ROR. The intracellular signaling through MT1and MT2 receptors is transmited via G proteins which activate Phospholipase C (PLC) and subsequent activation of IP3 and DAG pathways and inhibit cAMP or cGMP.Through the inhibition of inflammasome activation, melatonin exerts anti inflamatory effects, blocking the caspase 1 cleavage, cytokines release and nuclear NF-kB translocation oncostatic effects in various cancer models. Depending on the cellular activity, the receptors can form homo- or heteromeric complexes to promote a diversity of signaling capacity [42].
-
3.
Melatonin acts as an inhibitor of cytosolic glycolysis in cancer cells by downregulation of the enzyme pyruvate dehydrogenase complex (PDC) which converts pyruvate to acetyl CoA in the mitochondria,. therefore, inhibits the proliferative activity of cancer cells, reduces their metastatic potential and induces apoptosis. Whereas melatonin is synthesized in the mitochondria of normal cells, the inhibition of its synthesis in cancer cell mitochondria is due to the depressed acetyl CoA synthesis and in parallel the oxidative phosphorylation and ATP production is compromised. Therefore, the ability of melatonin to switch glucose oxidation from the cytosol to the mitochondria may explain why and how tumors resistant to conventional chemotherapies can be re-sensitized by external melatonin. Such metabolic changes in mitochondrial functions induces tumor cell apoptosis and via PDC induce the glycolytic shift from mitochondria to cytosol (Warburg effect) [69]
-
4.
Meanwhile melatonin significantly influences T cell-mediated immune responses, which are crucial to protect mammals against cancers and infections [70], melatonin is highly effective in modulating T cell activation and differentiation, especially for Th17 and Treg cells, and also memory T cells. Mechanistically, the influence of melatonin in T cell biology is associated with membrane and nuclear receptors as well as receptor independent pathways [70, 72].
Recent findings in vitro and in vivo have reported that melatonin regulates also autophagy-related processes due to its role as a metabolic regulator and mitochondrial protector against oxidative damage in the electron transport chain and mitochondrial DNA, finalized by increased ATP production. Melatonin can regulate autophagy both directly by improving the proteolysis pathway, and indirectly by reduction of excessive oxidative stress, resulting in a reduction of misfolded proteins [73]. Beside these mechanisms, melatonin modulates the linoleic acid uptake, the cAMP levels, cellular dynamics and other intracellular events [74].
Since melatonin mechanisms are overcrossing glucose metabolism; therefore, melatonin vs glycemic challenge should be considered to understand the mechanisms and therapeutic opportunities to develop melatonin-based therapies for type 2 diabetes. [75].
New findings have been reported related to the involvement of melatonin in degenerative diseases, including different types of cancer, trying to elucidate the potential mechanisms of the metabolic and molecular events associated with melatonin’s role and effects.
Table 1 includes a selection of new and relevant data related to melatonin involvement and effects on degenerative diseases.
The light suppression of human nocturnal melatonin levels may be consequential in the cancer cell proliferation and tumor growth as suggested by a large number of experimental studies. Since 2007 the circadian disruption and melatonin suppression is considered to be associated with a generalized elevation of all cancer subtypes, according to IARC (International Agency for Cancer Research) of the World Health Organization, the shift work that involves circadian disruption being classified in the Group 2A of risks (possibly carcinogenic) [92, 93].
Within the last few decades, melatonin has increasingly emerged in clinical oncology as a bioactive molecule with substantial anticancer properties and a pharmacological profile. Despite the substantial body of evidence linking chrono disruption and melatonin suppression to a higher prevalence of cancer, some experimental data showed that melatonin onset may act as an endogenously produced oncostatic agent [94,95,96,97,98,99]. A variety of mechanisms have been described by which a reduction of pineal or mitochondrial melatonin, levels may stimulate tumor growth [97, 100,101,102,103].
Melatonin interferes in the desmoplastic reaction by regulating the vascular endothelial growth factor and cytokine production, as shown in breast cancer (BC) [104, 105]. Melatonin inhibits the growth and sometimes the metastasis of experimental lung, liver, ovary, pituitary and prostate cancers, melanoma and leukemia [68, 106], modulating the cellular cytoskeleton [107].
Melatonin may regulate the tumor metabolism, gene expression or epigenetic modifications, impairs cell growth and inhibit angiogenesis and metastasis. In the last decade, many reports have demonstrated that exogeneous melatonin is a promising adjuvant molecule with many potential benefits when included in chemotherapy or radiotherapy protocols designed to treat endocrine-responsive tumors. Recently, a state-of-the-art review, summarized the knowledge about its oncostatic actions in hormone-dependent tumors, and the latest findings concerning its actions when administered in combination with radio- or chemotherapy in breast, prostate, and ovary cancers [108, 109].
Circadian rhythm disruption is associated especially with hormone-related cancers that are frequent in shift workers, as supported by epidemiological studies which observed the reduction in endogeneous melatonin levels by light-at-night (LAN) shifts. Melatonin regulates the production of estradiol and downregulates estrogen receptors, acting as a selective estrogen enzyme modulator via MT receptors in human ovarian cells, with a direct influence on the estrogens production. Data from several clinical trials and multiple experimental studies performed both in vivo and in vitro have documented melatonin inhibition on endocrine-dependent mammary tumors by interfering with the estrogen signaling. Similar results have been obtained in prostate, involving the androgen synthesis and modulation of androgen receptor levels and activity. According to the American Cancer Society, more than 70% of newly BC diagnosed cases are, at their initial stages, hormone-dependent. Estrogens play a crucial role in tumor genesis and progression, the alpha receptor being usually overexpressed (ER+ BC) [110, 111].
Since decades it was supposed that a diminished function of the pineal gland might promote an increase in the risk of BC, as a consequence of a prolonged time of exposure to circulating estrogens [112]. This hypothesis was based on several observations: (i) the incidence of BC is lowest in countries with low incidence of pineal calcification; (ii) patients taking chlorpromazine, a drug that increase melatonin levels, have lower rates of BC; (iii) In vitro data demonstrated the melatonins’ direct effect on BC cells. (iv) Plasma melatonin levels at night were lower in women with estrogen receptor-positive breast (ER+-BC) malignancy [110, 113, 114]. The circadian amplitude of melatonin was reduced by more than 50% in patients with BC vs patients with nonmalignant breast disease and higher melatonin levels have been found in morning urine of BC patients, suggesting a circadian disorganization [115].
