Abstract
Historic discoveries and key observations related to Rubisco (Ribulose 1,5-bisphosphate carboxylase/oxygenase), from 1947 to 2006, are presented. Currently, around 200 papers describing Rubisco research are published each year and the literature contains more than 5000 manuscripts on the subject. While trying to ensure that all the major events over this period are recorded, this analysis will inevitably be incomplete and will reflect the areas of particular interest to the authors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In photosynthetic carbon fixation, Rubisco (Ribulose 1,5-bisphosphate carboxylase/oxygenase, EC 4.1.1.39) catalyzes the combination of 1011 metric tons of CO2 each year with ribulose 1,5-bisphosphate (RuBP) (Siegenthaler and Sarmiento 1993). For an overview of the role and functioning of Rubisco in the context of photosynthetic carbon metabolism, see Buchanan et al. (2000), Lodish et al. (2000) and Heldt (2005). Each CO2 fixed in this way forms two molecules of d-3-phosphoglyceric acid (PGA). Rubisco also initiates photorespiration by catalyzing the oxygenation of RuBP to form one molecule each of 2-phosphoglycolate and PGA. In order to catalyze photosynthetic CO2 fixation at high rates, large amounts of Rubisco are needed to compensate for the enzyme’s slow catalytic rate (3–10 s−1). Rubisco accounts for a quarter of leaf nitrogen and up to half of the soluble protein in the leaves of C3 plants. Nevertheless, in bright light, Rubisco usually limits CO2 fixation. Rubisco activity is regulated to match RuBP regeneration with RuBP utilization. Gross regulation of Rubisco activity can be accomplished through alteration of the abundance of the enzyme. The catalytic activity of Rubisco can also be regulated by: (a) the reversible carbamylation of an essential lysine residue on the large subunit, which is subsequently stabilized by a Mg2+ ion forming a catalytically active ternary complex; and (b) by the interaction of both carbamylated and non-carbamylated Rubisco with certain phosphorylated metabolites, which block the active site of the enzyme. Such inhibition is rendered reversible by the action of the ancillary enzyme, Rubisco activase, whose activity promotes the release of such compounds.
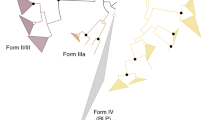
There are two distinct forms (Forms I and II) of Rubisco among photosynthetic Bacteria and Eucarya (Fig. 1). Form II Rubiscos are the simplest, comprised of two identical large subunits, and confined to certain photosynthetic bacteria. Form I Rubiscos contain a central core, composed of four large subunit dimers, as well as eight additional small subunits. Form I Rubiscos are found in plants, algae, cyanobacteria, and autotrophic proteobacteria. In higher plants the small subunits are nuclear encoded, but those for the large subunit are chloroplast encoded. The synthesis and assembly of the subunits is complex and involves molecular chaperones and post-translational modification (Fig. 2). In addition to Forms I and II, there are two groups of structurally related proteins that have been designated as Form III and Form IV Rubiscos which consist only of large subunits (Hanson and Tabita 2001). Form III Rubiscos are found only in Archea and catalyze the carboxylation of RuBP after its formation via alternative pathways to the Calvin–Benson–Bassham cycle. Form IV Rubiscos are found in Bacteria and Archea and are also called Rubisco-like proteins because they lack several key active-site residues necessary for the carboxylation of RuBP.
Molecular structures of photosynthetic Rubiscos—Forms I (L8S8, 8RUC) and II (L2, 9RUB). The other Rubisco forms (III and IV) also have L2 structures, which in one case are arranged in a pentagonal ring (Kitano et al. 2001). Large subunits (L) are colored green or red and the small subunits (S) are colored blue or yellow. Image created with RasWin
Rubisco processing and regulation. Redrawn after Houtz and Portis (2003)
This review provides a historical timeline of important discoveries and observations in Rubisco. We have elected to start in 1947 with the first purification of Rubisco, although by doing so, we realize that we will omit some significant earlier work, later shown to be directly relevant to Rubisco (e.g., Warburg 1920). In addition to the literature provided by this timeline, readers are encouraged to consult some of the earlier personal historical perspectives (e.g., Bassham 2003; Benson 2002; Bogorad 2003; Ellis 2004; Ogren 2003; Tabita 2004; Wildman 2002), comprehensive reviews on Rubisco (Jenson and Bahr 1977; Ellis 1979; Lorimer 1981; Miziorko and Lorimer 1983; Andrews and Lorimer 1987; Morell et al. 1992; Hartman and Harpel 1994; Gutteridge and Gatenby 1995; Spreitzer 1993; Spreitzer 1999; Cleland 1998; Tabita 1999; Spreizer and Salvucci 2002), Rubisco regulation (Portis 1992; Salvucci and Ogren 1996; Parry et al. 1999; Portis 2003) and most recently, reviews evaluating the prospects for increasing photosynthesis by overcoming the limitations of Rubisco by a transgenic approach (Zhu et al. 2004; Raines 2006; Long et al. 2006; Parry et al. 2007).
For timelines of oxygenic and anoxygenic photosynthesis, see Govindjee and Krogmann (2005) and Gest and Blankenship (2005), respectively.
1948–1950
Melvin Calvin (Director of Bio-Organic Chemistry Group) and Andrew Benson (Director of the Photosynthesis Laboratory) at the University of California, Berkeley resume the earlier attempts by Sam Ruben and Martin Kamen using carbon-11 to identify the early products of photosynthesis. They use 14CO2 coupled with anion exchange fractionation initially and subsequently paper chromatography to separate the early products of photosynthesis and identify PGA as the earliest product (Calvin and Benson 1948, 1949; Benson et al. 1950). This and subsequent work resulted in Calvin being awarded the Nobel Prize in Chemistry in 1961. For personal accounts of these experiments, see Calvin (1989), Benson (2002) and Bassham (2003).
Tamiya and Huzisige (1949) conduct studies that lead them to conclude that CO2 and O2 compete at a common site during photosynthesis. Benson and Calvin (1950) observe that oxygen enhances the formation of glycolate from 14CO2 in the light. Intrigued by this report, Hiroshi Tamiya visits Benson in 1952 and they conduct additional experiments using 14CO2 but the results are not published (see Benson 2002).
1951
Benson (1951) identifies ribulose bisphosphate as an early product of photosynthesis and notes that its oxidation in air produces PGA and phosphoglycolate.
1954
Rapid labeling with 14CO2 and analysis of the incorporation patterns in the sugar phosphates results in completion of a cyclic path for the incorporation of carbon in photosynthesis by Calvin’s group (Bassham et al. 1954). Addition of CO2 to the enediol form of RuBP is proposed as the carboxylation step. Rabinowitch (1956) provides a contemporary review of the difficulties and uncertainties along the way in the elucidation of the pathway and the competition between Calvin and other groups particularly that at the University of Chicago (E.W. Fager, J.L. Rosenberg, and Hans Gaffron; see Gaffron 1960), not discussed here. For personal accounts of this research, see Bassham (2003) and Benson (2002).
Using extracts from Chlorella (Quayle et al. 1954) and spinach (Weissbach et al. 1954), simultaneous reports of CO2 fixation appear in the Journal of the American Chemical Society.
1955
Ephraim Racker and Bernard Horecker purify and characterize key enzymes in the “pentose phosphate pathway,” which were also proposed components in Calvin–Benson’s cyclic pathway (see Horecker 2002). Racker (1955) reconstitutes the proposed cyclic pathway using purified preparations of the key enzymes.
Studies of CO2-dependent transients of the photosynthetic intermediates by Wilson and Calvin (1955) reveal a reciprocal relationship between RuBP and PGA and thus provide key support for the proposal that RuBP is the CO2 acceptor in the pathway. Wilson and Calvin (1955) propose that the glycolic acid produced during photosynthesis at low CO2 concentrations is derived from a transketolase reaction.
1956
In a tour de force, Horecker’s lab publishes three consecutive papers reporting: (a) the purification of phosphoribulokinase, a key enzymatic activity required for the cycle (Hurwitz et al. 1956); (b) the enzymatic synthesis of substrate quantities of RuBP using this enzyme (Horecker et al. 1956); and (c) the purification to near homogeneity of an enzyme from spinach that catalyzes the formation of PGA from RuBP and CO2 (Weissbach et al. 1956). Horecker (2002) provides a personal account of the work and states that “with this work and our earlier demonstration of the reversible reactions for the interconversion of pentose and hexose phosphates, with sedoheptulose phosphate as a prominent intermediate, all of the enzymes for the reactions of the Calvin [–Benson] Cycle were identified.”
Horecker’s group uses the term “carboxylation enzyme” in contrast to Calvin’s choice of carboxydismutase (Benson 2002). Further contributing to controversy in naming of the enzyme (including a change from RuDP to RuBP) the International Union of Biochemistry introduces, in 1961, the name 3-phospho-d-glycerate carboxy-lyase (dimerizing) (see Report of the Commission on Enzymes of the International Union of Biochemistry, Oxford. Pergamon, p. 121, 1961). Curiously Horecker’s group does not consider that their purified enzyme might be identical to the Fraction I protein of Sam Wildman.
Immediately following these papers in Journal of Biological Chemistry, a paper from Severo Ochoa’s lab (Jakoby et al. 1956) also reports the formation of RuBP and PGA with partially purified spinach leaf enzymes.
1957
In a report (Dorner et al. 1957) on studies of leaf senescence and the decay of Fraction I protein, Wildman’s group speculates that based on similar physical properties, Fraction I protein and the carboxylation enzyme isolated by Weissbach et al. (1956) are the same. Benson, working with others in Calvin’s group, had concluded that Fraction I protein had carboxylation activity in 1954, but had left the lab and the work was not published until 1957 (Mayaudon 1957; Mayaudon et al. 1957; see Benson 2002).
1958
Moses and Calvin (1958) attempt to trap and identify the proposed six-carbon carboxylation intermediate, 2-carboxy-3-ketopentitol 1,5-bisphosphate. After dephosphorylation, evidence for 2-carboxy-3-ketopentitol was obtained, but 2-carboxy-4-ketopentitol was much more abundant. Almost a quarter of a century passed before the intermediate was successfully trapped (Schloss and Lorimer 1982).
1961
Electron micrographs of spinach chloroplasts show Fraction I protein located in the chloroplast stroma. Fraction I protein in dried samples appeared as oblate spheres 100 Å in height and 200 Å in diameter (Park and Pon 1961). Park and Epstein (1961) demonstrate that Rubisco plays a key role in carbon isotope fractionation by plants.
1962
Bassham and Kirk (1962) report that the inhibition of photosynthesis and formation of glycolate and phosphoglycolate are well correlated with the presence of O2 during photosynthesis by Chlorella. They note that the stimulation of gycolate formation is consistent with the oxidation of the glycolaldehyde-thiamine pyrophosphate addition compound in the transketolase reaction in a metabolic path competing with the formation of RuBP. They also suggest that the increased phosphoglycolate could be explained by oxidation of an early intermediate during the carboxylase reaction.
1963
Calvin’s group discovers “activation” of Rubisco by preincubation with magnesium ion and bicarbonate as well as inhibition with RuBP. An enzyme—Mg—bicarbonate/CO2 complex is proposed as the active form of the enzyme and formation of enzyme-bound CO2 as an intermediate in the reaction is also proposed (Pon et al. 1963; Akoyunoglou and Calvin 1963).
1964
Müllhofer and Rose (1964) demonstrate that cleavage of RuBP in catalysis occurs between carbons two and three by using 14CO2 and 2H2O.
1965
Haselkorn et al. (1965) report the first high-resolution electron micrographs of Fraction I protein (Fig. 3) and they reveal a “central depression” which we now recognize is the “solvent channel hole.” The results were interpreted to suggest a structural model consisting of 24 subunits; later this proposed structure was shown to be incorrect (Sugiyama and Akazawa 1970; Knight et al. 1989, 1990).
Electron micrograph images of Rubisco. (From Haselkorn et al. 1965. Reprinted with permission from American association of Advancement of Science)
Trown (1965) convincingly demonstrates with spinach that Fraction I is crude Rubisco. Thornber et al. (1965) use gel electrophoretic and amino acid analysis to assess homogeneity and reach similar conclusions.
Levine and Togaski (1965) identify the first mutant lacking Rubisco in the green alga Chlamydomonas.
1966
Paulsen and Lane (1966) purify Rubisco to homogeneity from spinach (free of phosphoriboisomerase and phosphoribulokinase). A molecular weight of 557,000 and basic kinetic parameters are reported.
Based on studies of light–dark transients in metabolite levels with Chlorella, Al Bassham’s group (Pedersen et al. 1966) concludes that there is “light activation” of Rubisco and the two bisphosphatase enzymes. Light activation of Rubisco is also indicated by subsequent studies with isolated spinach chloroplasts (Jensen and Bassham 1968). Richard Jensen and Al Bassham suggest that stromal changes in pH and Mg2+ are responsible for the light–dark regulation.
1967
Gel electrophoresis and gel permeation chromatography in the presence of sodium dodecyl sulfate show that Rubisco from spinach is composed of two non-identical subunits (Rutner and Lane 1967; Sugiyama and Akazawa 1967).
Irwin Rose’s group (Fiedler et al. 1967) uses RuBP labeled with tritium at carbon three to demonstrate that proton abstraction is the slow step in the overall reaction and thus provide support for the Calvin group’s proposed reaction mechanism of enolization followed by addition of CO2.
1968
The first reports that Rubisco and not the physical resistance to CO2 diffusion can be a limiting component in leaf photosynthesis appear (Waring et al. 1968; Björkman 1968a, b)
Neales and Incoll (1968) hypothesize that photosynthetic rate is controlled by the amount of assimilate in a leaf.
Rubisco in some microorganisms is found to have a molecular size that is significantly different from Fraction I protein (Anderson et al. 1968).
Evidence is obtained that phytochrome regulates the synthesis of Rubisco and other enzymes (Graham et al. 1968).
Bender (1968, 1971) shows that C3 and C4 plants can be distinguished by carbon isotope content, providing a simple method to classify plants with unknown pathways. These studies apparently were the result of a chance conversation between Margaret Bender and R.H. Burris about technical difficulties in 14C dating of artifacts from plants (Osmond et al. 1994). Carbon isotope fractionation soon became an important tool for understanding photosynthesis and water use efficiency in ecological and physiological studies (see Farquhar et al. 1989 for details).
1969
CO2 and not bicarbonate is shown to be the substrate for Rubisco (Cooper et al. 1969).
