Abstract
Failures in either water systems or food systems, or a combination of system failures, could provide the underlying explanation for continued high levels of malnutrition in many regions. We focus on child health and offer the first spatially explicit analysis of the interaction between water source and food insecurity on children’s health in Burkina Faso, an African nation that continues to struggle with poor children’s health. We combine data from the 2010 Demographic and Health Survey, a small USAID water quality survey collected from community wells, and remotely sensed imagery. Results suggest that, in a few cases, reliable and clean water sources are positively correlated to children’s linear growth and weight gain, although in many regions, the interaction with community-level food production is critical to understanding health outcomes. The results also suggest that maternal health and nutrition during pregnancy and breastfeeding are foundational to the healthy development of young children. In all, the findings provide evidence of the importance of multi-sectoral interventions targeted at improving children’s health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the global climate changes and the populations of many African countries grow, ensuring clean drinking water and nutritionally adequate food has become a pressing concern. Because of their vulnerability to malnutrition and food insecurity, children face the greatest risk for adverse health outcomes related to climate change (Grace et al. 2012). Vulnerability, however, is highly variable, with some children in agriculturally productive communities showing signs of malnutrition, while other children in communities characterized by less agricultural production show signs of healthy growth (for example, Grace et al. 2016). Because agriculture production does not explain all of the malnutrition variability, the reasons underlying malnutrition may be related to other aspects of food security beyond local production, for example, food accessibility or illnesses (see also Ruel et al. 2013).
West Africa is one of two regions in the world where malnutrition has actually been increasing (WHO/UNICEF 2015). And while Burkina Faso does not have the highest share of malnutrition in the West African region, chronic malnutrition is common and is characteristic of approximately 1 in 3 children (WHO/UNICEF 2015; Beiersmann et al. 2012). One potential explanation for the observed levels of malnutrition is contaminated drinking water. If the available water for drinking is contaminated with bacteria or heavy metals, then food that is ingested might not be properly absorbed by the body—contributing to malnutrition (Blanton et al. 2016; Humphrey 2009). Recently, and as part of a broader plan to achieve the Millennium Development Goals of reducing child mortality, Burkina Faso has made great strides in reducing the number of people dependent on sub-optimal sources of surface water that may have contamination issues (WHO/UNICEF 2015).
However, water source is not the only piece of the malnutrition puzzle. The overwhelming majority of Burkinabé rely on locally grown, rain-fed agriculture in addition to locally procured shallow well or surface water. Therefore, even if the water source is not contaminated, if local food production is not sufficient or the food that is locally available is too costly, then people may face nutrition or caloric deficits that ultimately lead to malnutrition. Thus, failures in either the food system or the water system could provide the underlying explanation for the levels of malnutrition. Further, these two separate but related systems could act in concert to have dramatic negative impacts on children’s health. Despite the importance of understanding the linkages between water and malnutrition, no spatially relevant analysis of the combined interaction between water source and food insecurity and children’s health exists for Burkina Faso.
The goal of this research is to examine how unsafe drinking water and children’s anthropometry correlate after adjusting for community-level food availability. Because of the vulnerability and risks faced by very young children as they transition from ingesting breastmilk to water and complementary foods (Jones et al. 2003; Marino 2007; WHO/UNICEF 2003), we focus on Burkinabé children under 2 years of age (complementary foods are recommended to be introduced when the child reaches 6 months of age with breastfeeding recommended to at least age 2 years). To accomplish this goal, we rely on a publicly available, highly detailed, geo-referenced data set, the Demographic and Health Survey (DHS), to provide information on measures of childhood malnutrition and details on parental characteristics related to children’s health. We evaluate specific children’s anthropometric measures that are related to undernutrition—weight and height—and include measures of local food production as well as measures of both water quality and household drinking water source. Information on water source (covered/uncovered well, piped water, etc.) and water quality (measures of arsenic and bacteria) comes from DHS along with recently collected geo-referenced data from the US Agency for International Development (USAID). As no micro-level measures of food production exist, we estimate this critical information using remotely sensed vegetation data as has been done in other studies (see Shively et al. 2015; Brown et al. 2014; Grace et al. 2016).
Setting
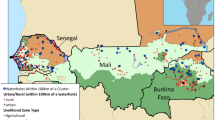
In terms of the Human Development Index, Burkina Faso ranks 181 (out of 187), making it one of the poorest nations in the world. Roughly 44% of the Burkinabé population live below the poverty level (UNDP 2015). The majority of Burkinabé are poor, rural semi-subsistence farmers who are heavily reliant on rain-fed crops—sorghum, millet, and corn—and some livestock to meet their dietary needs (FEWS NET 2010). In 1990, an estimated 44% of the population had access to improvedFootnote 1 drinking water. However, Burkina Faso has made rapid strides in improving access to quality drinking water, and now, it is estimated that 82% of the population has access to improved drinking water (WHO/UNICEF 2015). And while Burkina Faso is “on track” to meet the Millennium Development Goals for drinking water, malnutrition is consistently high, and overall levels of poor health in children are relatively unchanging (INSD 2012). Please see Fig. 1 for a map of drinking water characteristics across the country.
The reason Burkina Faso is the focus of this study on water, food, and children’s health is because of the persistent levels of child malnutrition despite the dramatic improvements made in access to improved sources for drinking water. One potential reason underlying this chronic and persistent malnutrition in the context of access to improved water is insufficient food availability (Olney et al. 2015). If a community has clean drinking water but insufficient food availability, then undernutrition may persist. Alternatively, in the case that there is adequate food availability but the drinking water is contaminated (leading to diarrhea, for example), children can also show signs of undernutrition (Marino 2007). Studies focusing on different aspects of undernutrition in Burkina Faso have highlighted the importance of community-level factors, including those that might relate to annual food or agricultural production (Olney et al. 2015) as well as other socio-cultural characteristics (Beiersmann et al. 2013).