It was shown years ago that melatonin inhibits ER+-BC growth and complements the oncostatic action of antiestrogen drugs (tamoxifen), suggesting an action as a “natural antiestrogen” [116, 117]. Melatonin has been shown to shift forskolin- and estrogen-induced elevation of cyclic adenosine monophosphate (cAMP) levels by 57% and 45%, respectively [102, 118]. The antiestrogenic actions of melatonin in ER+-BC cells was confirmed recently [108, 109, 119] and reflects its impact on the downregulation of the neuroendocrine/ reproductive axis via the inhibition of enzymes responsible for the conversion of adrenal androgens into estrogens and surrounding tissues. The inhibition of adenylate cyclase (AC) through binding to the melatonin receptor MT1 in malignant cells, counteracts the stimulatory effect of estradiol (See Fig.3). As a consequence, it is stimulated the cell differentiation, the immune system response and apoptosis via peroxisome proliferator-activated (PPAR) and retinoid X receptors (RXR) and downregulation of the angiogenesis and oxidative stress (See Fig.3) [119, 120].
The vascular endothelial growth factor released by ER+-BC cells binds to its receptor expressed in the membranes of adjacent endothelial cells. As a result, endothelial cells undergo proliferation, migration, and reorganization in a typical process of angiogenesis. Melatonin (1 mM) can regulate the paracrine mechanisms responsible for the interplay between mammary tumor cells and surrounding fibroblasts and endothelial cells, in experiments performed in vivo, by downregulating the levels of VEGF and by inhibiting the production of aromatase in those cell lines [104, 105].
Some recent studies have evaluated the association between residential outdoor light during sleeping time, with breast and prostate cancer, concluding that both prostate and invasive BC were associated with high exposure to outdoor light intensity and disruption of the circadian oncostatic actions of melatonin [118, 121, 122]. The majority of these observations claimed an elevated incidence of BC in women who commonly disturbed their circadian/melatonin cycles, these findings from preclinical studies being summarized in many reviews the last years [110, 119, 123,124,125]. The circadian disruption of nocturnal melatonin in nude rats bearing ER+ BC human MCF-7 xenografts induced a complete loss of tumor sensitivity to doxorubicin, indicating that chemotherapy resistance might be accentuated by a lower level of melatonin at night [126]. Melatonin alone or in combined administration seems to be an appropriate drug for the treatment of early stages of BC with documented low toxicity over a wide range of doses, in parallel with standard oncologic treatment regimens [115].
Many questions still remain concerning the mechanisms which may explain the anticancer effects of melatonin, and data from larger, randomized clinical trials are required before melatonin can be universally accepted as an anticancer drug.
5.2 Melatonin and its therapeutic potential
Extensive experimental data showed that melatonin exerts oncostatic effects throughout all stages of tumor growth, from initial cell transformation to mitigation of malignant progression and metastasis additionally, melatonin alleviating the side effects and improves the welfare of radio/chemotherapy-treated patients. Thus, the support of clinicians and oncologists for the use of melatonin in the treatment and proactive prevention of cancer is gaining importance.
The melatonin intrinsic properties, including high cell permeability, the ability to cross the blood–brain and placenta barriers, and its role as an endogenous free radical scavenger, explain its beneficial uses as an adjuvant in biomedical treatments via exogenous administration. In animal studies, melatonin treatment inhibited the incidence of tumors, induced by pineal suppression or pinealectomy [127].
Recently, new data about the therapeutic potential and clinical utilization of melatonin and its oncostatic action in humans were reviewed [128,129,130,131].
Melatonin and resveratrol treatments induced cardioprotection and diminished the cardiovascular risk factors [132], protecting also against doxorubicin-induced cardiotoxicity [133]. The effects of melatonin on metabolic syndrome were presented in a comprehensive review [134] and the tumor prevention by 9-cis-retinioic acid in animal models of mammary carcinogenesis were potentiated by melatonin [117] These results are promising, since melatonin synergizes the chemotherapy effects, allows to use lower doses with better tolerance and protects against the undesirable side effects of radiotherapy and chemotherapy, being considered a potential agent for cancer treatments [108, 109].
A recent meta-analysis using random-effect model (13 studies with 22 datasets and 749 participants) evaluated the effect of melatonin supplementation on inflammatory biomarkers It was concluded that melatonin supplementation significantly decreased TNF-alpha and IL-6 levels while the effect on C reactive protein was marginal, therefore melatonin supplementation may be effective on ameliorating inflammatory mediators. [135]
In vitro experiments with leukemia cells showed that 1 mM melatonin increased the rate of apoptosis induced by radiotherapy, an effect that seemed to be dependent of p53 [136]. In the ER+ MCF-7 BC cell line, melatonin treatment at physiological and pharmacological doses (1 nM, 10 μM, 1 mM) prior to radiation induced an inhibition of cell proliferation, the cell cycle arrest, and downregulation of DNA-protein kinases [108]. Additionally, the levels of p53 were much higher in pretreated cells with 1 nM melatonin [109]. Therefore, melatonin is an antiestrogen agent able to sensitize BC cells to radiotherapy, similar results being obtained with aromatase inhibitors administered to BC patients treated with ionizing radiation [137].
When a physiological dose of melatonin (1 nM or 10 nM) was combined in vitro with valproic acid, the melatonin MT1 receptor expression was upregulated and the antiproliferative effect of valproic acid was more effective [138]. Melatonin at 10–50 mg/day, at 8 pm, potentiated IL-2 immunotherapy of pulmonary metastases [46]. Constant treatment with melatonin reduced the incidence and size of breast carcinomas and lowered the incidence of lung metastasis, while interrupted treatment promoted mammary carcinogenesis in transgenic mice [139].
The anticancer effects of melatonin suggest its use as an adjuvant in combination with chemotherapy, radiotherapy, immunotherapy and tumor vaccination. Recently, based on PubMed database, it was reviewed the synergistic effect of melatonin combination with chemotherapy, the antitumoral outcomes suggesting to resolve the drug resistance of patients, reducing the dosage for chemotherapeutic agents and increasing the rate of survival [140] Protective and sensitive effects of melatonin combined with Adriamycin on ER + -BC. Combined with doxorubicin, melatonin (10 mg/kg body weight, for 15 days, daily injected) enhanced BC cell apoptosis in vivo in rats who showed the highest survival rate and lowest tumor weights [141]. Doxorubicin and 0.3 nM melatonin in vitro in MCF-7 cells had a synergic effect on apoptosis (higher procaspase-9, caspase-3, and caspase-9 activities) [142]. It has been reported also that BC patients receiving melatonin at therapeutic doses of 20 mg/day administered in the evening for at least 2 months, during chemotherapy, showed less side effects than controls receiving only chemotherapy [140, 143].
6 Metabolomics: Advanced technology for melatonin evaluation
Melatonin was firstly identified in plants by radioimmunoassays [144] and later from animal fluids by enzyme radioimmunoassay and immunoassay (EIA) [145]. Immunoassays are still widely used in the determination of melatonin, but it has the disadvantage of cross reactivity affected by similar compounds.