1970
Wishnick et al. (1970) report potent tight-binding inhibition by 2-carboxy-d-ribitol 1,5-bisphosphate, an analogue of the proposed six-carbon intermediate in the carboxylation reaction (Calvin 1954). From binding studies of the inhibitor and RuBP, Wishnick et al. (1970) propose a basic structure of eight large and eight small subunits (L8S8) for Rubisco from higher plants. This structure is consistent with contemporary studies determining molecular weight and amino acid composition (see e.g., Sugiyama and Akazawa 1970).
Using the classic inhibitors of protein synthesis, chloramphenicol and cycloheximide, Criddle et al. (1970) conclude that large subunit synthesis occurs in the chloroplast, whereas the small subunit is synthesized in the cytoplasm.
Andersen et al. (1970) identify the first Rubisco mutant in higher plants.
Huffaker et al. (1970) undertake the first studies of the effects of water stress on Rubisco but find little effect on Rubisco activity.
1971
Studies of the oxygen inhibition of isolated soybean Rubisco and comparison with earlier studies (Forrester et al. 1966) of the O2 inhibition of soybean leaf photosynthesis lead Ogren and Bowes (1971) to conclude that Rubisco is responsible for photorespiration. Along with R.H. Hageman, they then discover that Rubisco catalyzes a reaction of the RuBP substrate with oxygen, producing phosphoglycolate (Bowes et al. 1971). Acceptance by some scientists that the oxygenase activity accounted for almost all of the glycolate entering the photorespiratory pathway was slow and controversial, partly because other proposed routes for glycolate formation had earlier received experimental support. Ogren (2003) provides a personal account of this and later contributions by his group.
Holdsworth (1971) reports that pyrenoids present in many algae contain Rubisco.
1972
Using crosses between various tobacco species and tryptic peptide mapping, Sam Wildman’s group demonstrates that the small subunit is encoded in nuclear DNA (Mendelian inheritance), whereas the large subunit is encoded in chloroplast DNA (maternal inheritance) (Kawashima and Wildman 1972; Chan and Wildman 1972).
Zeigler (1972) proposes that SO2 decreases photosynthetic rate by competitive inhibition of Rubisco.
1973
Ed Tolbert’s group confirms that the Rubisco oxygenase reaction produces glycolate in studies using enzymatic synthesis of 14C-RuBP and 18O2/mass spectrometry to characterize the products (Andrews et al. 1973; Lorimer et al. 1973).
Key support for Calvin–Benson’s proposed mechanism of the carboxylation reaction is provided by the chemical synthesis and studies with a key intermediate, 2-carboxy-3-ketoribitol-1,5-bisphosphate, by Siegel and Lane (1973).
Using 35S-methionine, Blair and Ellis (1973) demonstrate the synthesis of the large subunit with isolated pea chloroplasts. This work removed lingering doubts about the previous use of protein synthesis inhibitors to determine sites of Rubisco synthesis.
Shively et al. (1973) discover that the polyhedral inclusions commonly found in blue-green algae (cyanobacteria) and chemoautotrophic bacteria contain Rubisco and propose the name “carboxysomes.”
1974
Tabita and McFadden (1974a, b) report the first homogeneous preparation of single subunit (L2) Rubisco from Rhodospirillum rubrum, which allows a detailed analysis of its properties (see Tabita 2004).
Laing et al. (1974) develop and validate competitive inhibition equations for Rubisco kinetics with CO2 and O2 as substrates by comparing the kinetic properties of soybean net photosynthesis with those of isolated soybean Rubisco. They also examine the effect of temperature on the relative affinities of Rubisco for CO2 and O2 and the relative carboxylase and oxygenase activities, allowing them to account for previous reports of the increase in O2 inhibition and photorespiration with temperature.
Long-standing discrepancies in apparent affinity for CO2 and maximal velocities of carboxylation between the isolated enzyme and chloroplasts/leaves are largely resolved by Bahr and Jensen (1974) and Lilley and Walker (1975).
Sam Wildman and co-workers (Kung et al. 1974) obtain evidence that the Rubisco small subunits might be encoded by more than one gene. This was later confirmed with Southern blotting of restriction digests of soybean nuclear DNA to reveal a multigene family of more than ten members (Berry-Lowe et al. 1982).
1975
John Ellis and co-workers (Hartley et al. 1975) report the first in vitro translation of mRNA for a plant enzyme and it results in Rubisco large subunit synthesis.
David Eisenberg’s group (Baker et al. 1975) obtains a low-resolution structure of tobacco Rubisco that reveals a two-layered structure with 4-2-2 symmetry and a central channel. However, it would be over 10 years before a high-resolution tobacco Rubisco structure is obtained.
Peterson and Huffaker (1975) show that the total soluble protein lost during senescence is accounted for by a decrease in Rubisco amount but that Rubisco is still being synthesized during senescence.
1976
After years of confusion, Lorimer et al. (1976) clearly elucidate the mechanism by which pre-incubation with CO2 and Mg2+ increases the activity of Rubisco.
In a review of Fraction I protein, Kung (1976) states that it is “the most abundant protein in nature.” A subsequent review by Ellis (1979) popularizes this view.
O’Toole et al. (1976) show that Rubisco activity is sensitive to changes in leaf water potential and declines rapidly under severe stress.
Huber et al. (1976) discover that Rubisco is confined to the bundle sheath chloroplasts in C4 plants.
1977
Dobberstein et al. (1977) use a cell-free wheat germ system to translate polyadenylated mRNA from Chlamydomonas and identify a small subunit precursor among the products, which can be processed to the correct size. They propose that the small peptide portion of the precursor plays a role in the transfer of the small unit into the chloroplast. Highfield and Ellis (1978) report similar studies of small subunit synthesis and transport into pea chloroplasts. They postulate a general model for Rubisco synthesis that proves to be applicable to other chloroplast proteins.
1978
Tobin (1978) shows that light increases the small subunit transcript.
In what might be considered an early biotechnology application, Wildman and Kwanyuen (1978) attempt to develop the ease of crystallization of the tobacco enzyme and exploit the abundance of Rubisco for commercial applications.
Several groups study the relationship between loss of Rubisco and the decline in photosynthesis during leaf senescence (Hall et al. 1978; Wittenbach 1979; Wittenbach et al. 1980; Friedrich and Huffaker 1980; Peoples et al. 1980). These studies built upon the earlier studies of the decline of Fraction I protein during senescence (see e.g., Dorner et al. 1957).
Nakamura and Saka (1978) report that ozone exposure reduces the Rubisco activity in rice leaves.
The first symposium devoted exclusively to Rubisco is held at the Brookhaven National Laboratory, Upton, NY with over 150 participants and the proceedings are published (Siegelman and Hind 1978).
1979
David Eisenberg coins the term “Rubisco” at a seminar honoring Sam Wildman (Wildman 2002).
Wong (1979) provides the first report that growth at high-CO2 concentrations causes reduced Rubisco activity.
Pell (1979) suggests that the crystalline structures found in the chloroplast stroma of bean and cotton following ozone fumigation are Rubisco crystals.
Ku et al. (1979) find that C3 species have 3- to 6-fold higher concentrations of Rubisco than C4 species.
1980
Full length nucleotide sequences for the maize large subunit (McIntosh et al. 1980) and pea small subunit (Bedbrook et al. 1980) are obtained. One year earlier, Martin (1979) obtained the amino acid sequence of the small subunit by the classic means of Edman degradation.
Barraclough and Ellis (1980) report the assembly of newly synthesized large subunits into Rubisco in isolated pea chloroplasts.
Using Chlamydomonas, the first structural mutation (in the large subunit) of Rubisco is obtained by Spreitzer and Mets (1980). The mutant has normal levels of Rubisco, but low rates of photosynthesis. Earlier, Nelson and Surzycki (1976) had reported the isolation of a Rubisco mutant, but it was subsequently lost and never fully characterized.
Farquhar et al. (1980) develop their now-classic biochemical model of leaf photosynthesis that merges Rubisco kinetics (developed by Laing et al. 1974), the energy requirements of carbon reduction and photorespiratory carbon oxidation and electron transport, and the dependence of electron transport on light.
Lorimer and Miziorko (1980) follow up their independent reports the previous year that the activator CO2 is discrete from the substrate CO2 and show that carbamate formation on a lysine residue is the basis for activation of the enzyme by CO2 and Mg2+.
Pierce et al. (1980) extend the earlier work by Wishnick et al. (1970) with epimeric transition-state analog mixtures by chemically synthesizing various transition-state analogs of the carboxylation reaction and characterize their interactions in detail. 2-carboxy-d-arabinitol 1,5-bisphosphate (CABP) is found to bind with the highest affinity and almost irreversibly.
1981
Perchorowicz et al. (1981) find that in limiting light, the extractable activity (i.e., activation state) of Rubisco and not the RuBP level changes proportionally with the steady-state photosynthetic rate of leaves. Jensen (2004) later provides a personal account of his contributions. Mächler and Nösberger (1980) performed similar studies examining the effects of light, CO2, and temperature on extractable activity.
Using a sensitive dual-label assay, Jordan and Ogren (1981) show that the relative rates of carboxylation and oxygenation vary in different species, reflecting evolutionary change.
Weis (1981a, b) observes that the light activation of Rubisco is more temperature sensitive (and largely reversible) than electron transport and thus proposes that it plays a role in the temperature regulation of the overall photosynthetic process.
Smith and Ellis (1981) observe that light regulates the accumulation of mRNAs encoding both genes of Rubisco in peas.
Tobin (1981) finds that phytochrome regulates the expression of the small subunit of Rubisco.
Sasaki et al. (1981) show that light increases small subunit mRNA level and the rates of small subunit and Rubisco synthesis proportionately, suggesting nuclear control of Rubisco synthesis by small subunit mRNA level.
Work by several groups (Hatch and Jensen 1980; McCurry et al. 1981; Badger and Lorimer 1981; Jordan et al. 1983) clarifies the role of binding of various sugar phosphates to Rubisco in promoting either increased or decreased activation/carbamylation.
Simpson et al. (1981) follow Rubisco degradation in maize leaves and determine half lives for Rubisco ranging from 5 to 8 days that apparently vary little with the photoperiod.
The first extensive survey (Yeoh et al. 1981) of Rubisco kinetic parameters in diverse plants indicates that Rubiscos in C4 species, aquatic plants, and algae have lower affinity for CO2 than do C3 and CAM species. In contrast, the variation in RuBP affinity does not correlate with the photosynthetic pathway.
1982
Schloss and Lorimer (1982) demonstrate that the transition-state intermediate of the carboxylase reaction is 2-carboxy-3-keto-d-arabinitol 1,5-bisphosphate.
Recognition that enzyme in the absence of CO2 is inactive allows Saver and Knowles (1982) to verify that enolization of RuBP is an intermediate step in the reaction. These results are soon confirmed by 1H NMR (Gutteridge et al. 1984a).
Somerville et al. (1982) isolate an Arabidopsis thaliana mutant that is defective in Rubisco activation in vivo. This mutant soon leads to the discovery of Rubisco activase (Salvucci et al. 1985).
Winter et al. (1982) conduct the first detailed analysis of the amounts of Rubisco and PEP (phosphoenol pyruvate) carboxylase in CAM (Crassulacean Acid Metabolism) plants.
1983
In contrast to previous observations with many species, the first reports (Vu et al. 1983; McDermitt et al. 1983) appear indicating that the “total” extractable Rubisco activity of soybean leaves varies diurnally due to light intensity. These findings would later be extended to several other species and led, in 1986, to the discovery of the “nocturnal inhibitor,” 2-carboxy-d-arbinitol 1-phosphate.
Jordan and Chollet (1983) characterize the tight binding of RuBP to the uncarbamylated and inactive form of Rubisco, clearly showing that it severely inhibits activation by CO2 and Mg2+ and binding constants are determined.
Schmidt and Mishkind (1983) show that there is a proteolytic mechanism in chloroplasts that selectively degrades the imported but unassembled small subunits so that a precise stoichiometric accumulation of both subunits occurs.
Ozone fumigation is shown to lead to a loss of Rubisco protein (Pell and Pearson 1983).
Sasaki et al. (1983) show that phytochrome regulates the expression of both large subunit and small subunit mRNAs.
1984
Coruzzi et al. (1984) show the tissue specific and light regulated expression of one of the small unit genes present in peas.
A pea small unit gene is inserted into the Ti plasmid of Agrobacterium tumefaciens and transferred into petunia cells by in vitro transformation by Broglie et al. (1984). The transcripts were processed correctly and translated to yield a peptide that was located in the chloroplasts. Although the amounts of the foreign small subunit were very low relative to the native small subunit, this work raised the possibility for genetic engineering of the small subunit as well as the expression of foreign genes targeted to the chloroplasts.
Following the cloning and sequencing of the L2 Rubisco found in Rhodospirillum rubrum and its successful expression in E. coli by Chris Somerville’s group (Somerville and Somerville 1984; Nargang et al. 1984), the first site-directed mutagenesis study of Rubisco is reported by Gutteridge et al. (1984b).
Using a sensitive assay procedure, Jordan and Ogren (1984) report that the CO2/O2 specificity of Rubisco decreases with temperature, thus largely explaining the increased inhibition of net photosynthesis by oxygen with increasing temperature. Jordan and Ogren (1984) also obtain a complete data-set on the effects of temperature on the kinetic constants. These results are soon confirmed in vivo by Brooks and Farquhar (1985) using gas-exchange measurements on spinach.
Parry and Gutteridge (1984) report reversible ‘fallover’ inhibition during catalysis by fully activated Rubisco.
Seemann et al. (1984) find that the specific activities of Rubiscos from C4 species and the alga Chlamydonomas are about 2-fold higher than those from C3 species.
1985
Rubisco activase is discovered by Salvucci et al. (1985). Portis and Salvucci (2002) have provided a personal account of the research.
Servaites (1985) discovers that the diurnal variation in Rubisco activity in tobacco is caused by the binding of a phosphorylated inhibitor at night. A similar inhibitor is discovered in garden bean (Phaseolus vulgaris) and characterized (Seemann et al. 1985).
Expression of an active L8S8 enzyme in E. coli is reported by three groups (Gatenby et al. 1985; Tabita and Small 1985; Gutteridge et al. 1986b), allowing future site-directed mutagenesis studies of this form of the enzyme.
Van den Broeck et al. (1985) fuse the transit peptide of the Rubisco small subunit to a foreign protein allowing its expression in the chloroplasts of tobacco. In vitro uptake and processing by isolated pea chloroplasts is also demonstrated by expression of the fusion protein in E. coli.