Breastfeeding offers an important and widely recommended strategy for protecting the very youngest children from contaminants and also providing them high-quality nutrition (WHO/UNICEF 2003). However, anecdotal reports from Burkinabé women suggest that breastfeeding can be both physically and psychologically taxing on the mothers themselves if there is no adequate food to support their increased caloric needs. And while food insecure Burkinabé mothers do not report modifying their breastfeeding behaviors because of inadequate food, they do report a decrease in milk production and milk quality during periods of prolonged food shortages (ibid). Indeed, if mothers are producing less milk (in terms of quantity or quality), this may lead their children to rely more heavily on water and complementary foods—placing their children at risk. Therefore, decreased food availability may result in a change in the amount or type of nutrition available to a child directly or indirectly, through the mother’s constraints. Unsafe water may deprive the child of nutrients even in the context of adequate food availability. While the cross-sectional data employed here cannot distinguish the causal effects relating to inadequate food and unclean water, we can use the data to investigate the role of potential factors. Evaluating the relationship between food, water, and children’s health outcomes is necessary to determine the reasons for, and the potential solutions to, chronic malnutrition in this context.
Food, water, and children’s health
We develop our research framework, integrating water source/quality and food availability, around the widely used conceptual model of food security combined with the UNICEF conceptual framework of children’s undernutrition (Black et al. 2008). Food security is defined as “when people, at all times, have physical, social and economic access to sufficient, safe and nutritious food to meet their dietary needs and food preferences for an active and healthy life” (World Food Summit 1996). For both policy and research, this definition provides the basis for the four pillars of food security—access to food, availability of food, utilization of food, and stability of food (see also the USDA report by Brown et al. 2015). In the event that any of these pillars is insufficient, that person, household, or community is considered to be food insecure. UNICEF develops their undernutrition framework with the idea that there are multiple levels of influential factors—immediate factors, underlying causes and broad factors related to the social/economic/political context (Black et al. 2008). The immediate factors are insufficient dietary intake and disease; the underlying causes refer to household and community factors relating to food, care, and health. This research primarily considers the underlying factors with attention to the variability in anthropometric outcomesFootnote 2 related to food insecurity and health of individual children.
In terms of the four pillars of food insecurity, the third pillar, utilization, refers to the body’s ability to process nutrients and can be affected by disease or by insufficient nutrients in the food (Brown et al. 2015). Utilization is the main focus of our analysis. In the developed world and in some developing countries, the utilization pillar can be associated with poor quality foods—high sugar, fat, or processed foods (Kennedy et al. 2006; Doak et al. 2005). Utilization can also be associated with a variety of illnesses that is an important component of both the underlying and immediate causes of child undernutrition in the UNICEF model. In the case of illness, even if adequate calories of high nutritive quality are ingested, and the body is unable to process these calories because the individual is afflicted with some illness, then the individual is considered food insecure.
Contaminated drinking water has been shown to provide one source of illness that leads to the inability of an individual to absorb the macronutrients and micronutrients needed in their diet and may be related to environmental enteropathy (EE) (Donowitz and Petri 2015; Gilmartin and Petri 2015). EE is any disease of the intestine that reduces intestinal nutrient absorption while creating short-term inflammatory sites associated with catabolic metabolism (Weisz et al. 2012; Bhutta et al. 2008; Gilmartin and Petri 2015). In Bangladesh, children (including those recently weaned) were determined to have negative growth patterns as a result of EE (Einstein et al. 1972). In Malawi, Weisz et al. (2012) examined the effects of EE due to a lack of unsafe drinking water. The authors found that even when children had enough to eat, they were not able to absorb the nutrients in the food. Similarly, acute infections due to unclean drinking water affected linear growth of children substantially.
While not specifically focusing on EE, other studies have focused more on the water source. Nagahori et al. (2015) found that the source of household drinking water was a significant indicator for stunting, where 58% of the children whose mothers used water from village wells were malnourished. The specific type of food given to the children such as fruits, cereals, or dairy products had no significant impact on child’s health status after accounting for water source.
The influence of water quality on health is complex to accurately measure because the collection and storage of water may allow for contamination even if the source of the drinking water is optimal (Wright et al. 2004; Kremer et al. 2007). While acknowledging these limitations, researchers have found a number of linkages between type of water source and children’s health outcomes that suggest that water source may explain at least part of children’s negative health outcomes (Günther and Fink 2010). The WHO/UNICEF Joint Monitoring Program identifies “improved” and “un-improved” water sources with the suggestion that improved water sources are more likely to minimize health risks and negative health outcomes. We use these broad categories to group the water sources in our analysis.
Specific sources of contamination found in water
A portion of this analysis is focused on examining specific types of contaminants—anthropogenic in origin as well as natural. Here, we provide a brief overview of the related research on these two different sources of contamination.
Microbial water contamination
Microbial contamination is typically anthropogenic in origin and generally results from poor water management where fecal disposal is too close to the drinking water source (Ashbolt 2004). This contamination can result in the presence of various bacteria or viruses living and breeding at the point-of-use. In developing countries, the microbial contamination of household drinking water is implicated in the prevalence of various diseases. One of the most commonly occurring and studied bacteria is E. coli (Gundry et al. 2004), which is often used as an indicator for water quality, along with fecal coliforms (thermo-tolerant). In addition to causing diarrhea, E. coli can cause serious illness, such as urinary tract infections, bacteremia, and meningitis (WHO 2008).
While the WHO does not have a standard on the amount of total coliforms (combined measure of bacterial contamination including fecal and E. coli) in drinking water, the United States and Canada both have limits of <1 CFU per 100 mL and 0 CFU per 100 mL, respectively (WHO 2008). Like E. coli and fecal coliforms, the presences of Streptococcus in drinking water can also indicate fecal contamination and can cause strep throat in children and adults alike. Multiple studies have shown the effect of bacterial contamination on the long-term health of children (Donowitz and Petri 2015; Khin-Maung-U et al. 1990; Omoike and Abiodun 1989), commonly causing diarrhea and other digestive disorders. Small intestine bacterial overgrowth (SIBO), a particularly prevalent digestive disorder, causes impaired micronutrient absorption and increased gastrointestinal permeability (Donowitz and Petri 2015). Donowitz and Petri (2015) found high rates of SIBO in children found living in unsanitary conditions with low access to clean water. Even if a high level of food is available, SIBO diminishes the absorptive surface in the intestines leading to deficiencies in B12, fat-soluble vitamins, and carbohydrates. In addition to causing SIBO, bacterial consumption from water source has been linked to undernutrition, as measured by children’s weight and height (Esrey et al. 1988; Omoike and Abiodun 1989).