Metabolomics is a new, emerging technology, increasingly used for the separation and identification of a cohort of molecules targeting some specific compounds to be considered biomarkers for (early) diagnosis, monitoring pain and therapies for degenerative diseases. Some relevant data are available from clinical studies on Alzheimer’s disease, as well as the effect of sleep deprivation on plasma melatonin and cortisol [146,147,148]. It is based on analytical procedures using techniques like high performance liquid chromatography (HPLC) or gas-chromatography (GC) coupled with different detection ways, such as fluorescence detection [149, 150], electrochemical detection [151, 152], but more and more, mass spectrometry. Several methods for the determination of melatonin in biological fluids have been reported recently, such as gas chromatography–mass spectrometry (GC-MS/MS) [153,154,155] and liquid chromatography coupled with tandem mass spectrometry (HPLC-MS/MS) [156,157,158].
While HPLC coupled with fluorescence or electrochemical detection, Capillary Electrophoresis with UV detection or GC-MS were not adequate to perform pharmacokinetic studies of melatonin because of difficult preparation procedures, the HPLC-MS/MS technique demonstrated great selectivity and relatively good sensitivity [159, 160].
Recently, highly sensitive liquid chromatography-mass spectrometry methods were established and validated for the determination of melatonin (MW = 232.2) in dog plasma, using a liquid–liquid extraction followed by C18 column separation and positive ionization (ESI+) and MS detection by multiple reaction monitoring (MRM) Plasma metabolomic patterns in patients with exhaustion disorder, sleepiness and sleep apnea were also reported recently [161, 162]. The main metabolite of melatonin in urine is 6-Sulfatoxymelatonin (aMT6s), considered as a reliable surrogate biomarker reflecting the blood melatonin concentration and possible association with BC incidence. Recently, preliminary identification and validation of this metabolite and other plasma metabolome-based biomarkers (6 hydroxy melatonin in plasma) for circadian phase in humans were reported [160, 163, 164].
7 Conclusion
Melatonin is a pleiotropic molecule, a metabolic regulator having key-roles and functions not only in the plant kingdom, but mostly in animals and humans’ metabolism. It is considered to act as a “master hormonal regulator” of the circadian rhythm, the aging process and cells activity but also a promising pharmacological agent due to its anti-inflammatory, antiapoptotic and powerful antioxidant properties. Melatonin proved, as demonstrated by many experiments over time, that it can interfere with signals transduction mediated or not by specific receptors, as well to regulate specific metabolic pathways in cytoplasm and mitochondria. Related to degenerative pathologies, including hormone-dependent cancers (especially breast and prostate). The exogeneous administration of melatonin has high beneficial effects by reversing pathologic pathways, preventing the progression of manydegenerative diseases. The supervision of the melatonin turnover and its metabolites in different biological samples can be made by advanced technologies like metabolomics, an emerging technique facilitated by new, high performance liquid chromatography coupled with mass spectrometry, able to fingerprint the metabolic profile and to identify and monitor the turnover of melatonin and its metabolites in normal and pathological circumstances.
References
Lynch HJ, Wurtman RJ, Moskowitz MA, Archer MC, Ho MH. Daily rhythm in human urinary melatonin. Science. 1975;187(4172):169–71.
Lewis A. Melatonin and the biological clock. McGraw-Hill 1999. ISBN 978-0-87983-734-1.
Salehi B, Sharopov F, Tsouh Fokou PV, Kobylinska A, de Jonge L, Tadio K, Sharifi-rad J, Posmyk MM, Martorell M, Martins N, Iriti M. Melatonin in medicinal and food plants: occurrence, bioavailability, and health potential for humans. Cells. 2019;8(7):681–98.
Tan DX, Manchester LC, Liu X, Rosales-Corral SA, Acuna-Castroviejo D, Reiter RJ. Mitochondria and chloroplasts as the original sites of melatonin synthesis: a hypothesis related to melatonin's primary function and evolution in eukaryotes. J Pineal Res. 2013;54:127–38.
Hardeland R. Melatonin in plants and other phototrophs: advances and gaps concerning the diversity of functions. J Exp Botany. 2015;66:627–46.
Hattori A, Migitaka H, Iigo M, Itoh M, Yamamoto K, Ohtani-Kaneko R, Hara M, Reiter RJ. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem Mol Biol Int. 1995;35:627–34.
Reiter RJ. Pineal melatonin: cell biology of its synthesis and of its physiological interactions. Endocrine Rev. 1991;12:151–80.
Reiter RJ, Rosales-Corral S, Tan DX, Jou MJ, Galano A, Xu B. Melatonin as a mitochondria-targeted antioxidant: one of evolution's best ideas. Cell Mol Life Sci. 2017;74:3863–81.
Zhao D, Yu Y, Shen Y, et al. Melatonin synthesis and function: evolutionary history in animals and plants. Front Endocrinol (Lausanne). 2019;10:249.
Tan DX, Hardeland R, Manchester LC, Korkmaz A, Ma S, Rosales-Corral S, Reiter RJ. Functional roles of melatonin in plants, and perspectives in nutritional and agricultural science. J Exp Botany. 2012;63:577–97.
Paredes SD, Korkmaz A, Manchester LC, Tan DX, Reiter RJ. Phytomelatonin: a review. J Exp Botany. 2009;60:57–69.
Nawaz MA, Huang Y, Bie Z, Ahmed W, Russel J, Reiter RJ, Niu M, Hameed S. Melatonin: current status and future perspectives in plant science. Front Plant Sci. 2016;6:1230–41.
Reiter RJ, Tan DX, Zhou Z, Cruz MH, Fuentes-Broto L, Galano A. Phytomelatonin: assisting plants to survive and thrive. Molecules. 2015;20:7396–437.
Arnao MB, Hernández-Ruiz J. The physiological function of melatonin in plants. Plant Signal Behav. 2006;1:89–95.
Arnao MB, Hernández-Ruiz J. Functions of melatonin in plants: a review. J Pineal Res. 2015;59:133–50.
Boutin JA, Audinot V, Ferry G, Delagrange P. Molecular tools to study melatonin pathways and actions. Trends Pharmacol Sci. 2005;26:412–9.
Zhu Y, Gao H, Lu MX, Hao CY, Pu ZQ, Guo MJ, Hou DR, Chen LY, Huang X. Melatonin-nitric oxide crosstalk and their roles in the redox network in plants. Int J Mol Sci. 2019;20:6200.
Tan DX, Manchester LC, Terron MP, Flores LJ, Reiter RJ. One molecule, many derivatives: a never-ending interaction of melatonin with reactive oxygen and nitrogen species? J Pineal Res. 2007;42:28–42.
Hardeland R. Antioxidative protection by melatonin: multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine. 2005;27:119–30.