In a comprehensive study, John Bedbrook’s group (Dean et al. 1985) finds wide variation in small subunit mRNA levels corresponding to the eight genes in petunia in both leaves and other organs.
J.O. Berry et al. (1985) measure polypeptide and mRNA levels of both subunits and conclude that both transcriptional and post-transcriptional controls modulate Rubisco expression in developing amaranth cotyledons.
The second major meeting devoted to Rubisco is held at a Royal Society Discussion Meeting in London, England and the proceedings are published (Ellis and Gray 1986).
1986
The “nocturnal” inhibitor is isolated from potato (Gutteridge et al. 1986a) and garden bean (J.A. Berry et al. 1987) and identified as being 2-carboxy-d-arabinitol 1-phosphate (CA1P).
Servaites et al. (1986) determine the species variation in the predawn inhibition of Rubisco which suggests that CA1P is not produced in the common crop grasses.
The first chloroplast genome is fully sequenced by Shinozaki et al. (1986).
Fluhr and Chua (1986) show that the effects of light on small subunit gene expression depend on the developmental stage of the leaves and to involve both phytochrome and a putative blue-light receptor.
Schneider et al. (1986) obtain a high-resolution structure for Rhodospirillum rubrum Rubisco which consists only of a dimer of large subunits.
Portis et al. (1986) obtain maximal and light-dependent activation of Rubisco at physiological concentrations of CO2 and RuBP with a reconstituted system containing Rubisco, activase and thylakoid membranes and thus resolve the apparent discrepancy between the CO2 dependence of Rubisco activation in vivo and in vitro.
1987
Ritland and Clegg (1987) demonstrate the usefulness of the large subunit gene to reconstruct phylogenetic topologies. A large co-operative effort then emerges to produce a detailed phylogeny of seed plants as reviewed later by Clegg (1993).
Higher resolution (3.0 and 2.6 Å) structures of tobacco Rubisco (L8S8) are obtained by David Eisenberg’s group (Chapman et al. 1987, 1988), but the fold of the small subunit proves to be incorrect.
Richard Jensen, Hans Bohnert, and George Lorimer organize a Rubisco workshop at the University of Arizona, USA with over 40 invited speakers (Fig. 4). Plans for publication of the contributions do not materialize.
1988
Boynton et al. (1988) report plastid gene transformation which raises the prospect of the genetic engineering of the large Rubisco subunit.
John Ellis’ group (Hemmingsen et al. 1988) discovers that an abundant chloroplast protein, involved in the assembly of Rubisco, is related to Escherichia coli groEL and the term “chaperonins” is introduced to describe these proteins (see Ellis 2004).
Werneke et al. (1988) clone the gene encoding Rubisco activase and obtain evidence of aberrant processing in the Arabidopsis rca (activase null) mutant.
Robinson and Portis (1988) show that Rubisco activase can release CA1P from activated Rubisco, restoring its activity.
Brooks and Portis (1988) show that the changes in Rubisco activation state with light intensity in Arabidopsis are correlated with changes in the amount of inactive Rubisco with tightly bound RuBP.
Kobza and Seemann (1988) document variations in the mechanism (nature and amount of the inhibitor) for the light-dependent regulation of Rubisco using three different plant species.
Studies by Sage et al. (1988) of the short-term in vivo response to elevated CO2 of Rubisco and levels of RuBP and other metabolites indicate that changes in Rubisco activation state occur to regulate the pool sizes of key photosynthetic intermediates.
Lawrence Bogorad’s group (Rodermel et al. 1988; also see Bogorad 2003) creates transgenic tobacco plants containing antisense small subunit DNA in the nuclear genome and shows that Rubisco levels and plant growth, but not large subunit mRNA, is reduced. This is one of the earliest applications of antisense technology to reduce native protein expression in plants. These plants are later used in numerous investigations of photosynthesis, plant growth, and development.
1989
A high-resolution structure (2.4 Å) of spinach Rubisco is obtained by Carl-Ivar Brändén’s group (Knight et al. 1989, 1990), which shows the correct fold of the small subunit and gives better assignments of the structural features for L8S8 Rubiscos in plants.
George Lorimer’s group (Goloubinoff et al. 1989) achieves in vitro assembly of a prokaryotic L2 Rubisco (Rhodospirillum rubrum) using E. coli chaperonins (cpn60 and cpn10) and Mg-ATP.
Ed Tolbert’s group (Houtz et al. 1989) establishes the different nature of several post-translational modifications of the Rubisco large subunit using several species.
During a sabbatical in Archie Portis’ lab, Simon Robinson characterizes several key activities of Rubisco activase including ATP hydrolysis (Robinson and Portis 1989a), release of the nocturnal inhibitor, CA1P (Robinson and Portis 1988) and restoration of the slow decline of Rubisco activity with reaction time in vitro (Robinson and Portis 1989b) due to what will eventually be shown to be inhibitor formation during catalysis.
A variety of approaches (Price and Badger 1989; Pierce et al. 1989; Friedberg et al. 1989) demonstrate that Rubisco and carbonic anhydrase localization in the carboxysomes is essential for the CO2 concentrating mechanism in cyanobacteria as proposed by a theoretical model from Aaron Kaplan’s group (Reinhold et al. 1987).
Woodrow and Mott (1989) show that the rate limitation to the increase in photosynthesis, following a step increase in light intensity, is the activation of Rubisco. Subsequently, measurements on plants expressing reduced amounts of activase demonstrate that the rate of increase is proportional to activase content (Mott et al. 1997; Hammond et al. 1998a).
A convincing early report of alternative splicing in plants explains the occurrence of two polypeptides for Rubisco activase of different sizes (Werneke et al. 1989).
Gutteridge and Julien (1989) and Holbrook et al. (1989) demonstrate that the Rubisco inhibitor, CA1P, is degraded in vivo by a specific chloroplast phosphatase to yield 2-carboxyarabinitol (CA). The enzyme is purified to homogeneity by Salvucci and Holbrook (1989).
In an extensive, comparative study of acclimation of photosynthesis to elevated CO2 in five C3 species (Sage et al. 1989), variations in the loss of Rubisco and decline in its activation state are observed.
1990
John Andrew’s group characterizes the biochemical basis of “fallover”, the slow decline in the activity of some Rubiscos with reaction time as being due to side reactions that form the inhibitors, xylulose 1,5-bisphosphate and 3-ketoarabinitol 1-5-bisphosphate, during catalysis (Edmondson et al. 1990a, b, c, d). Zhu and Jenson (1991a, b) make similar observations. Later it becomes clear that 3-ketoarabinitol 1,5-bisphosphate was likely mis-identified in these studies and that the second inhibitor is d-glycero-2,3-pentodiulose 1,5-bisphosphate (Kane et al. 1998).
Pal Maliga’s group creates tobacco lines with stably transformed plastids (Svab et al. 1990) and opens the way to modify the Rubisco large subunit in transgenic plants.
J.O. Berry et al. (1990) find that the mRNAs for both Rubisco subunits do not associate with polysomes in the dark and that neither subunit is synthesized in the dark. These results indicate a lack of translational initiation of both subunits in the dark.
1991
Transgenic plants expressing varying levels of Rubisco are used for a Control Theory analysis of the flux-control coefficients of Rubisco under various environmental conditions (Stitt et al. 1991).
Andrews and Kane (1991) discover that pyruvate is a minor byproduct of Rubisco carboxylation and results from the β-elimination of a phosphate ion.
For the five tomato small subunit (rbcS) genes, Wanner and Gruissem (1991) establish a general correlation between rbcS transcription and abundance but also significant post-translational control of gene expression during development.
Bartholomew et al. (1991) show that abscisic acid, increased by water stress, inhibits rbcS transcription in tomato plants.
Bowes (1991) provides an early, comprehensive review of the role of Rubisco in the responses of plants to elevated CO2.
1992
Read and Tabita (1992) discover a Rubisco from a marine alga with an unexpectedly high CO2/O2 specificity, significantly greater than that observed in terrestrial plants. (See Tabita (2003) for his personal perspective.)
In a comprehensive study (Holiday et al. 1992) of changes in enzyme activity and metabolites in leaves during short and long-term exposure of spinach and bean (chilling-sensitive) to low temperature, Rubisco amount increased in both, but activation state increased in spinach and declined in bean.
1993
Mark Stitt’s group reports (Krapp et al. 1993) detailed experiments to support the hypothesis that photosynthetic gene expression (including the transcription of the Rubisco small subunit) is inhibited by metabolic factors related to a high-carbohydrate content, thus representing a basic mechanism for the ‘sink regulation’ of photosynthesis.
Guy et al. (1993) characterize oxygen isotope discrimination by Rubisco and show it to be an important factor contributing to the ‘Dole effect’—the steady-state difference in the oxygen isotope composition of the atmosphere and seawater.
John Andrew’s group (Mate et al. 1993; Hammond et al. 1998b) demonstrates the role of activase in facilitating the rapid release of CA1P (the nocturnal inhibitor) from Rubisco in vivo by studies of tobacco plants expressing reduced levels of activase via antisense RNA transformation.
In an extensive survey, Moore et al. (1993) find that carboxyarabinitol is present in all of the vascular plants examined but it is not detected in non-vascular plants. Although the highest amounts are in the leaves, carboxyarabinitol is also present in other tissues.
In support of earlier work showing a decrease in Rubisco activity (Nakamura and Saka 1978) and content (Pell 1979) following ozone exposure, Reddy et al. (1993) measure decreased amounts of rbcL and rbcS transcripts.
Parry et al. (1993) measure the diurnal activity of Rubisco in water stressed tobacco plants and suggest a role for inhibitors other than CA1P in Rubisco regulation.
Pilgrim and McClung (1993) demonstrate differential involvement of the circadian clock in the expression of the genes involved in Rubisco synthesis (small subunit), assembly (alpha and beta chaperonin) and activation (Rubisco activase) in Arabidopsis.
Hartman and Harpel (1993) provide a comprehensive review of the numerous chemical modification and site-directed mutagenesis studies of the active site of Rubisco.
1994
Transgenic plants expressing reduced levels of Rubisco allow its kinetics to be inferred from gas-exchange analysis (von Caemmerer et al. 1994), further validating the mechanistic model of Farquhar et al. (1980).
Nuclear transformation of a tobacco line lacking the chloroplast rbcL gene with the rbcL preceded by a plastid targeting sequence functionally complements the defective plastids which partially restores Rubisco activity (Kanevski and Maliga 1994).
1995
Fred Hartman’s group (Harpel et al. 1995) provides evidence for presumed intermediates in the oxygenase reaction by showing that hydrogen peroxide can be eliminated from an improperly stabilized peroxy adduct of the enediol to form d-glycero-2,3-pentodiulose 1,5-bisphosphate in a Rubisco mutant. Kim and Portis (2004) later use hydrogen peroxide formation to characterize oxygen-dependent Rubisco fallover with wildtype enzymes.
In a review, Bainbridge et al. (1995) report the inverse correlation between specificity factor and catalytic rates and the uncertainty surrounding the extent to which these two components can be uncoupled.
At a meeting at Institute of Arable Crops Research-Rothamsted to mark his contributions and retirement, Keys suggests that inhibitors other than CA1P and RuBP regulate Rubisco in vivo (Keys et al. 1995). Speakers at the meeting are shown in Fig. 5.
Speakers at the IACR-Rothamsted meeting in 1994. Front row (left to right): Roger Leigh, Bill Ogren, Peter Lea, Ulf-Ingo Flűgge, Christine Foyer, Alfred Keys, David Walker, Stephen Rawsthorne, Dimah Habash and Tristan Dyer. Middle row: Jerry Servaites, Paul Quick, Hipolito Medrano, Archie Portis, Steve Gutteridge, Barry Osmond, Ulrich Lűttge and John Andrews. Back row: Martin Parry, Richard Leegood, Howard Griffiths, J.J. van Oosten, John Gray, Nick Kruger, Matthew Paul, David Lawlor and Chris Pollock. Not pictured: D.H. Bringlow, H. Jones, M. Chaumont and G.D. Price
1996
Andralojc et al. (1996) propose that CA1P is synthesized from newly assimilated CO2 via hamamelose and 2-carboxyarabinitol.
Studies (Furbank et al. 1996) of transgenic C4 plants expressing reduced levels of Rubisco demonstrate that Rubisco activity is also a major determinant of photosynthesis in C4 plants under high light and air levels of CO2.
Further analysis of rbcS antisense lines allows Rodermel et al. (1996) to clearly show that the abundance of small subunits specifically contributes to the regulation of large subunit accumulation at the level of rbcL translation initiation.
Active oxygen species are shown to stimulate the association of Rubisco with the insoluble fraction of chloroplasts and the specific degradation of Rubisco large subunits into an N-terminal 37-kDa (Desimone et al. 1996) and a C-terminal 16-kDa fragment products (Ishida et al. 1997).
An extensive phylogenetic analysis of Rubisco sequences indicates rampant horizontal transfer and duplication in eubacteria and plastids (Delwiche and Palmer 1996).
1997
Parry et al. (1997) devise measurements of ‘maximal’ Rubisco activity and provide further evidence for the regulation of Rubisco activity by inhibitors other than CA1P but which, like CA1P, can be displaced by SO 2−4 ions.
The research groups of Archie Portis and Robert Spreitzer use site-directed mutagenesis of the large subunit in Chlamydomonas to identify a residue conferring activase preference in its interaction with Rubisco (Larson et al. 1997). Later the same approach identifies another residue (Ott et al. 2000).
1998
Cleland et al. (1998) provide a comprehensive review of all the important work contributing to our current detailed understanding of the carboxylase and oxygenase reaction mechanisms.
Kane et al. (1998) discover that non-enzymatic oxidation of RuBP produces a tight-binding inhibitor of Rubisco, d-glycero-2,3-pentodiulose 1,5-bisphosphate (PDBP). The properties of PDBP match that of the daytime inhibitor reported by Keys et al. (1995). Degradation of RuBP to form inhibitors was first systematically studied by Paech et al. (1978).
Feller et al. (1998) provide evidence that the decline in Rubisco activation state at moderately high temperatures is associated with direct effects of temperature on Rubisco activase.
1999
Zhang and Portis (1999) report redox regulation of the ATP/ADP response of the larger activase isoform by thioredoxin-f and use site-directed mutagenesis to identify the critical cysteines. The results indicate that both ATP/ADP ratio and the redox state of the stroma are used to control Rubisco activity and this provides a mechanism for the light intensity dependence of Rubisco activation (see Perchorowicz et al. 1981).