Trace element water contamination
While microbial water contamination is anthropogenic in origin, trace element contamination, which is often naturally occurring, can also have negative effects on children’s health and well-being. One element of increasing interest in terms of children’s health is arsenic, which has been found in deep, borehole wells in northern Burkina Faso (Smedley et al. 2007). The world health organization prescribes a maximum concentration of 10 ppb arsenic in safe drinking water (WHO 2008). This maximum limit was set because of the negative health effects that come from consuming arsenic. The effects of high arsenic in drinking water on human health have been well studied for the past few decades, with a small selection of studies including the effects on children’s health (Wasserman et al. 2004). Increased ingestion of arsenic in drinking water can lead to skin lesions, hyper-keratosis, skin cancer, and liver disease (Karim 2000).
As a teratogen, arsenic is capable of crossing the placental membrane from a mother to the metabolic system of an unborn child and therefore may impact children from birth (Karim 2000). An intellectual function test of 201 children, age 10 from Araihazar, Bangladesh, showed that children exposed to arsenic through their drinking water had reduced intellectual function. This reduction followed a dose-response manner, where children with exposures <50 ppb arsenic received significantly lower performance and full-scale scores (Wechsler Intelligence Scale for Children version III) than did children with exposures to <5.5 ppb arsenic (Wasserman et al. 2004). In addition to the effects on children’s intelligence, arsenic consumption has been found to influence stunting and wasting in children (Gardner et al. 2012; Saha et al. 2012; Minamoto et al. 2005). Interestingly, the importance of breastfeeding duration in early childhood development has also become apparent as a means of protection from arsenic exposure in early childhood (Saha et al. 2012).
Some of the major challenges to advances in research of children’s health, water quality, and contaminated water are lack of necessary data. Here, we are faced with many of the same limitations but seek to provide evidence as to the importance of examining the interactions and linkages between food and water. We provide an analysis of the linkages of both microbial and trace element contamination to children’s health with several caveats that will be discussed in more detail in the following section.
Data
The three data sources used in this analysis are the Demographic and Health Survey data, a small set of well data from the USAID West Africa Water Sanitation and Hygiene (WA-WASH) program, and satellite remote sensing information. These data are described below.
DHS data
The data for children’s health variables and household water source come from the 2010 Burkina Faso Demographic and Health Survey (DHS). This is the most recently collected, country representative health data. The data contain information on household water source for a representative sample of households across the country. The data also contain detailed health information for the children living within the household as well as detailed information on parental and household characteristics relating to household size, household wealth, and other socio-demographic factors. The data are spatially referenced at the level of the “cluster.” A DHS cluster contains approximately 20 households, and we use the spatial information of the cluster to merge the DHS data with additional sources of data on water quality and food production.
We select a subset of the full data and include only children 2 years old and younger.Footnote 3 This restriction is made because of the breastfeeding norms in the country—where the overwhelming majority of children are breastfed through their second year of life. Because of the potential for correlation between children within the same household, we select only one child, the youngest child from each household meeting the 25 months or younger criterion.
WA-WASH well data
In the summer of 2014, the USAID funded WA-WASH to collect water quality samples from wells in subsistence-farming communities in central Burkina Faso. They collected 217 samples in 15 different communities focusing on measuring microbial pathogens (E. coli, fecal coliforms, total coliforms, and streptococcus) as well as arsenic. This data covers only a small area of Burkina Faso and provides information at a later date than the DHS survey. The water samples do not include additional information about health and community characteristics; therefore, to evaluate human health and water quality, we must link the data with health information from the DHS. We primarily use this data to provide contextual information for the DHS villages nearby to those of the WA-WASH well sampled villages. While an ideal dataset would contain both health and water quality information collected together, we build on geographic theory, specifically Tobler’s First Law, and assume that villages near each other are going to be more alike than distant villages (Tobler 1970; Miller 2004). Given that some of the underlying aspects of contamination reflect cultural practices and/or geophysical properties, assuming similarities between neighboring villages is not unreasonable. In this way, we “borrow” information from a WA-WASH village to provide the additional water quality information to a nearby DHS cluster.
Remotely sensed data
As is often the case in developing countries, there are no readily available, yearly estimates of micro-level food production in Burkina Faso. To approximately measure food production, we rely on multispectral remotely sensed data. Specifically, we use the normalized difference vegetation index (NDVI), a measure widely used to provide an indication of “greenness” or vegetation density and health (de Beurs and Henebry 2004; Funk and Budde 2007; Grace et al. 2014; Townshend and Justice 1986; Tucker 1979; Grace et al. 2012). The NDVI data are gathered from the NASA Terra satellite (Huete et al. 2002; Justice et al. 1998). Moderate Resolution Imaging Spectroradiometer (MODIS)-based NDVI have been collected since 2000 and are available at a spatial resolution of 250 m for all of Burkina Faso.
Vegetation indexes, including NDVI, are frequently used to calculate surrogate measures of cultivation and are therefore often linked to food production and can serve as proxies for local food availability (Grace et al. 2016; Grace et al. 2014; Shively et al. 2015; Brown et al. 2014). Maximum vegetation (when the NDVI value is at its peak) typically occurs later during the growing season, near the time of harvest. By evaluating the maximum NDVI value during the growing season of a given year at each pixel, we can determine if a given area is likely to be cultivated, and in this way, we can determine the density of cultivation in the area around the DHS cluster. NDVI maximum values are available every 10 days at each pixel. A standard approach when using NDVI, which we use here, is to temporally aggregate the 10-day averages into a seasonal maximum value for each pixel (Shively et al. 2015; Grace et al. 2014). Finally, consistent with the recommendations from DHS, we spatially aggregate the NDVI values for each DHS cluster using a 10-km buffer (Burgert et al. 2013).