Reiter RJ, Mayo JC, Tan DX, Sainz RM, Alatorre-Jimenez M, Qin L. Melatonin as an antioxidant: under promises but over delivers. J Pineal Res. 2016;61:253–78.
Illnerova H, Buresova M, Presl J. Melatonin rhythm in human milk. J Clin Endocrinol Metab. 1993;77:838–41.
Sae-Teaw M, Johns J, Johns NP, Subongkot S. Serum melatonin levels and antioxidant capacities after consumption of pineapple, orange, or banana by healthy male volunteers. J Pineal Res. 2013;55:58–64.
Yilmaz C, Gokmen V. Neuroactive compounds in foods: occurrence, mechanism and potential health effects. Food Res Int. 2020;128:108744.
Pereira N, Naufel MF, Ribeiro EB, Tufik S, Hachul H. Influence of dietary sources of melatonin on sleep quality: A review. J Food Sci. 2020;85:5–13.
Hardeland R. Melatonin, hormone of darkness and more – occurrence, control mechanisms, actions and bioactive metabolites. Cell Mol Life Sci. 2008;65:2001–18.
Zisapel N. New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br J Pharmacol. 2018;175:3190–9.
Emet M, Ozcan H, Ozel L, Yayla M, Halici Z, Hacimuftuoglu A. A review of melatonin, its receptors and drugs. Eurasian J Med. 2016;48:135–41.
Poeggeler B, Saarela S, Reiter RJ, Tan DX, Chen LD, Manchester LC, Barlow-Walden LR. Melatonin, a highly potent endogenous radical scavenger and electron donor: new aspects of the oxidation chemistry of this indole accessed in vitro. Ann N Y Acad Sci. 1994;738:419–20.
Pieri C, Marra M, Moroni F, Recchioni R, Marcheselli F. Melatonin: a peroxyl radical scavenger more effective than vitamin E. Life Sci. 1994;55:271–6.
Manchester LC, Coto-Montes A, Boga JA, Andersen LP, Zhou Z, Galano A, Vriend J, Tan DX, Reiter RJ. Melatonin: an ancient molecule that makes oxygen metabolically tolerable. J Pineal Res. 2015;59:403–19.
Peyrot F, Ducrocq C. Potential role of tryptophan derivatives in stress responses characterized by the generation of reactive oxygen and nitrogen species. J Pineal Res. 2008;45:235–46.
Reiter RJ, Paredes SD, Korkmaz A, Jou MJ, Tan DX. Melatonin combats molecular terrorism at the mitochondrial level. Interdisc Toxicol. 2008;1:137–49.
Sharafati-Chaleshtori R, Shirzad H, Rafieian-Kopaei M, Soltani A. Melatonin and human mitochondrial diseases. J Res Med Sci. 2017;22:2–10.
Reiter RJ, Paredes SD, Manchester LC, Tan DX. Reducing oxidative/nitrosative stress: a newly-discovered genre for melatonin. Crit Rev Biochem Mol Biol. 2009;44:175–200.
Tordjman S, Chokron S, Delorme R, Charrier A, Bellissant E, Jaafari N, Fougerou C. Melatonin: pharmacology, functions and therapeutic benefits. Curr Neuropharmacol. 2017;15:434–43.
Shirazi A, Ghobadi G, Ghazi-Khansari M. A radiobiological review on melatonin: a novel radioprotector. J Rad Res. 2007;48:263–72.
Meltz ML, Reiter RJ, Herman TS, Kumar KS. Melatonin and protection from whole-body irradiation: survival studies in mice. Mutation Res. 1999;425:21–7.
Reiter RJ, Herman TS, Meltz ML. Melatonin and radioprotection from genetic damage: in vivo/in vitro studies with human volunteers. Mutation Res. 1996;371:221–8.
Reiter RJ, Herman TS, Meltz ML. Melatonin reduces gamma radiation-induced primary DNA damage in human blood lymphocytes. Mutation Res. 1998;397:203–8.
Schomerus C, Korf HW. Mechanisms regulating melatonin synthesis in the mammalian pineal organ. Ann N Y Acad Sci. 2005;1057:372–83.
Jockers R, Delagrange P, Dubocovich ML, Markus RP, Renault N, Tosini G, Cecon E, Zlotos DP. Update on melatonin receptors: IUPHA review 20. Br J Pharmacol. 2016;173:2702–25.
Tarocco A, Caroccia N, Morciano G, Wieckowski MR, Ancora G, Garani G, Pinton P. Melatonin as a master regulator of cell death and inflammation: molecular mechanisms and clinical implications for newborn care. Cell Death Dis. 2019;10:317–26.
Liu L, Labani N, Cecon E, Jockers R. Melatonin target proteins: too many or not enough? Front Endocrinol. 2019;10:791.
Carrillo-Vico A, Guerrero JM, Lardone PJ, Reiter RJ. A review of the multiple actions of melatonin on the immune system. Endocrine. 2005;27:189–200.
Carrillo-Vico A, Reiter RJ, Lardone PJ, Herrera JL, Fernández-Montesinos R, Guerrero JM, Pozo D. The modulatory role of melatonin on immune responsiveness. Curr Opinion Invest Drugs. 2006;7:423–31.
Maestroni GJ. The immunotherapeutic potential of melatonin. Expert Opin Investig Drugs. 2001;10:467–76.
Armstrong SM, Redman JR. Melatonin: a chronobiotic with anti-aging properties? Med Hypotheses. 1991;34:300–9.
Reiter RJ. The melatonin rhythm: both a clock and a calendar. Experintia. 1993;49:654–64.
Kayumov L, Casper RF, Hawa RJ, Perelman B, Chung SA, Sokalsk S, Shapiro CM. Blocking low-wavelength light prevents nocturnal melatonin suppression with no adverse effect on performance during simulated shift work. J Clin Endocrinol Metab. 2005;90:2755–61.
Hardeland R. Melatonin in aging and disease:multiple consequences of reduced secretion, options and limits of treatment. Aging Dis. 2012;3:194–225.
Hardeland R, Madrid JA, Tan DX, Reiter RJ. Melatonin, the circadian multioscillator system and health: the need for detailed analyses of peripheral melatonin signaling. J Pineal Res. 2012;52:139–66.
Abbott SM, Malkani RG, Zee PC. Circadian disruption and human health: A bidirectional relationship. Eur J Neurosci. 2020;51:567–83.
Brasure M, MacDonald R, Fuchs E, Olson CM, Carlyle M, Diem S, Koffel E, Khawaja IS, Ouellette J, Butler M, Kane RL, Wilt TJ. Management of insomnia disorder. AHRQ Comparative Effectiveness Reviews. No. 159 2015.