Chloroplast transformation is used for targeted replacement of the large subunit of Rubisco in a higher plant (tobacco), advancing the prospect of improving photosynthesis by the genetic engineering of Rubisco (Kanevski et al. 1999).
Khan et al. (1999) show that CA1P binding protects activated Rubisco against proteolytic breakdown.
Two groups (Watson et al. 1999; Ezaki et al. 1999) characterize novel forms of Rubisco (later called Form III) from the Archaea.
2000
Crafts-Brandner and Salvucci (2000) demonstrate that at elevated temperatures the rate of Rubisco deactivation exceeds the capacity of Rubisco activase to promote activation. Gas-exchange studies show that when adjustments are made for the decline in activation state, the kinetic properties of Rubisco predict the response of photosynthesis to high temperature and CO2.
A comparison by John Andrews group (Duff et al. 2000) of high-resolution structures for Rubisco complexed with various substrate analogs leads them to conclude that phosphate binding per se does not trigger closure, but that if the distance between the terminal phosphates (P1 and P2) falls below 9.1 Å the active site closes.
The “International Symposium on Photosynthetic CO2-Assimilating Enzymes: RuBisCO and PEPC” is held at Hakone, Japan.
2001
Gas-exchange analysis of transgenic tobacco plants, with a low level of Rubisco, allows improved temperature response functions to be derived for models of Rubisco-limited photosynthesis (Bernacchi et al. 2001).
Hanson and Tabita (2001) identify a fourth form of Rubisco that lacks several conserved active-site residues. The Rubisco-like protein (RLP) in Chlorobium tepidum is shown to have no carboxylase activity and mutants have a pleiotropic phenotype suggesting that the RLP is involved in sulfur metabolism and the response to oxidative stress.
The structure of a Form III Rubisco with pentagonal symmetry (L2 × 5) from the hyperthermophilic archaeon Thermococcus kodakaraensis is reported (Kitano et al. 2001).
To test whether a foreign Rubisco can support photosynthesis in a transgenic plant, Whitney and Andrews (2001a) employ plastid transformation to replace tobacco L8S8 Rubisco with the simple L2, R. rubrum Rubisco and confirm that it can function effectively within the tobacco chloroplast.
Whitney and Andrews (2001b) show that rbcS gene can be relocated to the chloroplast, expressed, and correctly assembled into functional Rubisco. However the plastid transgenes never exceeded 1% of the total small subunits.
2002
Transformation of an Arabidopsis mutant (rca) lacking Rubisco activase with cDNAs encoding different forms of Rubisco activase definitively shows that redox regulation of the larger activase isoform is responsible for the regulation of Rubisco activity by limiting light in species containing both isoforms (Zhang et al. 2002).
Using classic radio-tracer studies and the analysis of transgenic plants with decreased chloroplastic fructose 1,6-bisphosphate phosphatase expression, Andralojc et al. (2002) confirm the biosynthetic pathway for CA1P from newly assimilated carbon.
Sage (2002) provides an insightful review of the current understanding of Rubisco control over C3 and C4 photosynthesis at different temperature and CO2 conditions and documents how Rubisco kinetic properties evolved to improve performance in the environment that the plants normally experience.
2003
A Rubisco-like protein (Form IV) in a non-photosynthetic bacteria (Bacillus subtilis), which lacks several critical residues for carboxylation, is shown to catalyze an enolase reaction in a methionine salvage pathway. Furthermore a growth-defective mutant is rescued by the presence of the Form II Rubisco from Rhodospirillum rubrum. It is suggested that Rubisco-like proteins may be the ancestral enzymes of those found in photosynthetic bacteria (Ashida et al. 2003).
2004
Rubiscos (Form III) in some anaerobic archaea are catalytically active but lack the enzyme phosphoribulokinase, necessary to form RuBP. Finn and Tabita (2004) provide evidence for an alternative route for RuBP formation using 5-phospho-d-ribose-1-pyrophosphate. Most recently, another Form III Rubisco was found to participate in adenosine 5′-monophosphate metabolism (Sato et al. 2007).
Rubisco without a complete Calvin–Benson–Bassham cycle is shown to improve the carbon efficiency of Brassica nappus L. embryos in the conversion of carbohydrate to oil (Schwender et al. 2004).
Onizuka et al. (2004) discover that the rbcX gene product can function as a Rubisco chaperonin by promoting the production and assembly of Synechococcus Rubisco expressed in E. coli.
Kim and Portis (2004) report oxygen-dependent H2O2 production by Rubisco.
2005
Robert Spreitzer’s group identifies a small set of residues that can impart land-plant kinetic properties to Chlamydomonas Rubisco (Spreizter et al. 2005).
Li et al. (2005) identify two residues of Rubisco activase that are involved in substrate recognition of Rubisco.
Consistent with the hypothesis that C3 plants growing in hot, arid conditions may have evolved forms of Rubisco with higher specificity factors, Galmes et al. (2005) identify two Mediterranean species of the genus Limonium whose constituent Rubiscos have high specificity factors.
Figure 6 shows a photograph of Spencer Whitney, Martin Parry, Archie Portis, Akiho Yokota, Inger Andersson, and Mike Salvucci, at an informal workshop, discussing highlights of research on Rubisco.
2006
Mutation and directed evolution of recombinant Synechococcus with selection in an E. coli strain engineered to require Rubisco catalytic activity, identifies variants with increased catalytic activity (Parikh et al. 2006).
Carré-Mlouka et al. (2006) discover that the planktonic cyanobacterium Microcystis contains both Form I and Form IV enzymes. The Form IV enzyme does not display CO2 fixation activity but catalyzes the enolization of 2,3-diketo-5-methylthiopentyl-1-phosphate, a key step in the methionine salvage pathway in bacteria.
Tcherkez et al. (2006) re-examine the inverse correlation between specificity factor and catalytic rates reported by Bainbridge et al. (1995) and conclude that Rubiscos are well adapted to their differing subcellular environments.
Interest in catalytic fallover is renewed. Consistent with earlier experiments (Salvucci and Crafts-Brandner 2004) showing that the synthesis of the catalytic misfire product, xylulose-1,5-bisphosphate, increases with temperature, Kim and Portis (2006) demonstrate that Rubisco fallover is facilitated by high temperature, a low concentration of CO2 or Mg++, and high O2. Schrader et al. (2006) also report increased production of misfire products at elevated temperatures but suggest that structural flexibility at higher temperatures releases the inhibitors more easily and decreases fallover.
Diversions and dead-ends
In many areas of research, provocative papers appear that later become discredited or remain unresolved. Some of the more interesting ones in Rubisco research are presented below.
-
As an alternative to the ene-diol intermediate, the formation of covalent bond between the RuBP substrate and enzyme via formation of a thiohemiacetal intermediate is proposed (Rabin and Trown 1964).
-
Stimulation of Rubisco activity by a small pigment-containing protein (absorbance maximum at 325 nm) is reported by Wildner and Criddle (1969).
-
Kung and Marsho (1976) suggest that the small subunit modifies the relative rates of carboxylation and oxygenation and that the oxygenase activity is probably not the principal source of phosphoglycolate in vivo.
-
Garrett (1978) suggests that increases in ploidy increases the specificity of Rubisco thereby decreasing photorespiratory losses.
-
Several groups claim that chemicals and other treatments can modify the relative rates of carboxylation and oxygenation (Wildner and Henkel 1976; Bhagwat and Sane 1978; Bhagwat et al. 1978; Okabe et al. 1979; Wildner and Henkel 1980; Van Assche et al. 1987; Schmid et al. 1987).
-
Brändén (1978) claims that the carboxylase and oxgenase activities are not catalyzed by the same enzyme and that l-3-phosphoglyceric acid formed by the enzyme is the primary substrate for photorespiration (Brändén 1978; Brändén et al. 1980a, b).
-
Kaul et al. (1986) claim that dephosphorylation of the Rubisco small subunits results in dissociation of the small subunits from the large subunits and a large loss in catalytic activity.
-
A non-cyclic pathway for carbon fixation is proposed by Fong and Butcher (1988).
-
Anwaruzzaman et al. (1996) propose a non-chloroplast location for CA1P.
-
Supercomplexes containing variable amounts of Rubisco and other stromal proteins that might allow metabolic channeling and/or regulation have been a recurring topic in Rubisco research. A review of research in this area is provided by Winkel (2004). However a general consensus on the significance, composition, and function of the Rubisco-containing complexes awaits future research.
We end this historical perspective by honoring several key players in Rubisco research (Sam G. Wildman, N. E. (Ed) Tolbert, Richard G. Jensen, William L. Ogren, T. John Andrews, George H. Lorimer, and Fred C. Hartman) by showing their photographs (Fig. 7).
References
Akoyunoglou G, Calvin M (1963) Mechanism of the carboxydismutase reaction. II. Carboxylation of the enzyme. Biochem Zeits 338:20–30
Anderson LE, Price GB, Fuller RC (1968) Molecular diversity of the ribulose-1,5-diphosphate carboxylase from photosynthetic microorganisms. Science 161:482–484
Andersen WR, Wildner GF, Criddle RS (1970) Ribulose diphosphate carboxylase: III. Altered forms of ribulose diphosphate carboxylase from mutant tomato plants. Arch Biochem Biophys 137:84–90
Andralojc PJ, Keys AJ, Martindale W, Dawson GW, Parry MAJ (1996) Conversion of d-hamamelose into 2-carboxy-d-arabinitol and 2-carboxy-d-arabinitol 1-phosphate in leaves of Phaseolus vulgaris L. J Biol Chem 271:26803–26809
Andralojc PJ, Keys AJ, Kossman J, Parry MAJ (2002) Elucidating the biosynthesis of 2-carboxyarabinitol 1-phosphate through reduced expression of chloroplastic fructose 1,6-bisphosphate phosphatase and radio tracer studies using 14CO2. Proc Natl Acad Sci USA 99:4742–4747
Andrews TJ, Kane HJ (1991) Pyruvate is a by-product of catalysis by ribulosebisphosphate carboxylase/oxygenase. J Biol Chem 266:9447–9452
Andrews TJ, Lorimer GH (1987) Rubisco: structure, mechanisms, and prospects for improvement. In: Hatch MD, Boardman NK (eds) The biochemistry of plants, vol 10. Academic Press, New York, pp 131–218
Andrews TJ, Lorimer GH, Tolbert NE (1973) Ribulose diphosphate oxygenase. I. Synthesis of phosphoglycolate by fraction 1 protein of leaves. Biochemistry 12:1–18
Anwaruzzaman, Nakano Y, Yokota A (1996) Different location in dark-adapted leaves of Phaseolus vulgaris of ribulose-1,5-bisphosphate carboxylase/oxygenase and 2-carboxyarabinitol 1-phosphate. FEBS Lett 388:223–227
Ashida H, Saito Y, Kojima C, Kobayashi K, Ogasawara N, Yokota A (2003) A functional link between RuBisCO-like protein of Bacillus and photosynthetic RuBisCO. Science 302:286–290
Badger MR, Lorimer GH (1981) Interaction of sugar phosphates with the catalytic site of ribulose-1,5-bisphosphate carboxylase. Biochemistry 20:2219–2225
Bahr JT, Jensen RG (1974) Ribulose disphosphate carboxylase from freshly ruptured spinach chloroplasts having an in vivo Km[CO2]. Plant Physiol 53:39–44
Bainbridge G, Madgwick P, Parmar S, Mitchell R, Paul M, Pitts J, Keys AJ, Parry MAJ (1995) Engineering Rubisco to change its catalytic properties. J Exp Bot 46:1269–1276
Baker TS, Eisenberg D, Eiserling FA, Weissman L (1975) The structure of form I crystals of d-ribulose-1,5-diphosphate carboxylase. J Mol Biol 91:391–398
Barraclough R, Ellis RJ (1980) Protein synthesis in chloroplasts. XI. Assembly of newly-synthesized large subunits into ribulose bisphosphate carboxylase in isolated pea chloroplasts. Biochim Biophys Acta 608:19–31
Bartholomew DM, Bartley GE, Scolnik P (1991) Abscisic acid control of rbcS and cab transcription in tomato leaves. Plant Physiol 96:291–296
Bassham JA (2003) Mapping the carbon reduction cycle: a personal retrospective. Photosynth Res 76:35–52
Bassham JA, Kirk M (1962) The effect of oxygen on the reduction of CO2 to glycolic acid and other products during photosynthesis by Chlorella. Biochem Biophys Res Commun 9:376–380
Bassham JA, Benson AA, Kay LD, Harris AZ, Wilson AT, Calvin M (1954) The path of carbon in photosynthesis. XXI. The cyclic regeneration of carbon dioxide acceptor. J Am Chem Soc 76:1760–1770
Bedbrook JR, Smith SM, Ellis RJ (1980) Molecular cloning and sequencing of cDNA encoding the precursor to the small subunit of chloroplast ribulose-1,5-bisphosphate carboxylase. Nature 287:692–697
Bender MM (1968) Mass spectrophotometric studies of carbon-13 variation in corn and other grasses. Radiocarbon 10:468–472