There are two important limitations in our use of NDVI as a proxy for local food production: (1) NDVI is not capable of differentiating between cash and food crops and (2) community-level cultivation does not necessarily indicate shared access to the agricultural production. For the first concern, we hypothesize that if cultivation is high in a community, then either the community has more available food (if engaged in food crops) or more available income (if engaged in cash crops). In either case, a higher NDVI will still be indicative of more food in the community. For the second concern, qualitative interviews with women and men in Burkina Faso and anecdotes shared with the first author suggest that feeding children is often the responsibility of the community. In the case that a particular house has a hungry child, the parents will send the child to neighbors to eat. Given this widespread cultural attitude towards children and food, we theorize that access to food is roughly equivalent among children in a particular village. However, we do note the potential for certain groups of children to be more vulnerable to food insecurity within villages (see Grace et al. 2016).
Methods and measures
Country-level analysis
We use two different categories of anthropometric information—a measure associated with chronic malnutrition and one associated with moderate malnutrition. To measure chronic undernutrition, we focus on the height-for-age z-score (HAZ) of the youngest child in each family. To measure moderate malnutrition, we focus on the weight-for-age z-score (WAZ) of the youngest child in the household. HAZ and WAZ are both continuous variables and are analyzed using standard normal regression (McCullagh and Nelder 1989). Our primary goal is to evaluate how HAZ and WAZ vary according to household water source, with attention to local food production. DHS provides detailed information on the source of water for each household. We rely on community-level NDVI to estimate food availability in the community. Depending on the age of the child, each child has been exposed to either one or two growing seasons that may have impacted the child’s health status.
Because of different breastfeeding needs of infants as compared to toddlers as well as different nutritional demands and relevant harvest seasons of these very young children, we opted to consider this population separately from the older children. To account for the differential exposures and breastfeeding norms, we therefore analyze two different sub-populations of children—those under 13 months old and those between 13 and 25 months old. We include an average of both the 2008 and 2009 community food-production proxy for the older group as we assume that the food harvested in 2008 would be among the first foods that they would eat as they are introduced to solid foods. For the younger group of children, we include only the 2009 food production measure as the 2008 production would have no direct impact on the local food available to them.
In addition to independent variables of primary focus to the analysis, household water source, and community food production, we also include control variables that correspond to our theoretical framing and that are known to correlate with undernutrition at the individual, household, and community levels. The control variables included in the analysis are further explained here.
Maternal characteristics such as maternal age, education, and weight (at the time of survey) are included in the analysis. Younger mothers may have fewer resources and also be less experienced at caring for children—these factors may impact children’s health in a negative way (Black et al. 2008). Maternal education may relate to a mother’s understanding of nutrition, water quality, and sanitation as well as her ability to identify that one of her children may be undernourished. It may also serve as a measure of household wealth (Walker et al. 2011; Grace et al. 2015; Engle et al. 1999). Because of the potential link with household wealth, if a community’s education level is higher than other communities, improved water sources may be available and therefore potentially accessible to less educated people in the community. To capture this potential source of variation, we also include average community-level education. Maternal weight can serve as a measure of household resources at the time of the survey, with the assumption that a heavier mother may be in a household with more food resources or it may capture some biological aspect of growth (Black et al. 2008). Child characteristics are also included as controls. As is standard in most studies of children’s health and nutrition, we include children’s age and sex. Additionally, we include the birth order with the assumption that a higher birth order, indicative of a larger family, may potentially reduce the resources available to a young member of the household. We also include the child’s birth weight, which may correlate to early feeding practices, care behavior as well as relate to the child’s weight and weight gain (Walker et al. 2011; Black et al. 2008)
We also include a variable relating to breastfeeding behaviors. As we are examining children who are about 25 months or younger, breastmilk is anticipated to remain a potentially important component of their diet with potentially important impacts on the pace of growth of infants (see Kramer et al. 2011). Breastmilk is free of biological contaminants and is encouraged by the WHO and other similar health organizations because it provides a highly nutritious, reliable, and important food in the early years of a child’s life that may protect them against illnesses related to malnutrition (Afifeen et al. 2000; Huttly et al. 1997; Slusser 2007). The WHO recommends exclusive breastfeeding until 6 months of age followed by breastfeeding in combination with complementary foods until 2 years of age. The relationship between a child’s growth (weight or height) and breastfeeding behaviors generally suggests that exclusively breastfed children may grow more slowly during the first year of life (Kramer et al. 2011; Kramer et al. 2007). As breastfeeding practices can vary depending on a child’s age—older children may rely heavily on foods other than breast milk as they near weaning age which may make them more vulnerable to contaminants—we include a measure of the frequency of ingesting something other than breastmilk over the previous 24 h. Finally, to account for infrastructure that may assist in providing cleaner water as well as variations in population density, we include information on if the household is in an urban or rural area and the type of toilet facility the household uses. These variables are highlighted in Table 1.
Community-focused analysis
In this analysis, we use the information from the WA-WASH well data to inform our investigation of water quality and children’s health. Because the USAID water quality data has only been gathered for a small number of villages, we must estimate the arsenic and bacteria levels of villages that were not sampled. We specifically evaluate villages that are within 10 and 20 km of the sampled areas, Fig. 2. We assume that villages near to the sampled areas may have similar water quality characteristics as the sampled areas. As DHS does not collect water quality information and WA-WASH does not collect household health information, we must qualitatively extrapolate information from the WA-WASH survey to help inform the interpretation of the results. We link DHS clusters to sampled sites by using a buffer—any cluster within 20 km is included in the analysis and is identified as either within 20 or 10 km of a specific village.
Because DHS data is only available at the spatial resolution of 10 km, we are not able to estimate on a finer spatial scale. We then conduct a regression analysis of the individuals who live within one of the spatially relevant villages, and we include village of nearest proximity, one of the eight listed in Table 2, as one of the fixed effects in the regression analysis. We also include the approximate distance to the sampled site, either 10 or 20 km. The sample size in this analysis, restricted to children 25 months or younger who live within 20 km of one of the sampled villages, is 211. For this portion of the analysis because of the smaller sample size, we restrict the number of independent variables included in the analysis to those that were typically significant in the larger country-level analysis.
Results
Because this analysis is cross-sectional, we do not make any causal claims. Our results present correlations between factors and are useful in considering different sources of variation.