Auld F, Maschauer EL, Morrison I, Skene DJ, Riha RL. Evidence for the efficacy of melatonin in the treatment of primary adult sleep disorders (PDF). Sleep Med Reviews. 2017;34:10–22.
Abdelgadir IS, Gordon MA, Akobeng AK. Melatonin for the management of sleep problems in children with neurodevelopmental disorders: a systematic review and meta-analysis (PDF). Arch Disease Childhood. 2018;103:1155–62.
Matheson E, Hainer BL. Insomnia: pharmacologic therapy. Am Fam Physician. 2017;96:29–35.
Liira J, Verbeek JH, Costa G, Driscoll TR, Sallinen M, Isotalo LK, Ruotsalainen JH. Pharmacological interventions for sleepiness and sleep disturbances caused by shift work. Cochrane Database Syst Rev. 2014;8:CD009776.
Besag FMC, Vasey MJ, Lao KSJ, Wong ICK. Adverse events associated with melatonin for the treatment of primary or secondary sleep disorders: A systematic review. CNS Drugs. 2019;33:1167–86.
Buscemi N, Vandermeer B, Hooton N, Pandya R, Tjosvold L, Hartling L, Vohra S, Klasen TP, Baker B. Efficacy and safety of exogenous melatonin for secondary sleep disorders and sleep disorders accompanying sleep restriction: meta-analysis. Br Med J. 2006;332:385–93.
Giannotti F, Cortesi F, Cerquiglini A, Bernabei P. An open-label study of controlled-release melatonin in treatment of sleep disorders in children with autism. J Autism Dev Disorders. 2006;36:741–52.
Jonsdottir IH, Dahlman AS. Endocrine and immunological aspects of burnout: a narrative review. Eur J Endocrinol. 2019;180:R147–58.
Bendz LM, Scates AC. Melatonin treatment for insomnia in pediatric patients with attention-deficit/hyperactivity disorder. Ann Pharmacotherapy. 2010;44:185–91.
Valdés-Tovar M, Estrada-Reyes R, Solís-Chagoyán H, Argueta J, Dorantes-Barrón AM, Quero-Chávez D, Cruz-Garduno R, Cercos MG, Trueta C, Oikawa-Sala J, Dubocovich ML, Benitez-King G. Circadian modulation of neuroplasticity by melatonin: a target in the treatment of depression. Br J Pharmacol. 2018;175:3200–8.
Bonnefont-Rousselot D, Collin F. Melatonin: action as antioxidant and potential applications in human disease and aging. Toxicology. 2010;278:55–67.
Reiter RJ, Tan DX, Korkmaz A, Erren TC, Piekarski C, Tamura H, Manchester LC. Light at night, chronodisruption, melatonin suppression, and cancer risk: a review. Crit Rev Oncol. 2007;13:303–28.
Chuffa L, Seiva F, Cucielo M, Silveira H, Reiter RJ, Lupi LA. Clock genes and the role of melatonin in cancer cells: an overview. Melatonin Res. 2019;2:133–57.
Tan D-X. Reiter RJ mitochondria: the birth place, battle ground and the site of melatonin metabolism in cells. Melatonin Res. 2019;2:44–66.
Reiter RJ, Rosales-Corral SA, Tan DX, Acuna-Castroviejo D, Qin L, Yang SF, Xu K. Melatonin, a full service anticancer agent: inhibition of initiation, progression and metastasis. Int J Mol Sci. 2017;18:4.
Reiter RJ, Sharma R, Ma Q, Rorsales-Corral S, Chuffa LGD. Melatonin inhibits Warburg-dependent cancer by redirecting glucose oxidation to the mitochondria: a mechanistic hypothesis. Cell Mol Life Sci. 2020:1–12.
Ren W, Liu G, Chen S, Yin J, Wang J, Tan B, Wu G, Bazer FW, Peng Y, Tiejun L. Melatonin signaling in T cells: functions and applications. J Pineal Res. 2017;62:e12394.
Liu Z, Yu K, Zheng J, Lin H, Zhao Q, Zhang X, Feng W, Wang L, Xu J, Xie D, Zuo Z-X, Liu Z-X, Zheng Q. Dysregulation, functional implications, and prognostic ability of the circadian clock across cancers. Cancer Med. 2019;8:1710–20.
Mortezaee K, Potes Y, Mirtavoos-Mahyari H, Motevaseli E, Shabeeb D, Musa AE, Najafi M, Farhood B. Boosting immune system against cancer by melatonin: A mechanistic viewpoint. Life Sci. 2019;238:116960.
Boga JA, Caballero B, Potes Y, Perez-Martinez Z, Reiter RJ, Vega-Naredo I, Coto-Montes A. Therapeutic potential of melatonin related to its role as an autophagy regulator: A review. J Pineal Res. 2019;66:e12534.
Giudice A, Crispo A, Grimaldi M, et al. The effect of light exposure at night (LAN) on carcinogenesis via decreased nocturnal melatonin synthesis. Molecules. 2018;23:E1308.
Garaulet M, Qian J, Florez JC, Arendt J, Saxena R, Scheer FAJL. Melatonin effects on glucose metabolism: time to unlock the controversy. Trends Endocrinol Metab. 2020;31(3):192–204.
Tamtaji OR, Reiter RJ, Alipoor R, Dadgostar E, Kouchaki E, Asemi Z. Melatonin and Parkinson disease: current status and future perspectives for molecular mechanisms. Cell Mol Neurobiol. 2020;40:15–23.
Shukla M, Chinchalongporn V, Govitrapong P, Reiter RJ. The role of melatonin in targeting cell signaling pathways in neurodegeneration. Ann N Y Acad Sci. 2019;1443:75–96.
Zhang TW, Li ZF, Dong J, Jiang LB. The circadian rhythm in intervertebral disc degeneration: an autophagy connection. Exp Mol Med. 2020;52:31–40.
Ma Q, Reiter RJ, Chen YD. Role of melatonin in controlling angiogenesis under physiological and pathological conditions. Angiogenesis. 2020;23:91–104.
Bartsch C, Bartsch H, Fluchter SH, Lippert TH. Depleted pineal melatonin production in primary breast and prostate cancer is connected with circadian disturbances: possible role of melatonin for synchronization of circadian rhythmicity. In: Touitou Y, Arendt J, Pevet P, editors. Melatonin and the pineal gland - from basic science to clinical application. Elsevier: New York; 1993. p. 311–6.
Kos-Kudla B, Ostrowska Z, Kozlowski A, Marek BM, Ciesielska-Kopacz N, Kudla M, Kajdaniuk D, Strzelczyk J, Staszewicz P. Circadian rhythm of melatonin in patients with colorectal carcinoma. Neuroendocrinol Lett. 2002;23:239–42.
