Abstract
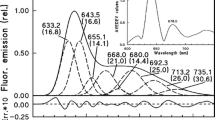
Cuttings of grapevine (Vitis vinifera L. cv. Chardonnay) were dark-forced at least three weeks. Pigment contents, 77 K fluorescence emission, excitation spectra of the leaves, petioles, stems, transmission electron micrographs of the etioplasts from leaves, the chlorenchyma tissues of the stems were analysed. The dark-grown leaves, stems contained 8 to 10, 3 to 5 μg/g fresh weight protochlorophyllide, its esters, respectively. HPLC analysis showed that the molar ratio of the unesterified, esterified pigments was 7:3 in the shoot developed in darkness. The dark-forced leaves contained carotenoids identified as: neoxanthin, violaxanthin, antheraxanthin, lutein, β-carotene. Detailed analyses of the fluorescence spectra proved that all tissues of the dark-forced shoots had protochlorophyllide or protochlorophyll forms with emission maxima at 628, 636, 644, 655, 669 nm. The 628, 636 nm emitting forms were present in all parts of the dark-forced shoot, but dominated in the stems, which may indicate an organ specificity of the etioplast development. Variations in the distribution of the pigment forms were even found in the different tissues of the stem. The subepidermal layers were more abundant in the 655 nm form than the parenchyma cells of the inner part of the cortex, the pith. In the latter cells, the plastid differentiation stopped in intermediary stages between proplastids, etioplasts. The plastids in the subepidermal layers had developed prolamellar body structures, which were similar to those of etiolated leaves. The results highlight the importance of organ-, tissue specificity of plastid differentiation for chlorophyll biosynthesis, greening of different plant organs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Albrecht M and Sandman G (1994) Light-stimulated carotenoid biosynthesis during transformation of maize etioplasts is regulated by increased activity of isopentenyl pyrophosphate izomerase. Plant Physiol 105: 529-534
Armstrong GA, Apel K and Rüdiger W (2000) Does a lightharvesting protochlorophyllide a/b-binding protein complex exist? Trends Plant Sci 5: 40-44
Avarmaa R, Renge I and Mauring K (1984) Sharp-line structure in the fluorescence and excitation spectra of greening etiolated leaves. FEBS Lett 167: 186-190
Bergweiler P, Röper U and Lütz C (1984) The development and ageing of membranes from etioplasts of Avena sativa. Physiol Plant 60: 395-400
Böddi B, Lindsten A, RybergM and Sundqvist C (1989) On the aggregational states of protochlorophyllide and its protein complexes in wheat etioplasts. Physiol Plant 76: 135-143
Böddi B, Ryberg M and Sundqvist C (1992) Identification of four universal protochlorophyllide forms in dark-grown leaves by analyses of the 77 K fluorescence emission spectra. J Photochem Photobiol 12: 389-401
Böddi B, Ryberg M and Sundqvist C (1993) Analysis of the 77 K fluorescence emission and excitation spectra of isolated etioplast inner membranes. J Photochem Photobiol B Biol 21: 125-133
Böddi B, Mc Ewen B, Ryberg M and Sundqvist C (1994) Protochlorophyllide forms in non-greening epicotyls of darkgrown pea (Pisum sativum). Physiol Plant 92: 160-170
Böddi B, Evertsson I, Ryberg M and Sundqvist C (1996) Protochlorophyllide transformations and chlorophyll accumulation in epicotyls of pea (Pisum sativum). Physiol Plant 96: 706-713
Böddi B, Kis-Petik K, Kaposi AD, Fidy J and Sundqvist C (1998) The two short wavelength protochlorophyllide forms in pea epicotyls are both monomeric Biochimica et Biophysica Acta 1365, 531-540
Breithaupt DE and Bamedi A (2002) Carotenoids and carotenoid esters in potatoes (Solanum tuberosum L): new insights into an ancient vegetable. J Agric Food Chem 50: 7175-7181
Dobek A, Dujardin E, Franck F, Sironval C, Breton J and Roux E (1981) The first events of protochlorophyll(ide) photoreduction investigated in etiolated leaves by means of the fluorescence excited by short, 610 nm laser flashes at room temperature. Photobiochem Photobiophys 2: 35-44
Franck F and Inoue Y (1984) Light-driven reversible transformation of chlorophyllide P696,684 into P688,678 in illuminated etiolated bean leaves. Photobiochem Photobiophys 8: 85-96
Franck F, Bereza B and Böddi B (1999) Protochlorophyllide-NADP+ and protochlorophyllide-NADPH complexes and their regeneration after flash illumination in leaves and etioplast membranes of dark-grown wheat. Photosynth Res 59: 53-61
Fujita Y (1996) Protochlorophyllide photoreduction: a key step in the greening of plants. Plant Cell Physiol 37: 411-421