Bender MM (1971) Variations in the 13C/12C ratios of plants in relation to the pathway of photosynthetic carbon dioxide fixation. Phytochemistry 10:1239–1244
Benson AA (1951) Identification of ribulose in C14O2 photosynthesis products. J Am Chem Soc 73:2971–2972
Benson AA (2002) Following the path of carbon in photosynthesis: a personal story. Photosynth Res 73:29–49
Benson AA, Calvin M (1950) The path of carbon in photosynthesis. VII. Respiration and photosynthesis. J Exp Bot 1:63–68
Benson AA, Bassham JA, Calvin M, Goodale TC, Hass VA, Stepka W (1950) The path of carbon in photosynthesis. V. Paper chromatography and radioautography of the products. J Am Chem Soc 72:1710–1718
Bernacchi CJ, Singsaas EL, Pimentel C, Portis AR Jr, Long SP (2001) Improved temperature response functions for models of Rubisco-limited photosynthesis. Plant Cell Environ 24:253–259
Berry JA, Lorimer GH, Pierce J, Seemann JR, Meek J, Freas S (1987) Isolation, identification, and synthesis of 2-carboxyarabinitol 1-phosphate, a diurnal regulator of ribulose-bisphosphate carboxylase activity. Proc Natl Acad Sci USA 84:734–738
Berry JO, Nikolau BJ, Carr JP, Klessig DF (1985) Transcriptional and post-transcriptional regulation of ribulose 1,5-bisphosphate carboxylase gene expression in light- and dark-grown amaranth cotyledons. Mol Cell Biol 5:2238–2246
Berry JO, Breiding DE, Klessig DF (1990) Light-mediated control of translational initiation of Ribulose-1,5-bisphosphate carboxylase in Amaranth Cotyledons. Plant Cell 2:795–803
Berry-Lowe SL, McKnight TD, Meagher RB (1982) The nucleotide sequence, expression and evolution of one member of a multigene family encoding the small subunit of ribulose-1,5-bisphosphate carboxylase in soybean. J Mol Appl Genet 1:483–498
Bhagwat AJ, Sane PV (1978) Evidence for the involvement of superoxide anions in the oxygenase reaction of ribulose-1,5-diphosphate carboxylase. Biochem Biophys Res Commun 84:865
Bhagwat AJ, Ramakrishna J, Sane PV (1978) Specific inhibition of oxygenase activity of ribulose-1,5-diphosphate carboxylase by hydroxylamine. Biochem Biophys Res Commun 83:954–962
Björkman O (1968a) Carboxydismutase activity in shade-adapted and sun-adapted species of higher plants. Physiol Plant 21:1–10
Björkman O (1968b) Further studies of photosynthetic properties in sun and shade ecotypes of Solidago virgaurea. Physiol Plant 21:84–99
Blair GE , Ellis RJ (1973) Protein synthesis in chloroplasts. I. Light-driven synthesis of the large subunit of Fraction I protein by isolated pea chloroplasts. Biochim Biophys Acta 319:223–224
Bogorad L (2003) Photosynthesis research: advances through molecular biology—the beginnings, 1975–1980s and on. Photosynth Res 76:13–33
Bowes G (1991) Growth at elevated CO2: photosynthetic responses mediated through Rubisco. Plant Cell Environ 14:795–806
Bowes G, Ogren WL, Hageman RH (1971) Phosphoglycolate production catalyzed by ribulose 1,5-diphosphate carboxylase. Biochem Biophys Res Commun 45:716–722
Boynton JE, Gillham NW, Harris EH, Hosler JP, Johnson AM, Jones AR, Randolphanderson BL, Robertson D, Klein TM, Shark KB, Sanford JC (1988) Chloroplast transformation in Chlamydomonas with high velocity microprojectiles. Science 240:1534–1538
Brändén R (1978) Ribulose-1,5-bisphosphate carboxylase and oxygenase from green plants are two different enzymes. Biochem Biophys Res Commun 81:539–546
Brändén R, Nilsson T, Styring S (1980a) The formation of l-3-phosphoglyceric acid by ribulose-1,5-bisphosphate carboxylase. Biochem Biophys Res Commun 92:1297–1305
Brändén R, Nilsson T, Styring S, Ångström J (1980b) l-3-phosphoglyceric acid, formed by ribulose-1,5-bisphosphate carboxylase, is the primary substrate for photorespiration. Biochem Biophys Res Commun 92:1306–1312
Broglie R, Coruzzi G, Fraley RT, Rogers SG, Horsch RB, Niedermeyer JG, Fink CL, Flick JS, Chua N-H (1984) Light-regulated expression of a pea ribulose-1,5-bisphosphate carboxylase small subunit gene in transformed plant cells. Science 224:838–843
Brooks A, Farquhar GD (1985) Effect of temperature on the CO2/O2 specificity of ribulose 1,5-bisphosphate carboxylase/oxygenase and the rate of respiration in the light. Estimates from gas-exchange measurements on spinach. Planta 165:397–406
Brooks A, Portis AR Jr (1988) Protein-bound ribulose bisphosphate correlates with deactivation of ribulose bisphosphate carboxylase in leaves. Plant Physiol 87:244–249
Buchanan BB, Gruissem W, Jones RL (eds) (2000) Biochemistry & molecular biology of plants. American Society of Plant Physiologists, Maryland
Calvin M (1954) Chemical and photochemical reactions of thioctic acid and related disulfides. Fed Proc 13:697–711
Calvin M (1989) Forty years of photosynthesis and related activities. Photosynth Res 21:3–16
Calvin M, Benson AA (1948) The path of carbon in photosynthesis. Science 107:476–480
Calvin M, Benson AA (1949) The path of carbon in photosynthesis IV: the identity and sequence of the intermediates in sucrose synthesis. Science 109:140–142
Carré-Mlouka A, Méjean A, Quillardet P, Ashida H, Saito Y, Yokota A, Callebaut I, Sekowska A, Dittmann E, Bouchier C, Tandeau de Marsac N (2006) A new Rubisco-like protein coexists with a photosynthetic Rubisco in the planktonic cyanobacteria Microcystis. J Biol Chem 281:24462–24471
Chan P-K, Wildman SG (1972) Chloroplast DNA codes for the primary structure of the large subunit of Fraction I protein. Biochim Biophys Acta 277:677–680
Chapman M, Suh SW, Cascio D, Smith WW, Eisenberg D (1987) Sliding-layer conformational change limited by the quaternary structure of plant Rubisco. Nature 329:354–356
Chapman MS, Shu SW, Curmi PMG, Cascio D, Smith WW, Eisenberg DS (1988) Tertiary structure of plant Rubisco—Domains and their contacts. Science 24:71–74
Clegg MT (1993) Chloroplast gene sequences and the study of plant evolution. Proc Natl Acad Sci USA 90:363–367
Cleland WW, Andrews TJ, Gutteridge S, Hartman FC, Lorimer GH (1998) Mechanism of Rubisco: the carbamate as general base. Chem Rev 98:549–561
Cooper TG, Filmer D Wishnick M, Lane MD (1969) The active species of “CO2” utilized by ribulose diphosphate carboxylase. J Biol Chem 244:1081–1083
Coruzzi G, Broglie R, Edwards C, Chua N-H (1984) Tissue-specific and light-regulated expression of a pea nuclear gene encoding the small subunit of ribulose-1,5-bisphosphate carboxylase. EMBO J 3:1671–1679
Crafts-Brandner SJ, Salvucci ME (2000) Rubisco activase constrains the photosynthetic potential of leaves at high temperature and CO2. Proc Natl Acad Sci USA 97:13430–13435
Criddle RS, Dau B, Kleinhof GE, Huffaker RC (1970) Differential synthesis of ribulosediphosphate carboxylase subunits. Biochem Biophys Res Commun 41:621–627
Dean C, van den Elzen P, Tamaki S, Dunsmuir P, Bedbrook J (1985) Differential expression of the eight genes of the petunia ribulose bisphosphate carboxylase small subunit multi-gene family. EMBO J 4:3055–3061
Delwiche CF, Palmer JD (1996) Rampant horizontal transfer and duplication of rubisco genes in eubacteria and plastids. Mol Biol Evol 13:873–882
Desimone M, Henke A, Wagner E (1996) Oxidative stress induces partial degradation of the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase in isolated chloroplasts of barley. Plant Physiol 111:789–796
Dobberstein B, Blobel G, Chua N-H (1977) In vitro synthesis and processing of a putative precursor for the small subunit of ribulose-1,5-bisphosphate carboxylase of Chlamydomonas reinhardtii. Proc Natl Acad Sci USA 74:1082–1085
Dorner RW, Kahn A, Wildman SG (1957) The proteins of green leaves. VII. Synthesis and decay of the cytoplasmic proteins during the life of the tobacco leaf. J Biol Chem 229:945–952
Duff AP, Andrews TJ, Curmi PMG (2000) The transition between the open and closed states of rubisco is triggered by the inter-phosphate distance of the bound bisphosphate. J Mol Biol 298:903–916
Edmondson DL, Badger MR, Andrews TJ (1990a) A kinetic characterization of slow inactivation of ribulosebisphosphate carboxylase during catalysis. Plant Physiol 93:1376–1382
Edmondson DL, Badger MR, Andrews TJ (1990b) Slow inactivation ribulosebisphosphate carboxylase during catalysis is not due to decarbamylation of the catalytic site. Plant Physiol 93:1383–1389
Edmondson DL, Badger MR, Andrews TJ (1990c) Slow inactivation ribulosebisphosphate carboxylase during catalysis is caused by accumulation of a slow, tight-binding inhibitor at the catalytic site. Plant Physiol 93:1390–1397
Edmondson DL, Kane HJ, Andrews TJ (1990d) Substrate isomerization inhibits ribulosebisphosphate carboxylase during catalysis. FEBS Lett 260:62–66
Ellis RJ (1979) The most abundant protein in the world. Trends Biochem Sci 4:241–244
Ellis RJ (2004) Chloroplasts to chaperones: how one thing led to another. Photosynth Res 80:333–343
Ellis RJ, Gray JC (eds) (1986) Ribulose bisphosphate carboxylase-oxygenase. The Royal Society, London
Ezaki S, Maeda N, Kishimoto T, Atomi H, Imanaka T (1999) Presence of a structurally novel type ribulose-bisphosphate carboxylase/oxygenase in the hyperthermophilic archaeon, Pyrococcus kodakaraensis KOD1. J Biol Chem 274:5078–5082
Farquhar GD, von Caemmerer, Berry JA (1980) A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149:78–90
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 40:503–537
Feller U, Crafts-Brandner SJ, Salvucci ME (1998) Moderately high temperatures inhibit ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) activase-mediated activation of Rubisco. Plant Physiol 116:539–546
Fiedler F, Müllhofer G, Trebst A, Rose IA (1967) Mechanism of ribulose-diphosphate carboxydismutase reaction. Eur J Biochem 1:395–399
Finn MW, Tabita FR (2004) Modified pathway to synthesize ribulose 1,5-bisphosphate in methanogenic archaea. J Bacteriol 186:6360–6366
Fluhr R, Chua N-H (1986) Developmental regulation of two genes encoding ribulose-bisphosphate carboxylase small subunit in pea and transgenic petunia plants: phytochrome response and blue-light induction. Proc Natl Acad Sci USA 83:2358–2362
Fong FK, Butcher KA (1988) Non-cyclic photoreductive carbon fixation in photosynthesis. Light and dark transients of the glycerate-3-P special pair. Biochem Biophys Res Commun 150:399–404
Forrester ML, Krotkov G, Nelson CD (1966) Effect of oxygen on photosynthesis, photorespiration, and respiration in detached leaves. I. Soybean Plant Physiol 41:422–427
Friedberg D, Kaplan A, Ariel R, Kessel M, Seijffers J (1989) The 5’-flanking region of the gene encoding the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase is crucial for growth of the cyanobacterium Synechococcus sp. strain PCC 7942 at the level of CO2 in air. J Bacteriol 171:6069–6076
Friedrich JW, Huffaker RC (1980) Photosynthesis, leaf resistances, and ribulose-1,5-bisphosphate carboxylase degradation in senescing barley leaves. Plant Physiol 65:1103–1107
Furbank RT, Chitty JA, von Caemmerer S, Jenkins CLD (1996) Antisense RNA inhibition of rbcS gene expression reduces Rubisco level and photosynthesis in the C4 plant Flaveria bidentis. Plant Physiol 111:725–734
Gaffron H (1960) Energy storage: photosynthesis. In: Steward FC (ed) Plant physiology. Academic Press, New York, pp 136–160
Galmes J, Flexas J, Keys AJ, Cifre J, Mitchell RAC, Madgwick PJ, Haslam RP, Medrano H, Parry MAJ (2005) Rubisco specificity factor tends to be larger in plant species from drier habitats and with persistent leaves. Plant Cell Environ 28:571–579
Garrett MK (1978) Different specificity/photorespiration in diploid vs. tetraploid wheat. Nature 274:913–915
Gatenby AA, van der Vies SM, Bradley D (1985) Assembly in E. coli of a functional multi-subunit ribulose bisphosphate carboxylase from a blue-green alga. Nature 314:617–620
Gest H, Blankenship RE (2005) Time line of discoveries: anoxygenic bacterial photosynthesis. In: Govindjee, Beatty JT, Gest H, Allen JF (eds) Discoveries in photosynthesis. Advances in photosynthesis and respiration, vol 20. Springer, Dordrecht, pp 51–62
Goloubinoff P, Christeller JT, Gatenby AA, Lorimer GH (1989) Reconstitution of active dimeric ribulose bisphosphate carboxylase from an unfolded state depends on two chaperonin proteins and Mg-ATP. Nature 342:884–889
Govindjee, Krogmann D (2005) Discoveries in oxygenic photosynthesis (1727–2003): a perspective. In: Govindjee, Beatty JT, Gest H, Allen JF (eds) Discoveries in photosynthesis. Advances in photosynthesis and respiration, vol 20. Springer, Dordrecht, pp 63–105
Graham D, Grieve AM, Smillie RM (1968) Phytochrome as the primary photoregulator of the synthesis of Calvin Cycle enzymes in etiolated pea seedlings. Nature 218:89–90
Gutteridge S, Gatenby AA (1995) Rubisco synthesis, assembly, mechanism, and regulation. Plant Cell 7:809–819
Gutteridge S, Julien B (1989) A phosphatase from chloroplast stroma of Nicotiana tabacum hydrolyses 2’-carboxyarabinitol 1-phosphate, the natural inhibitor of Rubisco to 2’-carboxyarabinitol. FEBS Lett 254:225–230