Iravani S, Eslami P, Moghadam AD, Moazzami B, Mehrvar A, Hashemi MR, Mansour-Ghanaei F, Mansour-Ghanaei A, Majidzadeh AK. The role of melatonin in colorectal cancer. J Gastrointest Canc. 2019; https://doi.org/10.1007/s12029-019-00336-4.
Gil-Martin E, Egea J, Reiter RJ, Romero A. The emergence of melatonin in oncology: focus on colorectal cancer. Med Res Rev. 2019;39:2239–85.
Aghazadeh-Attari M, Mohammadzadeh M, Mostavafi S, Mihanfar A, Ghazizadeh S, Sadighparvar S, Gholamzadeh S, Majidinia M, Yousefi B. Melatonin: an important anticancer agent in colorectal cancer. J Cell Physiol. 2020;235:804–17.
Karasek M, Carillo-Vico A, Guerrero JM, Winczyk K, Pawlikowsky M. Expression of melatonin MT(1) and MT(2) receptors, and ROR alpha(1) receptor in transposable murine Colon 38 cancer. Neuroendocrinol Lett. 2002;23:55–60.
Fathizadeh H, Mirzaei H, Asemi Z. Melatonin: an anti-tumor agent for osteosarcoma. Cancer Cell Int. 2019;19:319.
Neamati F, Asemi Z. The effects of melatonin on signaling pathways and molecules involved in glioma. Fund Clin Pharmacol. 2020; https://doi.org/10.1111/fcp.12538.
Mojaverrostami S, Asghari N, Khamisabadi M, Khoei HH. The role of melatonin in polycystic ovary syndrome: A review. Int J Reprod Biomed. 2019;17:865–82.
Rimler A, Lupowitz Z, Zisapel N. Differential regulation by melatonin of cell growth and androgen receptor binding to the androgen response element in prostate cancer cells. Neuroendocrinol Lett. 2002;23:45–9.
Kubo T, Ozasa K, Mikami E, Wakai K, Fujino Y, Watanabe Y, Miki T, Nakao M, Hayashi K, Suzuki K, Mori M, Washio M, Sakauchi F, Ito Y, Yoshimura T, Tamakoshi A. Prospective cohort study of the risk of prostate cancer among rotating-shift workers: findings from the Japan collaborative cohort study. Am J Epidemiol. 2006;164:549–55.
Shafabakhsh R, Reiter RJ, Mirzaei H, Teymoordash SN, Asemi Z. Melatonin: A new inhibitor agent for cervical cancer treatment. J Cell Physiol. 2019;234:21670–82.
Kerenyi NA, Pandula E, Feuera G. Why the incidence of cancer is increasing: the role of ‘light pollution’. Med Hypotheses. 1990;33:75–8.
Straif K, Baan R, Grosse Y, Secretan B, El Ghissassi F, Bouvard V, Altieri A, Benbrahim-Tallaa L, Cogliano V. On behalf of the WHO International Agency for Research on Cancer monograph working group, carcinogenicity of shift-work, painting, and fire-fighting. Lancet Oncol. 2007;8:1065–6.
Hill SM, Frasch T, Xiang S, Yuan L, Duplessis T, Mao L. Molecular mechanisms of melatonin anticancer effects. Integr Cancer Ther. 2009;8:337–46.
Sanchez-Barcelo E, Cos S, Mediavilla D, Martinez-Campa C, Gonzalez A, Alonso-Gonzalez C. Melatonin-estrogen interactions in breast cancer. J Pineal Res. 2005;38:217–22.
Shiu SY. Towards rational and evidence-based use of melatonin in prostate cancer prevention and treatment. J Pineal Res. 2007;45:1–9.
Blask DE, Dauchy RT, Sauer LA. Putting cancer to sleep at night: the neuroendocrine/circadian melatonin signal. Endocrine. 2005;27:179–88.
Blask DE, Sauer LA, Daunchy RT. Melatonin as a chronobiotic/anticancer agent: cellular, biochemical, and molecular mechanisms of action and their implication for circadian-based cancer therapy. Curr Top Med Chem. 2002;2:113–32.
Korkmaz A, Sanchez-Barcelo EJ, Tan DX, Reiter RJ. Role of melatonin in the epigenetic regulation of breast cancer. Breast Cancer Res Treat. 2009;115:13–27.
Leon-Blanco MM, Guerrero JM, Reiter RJ, Calvo JR, Pozo D. Melatonin inhibits telomerase activity in the MCF-7 tumor cell live both in vivo and in vitro. J Pineal Res. 2003;35:204–11.
Girgert R, Bartsch C, Hill SM, Kreienberg R, Hauf V. Tracking the elusive antiestrogenic effect of melatonin: a new methodological approach. Neuroendocrinol Lett. 2003;24:440–4.
Cos J, Fernandez R, Guezmes A, Sanchez-Barcelo EJ. Influcence of melatonin on invasive and metastic properties of MCF-7 human breast cancer cells. Cancer Res. 1998;58:4383–90.
Jung-Hynes B, Reiter RJ, Ahmad N. Sirtuins, melatonin and circadian rhythms: building a bridge between aging and cancer. J Pineal Res. 2010;48:9–19.
Alvarez-García V, González A, Alonso-González C, Martínez-Campa C, Cos S. Melatonin interferes in the desmoplastic reaction in breast cancer by regulating cytokine production. J Pineal Res. 2012;52:282–90.
Alvarez-García V, González A, Alonso-González C, Martínez-Campa C, Cos S. Regulation of vascular endothelial growth factor by melatonin in human breast cancer cells. J Pineal Res. 2013;54:373–80.
Grin W, Grünberger WA. Significant correlation between melatonin deficiency and endometrial cancer. Gynecol Obstet Investig. 1998;45:62–5.
Benitez-King G, Soto-Vega E, Ramirez-Rodriguez G. Melatonin modulates microfilament phenotypes in epithelial cells: implications for adhesion and inhibition of cancer cell migration. Histol Histopathol. 2009;24:789–99.
Alonso-González C, González A, Martínez-Campa C, Gómez-Arozamena J, Cos S. Melatonin sensitizes human breast cancer cells to ionizing radiation by downregulating proteins involved in double-strand DNA break repair. J Pineal Res. 2015;58:189–97.
Alonso-González C, González A, Martínez-Campa C, Menendez-Menendez J, Gomez-Arozamena J, Garcia-Vidal A, Cos S. Melatonin enhancement of the radiosensitivity of human breast cancer cells is associated with the modulation of proteins involved in estrogen biosynthesis. Cancer Lett. 2016;370:145–52.
Schernhammer ES, Kroenke CH, Laden F, Hankinson SE. Night work and risk of breast cancer. Epidemiology. 2006;17:108–11.
Menéndez-Menéndez J, Martínez-Campa C. Melatonin: an anti-tumor agent in hormone-dependent cancers, review article. Int J Endocrinol. 2018:3271948.
