Abstract
Hypocholesterolemic and hypoglycemic activities of Coptis chinensis franch inflorescence (Coptis inflorescence) were studied using animal models. Serum total and LDL cholesterol of rats fed a diet containing 1% cholesterol and 0.5% cholic acid increased, as compared with those of rats fed a normal diet. The level of total and LDL cholesterol were reduced markedly in a dose dependent manner, in rats given Coptis inflorescence extract orally at doses of 0.25, 0.5 g/kg.day for 4 weeks. In diabetic rats induced by alloxan, Coptis inflorescence extract showed a significant (p < 0.05) blood sugar lowering activity at all experimented doses (0.125, 0.25 and 0.5 g/kg.day). The highest reduction of blood sugar was about 58% when the rats were given Coptis inflorescence extract orally at a dose of 0.5 g/kg.day for 3 weeks. The 100 g dried water extract of Coptis inflorescence contained 8.11 g total alkaloid, 3.34 g berberin, 1.08 g palmatine and 0.66 g jatrorrhizine, which had long been identified as active compounds in Coptis chinensis franch root (Coptis root). Thus, the results suggest that Coptis inflorescence would be effective in the prevention and management of coronary artery disease by lowering serum cholesterol and blood sugar.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The root of Coptis chinensis Franch (Coptis root or Huanglian), is a widely used and important medicine herb in China. Many Chinese herbal formulas contain an extract of this root as their major ingredient; most well-known being oral liquid “shuang huanglian,” which is used for the treatment of fever, cough, angina [1]. In addition, Coptis root has long been prescribed in single or combination for the treatment of diabetes and several anti-diabetes drug products officially approved in China contain it as their main ingredients such as Shen-qi and Xiao-ke-an [2]. The constituents of Coptis root have been studied extensively with various alkaloids such as berberine, coptisine, palmatine, jatrorrhizine and magnoflorine being the major components [3, 4]. One of the major alkaloids, berberine has strong anti-inflammatory [5] and antimicrobial [6] activities. Antiprotozoal, antihypertensive [7], anticholinergic [8], antiarrhythmic [9] and anticancer [10, 11] activities of alkaloids from the root of Coptis chinensis Franch have been also reported. More recently, cholesterol-lowering and anti-diabetic effects of berberine have been approved [12–13]. Compared with the roots, the chemical composition and pharmacological properties of Coptis inflorescence have not been studied. A preinvestigation into the chemical compounds in the Coptis inflorescence showed that 100 g dry flower contained total alkaloid 2.85 g, berberin 1.32 g, palmatine 0.43 g and jatrorrhizine 0.26 g similar to those in its root [3–4]. The finding suggests that the Coptis inflorescence could have similar pharmaceutical use in the treatment of diabetes and hypercholesterol as that of the root. In fact, as a by-product generated in the production of Coptis root, the inflorescence had been used as folk medicine or diet supplement to treat heatstroke, bacterial diarrhea, inflammatory and cardiovascular disease. Therefore, this study was designed to investigate the hypoglycaemic and hypocholesterolemic activities for elucidation of their potential empirical use.
Materials and Methods
Chemicals
Reference berberine chloride, jatrorrhizine chloride, palmatine chloride were purchased from the National Institute for the Control of Pharmaceutical Biological Products (Beijing, China). Alloxan, cholesterol, cholic acid and casein were obtained from Pharmaceutical and Chemical Reagent Inc. (Chongqing, China).
Plant Materials
Coptis inflorescence was collected from a Huanglian GAP (good agricultural practice) plot at Shizhu County, China, in April 2004. It was immediately treated with 100 °C steam for 5 min after collection, and then dried at 75 °C in a vibrating blast drier (model VFB-A, Beijing, China). The dried inflorescence was ground to pass through a 60 mesh screen; the powder was stored in a desiccator at Chemistry Institute of Pharmacological Resource, Southwest University, China.
Animals
Healthy wistar rats weighing 180–220 g were purchased from Chinese Medicine Research Institute in Chongqing, China. All rats were accommodated for 72 h prior to the experiment in an environmentally controlled room (22±2 °C, 55±10% relative humidity, 14 times of air change in one hour and 12 h light cycle), and given free access to feed and water.