Gounaris I, Fässler L and Lichtenthaler HK (1983) Changes in plastid ultrastructure and fluctuations of cellular isoprenoid and carbohydrate compounds during continued etiolation of dark-grown oat seedlings. Physiol Plant 59: 347-354
Griffiths WT (1975) Characterization of the terminal stages of chlorophyll(ide) synthesis in etioplast membrane preparations. Biochem J 152: 623-655
Gunning BES (1975) Plastids. In: Gunning BES and SteereMW (eds) Ultrastructure and Biology of Plant Cells, pp 97-133. Edward Arnold, London
Houssier C and Sauer K (1970) Circular dichroism and magnetic circular dichroism of the chlorophyll and protochlorophyll pigments. J Am Chem Soc 92: 779-791
Kamiya A, Ikegami I and Hase E (1981) Effects of light on chlorophyll formation in cultivated tobacco cells. I. Chlorophyll accumulation and phototransformation of protochlorophyll( ide) in callus cells under blue and red light. Plant Cell Physiol 22: 1385-1396
KrólM, Ivanov AG, Jansson S, Kloppstech K and Huner NPA (1999) Greening under high light and cold temperature affects the level of xanthophyll cycle pigments, early light inducible proteins and light-harvesting polypeptides in wild-type barley and the chlorina f2 mutant. Plant Physiol 120: 193-204
Madsen A (1963) The time course for the photoconversion of protochlorophyll by flash illumination. Physiol Plant 16: 470-473
Mc Ewen B and Lindsten A (1992) Characteriztaion of protochlorophyllide and protochlorophyllide esters in roots of dark-grown plants. Physiol Plant 84: 343-350
Mc Ewen B, Sundqvist C and Younis S (1994) Protochlorophyll( ide) forms in hypocotyls of dark-grown bean (Phaseolus vulgaris). Physiol Plant 90: 396-407
Rascio N, Mariani P, Chitano P and Dalla Vecchia F (1986) An ultrastructural study of maize leaf etioplasts throughout their entire life-cycle. Protoplasma 130: 98-107
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17: 208-212
Robertson D and Laetsch WM (1974) Structure and function of developing barley plastids. Plant Physiol 54: 148-159
Ryberg M and Dehesh K (1986) Localization of NADPHprotochlorophyllide oxidoreductase in dark-grown wheat (Triticum aestivum) by immuno-electron microscopy before and after transformation of the prolamellar bodies. Physiol Plant 66: 616-624
Schiff JA and Epstein HT (1966) The relative aspect of chloroplast continuity in Euglena. In: Goodwin TW (ed) Biochemistry of Chloroplasts, Vol I, pp 341-353. Academic Press, London
Schoefs B (2001) The protochlorophyllide-chlorophyllide cycle. Photosynth Res 70: 257-271
Seyedi M, Timko MP and Sundqvist C (2001) The distribution of protochlorophyllide and chlorophyll within seedlings of the lip1 mutant of pea. Plant Cell Physiol 42: 931-941
Shibata K (1957) Spectroscopic studies on chlorophyll formation in intact leaves. J Biochem-Tokyo 44: 147-173
Sironval C and Brouers M (1970) The reduction of protochlorophyllide into chlorophyllide. II. The temperature depen-phototransformation. Photosynthetica 2: 268-287
Skribanek A and Böddi B (2001) Light and cold-stress effects on the greening process of epicotyls and young stems of red oak (Quercus rubra L.) seedlings. Tree Physiol: 21 549-554
Skribanek A, Apatini D, Inaoka M and Böddi B (2000) Protochlorophyllide and chlorophyll forms in dark-grown stems and stem-related organs. J Photoch Photobiol B Biol 55: 172-177
Younis S, Ryberg M and Sundqvist C (1995) Plastid development in germinating wheat (Triticum aestivum) is enhanced by gibberellic acid and delayed by gabaculine. Physiol Plant 95: 336-346
Virgin HI (1966) Carotenoid synthesis in leaves of wheat after irradiation by red light. Physiol Plant 19: 40-46
Virgin HI (1967) Carotenoid synthesis in leaves of etiolated wheat seedlings after varying light and dark treatments. Physiol Plant 20: 314-320
Virgin HI (1996) Pigments in mesocotyls of corn (Zea mays) and oats (Avena sativa) in darkness, and after irradiation and treatment with 5-aminolevulinic acid. Physiol Plant 98: 714-722
Virgin HI and Sundqvist C (1992) Pigment formation in potato tubers (Solanum tuberosum) exposed to light followed darkness. Physiol Plant 86: 587-592
Wiktorsson B, Engdahl S, Zhong LB, Böddi B, Ryberg M and Sundqvist C (1993) The effect of cross-linking of the subunits of NADPH-protochlorophyllide oxidoreductase on the aggregational state of protochlorophyllide. Photosynthetica 29: 205-218
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Böddi, B., Bòka, K. & Sundqvist, C. Tissue specific protochlorophyll(ide) forms in dark-forced shoots of grapevine (Vitis viniferaL.). Photosynthesis Research 82, 141–150 (2004). https://doi.org/10.1007/s11120-004-1061-3
Issue Date:
DOI: https://doi.org/10.1007/s11120-004-1061-3