Cohen M, Lippman M, Chabner B. Pineal gland and breast cancer. Lancet. 1978;312:P1381–2.
Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Colditz GA. Rotating night shifts and risk of breast cancer in women participating in the nurses’ health study. J Nat Cancer Inst. 2001;93:1563–8.
Tamarkin L, Danforth D, Lichter A, Cohen M, Chabner B, Lippman M. Decreased nocturnal plasma melatonin peak in patients with estrogen receptor positive breast cancer. Science. 1982;216:1003–5.
Bartsch C, Bartsch H, Karasek M. Melatonin in clinical oncology. Neuroendocrinol Lett. 2002;23:30–8.
Molis TM, Spriggs LL, Hill SM. Modulation of estrogen receptor mRNA expression by melatonin in MCF-7 human breast cancer cells. Mol Endocrinol. 1994;8:1681–90.
Nowfar S, Treplitzky SR, Melancon K, Kiefer TL, Chieng Q, Dwivedi PD, Bischoff ED, Moroz K, Anderson MB, Dai J, Lai N, Yuan L, Hill SM. Tumor prevention by 9-cis-retinioic acid in the N-nitroso-N-methylurea model of mammary carcinogenesis is potentiated by the pineal hormone melatonin. Breast Cancer Res Treat. 2002;72:33–43.
Cos S, González A, Martínez-Campa C, Mediavilla MD, Alonso-González C, Sánchez-Barceló EJ. Estrogen signaling pathway: a link between breast cancer and melatonin oncostatic actions. Cancer Detect Prev. 2006;30:118–28.
Kubatka P, Zubor P, Busselberg D, Kwon TK, Adamek M, Petrovic D, Opatrilova R, Gazdikova K, Caprnda M, Rodrigo L, Danko J, Kruzliak P. Melatonin and breast cancer: evidences from preclinical and human studies. Crit Rev Oncology/Hematol. 2018;122:133–43.
Cos S, Alvarez-García V, González A, Alonso-González C, Martínez-Campa C. Melatonin modulation of crosstalk among malignant epithelial, endothelial and adipose cells in breast cancer (review). Oncol Lett. 2014;8:487–92.
James P, Bertrand KA, Hart JE, Schernhammer ES, Tamimi RM, Laden F. Outdoor light at night and breast cancer incidence in the nurses’ health study II. Env Health Perspectives. 2017;125:087010.
Garcia-Saenz A, Sánchez de Miguel A, Espinosa A, Valentin A, Aragonés N, Llorca J, Amiano P, Sánchez VM, Guevara M, Capelo R, Tardon A, Peiró-Perez R, Jiménez-Moleón JJ, Roca-Barceló A, Pérez-Gómez B, Dierssen-Sotos T, Fernández-Villa T, Moreno-Iribas C, Moreno V, García-Pérez J, Castaño-Vinyals G, Pollán M, Aubé M, Kogevinas M. Evaluating the association between artificial light-at-night exposure and breast and prostate cancer risk in Spain (MCC-Spain study). Env Health Perspectives. 2018;126:047011.
Stevens RG, Rea MS. Light in a built environment: potential role of circadian disruption in endocrine disruption and breast cancer. Cancer Causes Control. 2001;12:279–87.
Hansen J. Increased breast cancer risk among women who work predominantly at night. Epidemiology. 2001;12:74–7.
Coleman MP, Reiter RJ. Breast cancer, blindness and melatonin. Eur J Cancer Clin Oncol. 1992;28:501–3.
Xiang S, Dauchy RT, Hauch A, Mao L, Yuan L, Wren MA, Belancio VP, Mondal D, Frasch P, Blask DE, Hill SM. Doxorubicin resistance in breast cancer is driven by light at night-induced disruption of the circadian melatonin signal. J Pineal Res. 2015;59:60–9.
Regelson W, Pierpaoli MD. Melatonin: A rediscovered antitumor hormone? Its relation to surface receptors sex steroid metabolism, immunologic response, and chronobiologic factors in tumor growth and therapy. Cancer Investig. 1987;5:379–85.
Mills E, Wu P, Seely D, Guyatt G. Melatonin in the treatment of cancer: a systematic review of randomized controlled trials and meta-analysis. J Pineal Res. 2005;39:360–6.
Altun A, Ugur-Altun B. Melatonin: therapeutic and clinical utilization. Int J Clinical Practice. 2007;61:835–45.
Pawlikowsky M, Winczyk K, Karasek M. Oncostatic action of melatonin: facts and question marks. Neuroendocrinol Lett. 2002;23:24–9.
Chao YH, Wu KH, Yeh CM, Su SC, Reiter RJ, Yang SF. The potential utility of melatonin in the treatment of childhood cancer. J Cell Physiol. 2019;234:19158–66.
Lamont KT, Somers S, Lacerda L, Opie LH, Lecour S. Is red wine a SAFE sip away from cardioprotection? Mechanisms involved in resveratrol- and melatonin-induced cardioprotection. J Pineal Res. 2011;50:374–80.
Najafi M, Shayesteh MRH, Mortezaee K, Farhood B, Haghi-Aminjan H. The role of melatonin on doxorubicin-induced cardiotoxicity: a systematic review. Life Sci. 2020;241:117173.
Imenshahidi M, Karimi G, Hosseinzadeh H. Effects of melatonin on cardiovascular risk factors and metabolic syndrome: a comprehensive review. Naunyn Schmiedebergs Archives Pharmacol. 2020;393:521–36.
Zarezadeh M, Khorshidi M, Emami M, Janmohammadi P, Kord-varkaneh H. Mousavi SM, Mohammed SH, Saedisomeolia A, Alizadeh S. Melatonin supplementation and pro-inflammatory mediators: a systematic review and meta-analysis of clinical trials. Eur J Nutr 2019; https://doi.org/10.1007/s00394-019-02123-0, 59, 1803, 1813.
Jang SS, Kim WD, Park WY. Melatonin exerts differential actions on X-ray radiation-induced apoptosis in normal mice splenocytes and Jurkat leukemia cells. J Pineal Res. 2009;47:147–55.
Ishitobi M, Shiba M, Nakayama T, Motomura K, Koyama H, Nishiyama K, Tamaki Y. Treatment sequence of aromatase inhibitors and radiotherapy and long-term outcomes of breast cancer patients. Anticancer Res. 2014;34:4311–4.
Jawed S, Kim B, Ottenhof T, Brown GM, Werstiuk ES, Niles LP. Human melatonin MT1 receptor induction by valproic acid and its effects in combination with melatonin on MCF-7 breast cancer cell proliferation. Eur J Pharmacol. 2007;560:17–22.