Country-level results
We first begin with the results of the country-level analyses of individual children’s health outcomes as they relate to household water source, while accounting for variation in community-level NDVI. Table 2 presents the results of the analysis—the regression coefficients, significance values, and standard errors. Note that mother’s educational attainment is not included in the table, as the correlation between household water source and maternal education attainment was too high to include both variables in the same model. We do, however, include the community average educational maternal attainment.
In the first column, we evaluate chronic malnutrition as measured by a child’s HAZ (height-for-age standard deviations from the reference median) among the older children in our sample. The results highlight the significance of maternal weight, child’s sex, child’s age, child’s birth weight, and child’s birth order. Female children who were heavier at birth and whose mothers are heavier are less likely to have a low HAZ value. Additionally, if children were fed something other than breastmilk only once in the previous 24-h period, they report lower HAZ values. Toilet facility and local, community-level food production (NDVI) do not have a significant correlation to the HAZ. Average educational attainment of women in the community is associated with healthier children.
In the third column, we evaluate the correlates of malnutrition as measured by WAZ (standard deviation of weight-for-age from a reference median) of the older children in the sample. Here, we again see the significance maternal weight, child’s sex, and birth weight. The direction of the relationships is the same as with HAZ—girls weighing more at birth who have heavier mothers are more likely to weigh more than their same height counterparts. Again, communities where mothers have a higher average level of educational attainment are more likely to have children who weigh more. Children from households who rely on surface water are more likely to weigh less for their heights.
We also analyzed the same malnutrition outcome variables using the younger population—children who were around 6 months old or younger. We separated out this age group because of the exclusive breastfeeding recommendations of the WHO (<6 months of age). Indeed, among this subset, less than 3% of the children consumed anything other than breastmilk in the previous 24 h and we consequently removed this variable from the analysis. In terms of the variables of focus in this analysis—water source and food production—food production was positively correlated to a child’s HAZ while there was no significant relationship between water source and HAZ. Child characteristics were significantly related to HAZ as was toilet facility. In terms of WAZ, we see the significance of child characteristics and maternal weight. Additionally, toilet facility is significant with children more likely to weigh less for their height in households without a flush toilet. Children who live in communities with more food production during the most recent growing season are also more likely to weigh more. Overall, for the very youngest children, a higher measure of community food production is correlated to greater HAZ and WAZ scores even after adjusting for individual, household, and community features related to food insecurity.
Community results
In this section, we focus on the results for DHS survey clusters in villages that are within 10 and 20 km of the WA-WASH sampled areas. Given the paucity of the WA-WASH data, we suggest care when interpreting the results and encourage the use of the results as descriptive and providing indication of potential relationships that require further study. Before adjusting for any effects and to provide descriptive detail, all eight villages had negative HAZ and WAZ values, with the village of Tama and Dalgane having the highest average HAZ (−1.39) and WAZ (−1.38), respectively. Dalgane has the second highest WAZ (−1.25) and Tama has the third highest WAZ (−1.22). Dalgane had an average measured arsenic concentration of 5.5 ppm, but with only 2 wells measured. Arsenic levels elevated above WHO recommendation of 10 ppb were reported in the villages of Lekoro, Bena, and Moulourou. The highest counts of total coliform (CFU) were reported in Biron-Marka and Lekoro (749.9 and 794.4 CFU, respectively). Additionally, the village of Lekoro had the highest reported incidences of Streptococus (600.8 CFU). Lekoro reported an average HAZ of −0.805 and average WAZ of −1.09. Table 3 summarizes the child health outcomes from the DHS data and the water quality data according to the villages used in this analysis.
In Table 4, we present the results for HAZ and WAZ, and because of the smaller sample sizes, we evaluate only the children between 7 and 25 months of age. We also restricted the number of independent variables of the variables used in the analysis. The smaller sample size produced some categories that were too small for inclusion. Ultimately, the results, as in the previous regression models, indicate the significance of individual child and maternal factors related to anthropometry. In households where shallow wells, protected or unprotected, provide the water, children are more likely to be shorter for their ages than in households with water provided by a tap in the dwelling. After controlling for the independent variables, children living in households in Tama are more likely to be short for age as compared to children living in Vipalogo (the most common community in our sample).
In terms of weight-for-age, maternal weight, child’s birth weight, and child’s birth order are significant. Children living in household where water is gathered from surface sources are more likely to weigh less as compared to those with water piped into their dwelling. In terms of variation due to village, our results suggest that there is no significant difference between villages. Our results do show, however, that an increase in food production and as well as a higher level of community educational attainment are positively related to weight for age.
Discussion
Water quality and food production can have important impacts on children’s health, and illness and undernutrition can occur together and create extreme challenges for early child health (Marino 2007; Peletz et al. 2011). We investigated the correlation between estimated community food production, water source, and very young children’s weight and height to better understand the correlations between these factors. To evaluate more fine scale information on water quality and health, we also conducted an analysis using more detailed water quality information from recent well surveys. The results of the analysis suggest the consistent and important role of individual and maternal characteristics and, in some cases, pointed to the important role of water source, food production, and the estimate of community-level educational attainment. The role of local food production or household water source was not consistent across the different groups analyzed; however, maternal anthropometric measures and child characteristics were significant across the range of models. The community-level measures of educational attainment and food production were correlated, in several instances, with the weight and heights of the very youngest children across Burkina Faso. The results therefore suggest that strategies to improve children’s height and weight gain should address issues of food availability, maternal nutritional and support and education, as well as household water source.
The source of a household’s water seemed to have a limited relationship to the height and weight outcomes of children. The models indicated that, in some cases, when children live in households that source their water from shallow wells (protected or unprotected) or from surface sources, the children have less healthy growth outcomes. Overall, the more consistent result from this analysis is the role of community—both in terms of food production and in terms of maternal education. In communities where there was more food, the very youngest children reported larger HAZ and WAZ than in communities with less food, regardless of the household water source. In virtually every country-level model, when the educational attainment of the sampled women in the community was higher, children’s HAZ and WAZ scores were higher. Community characteristics may indicate an increase in infrastructure or community resources and may contribute to the healthy development of children—even in the case where the particular household relies on an unimproved water source (see also Bryce et al. 2008).
