Abstract
Aims
Termites function as “soil engineers” in tropical agroforestry ecosystems. However, of their role in phosphorus (P) cycling little is known. We aimed to investigate the impact of termite activity on soil aggregate stability and P fractions at the aggregate level in a tropical rubber plantation.
Methods
Fungus-growing termite mounds (active and abandoned) involving both above- and belowground locations were studied in a 24-year-old rubber stand. The mass percentage and stability of aggregates, P fractions contents and other major chemical properties of soil aggregates were measured. Aggregate-associated P preservation capacity was also calculated.
Results
More aggregates < 1 mm in size were concentrated in active aboveground mounds than active belowground chambers, thus resulting in weaker stability and erosion resistance, whereas the opposite trend occurred in abandoned mounds. The concentrations of labile P (in > 2 mm aggregate size), moderately labile P (0.25–1 mm), and non-labile P (0.053–1 mm) in active aboveground mounds were significantly higher than other types. The changes in specific P forms enriched TPi in aggregates > 2 mm and TPo in 0.053–1 mm size of active aboveground mounds relative to others, implying the importance of Po storage in microaggregates induced by termite activity involved in long-term P transformation. Furthermore, middle-sized (0.25–2 mm) aggregates stored more P and represented the highest P storing capacity, especially for active belowground chambers.
Conclusions
These results suggest that in the presence of termite activity, P cycling is greatly enhanced in aboveground mounds despite the poor aggregate stability, whereas P forms are stable after mound abandonment, except for a higher H2O-Pi concentration aboveground. Our study provides an important reason why mound soils can be considered as fertility amendments for agroforestry practices in P-deficient tropical soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Termites are social insects of the infraorder Isoptera and are widely distributed across tropical and subtropical ecosystems (Muvengwi and Witkowski 2020). These insects are the most important soil bioturbators and have been called “soil engineers” (Jouquet et al. 2016b). To fully understand the impact of termites on soil properties, species are usually classified into two ecological groups (soil-feeding termites vs. fungus-growing termites) according to similarities in their food sources and nesting strategies as well as their effects on the surrounding environment (Jouquet et al. 2005, 2011, 2023). Soil-feeding termites consume organic matter (e.g., humus, minerals, and litter) and build their nests with feces mixed with coarse inorganic soil particles (Tuma et al. 2022). In contrast, fungus-growing termites develop an obligate ectosymbiosis relationship with the soil-borne fungus Termitomyces sp., and enrich their nests construction with saliva rather than feces (Jouquet et al. 2011). Termites can affect the physical, chemical, and biological properties of soils by creating a new microenvironment through nesting and foraging behavior, thus altering the soil enzymatic activity and microbial community structure (Ackerman et al. 2007). This in turn affects the mineralization and decomposition of soil organic matter, and thus the concentration and composition of soil nutrients (Bera et al. 2020; Chen et al. 2021; Rückamp et al. 2010). Generally, termite mounds have a higher nutrient concentration (Apori et al. 2020; Lejoly et al. 2019), cation concentration, pH value, and mineral availability in bulk soils (Bera et al. 2020; de Lima et al. 2018) than the surrounding soils; but they show a lower microbial diversity (Aguero et al. 2021). However, these effects vary with termite taxa, ecological groups, and habitat change (Holt, and Lepage 2000; Jouquet et al. 2022). For example, soil-feeding termites affect soil properties primarily through their feces regrowth in clay-organic complexes (Tuma et al. 2022), while fungus-growing termites often rely on symbiosis with the fungi to complete the degradation of the litter and thus alter soil environment (Menichetti et al. 2014). Meanwhile, their feeding strategies also have different functional consequences in terms of soil aggregate stability (Jouquet et al. 2023).
Soil aggregates, as the basic units of the soil structure, are generally divided into macroaggregates (> 0.25 mm) and microaggregates (< 0.25 mm). The former is mainly formed by fine roots and hyphae, and the latter are organized by microbial polysaccharides and smaller soil particles (e.g., sand and clay). They can help to maintain soil stability and increase nutrient retention and availability to plants due to improved soil structure through mutual processes of microaggregate formation and macroaggregate turnover (Lejoly et al. 2019). Aggregate stability is influenced by many factors including anthropogenic land use change (Davies et al. 2020; Liu et al. 2019a, b), changes in soil biological activity (Haydu-Houdeshell et al. 2018), development of roots and hyphae, and organic matter content (Jouquet et al. 2016a). The outer walls of termite mounds have lower soil macro- and microaggregate stability than the surrounding soil (Harit et al. 2017; Jouquet et al. 2016a, b). It has reported that soil-feeding termites substantially increase the stability of soil aggregates > 2 mm in size, while fungus-growing termites reduce it (Contour-Ansel et al. 2000; Garnier-Sillam and Harry 1995). Moreover, fungus-growing termites decrease the stability of all soil aggregates in the Ferralsol but they only decrease that of > 0.25 mm macroaggregates in the Vertisol (Jouquet et al. 2016a). Deep cracks in the nests of fungus-growing termites are caused by soil compaction during the dry season, which allows the rapid infiltration of large amounts of rainwater into the nests during the rainy season, thus destroying the unstable aggregates and their nutrients (Bera et al. 2020; Chen et al. 2019). As a result of distinct dry and wet seasons, there is a greater likelihood of variation between the above- and belowground parts of termite nests in tropical forest ecosystems (Davies et al. 2020; Muvengwi et al. 2017; Traore et al. 2019). However, the questions of how termite nesting affects soil aggregate stability under such climatic conditions and the underlying mechanisms behind this process are rarely addressed.