Preparation of Extract of Coptis Inflorescence
A known weight (100 g) of dried inflorescence was extracted three times, each time with 600 ml water for 1 h at 100 °C. The water extract was concentrated at 60 °C in vacuum condition to a volume of 100 ml, in which 1 ml extract equals approximately 1 g dry inflorescence. For the animal tests, this liquid extract was directly used and a dried sample was prepared under vacuum condition for the determination of alkaloids (31–32 g dry extract can be obtained from 100 g dry inflorescence).
Alkaloids Determination
To determine the total alkaloids, the dried water extract of Coptis inflorescence was dissolved in methanol and after brief centrifugation (1000 g, 10 min), a 5 ml aliquot of the supernatant was passed through a 2 ml clean-up column (filled with neutral aluminum oxide) and eluted with methanol. Collected eluant was diluted with 0.1 M of HCl−methanol and the absorbance was measured at 345 nm using a U-1800 UV spectrophotometer (HITACHI, Japan). Berberine was used as reference and the total alkaloids were expressed as berberin equivalent in each 100 g dried extract (g.berberine/100 g dried extract).
For quantification of berberine, jatrorrhizine, and palmatine, a Waters HPLC system equipped with 510 pump, UV detector and Symmetry® C18 column (4.6×250 mm, 5 μm) was used. Berberine chloride, jatrorrhizine chloride and palmatine chloride in methanol were injected into the column as external standard compounds. After injection, the column was eluted with a linear gradient of acetonitrile in water from 25–55% (containing 0.2% KH2PO4) over 30 min before returning to initial condition for 10 min. The flow rate was 1ml/min and effluent was monitored at 346 nm [3, 14].
Hypocholesterolemic Test
Hypercholesterolemia was induced by feeding the rats a diet containing 18% casein, 1% cholesterol and 0.5% cholic acid [15, 16]. The rats were divided into normal (cholesterol-cholic acid free), model (cholesterol diet), low dose treatment, medium dose treatment and high dose treatment groups (cholesterol+extract of Coptis inflorescence) and 10 rats were included in each group. The extract of Coptis inflorescence was diluted with water and given orally to rats at 0.125 (low dose), 0.25 (medium dose) or 0.5 (high dose) g/kg/day for 4 weeks using a stomach tube. Six hours after the last treatment, the rats were decapitated, their blood was collected and serum for the measurement of cholesterol levels was obtained by centrifugation. Each liver was removed, dried on tissue paper, weighed and stored at−80 °C until analysis.
The liver was homogenized and total cholesterol was extracted with a mixture of chloroform and methanol (v/v, 2:1). The total cholesterol levels in serum and liver tissue were determined using commercial kit (CHL-KIT, Antai clinical analysis reagent co., Beijing, China). Serum LDL cholesterol levels was determined according to the method of Kersher et al. [17].
Antihyperglycemic Test
Rats were divided into normal and hyperglycemic groups and 10 rats were included in each group. Hyperglycemia was induced by a total of three intraperitoneal injections (70 mg/kg per injection) administered as single doses of alloxan every third day [18]. At the end of the third injection blood samples were drawn from the caudal vein and blood sugar was measured. The animals with 10 mg/ml blood sugar or more were included in the hyperglycemic groups. The extract of Coptis inflorescence was administered orally at 0.125, 0.25 and 0.5 g/kg/day by using a stomach tube. A control test using distilled water and a positive control using Jin-qi, a Chinese medicine formula for the treatment of diabetes, were carried out. Blood sugar was measured using a sugar test kit (GLU-kit, Antai clinical analysis reagent co., Beijing, China).
Statistical Analysis
Statistical analysis was performed by the Duncan Multiple Range Test (SAS, version 6.12, Cary, NC) and those at p < 0.05 were accepted as significant.