The heights and weights of the very youngest children, those 6 months of age and younger, were positively related to community-level food production. While children under 6 months of age in Burkina Faso are not always exclusively breastfed, ingesting anything other than breastmilk was not common in our sample (see Sawadogo et al. 2010). Because their diets are dominated by breastmilk, the significance of food production requires some further investigation. Breastfeeding is potentially costly to the mother, both in terms of time, psychological status, and nutrition; these costs must be considered in the context of food insecurity, seasonal hunger periods, and changes in economic opportunities that might influence the caretaking of small children (see Rippeyoung and Noonan (2012) for discussions about the costs of breastfeeding to women). We theorize that in communities with more food production, that nursing mothers are potentially better nourished themselves which may facilitate nursing. It is also possible that in communities with less food, to produce enough food to feed themselves and any other dependents, women may work in the fields away from their children. Ultimately, however, the behavioral and biological components of breastfeeding in food insecure contexts in poor countries are not well understood.
Evaluating all of the results together, however, the importance of maternal physical characteristics and child’s birth weight—heavier mothers have healthier children and babies born weighing more are more likely to be taller and weigh more than their counterparts—reinforces the importance of focusing on women’s health and nutrition throughout a woman’s life. Birth weight is partially an outcome of conditions impacting the mother during pregnancy (Grace et al. 2014; Grace et al. 2015); thus, if the mother’s health and well-being is protected and her child is born at a healthy birth weight, this outcome will have an important impact through the second year of life for the child.
In addition to DHS variables, we were able to perform limited analysis with the WA-WASH water quality data. The villages of Tama and Dalgane had the highest that show a greater degree of delayed or impaired growth and weight gain. Even after accounting for variations in children’s and maternal characteristics, children from Tama remain significantly more likely to show signs of malnutrition. Due to the paucity of data, it is difficult to draw meaningful conclusions. However, it is interesting to note when comparing the water quality of Tama to that of the other seven villages, Tama only has a higher than average concentration of total coliforms, with the rest of the water quality characteristics lower than the averages for all of the villages. Conversely, the town of Dalgane, with similar anthropometric averages, reports higher than average values for fecal coliforms, total coliforms, Streptococcus, and arsenic.
Future research of household water storage and management behaviors, beyond simply just water source, will help to highlight the relationships between water, food, and malnutrition and will be particularly relevant as changes in climate lead to variability in water sources and food production. Additionally, further investigation in how families and communities manage food and water scarcity, storage, and sharing (factors that are potentially related to both the independent and dependent variables in this analysis) will help to reveal more specific mechanisms that individuals and communities use to protect their health and their children’s health (Kazianga et al. 2014). In the meantime, while investing in improved water sources is important, continued investment in maternal health, supporting women during pregnancy and breastfeeding, is bound to yield lasting and significantly positive health outcomes for children (see also Ruel et al. 2013). Our research shows that even in very poor communities with limited food and water resources, investing in community resources related to education and food production and in women’s health over the course of their lives can help to remove important environmental and developmental constraints.
Notes
Improved water sources include the following: covered wells, protected springs, and piped water (into community or dwelling); and unimproved water sources include the following: unprotected spring, unprotected shallow wells, and surface water.
Height and weight are commonly used measures of healthy growth. When height-for-age or weight-for-age z-scores fall below 2 standard deviations from a reference median, children are considered stunted or moderately malnourished, respectively. Both stunted and moderately malnourished children are at increased risk for illness and death (WHO 2006).
In the analysis, we select out all children who are 25 months and younger to include those who have just turned 2 years old as well.
References
Arifeen, Shams E., et al. (2000). "Infant growth patterns in the slums of Dhaka in relation to birth weight, intrauterine growth retardation, and prematurity." The American journal of clinical nutrition, 72.4, 1010–1017.
Ashbolt, N. J. (2004). Microbial contamination of drinking water and disease outcomes in developing regions. Toxicology, 198, 229–238.
Beiersmann, C., et al. (2012). Malnutrition in young children of rural Burkina Faso: comparison of survey data from 1999 with 2009. Tropical Med Int Health, 17.6(2012), 715–721.
Beiersmann, Claudia, et al. (2013). Malnutrition determinants in young children from Burkina Faso. Journal of Tropical Pediatrics: fmt037.
Bhutta, Z. A., Ahmed, T., Black, R. E., Cousens, S., Dewey, K., Giugliani, E., Haider, B. A., Kirkwood, B., Morris, S. S., Sachdev, H. P. S., & Sekar, M. (2008). What works? Interventions for maternal and child undernutrition and survival. Lancet, 371(9610), 417–440.
Black, R. E., Allen, L. H., Bhutta, Z. A., Caulfield, L. E., De Onis, M., Ezzati, M., Mathers, C., Rivera, J., & Maternal and Child Undernutrition Study Group. (2008). Maternal and child undernutrition: global and regional exposures and health consequences. Lancet, 371(9608), 243–260.
Blanton, L. V., et al. (2016). Gut bacteria that prevent growth impairments transmitted by microbiota from malnourished children. Science, 351(6275), aad3311.
Brown, M. E., Kathryn, G., Shively, G., Johnson, K. B., & Carroll, M. (2014). Using satellite remote sensing and household survey data to assess human health and nutrition response to environmental change. Popul Environ, 36(1), 48–72.
Brown, M. E., Antle, J. M., Backlund, P., Carr, E. R., Easterling, W. E., Walsh, M. K., Ammann, C., Attavanich, W., Barrett, C. B., Bellemare, M. F., Dancheck, V., Funk, C., Grace, K., Ingram, J. S. I., Jiang, H., Maletta, H., Mata, T., Murray, A., Ngugi, M., Ojima, D., ONeill, B., & Tebaldi, C. (2015). Climate change, global food security, and the U.S, Food System. Washington DC: United States Department of Agriculture. USDA Technical Document. doi:10.7930/J0862DC7.
Bryce, J., et al. (2008). Maternal and child undernutrition: effective action at national level. Lancet, 371(9611), 510–526.