Gutteridge S, Parry MAJ, Schmidt CNG, Feeney J (1984a) An investigation of ribulosebisphosphate carboxylase activity by high-resolution H1-NMR. FEBS Lett 170:355–359
Gutteridge S, Sigal I, Thomas B, Arentzen R, Cordova A, Lorimer G (1984b) A site-specific mutation within the active site of ribulose-1,5-bisphosphate carboxylase of Rhodospirillum rubrum. EMBO J 3:2737–2743
Gutteridge S, Parry MAJ, Burton S, Keys AJ, Mudd A, Feeney J, Servaites JC, Pierce J (1986a) A nocturnal inhibitor of carboxylation in leaves. Nature 324:274–276
Gutteridge S, Phillips AL, Kettleborough CA, Parry MAJ, Keys AJ (1986b) Expression of bacterial Rubisco genes in Escherichia coli. Phil Trans R Soc Lond B 313:433–445
Guy RD, Fogel ML, Berry JA (1993) Photosynthetic fractionation of the stable isotopes of oxygen and carbon. Plant Physiol 101:37–47
Hall NP, Keys AJ, Merrett MJ (1978) Ribulose-1,5-bisphosphate carboxylase protein during flag leaf senescence. J Exp Bot 29:31–37
Hammond ET, Andrews TJ, Mott KA, Woodrow IE (1998a) Regulation of Rubisco activation in antisense plants of tobacco containing reduced levels of Rubisco activase. Plant J 14:101–110
Hammond ET, Andrews TJ, Woodrow IE (1998b) Regulation of ribulose-1,5-bisphosphate carboxylase/oxygenase by carbamylation and 2-carboxyarabinitol 1-phosphate in tobacco: insights from studies of antisense plants containing reduced amounts of Rubisco activase. Plant Physiol 118:1463–1471
Hanson TE, Tabita FR (2001) A ribulose-1,5-bisphosphate carboxylase/oxygenase (RubisCO)—like protein from Chlorobium tepidum that is involved with sulfur metabolism and the response to oxidative stress. Proc Natl Acad Sci USA 98:4397–4402
Harpel MR, Serpersu EH, Lamerdin JA, Huang Z-H, Gage DA, Hartman FC (1995) Oxygenation mechanism of ribulose-bisphosphate carboxylase/oxygenase. Structure and origin of 2-carboxytetritol 1,4-bisphosphate, a novel O2-dependent side product generated by a site-directed mutant. Biochemistry 34:11296–11306
Hartley MR, Wheeler A, Ellis RJ (1975) Protein synthesis in chloroplasts. V. Translation of messenger RNA for the large subunit of fraction I protein in a heterologous cell-free system. J Mol Biol 91:67–77
Hartmen FC, Harpel MR (1993) Chemical and genetic probes of the active-site of d-ribulose-1,5-bisphosphate carboxylase oxygenase—a retrospective based on the 3-dimensional structure. Adv Enzymol 67:1–75
Hartman FC, Harpel MR (1994) Structure, function, regulation, and assembly of d-ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem 63:197–234
Haselkorn RR, Fernández-Morán H, Kieras FJ, van Bruggen EFJ (1965) Electron microscopic and biochemical characterization of Fraction 1 protein. Science 150:1598–1601
Hatch AL, Jensen RG (1980) Regulation of ribulose-1,5-bisphosphate carboxylase from tobacco: changes in pH response and affinity for CO2 and Mg2+ induced by chloroplast intermediates. Arch Biochem Biophys 205:587–594
Heldt HW (2005) Plant biochemistry, 3rd edn. Elsevier Academic Press, Amsterdam
Hemmingsen SM, Woolford C, van der Vies SM, Tilly K, Dennis DT, Georgopoulos CP, Hendrix RW, Ellis RJ (1988) Homologous plant and bacterial proteins chaperone oligomeric protein assembly. Nature 333:330–334
Highfield PE, Ellis RJ (1978) Synthesis and transport of the small subunit of chloroplast ribulose bisphosphate carboxylase. Nature 271:420–424
Holiday AS, Martindale W, Alred R, Brooks AL, Leegood RC (1992) Changes in activities of enzymes of carbon metabolism in leaves during exposure of plants to low temperature. Plant Physiol 98:1105–1114
Holbrook GP, Bowes G, Salvucci ME (1989) Degradation of 2-carboxyarabinitol 1-phosphate by a specific chloroplast phosphatase. Plant Physiol 90:673–678
Holdsworth RH (1971) The isolation and the partial characterization of the pyrenoid protein from Eremosphaera viridis. J Cell Biol 15:499–513
Horecker BL (2002) The pentose phosphate pathway. J Biol Chem 277:47965–47971
Horecker BL, Hurwitz J, Weissbach A (1956) The enzymatic synthesis and properties of ribulose 1,5-diphosphate. J Biol Chem 218:785–794
Houtz RL, Portis AR Jr (2003) The life of ribulose 1,5-bisphosphate carboxylase/oxygenase—posttranslational facts and mysteries. Arch Biochem Biophys 414:150–158
Houtz RL, Stults JT, Mulligan RM, Tolbert NE (1989) Post-translational modifications in the large subunit of ribulose bisphosphate carboxylase/oxygenase. Proc Natl Acad Sci USA 86:1855–1859
Huber SC, Hall TC, Edwards GE (1976) Differential localization of Fraction I protein between chloroplast types. Plant Physiol 57:730–733
Huffaker RC, Radin T, Kleinkop GE, Cox EL (1970) Effects of mild water stress on enzymes of nitrate assimilation of carboxylative phase of photosynthesis in barley. Crop Sci 10:471–474
Hurwitz J, Weissbach A, Horecker BL, Symrniotis PZ (1956) Spinach phosphoribulokinase. J Biol Chem 218:769–783
Ishida H, Nishimori Y, Sugisawa M, Makino A, Mae T (1997) The large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase is fragmented into 37-kDa and 16-kDa polypeptides by active oxygen in the lysates of chloroplasts from primary leaves of wheat. Plant Cell Physiol 38:471–479
Jakoby WB, Drummond DO, Ochoa S (1956) Formation of 3-phosphoglyceric acid by carbon dioxide fixation with spinach leaf enzymes. J Biol Chem 218:811–822
Jensen RG (2004) Activation of Rubisco controls CO2 assimilation in light. Photosynth Res 82:187–193
Jensen RG, Bahr JT (1977) Ribulose 1,5-bisphosphate carboxylase-oxygenase. Annu Rev Plant Physiol 28:379–400
Jensen RG, Bassham JA (1968) Photosynthesis by isolated chloroplasts. III. Light activation of the carboxylation reaction. Biochim Biophys Acta 153:227–234
Jordan DB, Chollet R (1983) Inhibition of ribulose bisphosphate carboxylase by substrate ribulose 1,5-bisphosphate. J Biol Chem 258:13752–13758
Jordan DB, Ogren WL (1981) Species variation in the specificity of ribulose bisphosphate carboxylase/oxygenase. Nature 291:513–515
Jordan DB, Ogren WL (1984) The CO2/O2 specificity of ribulose 1,5-bisphosphate carboxylase/oxygenase. Dependence on ribulosebisphosphate concentration, pH and temperature. Planta 161:308–313
Jordan DB, Chollet R, Ogren WL (1983) Binding of phosphorylated effectors by active and inactive forms of ribulose-1,5-bisphosphate carboxylase. Biochemistry 22:3410–3418
Kane HJ, Wilkin JM, Portis AR Jr, Andrews TJ (1998) Potent inhibition of ribulose-bisphosphate carboxylase by an oxidized impurity in ribulose-1,5-bisphosphate Plant Physiol 117:1059–1069
Kanevski I, Maliga P (1994) Relocation of the plastid rbcL gene to the nucleus yields functional ribulose-1,5-bisphosphate carboxylase in tobacco chloroplasts. Proc Nat Acad Sci USA 91:1969–1973
Kanevski I, Maliga P, Rhoades DF, Gutteridge S (1999) Plastome engineering of ribulose-1,5-bisphosphate carboxylase/oxygenase in tobacco to form a sunflower large subunit and tobacco small subunit hybrid. Plant Physiol 119:133–141
Kaul R, Saluja D, Sachar RC (1986) Phosphorylation of small subunit plays a critical role in the regulation of RuBPCase in moss and spinach. FEBS Lett 209:63–70
Kawashima N, Wildman SG (1972) Studies on Fraction I protein. IV. Mode of inheritance of the primary structure in relation to whether chloroplast or nuclear DNA contains the code for a chloroplast protein. Biochim Biophys Acta 262:42–49
Keys AJ, Major I, Parry MAJ (1995) Is there another player in the game of Rubisco regulation? J Exp Bot 46:1245–1251
Khan S, Andraloj PJ, Lea PJ, Parry MAJ (1999) 2’-carboxy-d-arabinitol 1-phosphate (CA1P) protects ribulose-1,5-bisphosphate carboxylase/oxygenase against proteolytic breakdown. Eur J Biochem 266:840–847
Kim K, Portis AR Jr (2004) Oxygen-dependent H2O2 production by Rubisco, FEBS Lett 571:124–128
Kim K, Portis AR Jr (2006) Kinetic analysis of the slow inactivation of Rubisco during catalysis: effects of temperature, O2 and Mg++. Photosynth Res 87:195–204
Kitano K, Maeda N, Fukui T, Atomi H, Imanaka T, Miki K (2001) Crystal structure of a novel-type archaeal Rubisco with pentagonal symmetry. Structure 9:473–481
Kobza J, Seemann JR (1988) Mechanisms for light-dependent regulation of ribulose-1,5-bisphosphate carboxylase activity and photosynthesis in intact leaves. Proc Natl Acad Sci USA 85:3815–3819
Knight S, Andersson I, Brändén C-I (1989) Re-examination of the three-dimensional structure of the small subunit of RuBisCo from higher-plants. Science 244:702–705
Knight S, Andersson I, Brändén C-I (1990) Crystallographic analysis of ribulose 1,5-bisphosphate carboxylase from spinach at 2.4 Å resolution: subunit interactions and active site. J Mol Biol 215:113–160
Krapp A, Hofmann B, Schäfer C, Stitt M (1993) Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates: a mechanism for the ‘sink regulation’ of photosynthesis? Plant J 3:817–828
Ku MSB, Schmitt MR, Edwards GE (1979) Quantitative determination of RuBP carboxylase–oxygenase protein in leaves of several C3 and C4 plants. J Exp Bot 30:89–98
Kung S (1976) Tobacco fraction 1 protein: a unique genetic marker. Science 191:429–434
Kung SD, Marsho TV (1976) Regulation of RuBP carboxylase/oxygenase activity and its relationship to plant photorespiration. Nature 259:352–356
Kung SD, Sakano K, Wildman SG (1974) Multiple peptide composition of the large and small subunits of Nicotiana tabacum fraction I protein ascertained by fingerprinting and electrofocusing. Biochim Biophys Acta 365:138–147
Laing WA, Ogren WL, Hageman RH (1974) Regulation of soybean net photosynthetic CO2 fixation by the interaction of CO2, O2 and ribulose 1,5-diphosphate carboxylase. Plant Physiol 54:678–685
Larson EM, O’Brien CM, Zhu G, Spreitzer RJ, Portis AR Jr (1997) Specificity for activase is changed by a pro-89 to arg substitution in the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. J Biol Chem 272:17033–17037
Levine RP, Togasaki RK (1965) A mutant strain of Chlamydomonas reinhardi lacking ribulose diphosphate carboxylase activity. Proc Natl Acad Sci USA 53:987–990
Li C, Salvucci ME, Portis AR Jr (2005) Two residues of Rubisco activase involved in recognition of the Rubisco substrate. J Biol Chem 280:24864–24869
Lilley RM, Wlaker DA (1975) Carbon dioxide assimilation by leaves, isolated chloroplasts, and ribulose bisphosphate carboxylase from spinach. Plant Physiol 55:1087–1092
Lodish H, Berk A, Zipursky LS, Matsudaria P, Baltimore D, Darnell J (eds) (2000) Molecular cell biology. WH Freeman & Co, New York
Long SP, Zhu XG, Naidu SL, Ort DR (2006) Can improvement in photosynthesis increase crop yields? Plant Cell Environ 29:315–330
Lorimer GH (1981) The carboxylation and oxygenation of ribulose 1,5-bisphosphate: the primary events in photosynthesis and photorespiration. Annu Rev Plant Physiol Plant Mol Biol 32:349–383
Lorimer GH, Miziorko H (1980) Carbamate formation of the epsilon-amino group of a lysl residue as the basis for the activation of ribulose bisphosphate carboxylase by CO2 and Mg2+. Biochemistry 19:5321–5328
Lorimer GH, Andrews TJ, Tolbert NE (1973) Ribulose diphosphate oxygenase. II. Further proof of reaction products and mechanism of action. Biochemistry 12:18–23
Lorimer GH, Badger MR, Andrews TJ (1976) The activation of ribulose-1,5-bisphosphate carboxylase by carbon dioxide and magnesium ions. Equilibria, kinetics, a suggested mechanism, and physiological implications. Biochemistry 15:529–536
Mächler F, Nösberger J (1980) Regulation of ribulose bisphosphate carboxylase activity in intact wheat leaves by light, CO2, and temperature. J Expt Bot 31:1485–1491
Mate CJ, Hudson GS, von Caemmerer S, Evans JR, Andrews TJ (1993) Reduction of ribulose-bisphosphate carboxylase activase levels in tobacco (Nicotiana tabacum) by antisense RNA reduces ribulose bisphosphate carboxylase carbamylation and impairs photosynthesis. Plant Physiol 102:1119–1128
Mayaudon J (1957) Study of association between the main nucleoprotein of green leaves and carboxydismutase. Enzymologia 18:345–354
Mayaudon J, Benson AA, Calvin M (1957) Ribulose-1,5-diphosphate from and CO2 fixation by Tetragonia expansa leaves extract. Biochim Biophys Acta 23:342–351
McCurry SD, Pierce J, Tolbert NE, Orme-Johnson WH (1981) On the mechanism of effector-mediated activation of ribulose bisphosphate carboxylase/oxygenase. J Biol Chem 256:6623–6628
McDermitt DK, Zeiher CA, Porter CA (1983) Physiological activity of RuBP carboxylase in soybeans. In: Randall DD, Blevins DG, Larson R (eds) Current topics in plant biochemistry and physiology, vol 1. University of Missouri Press, Columbia, p 230
McIntosh L, Poulsen C, Bogorad L (1980) Chloroplast gene sequence for the large subunit of ribulose bisphosphatecarboxylase of maize. Nature 288:556–560
Martin PG (1979) Amino-acid sequence of the small subunit of ribulose-1,5-bisphosphate carboxylase from spinach. Aust J Plant Physiol 6:401–408
Miziorko HM, Lorimer GH (1983) Ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem 52:507–535