Results
Alkaloids in Coptis Inflorescence
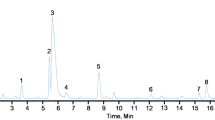
The HPLC chromatogram of water extract of Coptis inflorescence is shown in Fig.1. It was observed that the largest peak of our extract was berberine, which accounted for more than 40% of total alkaloid and 100 g dried extract contained total alkaloid value of 8.11±0.16 g, berberin 3.34±0.09 g, palmatine 1.08±0.15 g and jatrorrhizine 0.66±0.12 g. Nutrients analysis showed that this extract was rich in protein (amino acids) and minerals (data not shown).
HPLC chromatogram of water extract of Coptis inflorescence, 1: jatrorrhizine, 2: palmatine, 3: berberine. Symmetry® C18 column (4.6×250 mm, 5 μm) was used, elution was done by a linear gradient of acetonitrile in water from 25–55% (containing 0.2% KH2PO4) over 30 min before returning to initial condition for 10 min. The flow rate was 1ml/min and effluent was monitored at 346 nm.
Cholesterol Lowering Effect
The rats fed a high cholesterol diet had a markedly higher serum level of total and LDL cholesterol compared with those fed normal diet. The serum cholesterol level of rats given Coptis inflorescence extract were significantly (p < 0.05) reduced as compared with those of model rats; this hypocholesterolemic effect was enhanced when the oral dose administered increased (Figs. 2 and 3). Administration of Coptis inflorescence extract at a dose of 0.25, 0.5 g/kg body weight/day for 4 weeks reduced the total cholesterol by 23% and 28%, respectively. The LDL cholesterol after administration of 0.125, 0.25 and 0.5 g/kg body weight/day of the inflorescence extract for 4 weeks was reduced by 11%, 17% and 25%, respectively.
The total cholesterol levels of liver of rats fed cholesterol diet are markedly higher than those of rats fed normal diet (Table 1). The total cholesterol levels of liver of rats given Coptis inflorescence extract at dose of 0.125, 0.25 and 0.5 g/kg body weight/day for 4 weeks reduced by 6.21%, 14.89% and 22.61%, respectively.
Blood Sugar Lowering Effect
Table 2 shows that the extract of Coptis inflorescence possesses significant blood sugar lowering potential in alloxan-induced diabetic model rats throughout a 3-week experiment in a dose dependent manner. In particular, the administration of Coptis inflorescence extract at doses of 0.25 and 0.5 g/kg body weight/day for 21 d reduced the blood glucose by 48.3% and 58.3%, respectively. This finding is comparable to the results we obtained for the positive rats that were fed a diet containing commercial anti-diabetes herbal formula (Jin-qi), which reduced the blood glucose by 52.5% at 0.25 g kg body weight day after 21d.
Discussion
Alkaloids including berberin, palmatine, coptisine and jatrorrhine have long been realized as active compounds in Coptis root, a very important traditional Chinese medicine which is widely used singly or in formulas for the treatment of hypertension, hemoptysis, hematemesis, melena, cerebral hemorrhage, gastrointestinal disorders and inflammatory diseases including hyperglycemia [1–2]. The presence of alkaloids in Coptis inflorescence suggests that the flower may have similar pharmaceutical use as the root as evident in the hypocholesterolemic and antihyperglycemic effects obtained in this work.
A study using pure berberine demonstrated that berberine is an effective cholesterol-lowering drug with a mechanism of action different from that of statin drugs [18]. Alkaloids especially berberine in Coptis inflorescence may contribute most to the cholesterol lowering activity of the flower extract. The effect of Coptis inflorescence extract on total cholesterol level of liver indicates modulation of the synthesis and/or excretion of cholesterol.
It is well known that increased cholesterol concentration in plasma is associated with serious pathological conditions such as coronary atherosclerosis [19] and increase risk of coronary artery disease (CAD) [20–21]. Studies have demonstrated that cholesterol, especially LDL cholesterol and its oxidized derivatives play an important role in the pathogenesis of atherosclerotic conditions [22–23]. Other reports have used diet treatments or drug therapy to show that lowering cholesterol levels can reduce CAD-associated morbidity and mortality [24]. Accordingly, results from this study suggest that Coptis inflorescence extract may serve as an alternative natural health therapy to reduce CAD-related morbidity and mortality by lowering cholesterol levels in blood.