Burgert, C. R., Colston, J., Roy, T., & Zachary, B. (2013). Geographic displacement procedure and georeferenced data release policy for the demographic and health surveys. DHS spatial analysis reports no. 7. Calverton, Maryland, USA: ICF International.
de Beurs, K. M., & Henebry, G. M. (2004). Trend analysis of the pathfinder AVHRR land (PAL) NDVI data for the desert of Central Asia. IEEE Geosci Remote Sens Lett, 1, no. 4.
Doak, C. M., Adair, L. S., Bentley, M., Monteiro, C., & Popkin, B. M. (2005). The dual burden household and the nutrition transition paradox. Int J Obes, 29(1), 129–136.
Donowitz, J. R., & Petri Jr., W. A. (2015). Pediatric small intestine bacterial overgrowth in low-income countries. Trends Mol Med, 21.1, 6–15.
Einstein, L. P., Mackay, D. M., & Rosenberg, I. H. (1972). Pediatric xylose malabsorption in East Pakistan: correlation, with age, growth retardation, and weanling diarrhea. Am J Clin Nutr, 25(11), 1230–1233.
Engle, P. L., Menon, P., & Haddad, L. (1999). Care and nutrition: concepts and measurement. World Dev, 27(8), 1309–1337.
Esrey, S., Habicht, J., Latham, M., Sisler, D., & Casella, G. (1988). Drinking water source, diarrheal morbidity, and child growth in villages with both traditional and improved water supplies in rural Lesotho, southern Africa. Am J Public Health, 78(11), 1451–1455.
FEWS NET. 2010. Burkina Faso Livelihood Zones. Accessed November 25, 2015. http://www.fews.net.
Funk, C., & Budde, M. (2007). National MODIS NDVI-based production anomaly estimates for Zimbabwe. Climate Hazards Group: Department of Geography University of California, Santa Barbara.
Gardner, Renee M. Maria Kippler, Fahmida Tofail, Matteo Bottai, Jena Hamadani, Margaretha Grander, Barbro Nermell, Brita Palm, Kathleen M. Rasmussen, and Marie Vahter. (2012). Environmental exposure to metals and Children’s growth to age 5 years: a prospective cohort study. Am J Epidemiol 177, no. 2.
Gilmartin, A. A., & Petri, W. A. (2015). Exploring the role of environmental enteropathy in malnutrition, infant development and oral vaccine response. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 370 .no. 1671
Grace, K., Davenport, F., Funk, C., & Lerner, A. M. (2012). Child malnutrition and climate in sub-Saharan Africa: an analysis of recent trends in Kenya. Appl Geogr, 35, 405–413.
Grace, K., Brown, M., & McNally, A. (2014). Examining the link between food prices and food insecurity: a multi-level analysis of maize price and birthweight in Kenya. Food Policy, 46, 55–65.
Grace, K., Davenport, F., Hanson, H., Funk, C., & Shukla, S. (2015). Linking climate change and health outcomes: examining the relationship between temperature, precipitation and birth weight in Africa. Glob Environ Chang, 35, 125–137.
Grace, K., Nagle, N. N., & Husak, G. (2016). Can small-scale agricultural production improve Children’s health? Examining stunting vulnerability among very young children in Mali, West Africa. Annals of the American Association of Geographers, 106(3), 722–737.
Gundry, S., Wright, J., & Conroy, R. (2004). A systematic review of the health outcomes related to household water quality in developing countries Journal of Water and Health, 2 .no. 1
Günther, I., and G Fink. (2010). Water, sanitation and children’s health: evidence from 172 DHS surveys. World Bank Policy Research Working Paper Series.
Huete, A., Didan, K., Miura, T., Rodriguez, E. P., Gao, X., & Ferreira, L. G. (2002). Overview of the radiometric and biophysical performance of the MODIS vegetation indices. Remote Sens Environ, 83, 195–213.
Humphrey, J. H. (2009). Child undernutrition, tropical enteropathy, toilets, and handwashing. Lancet, 374(9694), 1032–1035.
Huttly, S. R., Morris, S. S., & Pisani, V. (1997). Prevention of diarrhoea in young children in developing countries. Bull World Health Organ, 75(2), 163.
INSD. (2012). Ministère de l’Économie et des Finances, Ouagadougou, Burkina Faso et ICF International, Institut National de la Statistique et de la Démographie. USA: Maryland.
Jones, G., Steketee, R. W., Black, R. E., Bhutta, Z. A., & Morris, S. S. (2003). Bellagio child survival study group. How many child deaths can we prevent this year? Lancet, 362, 65–71.
Justice, C. O., Vermote, E., Townshend, J. R. G., Defries, R., & Roy, D. P. (1998). The moderate resolution imaging Spectroradiometer (MODIS): land remote sensing for global change research. IEE Transactions on Geoscience and Remote Sensing, 36 .no. 4
Karim, M. (2000). Arsenic in groundwater and health problems in Bangladesh. Water Resources, 34(1), 304–310.
Kazianga, H., de Walque, D., & Alderman, H. (2014). School feeding programs, intrahousehold allocation and the nutrition of siblings: evidence from a randomized trial in rural Burkina Faso. J Dev Econ, 106(2014), 15–34.
Kennedy, G., Nantel, G., & Shetty, P. (2006). The double burden of malnutrition: case studies from six developing countries. Rome: Nutrition Planning, Assessment and Evaluation Service, Food and Agriculture Organization of the United Nations.
Khin-Maung-U, Pereira, S. P., & Bolin, T. D. (1990). The effect of rice carbohydrate malabsorption on child growth: a longitudinal study using the breath hydrogen test in Burmese village children. American Journal Clinical Nutrition, 52, 348–352.
Kramer, M. S., Matush, L., Vanilovich, I., Platt, R. W., Bogdanovich, N., Sevkovskaya, Z., Dzikovich, I. et al. (2007) Effects of prolonged and exclusive breastfeeding on child height, weight, adiposity, and blood pressure at age 6.5 y: evidence from a large randomized trial. The American Journal of Clinical Nutrition, 86(6), 1717–1721.
Kramer, M., et al. (2011). "Breastfeeding and infant size: evidence of reverse causality." American journal of epidemiology: kwq495. doi:10.1093/aje/kwq495.