Moore BD, Isidoro E, Seemann JR (1993) Distribution of 2-carboxyarabinitol among plants. Phytochem 34:703–707
Morell MK, Paul K, Kane HJ, Andrews TJ (1992) Rubisco: maladapted or misunderstood. Aust J Bot 40:431–441
Moses V, Calvin M (1958) The path of carbon in photosynthesis. XXII. The identification of carboxy-ketopentitol diphosphates as product of photosynthesis. Proc Natl Acad Sci USA 44:260–277
Mott KA, Snyder GW, Woodrow IE (1997) Kinetics of Rubisco activation as determined from gas-exchange measurements in antisense plants of Arabidopsis thaliana containing reduced levels of Rubisco activase. Aust J Plant Physiol 24:811–818
Müllhofer G, Rose IA (1964) The position of carbon-carbon bond cleavage in the ribulose diphosphate carboxydismutase reaction. J Biol Chem 240:1341–1346
Nakamura H, Saka H (1978) Photochemical oxidants injury in rice plants. III. Effect of ozone on physiological activities in rice plants. Jpn J Crop Sci 47:704–714
Nargang R, McIntosh L, Somerville CR (1984) Nucleotide sequence of the ribulosebisphosphate carboxylase gene from Rhodospirillum rubrum. Mol Gen Genet 193:220–224
Neales TF, Incoll LD (1968) Control of leaf photosynthesis rate by level of assimilate concentration in leaf—a review of the hypothesis. Bot Rev 34:107–125
Nelson PE, Surzycki SJ (1976) A mutant strain of Chlamydomonas reinhardi exhibiting altered ribulosebisphosphate carboxylase. Eur J Biochem 61:465–474
Ogren WL (2003) Affixing the O to rubisco: discovering the source of photorespiratory glycolate and its regulation. Photosynth Res 76:53–63
Ogren WL, Bowes G (1971) Ribulose diphosphate carboxylase regulates soybean photorespiration. Nature 230:159–160
Okabe KI, Codd GA, Stewart WDP (1979) Hydroxylamine stimulates carboxylase activity and inhibits oxygenase activity of cyanobacterial RuBP carboxylase/oxygenase. Nature 279:525–527
Onizuka T, Endo S, Akiyama H, Kanai S, Hirano M, Yokota A, Tanaka S, Miyasaka H (2004) The rbcX gene product promotes the production and assembly of ribulose-1,5-bisphosphate carboxylase/oxygenase of Synechococcus sp. PCC7002 in Escherichia coli. Plant Cell Physiol 45:1390–1395
Osmond B, Yakir D, Giles L, Morrison J (1994) From corn shucks to global greenhouse: stable isotopes as integrators of photosynthetic metabolism from tissue to planetary scale. In: Tolbert NE, Preiss J (eds) Regulation of atmospheric CO2 and O2 by photosynthetic carbon metabolism. Oxford University Press, NY, pp 249–265
O’Toole JC,Crookson RK, Treharne KJ, Ozbun JL (1976) Mesophyll resistance and carboxylase activity—comparison under water stress conditions. Plant Physiol 57:465–468
Ott CM, Smith BD, Portis AR Jr, Spreitzer RJ (2000) Activase region on chloroplast ribulose-1,5-bisphosphate carboxylase/oxygenase. Nonconservative substitution in the large subunit alters species specificity of protein interaction. J Biol Chem 275:26241–26244
Parikh MR, Greene DN, Woods KK, Matsumura I (2006) Directed evolution of RuBisCO hypermorphs through genetic selection in engineered E. coli . Protein Engineering Design and Selection 19:113–119
Park R, Epstein S (1961) Carbon isotope fractionation during photosynthesis. Geochim Cosmochim Acta 21:110–126
Park RB, Pon NG (1961) Correlation of structure with function in Spinacea oleracea chloroplasts. J Mol Biol 3:1–10
Parry MAJ, Gutteridge S (1984) The effect of SO 2− 3 and SO 2− 4 ions on the reactions of ribulosebisphosphate carboxylase. J Exp Bot 35:157–168
Parry MAJ, Delgado E, Vadell J, Keys AJ, Lawlor DW, Medrano H (1993) Water-stress and the diurnal activity of ribulose-1,5-bisphosphate carboxylase in field-grown Nicotiana-tabacum genotypes selected for survival at low CO2 concentrations. Plant Physiol Biochem 31:113–120
Parry MAJ, Andralojc PJ, Parmar S, Keys AJ, Habash D, Paul MJ, Alred R, Quick WP, Servaites JC (1997) Regulation of Rubisco by inhibitors in the light. Plant Cell Environ 20:528–534
Parry MAJ, Loveland JE, Andralojc PJ (1999) Regulation of Rubisco. In: Bryant JE, Burrell MM, Kruger NJ (eds) Plant carbohydrate biochemistry. Bios, Oxford, UK, pp 127–145
Parry MAJ, Madgwick PJ Carvahlo JFC, Andralojc PJ (2007) Prospects for increasing photosynthesis by overcoming the limitations of Rubisco. J Agric Sci 145:31–43
Paulsen JM, Lane MD (1966) Spinach ribulose diphosphate carboxylase. I. Purification and properties of the enzyme. Biochemisty 5:2350–2357
Paech C, Pierce J, McCurry SD, Tolbert NE (1978) Inhibition of ribulose-1,5-bisphosphate carboxylase/oxygenase by ribulose-1,5-bisphosphate epimerization and degradation products. Biochem Biophys Res Commun 83:1084–1092
Pell EJ (1979) How air pollutants induce disease. In: Horsafll J, Cowling E (eds) Plant disease, vol 4. Academic Press, New York, pp 179–194
Pell EJ, Pearson NS (1983) Ozone-induced reduction in quantity of ribulose-1,5-bisphosphate carboxylase in alfalfa foliage. Plant Physiol 73:185–187
Peoples MB, Beilharz VC, Waters SP, Simpson RJ, Dalling MJ (1980) Nitrogen redistribution during grain growth in wheat (Triticum aestivum L.). II. Chloroplast senescence and the degradation of ribulose-1,5-bisphosphate carboxylase. Planta 149:241–251
Perchorowicz JT, Raynes DA, Jensen RG (1981) Light limitation of photosynthesis and activation of ribulose bisphosphate carboxylase in wheat seedlings. Proc Natl Acad Sci USA 78:2985–2989
Peterson LW, Huffaker RC (1975) Loss of ribulose 1,5-diphosphate carboxylase and increase in proteolytic activity during senescence of detached primary barley leaves. Plant Physiol 55:1009–1015
Pierce J, Tolbert NE, Barker R (1980) Interaction of ribulosebisphosphate carboxylase/oxygenase with transition-state analogs. Biochemistry 19:934–942
Pierce J, Carlson TJ, Williams GK (1989) A cyanobacterial mutant requiring the expression of ribulose bisphosphate carboxylase from a photosynthetic anaerobe. Proc Natl Acad Sci USA 86:5753–5757
Pilgrim ML, McClung CR (1993) Differential involvement of the circadian clock in the expression of genes required for ribulose-1,5-bisphosphate carboxylase/oxygenase synthesis, assembly, and activation in Arabidopsis thaliana. Plant Physiol 103:553–564
Pon NG, Rabin BR, Calvin M (1963) Mechanism of the carboxydismutase reaction. I. The effect of preliminary incubation of substrates, metal ion, and enzyme on activity. Biochem Zeits 338:7–19
Portis AR Jr (1992) Regulation of ribulose 1,5-bisphosphate carboxylase/oxygenase activity. Annu Rev Plant Phys Plant Mol Biol 43:415–437
Portis AR Jr (2003) Rubisco activase—Rubisco’s catalytic chaperone. Photosynth Res 75:11–27
Portis AR Jr, Salvucci ME (2002) The discovery of Rubisco activase—yet another story of serendipity. Photosynth Res 73:257–264
Portis AR Jr, Salvucci ME, Ogren WL (1986) Activation of ribulosebisphosphate carboxylase/oxygenase at physiological CO2 and ribulosebisphosphate concentrations by Rubisco activase. Plant Physiol 82:967–971
Price GD, Badger MR (1989) Expression of human carbonic anhydrase in the cyanobacterium Synechococcus PCC7942 creates a high CO2-requiring phenotype. Evidence for a central role for carboxysomes in the CO2 concentrating mechanism. Plant Physiol 91:505–513
Quayle JR, Fuller RC, Benson AA, Calvin M (1954) Enzymatic carboxylation of ribulose diphosphate. J Am Chem Soc 76:3610–3611
Rabin BR, Trown PW (1964) Mechanism of action of carboxydismutase. Nature 202:1290–1291
Rabinowitch EI (1956) Photosynthesis and related processes vol II, Part 2. Interscience Publishers, New York, pp 1630–1713
Racker E (1955) Synthesis of carbohydrates from carbon dioxide and hydrogen in a cell-free system. Nature 175:249–251
Raines CA (2006) Transgenic approaches to manipulate the environmental responses of the C3 carbon fixation cycle. Plant Cell Environ 29:331–339
Read BA, Tabita FR (1992) A hybrid ribulosebisphosphate carboxylase oxygenase enzyme exhibiting a substantial increase in substrate-specificity factor. Biochemistry 31:5553–5560
Reddy G, Arteca RN, Dai Y-R, Flores HE, Negm FB, Pell EJ (1993) Changes in ethylene and polyamines in relation to mRNA levels of the large and small subunits of ribulose bisphosphate carboxylase/oxygenase in ozone-stressed potato foliage. Plant Cell Environ 16:819–826
Reinhold L, Zviman M, Kaplan A (1987) Inorganic carbon fluxes and photosynthesis in cyanobacteria—a quantitative model. In: Biggins J (eds) Progress in photosynthesis, vol IV. Martinus, Nijhoff, Dordrecht, The Netherlands, pp 6.289–6.296
Ritland K, Clegg MT (1987) Evolutionary analysis of plant DNA sequences. Am Nat 130:S74–S100
Robinson SP, Portis AR Jr (1988) Release of the nocturnal inhibitor, carboxyarabinitol-1-phosphate, from ribulose bisphoshate carboxylase/oxygenase by Rubisco activase. FEBS Lett 233:413–416
Robinson SP, Portis AR Jr (1989a) Adenosine triphosphate hydrolysis by purified Rubisco activase. Arch Biochem Biophys 268:93–99
Robinson SP, Portis AR Jr (1989b) Ribulose-1,5-bisphosphate carboxylase/oxygenase activase protein prevents the in vitro decline in activity of ribulose-1,5-bisphosphate carboxylase/oxygenase. Plant Physiol 88:1008–1014
Rodermel SR, Abbott MS, Bogorad L (1988) Nuclear-organelle interactions: nuclear antisense gene inhibits ribulose bisphosphate carboxylase enzyme levels in transformed tobacco plants. Cell 55:673–681
Rodermel SR, Haley J, Jiang CZ, Tsai CH, Bogorad L (1996) A mechanism for intergenomic integration—abundance of ribulose bisphosphate carboxylase small-subunit protein influences the translation of the large-subunit mRNA. Proc Natl Acad Sci USA 93:3881–3885
Rutner AC, Lane MD (1967) Nonidentical subunits of ribulose diphosphate carboxylase. Biochem Biophys Res Commun 28:531–537
Sage RF (2002) Variation in the kcat of Rubisco in C3 and C4 plants and some implications for photosynthetic performance at high and low temperature. J Expt Bot 53:609–620
Sage RF, Sharkey TD, Seemann JR (1988) The in-vivo response of the ribulose-1,5-bisphosphate carboxylase activation state and the pool sizes of photosynthetic metabolites to elevated CO2 in Phaseolus vulgaris L. Planta 174:407–416
Sage RF, Sharkey TD, Seemann JR (1989) Acclimation of photosynthesis to elevated CO2 in five C3 species. Plant Physiol 89:590–596
Salvucci ME, Holbrook GP (1989) Purification and properties of 2-carboxy-d-arabinitol 1-phosphatase. Plant Physiol 90:679–685
Salvucci ME, Ogren WL (1996) The mechanism of Rubisco activase: insights from studies of the properties and structure of the enzyme. Photosynth Res 47:1–11
Salvucci ME, Portis AR Jr, Ogren WL (1985) A soluble chloroplast protein catalyzes ribulosebisphosphate carboxylase/oxygenase activation in vivo. Photosynth Res 7:193–201
Salvucci ME, Crafts-Brandner SJ (2004) Mechanism for deactivation of Rubisco under moderate heat stress. Physiol Plant 122:513–519
Sasaki Y Ishiye M, Sakihama T, Kamikubo T (1981) Light-induced increase of mRNA activity coding for the small subunit of ribulose-1,5-bisphosphate carboxylase. J Biol Chem 256:2315–2320
Sasaki Y, Sakihama T, Kamikubo T, Shinozaki K (1983) Phytochrome-mediated regulation of two mRNAs, encoded by nuclei and chloroplasts of ribulose-1,5-bisphosphate carboxylase/oxgenase. Eur J Biochem 133:617–620
Sato T, Atomi H, Imanaka T (2007) Archaeal type III RuBisCOs function in a pathway for AMP metabolism. Science 315:1003–1006
Saver B, Knowles JR (1982) Ribulose-1,5-bisphosphate carboxylase: enzyme-catalysed appearance of solvent tritium at carbon 2 of ribulose 1,5-bisphosphate reisolated after partial reaction. Biochemistry 21:5398–5403
Schloss JV, Lorimer GH (1982) The stereochemical course of ribulosebisphosphate carboxylase. Reductive trapping of the 6-carbon reaction-intermediate. J Biol Chem 257:4691–4694
Schmidt GW, Mishkind ML (1983) Rapid degradation of unassembled ribulose 1,5-bisphosphate carboxylase small subunits in chloroplasts. Proc Natl Acad Sci USA 80:2632–2636
Schmid GH, Bader KP, Radunz A, van Assche CJ, Reinier N, Courtiade B (1987) Effect of hydroxylamine derivatives on photorespiration in the tobacco aurea mutant Nicotiana tabacum Su/su. Z Naturforsch 42c:965–969
Schneider G, Lindqvist Y, Brändén C-I, Lorimer GH (1986) Three-dimensional structure of ribulose-1,5-bisphosphate carboxylase/oxygenase from Rhodospirillum rubrum at 2.9 Å resolution. EMBO J 5:3409–3415
Schrader SM, Kane HJ, Sharkey TD, von Caemmerer S (2006) High temperature enhances inhibitor production but reduces fallover in tobacco Rubisco. Funct Plant Biol 33:921–929
Schwender J, Goffman F, Ohlrogge JB, Shachar-Hill Y (2004) Rubisco without the Calvin cycle improves the carbon efficiency of developing green seeds. Nature 432:779–782
Seemann JR, Badger MR, Berry JA (1984) Variations in the specific activity of ribulose-1,5-bisphosphate carboxylase between species utilizing differing photosynthetic pathways. Plant Physiol 74:791–794
Seemann JR, Berry JA, Freas SM, Krump MA (1985) Regulation of ribulose bisphosphate carboxylase activity in vivo by a light-modulated inhibitor of catalysis. Proc Natl Acad Sci USA 82:8024–8028