Although the Coptis root has long been used singly or in product formulations to effectively treat hyperglycemia, the active compounds and mechanism of this herbal plant remains uncertain. It is generally believed by most researchers that polysaccharides in the herbs may protect pancreatic islets and beta cells, help the regeneration of beta cells, and therefore, increase the insulin secretion from pancreas [25–26]. Other experiments carried out with pure berberine suggested that berberine was not only related to the property of stimulating insulin secretion but also modulating lipids [12]. Together with our result, further investigations into the blood sugar lowering mechanism and active compounds in this plant are required as other mechanisms such as inhibition of intestinal absorption of glucose [27], increase of availability of insulin may contribute to the observed hypoglycaemic effect.
Diabetes mellitus is a chronic condition in which patient may require long-term administration of drugs that help to improve glycemia control [28]. In addition, diabetic patients have an increased risk of cardiovascular disease and stroke [29]. Therefore, there is a strong need for safe and effective oral hypoglycemic agents. Hence herbal anti-diabetic agents, especially medical diets such as pumpkin, wheat, celery, wax gourd, lotus root and bitter melon with lower side effects when compared to drugs, have received great attention [30]. Our research result suggests that Coptis inflorescence would be effective in preventing, treating and managing diabetes although it remains unclear which of the components exhibit blood glucose lowering activity.
Reference
Jiangsu New Medical College (1986) Zhongyao Dacidian (Encyclopaedia of ChineseTraditional medicine). Shanghai, China: The People’s Publishing Company.
Jia W, Gao WY, Tang L (2003) Antidiabetic Herbal Drugs Officially Approved in China. Phytother Res 17: 1127–1134.
Wang JH, Chen DY, Su YY (1994) Study on the prepared products of Rhizoma coptidis by HPLC. Chin Tradit Herb Drug 25: 293.
Yang F, Zhang T, Zhang R, Ito Y (1998) Application of analytical and preparative high-speed counter-current chromatography for separation of alkaloids from Coptis chinensis Franch. J Chromatography A 829(1/2): 137–141.
Akhter MH, Sabir M, Bhide NK (1997) Anti-inflammatory effect of berberine in rats injected locally with cholera toxin. Indian J Med Res 65: 133–141.
Amin AH, Subbaiah TV, Abbasi KM (1969) Berberine sulfate: Antimicrobial activity, bioassay, and mode of action. Can J Microbiol 15: 1067–1076.
Bova S, Padrini R, Goldman WF, Berman DM, Cargnelli G (1992) On the mechanism of vasodilating action of berberine: Possible role of inositol lipid signaling system. J Pharmacol Exp Ther 261: 318–323.
Tsai CS, Ochillo RF(1991) Pharmacological effects of berberine on the longitudinal muscle of the guinea-pig isolated ileum. Arch Int Pharmacodyn Ther 310: 116–131.
Wang YX, Zheng YM (1997) Ionic mechanism responsible for prolongation of cardiac action-potential duration by berberine. J Cardiovasc Pharmacol 30: 214–222.
Anis KV, Kuttan G, Kuttan R (1999) Role of Berberine as An Adjuvant Response Modifier During Tumour Therapy in Mice. Pharm Pharmacol Commun 5: 697–700.
Lin SK, Tsai SC, Lee CC, Wang BW, Liou JY, Shyu KG (2004) Berberine Inhibits HIF-1α Expression via Enhanced Proteolysis. Mol Pharmacol 66: 612–619.
Leng SH, Lu FE, Xu LJ (2004) Therapeutic effects of berberine in impaired glucose tolerance rats and its influence on insulin secretion. Acta Pharmacologica Sinica 25: 496–502.
Kong WJ, Wei J, Abidi P, Lin M, Inaba S, Li C, Wang YL, Wang ZZ, Si SY, Pan HN, Wang SK, Wu JD, Wang Y, Li ZR, Liu JW, Jiang JD (2004) Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat Med 10: 1344–1351.