Kremer, M., J. Leino, E. Miguel, & A.P. Zwane. (2007). Spring cleaning: a randomized evaluation of source water quality improvement. Quarterly Journal of Economics.
Marino, D. D. (2007). Water and food safety in the developing world: global implications for health and nutrition of infants and young children. J Am Diet Assoc, 107(11), 1930–1934.
McCullagh, P., & Nelder, J. A. (1989). Generalized Linear Models 37. Boca Raton: CRC press.
Miller, H. J. (2004). Tobler’s first law and spatial analysis. Ann Assoc Am Geogr, 94(2), 284–289.
Minamoto, K., Mascie-Taylor, C. G., Moji, K., Karim, E., & Rahman, M. (2005). Arsenic-contaminated water and extent of acute childhood malnutrition (wasting) in rural Bangladesh. Environ Sci, 12(5), 283–291.
Nagahori, C., Tchuani, J. P., & Yamauchi, T. (2015). Factors associated with nutritional status in children aged 5–24 months in the Republic of Cameroon. Nursing & Health Sciences, 17(2), 229–235.
Olney, D. K., et al. (2015). A 2-year integrated agriculture and nutrition and health behavior change communication program targeted to women in Burkina Faso reduces anemia, wasting, and diarrhea in children 3–12.9 months of age at baseline: a cluster-randomized controlled trial. J Nutr, 145.6(2015), 1317–1324.
Omoike, I. U., & Abiodun, P. O. (1989). Upper small intestinal microflora in diarrhea and malnutrition in Nigerian children. J Pediatr Gastroenterol Nutr, 9(3), 314–321.
Peletz, R., et al. (2011). Drinking water quality, feeding practices, and diarrhea among children under 2 years of HIV-positive mothers in peri-urban Zambia. AmJTrop Med Hyg, 85(2), 318–326.
Rippeyoung, P. L., & Noonan, M. C. (2012). Is breastfeeding truly cost free? Income consequences of breastfeeding for women. Am Sociol Rev, 77, 244–267.
Ruel, M. T., Alderman, H., & Maternal and Child Nutrition Study Group. (2013). Nutrition-sensitive interventions and programmes: how can they help to accelerate progress in improving maternal and child nutrition? Lancet, 382(9891), 536–551.
Saha, K. K., Engstrom, A., Hamadani, J. D., Tofail, F., Rasmssen, K. M., & Vahter, M. (2012). Pre- and postnatal arsenic exposure and body size to 2 Years of age: a cohort study in rural Bangladesh. Environmental Health Perspectives: Children’s Health, 120 .no. 8
Sawadogo, S. P., et al. (2010). Late introduction and poor diversity were the main weaknesses of complementary foods in a cohort study in rural Burkina Faso. Nutrition, 26.7(2010), 746–752.
Shively, G., Sununtnasuk, C., & Brown, M. (2015). Environmental variability and child growth in Nepal. Health & Place, 35, 37–51.
Slusser, W. (2007). Breastfeeding and maternal and infant health outcomes in developed countries. AAP Grand Rounds, 18(2), 15–16.
Smedley, P. L., Knudsen, J., & Maiga, D. (2007). Arsenic in groundwater from mineralized Proterozoic basement rocks of Burkina Faso. Appl Geochem, 22, 1074–1092.
Tobler, W. R. (1970). A computer movie simulating urban growth in the Detroit region. Econ Geogr, 46, 234–240.
Townshend, J. R. G., & Justice, C. O. (1986). Analysis of the dynamics of African vegetation using the normalized difference vegetation index. Int J Remote Sens, 7(11), 1435–1445.
Tucker, C. J. (1979). Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens Environ, 8(2), 127–150.
UNDP. (2015). Human development report 2015: work for human development. United Nations Development Programme.
Walker, S. P., Wachs, T. D., Grantham-McGregor, S., Black, M. M., Nelson, C. A., Huffman, S. L., Baker-Henningham, H., Chang, S. M., Hamadani, J. D., Lozoff, B., Meeks Garner, J. M., Powell, C. A., Rahman, A., & Richter, L. (2011). Inequality in early childhood: risk and protective factors for early child development. Lancet, 378(9799), 1325–1338.
Wasserman, G. A., Liu, X., Parvez, F., Ahasan, H., Factor-Litvak, P., van Geen, A., Slavkovitch, V., Lolacono, N. J., Cheng, Z., Hussain, I., Momotaj, H., & Gaziano, J. H. (2004). Water arsenic exposure and Children’s intellectual function in Araihazar, Bangladesh. Environ Health Perspect, 112, 13.
Weisz, A. J., Manary, M. J., Stephenson, K., Agapova, S., Manary, F. G., Thakwalakwa, C., Shulman, R. J., & Manary, M. J. (2012). Abnormal gut integrity is associated with reduced linear growth in rural Malawian children. J Pediatr Gastroenterol Nutr, 55(6), 747–750.
WHO. (2006). Child growth standards: length/height-for-age, weight-for-age, weight-for-length, weight-for-height and body mass index-for-age: methods and development. Geneva: World Health Organization.
WHO. (2008). Guidelines for drinking-water quality, Third Edition Incorporating the First and Second Addenda. Volume 1, Recommendations. Geneva: World Health Organization.
WHO/UNICEF. (2003). Global strategy for infant and young child feeding. Geneva: World Health Organization.
WHO/UNICEF. (2015). Progress on sanitation and drinking water – 2015 update and MDG assessment. Geneva: World Health Organization.
World Food Summit. (1996). Rome declaration on world food security. Rome: Food and Agriculture Organization of the United Nations.
Wright, J., Gundry, S., & Conroy, R. (2004). Household drinking water in developing countries: a systematic review of microbiological contamination between source and point-of-use. Tropical Med Int Health, 9(1), 106–117.
Acknowledgements
This research was funded, in part, by The University of Utah's Primary Children's Fellowship and by NASA under award number NNX16AI02G.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grace, K., Frederick, L., Brown, M.E. et al. Investigating important interactions between water and food security for child health in Burkina Faso. Popul Environ 39, 26–46 (2017). https://doi.org/10.1007/s11111-017-0270-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11111-017-0270-6