Servaites JC (1985) Binding of a phosphorylated inhibitor to ribulose bisphosphate carboxylase/oxygenase at night. Plant Physiol 78:839–843
Servaites JC, Parry MAJ, Gutteridge S, Keys AJ (1986) Species variation in the predawn inhibition of ribulose-1,5-bisphosphate carboxylase oxygenase. Plant Physiol 82:1161–1163
Shinozaki K, Ohme M, Tanaka M, Wakasugi T, Hayashida H, Matsubayashi T, Zaita N, Chunwongse J, Obokata J, Yamaguchi-Shinozaki K, Ohto C, Torazawa K, Meng BY, Sugita M, Deno H, Kamogashira T, Yamada K, Kusuda J, Takaiwya F, Kato A, Tohdoh N, Shimada H, Sugiura M (1986) The complete nucleotide sequence of the tobacco chloroplast genome: its gene organization and expression. EMBO J 5:2043–2049
Shively JM, Ball F, Brown RE, Saunders RE (1973) Functional organelles in prokaryotes: polyhedral inclusions (carboxysomes) of Thiobacillus neapolitanus. Science 182:584–586
Siegel MI, Lane MD (1973) Chemical and enzymatic evidence for the participation of a 2-carboxy-3-ketoribitiol-1,5-diphosphate intermediate in the carboxylation of ribulose 1,5-diphosphate. J Biol Chem 248:5486–5498
Siegelman HW, Hind G (1978) Photosynthetic carbon assimilation. Plenum Press, New York
Siegenthaler U, Sarmiento JL (1993) Atmospheric carbon dioxide and the ocean. Nature 365:119–125
Simpson E, Cooke RJ, Davies DD (1981) Measurement of protein degradation in leaves of Zea mays using [3H]acetic anhydride and tritiated water. Plant Physiol 67:1214–1219
Smith SM, Ellis RJ (1981) Light stimulated accumulation of transcripts of nuclear and chloroplast genes for ribulosebisphosphate carboxylase. J Mol Appl Genet 1:127–137
Somerville CR, Portis AR Jr, Ogren WL (1982) A mutant of Arabidopsis-thaliana which lacks activation of RuBP carboxylase in vivo. Plant Physiol 70:381–387
Somerville CR, Somerville SC (1984) Cloning and expression of the Rhodospirillum rubrum ribulosebisphosphate carboxylase gene in E. coli. Mol Gen Genet 193:214–219
Spreitzer RJ (1993) Genetic dissection of Rubisco structure and function. Annu Rev Plant Physiol Plant Mol Biol 44:411–434
Spreitzer RJ (1999) Questions about the complexity of chloroplast ribulose bisphosphate carboxylase/oxygenase. Photosynth Res 60:29–42
Spreitzer RJ, Mets LJ (1980) Non-mendelian mutation affecting ribulose-1,5-bisphosphate carboxylase structure and activity. Nature 285:114–115
Spreizter RJ, Salvucci ME (2002) Rubisco: structure, regulatory interactions, and possibilities for a better enzyme. Annu Rev Plant Biol 53:449–475
Spreitzer RJ, Peddi SR, Satagopan S (2005) Phylogenetic engineering at an interface between large and small subunits imparts land-plant kinetic properties to algal Rubisco. Proc Natl Acad Sci USA 102:17225–17230
Stitt M, Quick WP, Schurr U, Schulze E-D, Rodermel SR, Bogorad L (1991) Decreased ribulose-1,5-bisphosphate carboxylase-oxygenase in transgenic tobacco transformed with ‘antisense’ rbcS. II. Flux-control coefficients for photosynthesis in varying light, CO2, and air humidity. Planta 183:555–566
Sugiyama T, Akazawa T (1967) Structure of chloroplast proteins I. Subunit structure of wheat Fraction-1 protein. J Biochem 62:474–482
Sugiyama T, Akazawa T (1970) Subunit structure of spinach leaf ribulose 1,5-diphosphate carboxylase. Biochemistry 9:4499–4504
Svab Z, Hajdukiewicz P, Maliga P (1990) Stable transformation of plastids of higher plants. Proc Natl Acad Sci USA 87:8526–8530
Tabita FR (1999) Microbial ribulose 1,5-bisphosphate carboxylase/oxygenase: a different perspective. Photosynth Res 60:1–28
Tabita FR (2004) Research on carbon dioxide fixation in photosynthetic microorganisms (1971–present). Photosyn Res 80:315–332
Tabita FR, McFadden BA (1974a) d-Ribulose 1,5-diphosphate carboxylase from Rhodospirillum rubrum I. Levels, purification, and effects of metallic ions. J Biol Chem 249:3453–3458
Tabita FR, McFadden BA (1974b) d-Ribulose 1,5-diphosphate carboxylase from Rhodospirillum rubrum. II. Quarternary structure, composition, catalytic, and immunological properties. J Biol Chem 249:3459–3464
Tabita FR, Small CL (1985) Expression and assembly of active cyanobacterial ribulose-1,5-bisphosphate carboxylase/oxygenase in Escherichia coli containing stoichiometric amounts of large and small subunits. Proc Natl Acad Sci USA 82:6100–6103
Tamiya H, Huzisige H (1949) Effect of oxygen on the dark reaction of photosynthesis. Acta Phytochim 15:83–104
Tcherkez GGB, Farquhar GD, Andrews TJ (2006) Despite slow catalysis and confused substrate specificity, all ribulose bisphosphate carboxylases may be nearly perfectly optimized. Proc Natl Acad Sci USA 103:7246–7251
Thornber JP, Ridley SM, Bailey JL (1965) Isolation and partial characterization of Fraction I protein from spinach-beet chloroplasts. Biochem J 96:29–31
Tobin EM (1978) Light regulation of specific mRNA species in Lemna gibba L. G-3. Proc Natl Acad Sci USA 75:4749–4753
Tobin EM (1981) Phytochrome-mediated regulation of messenger RNAs for the small subunit of ribulose 1,5-bisphosphate carboxylase and the light-harvesting chlorophyll a/b-protein in Lemna gibba. Plant Mol Biol 1:35–51
Trown PW (1965) An improved method for the isolation of carboxydismutase. Probable identity with Fraction 1 protein and the protein moiety of protochlorophyll holochrome. Biochemistry 4:908–918
Van Assche CJ, Reinier N, Courtiade B, Chancel A, Huber S (1987) Chemical control of photorespiration: steady-state kinetic and conformational changes of ribulose-1,5-bisphosphate carboxylase/oygenase obtained with O-p-nitrophenylhydroxylamine. Z Naturforsch 42c:837–844
Van den Broeck G, Timko MP, Kausch AP, Cashmore AR, Montau MV, Herrera-Estrella L (1985) Targeting of a foreign protein to chloroplast by fusion to the transit peptide from the small subunit of ribulose 1,5-bisphosphate carboxylase. Nature 313:358–363
von Caemmerer S, Evans JR, Hudson GS, Andrews TJ (1994) The kinetics of ribulose-1,5-bisphosphate carboxylase/oxygenase in vivo inferred from measurements of photosynthesis in leaves of transgenic tobacco. Planta 195:88–97
Vu CV, Allen LH Jr, Bowes G (1983) Effects of light and elevated atmospheric CO2 on the ribulose bisphosphate carboxylase activity and ribulose bisphosphate level of soybean leaves. Plant Physiol 73:729–734
Wanner LA, Gruissem W (1991) Expression dynamics of the tomato rbcS gene family during development. Plant Cell 3:1289–1303
Warburg O (1920) Über die Geschwindigkeit der photochemischen Kohlensäurezersetzung in lebenden Zellen. II. Biochem Z 103:188–217
Waring PF, Khalifa MM, Treharne KJ (1968) Rate-limiting processes in photosynthesis at saturating light intensities. Nature 220:453–457
Watson GFM, Yu J-P, Tabita FR (1999) Unusual ribulose 1,5-bisphosphate carboxylase/oxygenase of anoxic Archaea. J Bacteriol 181:1569–1575
Weis E (1981a) Reversible heat-inactivation of the Calvin cycle: a possible mechanism of the temperature regulation of photosynthesis. Planta 151:33–39
Weis E (1981b) The temperature sensitivity of dark-inactivation and light-activation of the ribulose-1,5-bisphosphate carboxylase in spinach chloroplasts. FEBS Lett 129:197–200
Weissbach A, Smyrniotis PZ, Horecker BL (1954) Pentose phosphate and CO2 fixation with spinach extracts. J Am Chem Soc 76:3611–3612
Weissbach A, Horecker BL, Hurwitz J (1956) The enzymatic formation of phosphoglyceric acid from ribulose diphosphate and carbon dioxide. J Biol Chem 218:795–810
Werneke JM, Zielinski RE, Ogren WL (1988) Structure and expression of spinach leaf cDNA encoding ribulosebisphosphate carboxylase/oxygenase activase. Proc Natl Acad Sci USA 85:787–791
Werneke JM, Chatfield JM, Ogren WL (1989) Alternative mRNA splicing generates the two ribulosebisphosphate carboxylase/oxygenase activase polypeptides in spinach and Arabidopsis. Plant Cell 1:815–825
Whitney SM, Andrews TJ (2001a) Plastome-encoded bacterial ribulose-1,5-bisphosphate carboxylase/oxygenase (RubisCO) supports photosynthesis and growth in tobacco. Proc Natl Acad Sci USA 98:14738–14743
Whitney SM, Andrews TJ (2001b) The gene for the ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) small subunit relocated to the plastid genome of tobacco directs the synthesis of small subunits that assemble into Rubisco. Plant Cell 13:193–205
Wildman SG (2002) Along the trail from Faction I protein to Rubisco (ribulose bisphosphate carboxylase-oxygenase). Photosynth Res 73:243–250
Wildman SG, Bonner J (1947) The proteins of green leaves. I. Isolation, enzymatic properties, and auxin content of spinach cytoplasmic proteins. Arch Biochem Biophys 14:381–413
Wildman SG, Kwanyuen P (1978) Fraction I protein and other products from tobacco for food. In: Siegelman HW, Hind G (eds) Photosynthetic carbon assimilation. Plenum Press, New York, pp 1–17
Wildner GF, Criddle RS (1969) Ribulose disphosphate carboxylase I. A factor involved in light activation of the enzyme. Biochem Biophys Res Commun 37:952–960
Wildner GF, Henkel J (1976) Specific inhibition of the oxygenase activity of ribulose-1,5-bisphosphate carboxylase. Biochem Biophys Res Commun 69:268–275
Wildner GF, Henkel J (1980) Preservation of RuBP carboxylase without oxygenase activity during anaerobiosis. FEBS Lett 113:81–84
Wilson AT, Calvin M (1955) The photosynthetic cycle. CO2 depedent transients. J Am Chem Soc 77:5948–5957
Winkel BSJ (2004) Metabolic channeling in plants. Annu Rev Plant Biol 55:85–107
Winter K, Foster JG, Schmitt MR, Edwards GE (1982) Activity and quantity of ribulose bisphosphate carboxylase- and phosphoenolpyruvate carboxylase-protein in two Crassulacean acid metabolism plants in relation to leaf age, nitrogen nutrition, and point in time during a day/night cycle. Planta 154:309–317
Wishnick M, Lane MD, Scrutton MC (1970) The interaction of metal ions with ribulose 1,5-diphosphate carboxylase from spinach. J Biol Chem 245:4939–4947
Wittenbach VA (1979) Ribulose bisphosphate carboxylase and proteolytic activity in wheat leaves from anthesis through senescence. Plant Physiol 64:884–887
Wittenbach VA, Ackerson RC, Giaquinta RT, Hebert RR (1980) Changes in photosynthesis, ribulose bisphosphate carboxylase, proteolytic activity, and ultrastructure of soybean leaves during senescence. Crop Sci 20:225–231
Wong SC (1979) Elevated atmospheric partial pressure of CO2 and plant growth. I. Interactions of nitrogen nutrition and photosynthetic capacity in C3 and C4 plants. Oecologia 44:68–74
Woodrow IE, Mott KA (1989) Rate limitation of non-steady-state photosynthesis by ribulose-1,5-bisphosphate carboxylase in spinach. Aust J Plant Physiol 16:487–500
Yeoh HH, Badger MR, Watson L (1981) Variations in the kinetic properties of ribulose-1,5-bisphosphate carboxylases among plants. Plant Physiol 67:1151–1155
Zhang N, Portis AR Jr (1999) Mechanism of light regulation of Rubisco: a specific role for the larger Rubisco activase isoform involving reductive activation by thioredoxin-f. Proc Natl Acad Sci USA 96:9438–9443
Zhang N, Kallis RP, Ewy RG, Portis AR Jr (2002) Light modulation of Rubisco in Arabidopsis requires a capacity for redox regulation of the larger Rubisco activase isoform. Proc Natl Acad Sci USA 99:3330–3334
Zhu X-G, Portis AR Jr, Long SP (2004) Would transformation of C3 crop plants with foreign Rubisco increase productivity? A computational analysis extrapolating from kinetic properties to canopy photosynthesis. Plant Cell Environ 27:155–165
Zeigler I (1972) The effect of SO −3 on the activity of ribulose-1,5-diphosphate carboxylase in isolated spinach chloroplasts. Planta 103:155–163
Zhu G, Jensen RG (1991a) Xylulose 1,5-bisphosphate synthesized by ribulose 1,5-bisphosphate carboxylase/oxygenase during catalysis binds to decarbamylated enzyme. Plant Physiol 97:1348–1353
Zhu G, Jensen RG (1991b) Fallover of ribulose 1,5-bisphosphate carboxylase/oxygenase activity. Decarbamylation of catalytic sites depends on pH. Plant Physiol 97:1354–1358
Acknowledgments
The authors thank Govindjee for editing and inviting us to write this historical minireview. We thank Alf Keys, Mike Salvucci and our reviewers for suggestions that led to the improvement of the timeline presented here. Archie Portis thanks the Agricultural Research Service, United States Department of Agriculture and the Office of Basic Energy Sciences, United States Department of Energy for their long-term support. Martin Parry thanks the Biotechnology and Biological Sciences Research Council of the United Kingdom for their grant-aided support.
We offer our sincere apologies to those whose contributions could not be included here.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Portis, A.R., Parry, M.A.J. Discoveries in Rubisco (Ribulose 1,5-bisphosphate carboxylase/oxygenase): a historical perspective. Photosynth Res 94, 121–143 (2007). https://doi.org/10.1007/s11120-007-9225-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-007-9225-6