Ikuta A, Kobayashi A, Itokawa H (1984) Studies on the quantitative analysis of protoberberine alkaloids in Japanese, Chinese and other countries Coptis rhizomes by thin-layer chromatography-densitometry. Shoyakugaku zasshi 38: 279–282.
Kalman J, Kudchodkar BJ, Krishnamoorthy R, Dory L, Kacko AG, Agarwal N (2001) High cholesterol diet down regulates the activity of activator protein-1 but not nuclear factor-kappa B in rabbit brain. Life Sci 68: 1495–1503.
Ha TY, Han SY, Kim SR, Kin IH, Lee HY, Kim HK (2005) Bioactive components in rice bran oil improve lipid profiles in rats fed a high-cholesterol diet. Nutr Res 25: 597–606.
Kersher L, Schiefer S, Draeger B, Maier J, Ziegenhorn J (1995) Precipitation methods for the determination of LDL-cholesterol. Clin Biochem 18: 118–125.
Anturlikar SD, Gopumadhavan S, Chauhan BL, Mitra SK (1995) Effect of D-400, a herbal Formulation, on blood sugar of normal and alloxan-induced diabetic rats. Indian J Physiol Pharmacol 39: 95–100.
Kannel WB, Castelli WP (1979) Cholesterol In the prediction of atherosclerotic disease: New perspectives based on the Framingham Study. Ann Intern Med 90: 85–91.
Steinberg D, Gotto AM (1999) Preventing coronary artery disease by lowering cholesterol levels. J Am Med Assoc 282: 2043–2050.
Castelli WP, Garrison RJ, Wilson PW, Abbott RD, Kalousdian S, kannel WB (1986) Incidence of coronary artery disease and lipoprotein cholesterol levels: the Framingham Study. J Am Med Assoc 256: 2855–2858.
Simon BC, Cummingham LD, Cohen RA (1990) Oxidized low density lipoproteins cause contraction and inhibit endothelium-dependent relaxation in the pig coronary artery. J Clin Invest 86: 75–79.
Berliner JA, Navab M, Fogelman AM, Frank JS, Demer LL, Edwards PA, Watson AD, Lusis AJ (1995) Atherosclerosis: Basic mechanisms. Oxidation, inflammation and genetics. Circulation 91: 2488–2496.
Neaton JD, Blackburn H, Jacobs D, Kuller L, Lee DJ, Sherwin R, Shih J, Stamler J, Wentworth D (1992) Serum cholesterol level and mortality findings for men screened in the multiple risk factor intervention trial.multiple risk factor intervention trial research group. Arch Intern Med 152: 1490–152.
Hong H (2001) Progress of experimental studies on hypoglycemi mechanisms by TCM. J Anhui TCM College 20: 59–63.
Wang L, Li SL, Wang T (2000) Progress on the study of polysaccharides and glucosides of hypoglycemic plants. Chin Med Mater 23: 575–577.
Pan GY, Huang ZJ, Wang GJ, Fawcett JP, Liu XD, Zhao XC, Sun JG, Xie YY (2003) The antihyperglycaemic activity of berberine arises from a decrease of glucose absorption. Planta Med 69: 632–636.
Tiwari AK, Rao JM (2002) Diabetes mellitus and multiple therapeutic approaches of phytochemicals: Present status and future prospects. Current Sci 83: 33–38.
Krolewski AS, Warram JH, Valsania P, Martin BC, Laffel LM, christlieb AR (1991) Evolving natural history of coronary artery disease in diabetes mellitus. Am J Med 90: 56s–61s.
Chattopadhyay RR (1999) A comparative evaluation of some blood sugar lowering agents of plant origin. J Ethnopharmacol 67: 367–362.
Acknowledgement
The authors wish to thank Dr. Rotimi Aluko (Department of Human Nutritional Science, University of Manitoba) for his constructive critique of the manuscript. This study was supported by China Scholarship Council (granted number: NSCIS No. 23110003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
YUAN, L., TU, D., YE, X. et al. Hypoglycemic and Hypocholesterolemic Effects of Coptis chinensis Franch Inflorescence. Plant Foods Hum Nutr 61, 139–144 (2006). https://doi.org/10.1007/s11130-006-0023-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-006-0023-7