Anisimov VN, Alimova IN, Baturin DA, Popovich IG, Zabezhinski MA, Manton KG, Semenchenko AV, Yashin AI. The effect of melatonin treatment regimen on mammary adenocarcinoma development in HER-2/neu transgenic mice. Int J Cancer. 2003;103:300–5.
Najafi M, Salehi E, Farhood B, Nashtaei MS, Khanlarkhani N, Namjoo Z, Keywan MK. Adjuvant chemotherapy with melatonin for targeting human cancers: a review. J Cell Physiol. 2019;234(3):2356–72. https://doi.org/10.1002/jcp.27259.
Ma C, Li LX, Zhang Y, Xiang C, Ma T, Ma ZQ, Zhang ZP. Protective and sensitive effects of melatonin combined with Adriamycin on ER+ (estrogen receptor) breast cancer. Eur J Gynaecol Oncol. 2016;36:197–202.
Kosar PA, Naziroglu M, Ovey IS, Cig B. Synergic effects of doxorubicin and melatonin on apoptosis and mitochondrial oxidative stress in MCF-7 breast cancer cells: involvement of TRPV1 channels. J Membrane Biol. 2016;249:129–40.
Lissoni P, Tancini G, Barni S, Paolorossi F, Ardizzoia A, Conti A, Maestroni G. Treatment of cancer chemotherapy-induced toxicity with the pineal hormone melatonin. J Pineal Res. 1997;5:126–9.
Dubbels R, Reiter RJ, Goebel A, Schnakenberg GE, Ehlers C, Schiwara HW, Schloot W. Melatonin in edible plants identified by radioimmunoassay and by high performance liquid chromatography-mass spectrometry. J Pineal Res. 1995;18:28–31.
Ferrua B, Masseyeff R. Immunoassay of melatonin with enzyme-labeled antibodies. J Immunoass. 1985;6:79–94.
Honma A, Revell VL, Gunn PJ, Davies SK, Middleton B, Raynaud FI, Skene DJ. Effect of acute total sleep deprivation on plasma melatonin, cortisol and metabolite rhythms in females. Eur J Neurosci. 2020;51:366–78.
Sengupta A, Weljie AM. Metabolism of sleep and aging: bridging the gap using metabolomics. Nutr Health Aging. 2019;5:167–84.
Xie S, Fan W, He H, Huang F. Role of melatonin in the regulation of pain. J Pain Res. 2020;13:331–43.
Mills MH, Finlay DC, Haddad PR. Determination of melatonin and mono-amines in rat pineal using reversed-phase ion-interaction chromatography with fluorescence detection. J Chromatogr A. 1991;564:93–102.
Peniston-Bird JF, Di WL, Street CA, Kadva A, Stalteri MA, Silman SE. HPLC assay of melatonin in plasma with fluorescence detection. Clin Chem. 1993;39:2242–7.
Chin JR. Determination of six indolic compounds, including melatonin, in rat pineal using high-performance liquid chromatography with serial fluorimetric-electrochemical detection. J Chromatogr. 1990;528:111–21.
Vieira R, Míguez J, Lerna M, Aldegunde M. Pineal and plasma melatonin a determined by high-performance liquid chromatography with electrochemical detection. Anal Biochem. 1992;205:300–5.
Skene DJ, Leone RM, Young IM, Silman RE. The assessment of a plasma melatonin assay using gas chromatography negative ion chemical ionization mass spectrometry. Biomed Mass Spectrom. 1983;10:655–9.
Fourtillan JB, Gobin P, Faye B, Girault J. A highly sensitive assay of melatonin at the femotogram level in human plasma by gas chromatography/negative ion chemical ionization mass spectrometry. Biol Mass Spectrom. 1994;23:499–509.
Simonin G, Bru L, Lelièvre E, Jeanniot J-P, Bromet N, Walther B, Boursier-Neyret C. Determination of melatonin in biological fluids in the presence of the melatonin agonist S 20098: comparison of immunological techniques and GC-MS methods. J Pharm Biomed Anal. 1999;21:591–601.
Yang S, Zheng X, Xu Y, Zhou X. Rapid determination of serum melatonin by ESI–MS–MS with direct sample injection. J Pharm Biomed Anal. 2002;30:781–90.
Jensen MA, Hansen AM, Abrahamsson P, Norgaard AW. Development and evaluation of a liquid chromatography tandem mass spectrometry method for simultaneous determination of salivary melatonin, cortisol and testosterone. J Chromatogr B Analyt Technol Biomed Life Sci. 2011;879:2527–32.
Carter MD, Calcutt MW, Malow BA, Rose KL, Hachey DL. Quantitation of melatonin and N-acetylserotonin in human plasma by nanoflow LC-MS/MS and electrospray LC-MS/MS. J Mass Spectrom. 2012;47:277–85.
Khan SA, George R, Charles BG, Taylor P, Heussler HS, Couper DM, McGuire TM, Pache D, Norris D. Monitoring salivary melatonin concentrations in children with sleep disorders using liquid chromatography-tandem mass spectrometry. Ther Drug Monit. 2013;35:388–95.
Wang H, Walaszczyk EJ, Li K, Chung-Davidson Y-W, Li W. High-performance liquid chromatography with fluorescence detection and ultra-performance liquid chromatography with electrospray tandem mass spectrometry method for the determination of indoleamine neurotransmitters and their metabolites in sea lamprey plasma. Anal Chim Acta. 2012;721:147–53.
Hadrevi J, Jonsdottir IH, Jansson P-A, Eriksson JW, Sjörs A. Plasma metabolomic patterns in patients with exhaustion disorder. Stress. 2019;22:17–26.
Diallo I, Pak VM. Metabolomics, sleepiness and sleep duration in sleep apnea. Sleep and Breath. 2020; https://doi.org/10.1007/s11325-019-01969-2.
Härtter S, Morita S, Bodin K, Ursing C, Tybring C, Bertilsson L. Determination of exogenous melatonin and its 6-hydroxy metabolite in human plasma by liquid chromatography–mass spectrometry. Ther Drug Monit. 2001;23:282–6.
Xu J, Huang L. Urinary 6-sulfatoxymelatonin level and breast cancer risk: systematic review and meta-analysis. Sci Rep. 2017;7:5353–63.
Acknowledgments
The logistic, administrative and technical support offered by the Research Center for Applied Biotechnology in Diagnosis and Molecular Therapy (BIODIATECH) Cluj-Napoca is acknowledged.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Not applicable. The authors declare that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Socaciu, A.I., Ionuţ, R., Socaciu, M.A. et al. Melatonin, an ubiquitous metabolic regulator: functions, mechanisms and effects on circadian disruption and degenerative diseases. Rev Endocr Metab Disord 21, 465–478 (2020). https://doi.org/10.1007/s11154-020-09570-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-020-09570-9