Phosphorus (P) occurs in soils in organic and inorganic forms, ranging from ionic forms in solution to highly stable compounds combined with organic matter and clay minerals (Bera et al. 2020). Nevertheless, in most cases, P is found mainly in inorganic form (60–85%) (Liu et al. 2018). However, P distribution as well as its recycling and availability in the soil is uneven, and its concentration can vary due to the influence of soil bioturbation (Rückamp et al. 2012), the stability of soil aggregates or their size (Cui et al. 2019), the distribution of nutrients within the aggregates (Li et al. 2016), the type of land use (Liu et al. 2018), and degree of soil weathering (Aleixo et al. 2020). The mineralization and decomposition of litter and organic matter are the main sources of P. P concentrations are usually low in highly weathered tropical acidic soils, but termite nests form bioaggregates that serve as carriers for P protection and stabilization. However, the effects of termites on total P (TP) and available P in whole soil vary greatly depending on their feeding groups, nest sites, and initial soil properties (Apori et al. 2020; Chisanga et al. 2020; Jouquet et al. 2015; Seymour et al. 2014). For instance, available P concentration in the bulk soil of soil-feeding termite mounds was substantially higher than that in the surrounding soil, whereas the fungus-growing termites tended to increase the adsorption of P in the surrounding soil (López‐Hernández et al. 2006; Mamo and Wortmann 2009; Rückamp et al. 2010).
Rubber (Hevea brasiliensis) is a typical tropical cash crop, and most of the large-scale rubber plantations in Xishuangbanna have resulted in the destruction of tropical forest. Deforestation in turn induces serious degradation of soil quality and causes soil P depletion (Liu et al. 2018, 2021b), organic matter reduction (Wang et al. 2020), and decreased diversity of soil microbes and arthropods (Hidayat et al. 2018; Liu et al. 2019a, b; 2021a). Termites (mainly fungus-growing termites), important decomposers in the tropics, are very abundant and extensively distributed in rubber plantations (Arifin et al. 2016; Seetapong et al. 2021). These termites can contribute 58–64% of global wood decay (Griffiths et al. 2019), which will increase with tropicalization (Zanne et al. 2022) and return the organic matter to the soil in the form of feces and saliva, thus altering the availability of P by affecting the formation and turnover of aggregates (Jouquet et al. 2018; Rückamp et al. 2010). However, few studies have focused on the changes in the composition and availability of aggregate-associated P within termite mounds.
In this study, we investigated the influence of termite nesting on the soil aggregate stability and aggregate-associated P cycle in different types of termite mounds in rubber monoculture. To achieve this, we measured the mass distribution and stability of aggregates and the P fraction concentrations in soil aggregates of termite mounds as well as their correlations with the main chemical properties. We hypothesized that: 1) abandoned termite mounds, particularly aboveground external walls, will improve build-up of soil structure due to prolonged leaching and bonding relative to active mounds, and 2) active termite mounds, especially underground chambers, will increase P availability and soil aggregate-associated P cycle in the presence of frequent nesting and foraging behavior.
Materials and methods
Site description
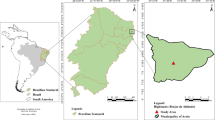
The study was conducted in Mengla Township (101°05′ E, 21°09′ N), which lies on the northern margin of Southeast Asia and the East Lancang River, and is a hotspot of biodiversity in the world. The climate in this region is influenced by the southwest tropical monsoon. The average altitude is 890 m and the mean annual temperature, sunshine duration, and precipitation are 21.5 °C, 1850 h, and 1480 mm, respectively. The region has a seasonal climate with three distinct seasons (dry-hot, wet, and fog-cool) alternating during the year. The dry-hot season (March to May) has a higher temperature and less rainfall. The wet season (June to October) is hot and humid, and 85% of the annual precipitation is concentrated during this period. The fog-cool season (November to February) receives less precipitation but there is thick fog in the morning and evening and the air humidity is high. The region was mainly covered by rubber monoculture plantation established in 1994 and arranged in a conventional spacing of 2.5 m × 8 m. All stands were managed under local agricultural practices, such as green manure, compost and biological pest management, without the use of pesticides or other chemicals (Liu et al. 2021b). The soil type in this region is laterite (Oxisol) according to the USDA soil taxonomy. The initial soil (0–30 cm) properties were as follows: total carbon (TC) of 10.92 g kg−1, total nitrogen (TN) of 1.27 g kg−1, TP of 0.26 g kg−1, available P of 0.57 mg kg−1, bulk density of 1.35 kg m−3, soil pH of 5.07, and exchangeable Al of 5.50 cmol kg−1 (Liu et al. 2018).
Experimental design and soil sampling
Termite mounds constructed by fungus-growing termite (i.e., Macrotermes annandalei) in a monoculture rubber plantation (24-year-old) were selected. Two types (active and abandoned mounds) were sampled in late April 2018 with four replicates for each type. This satisfies statistical duplication of sampling because the density of mounds is 6.0 ha–1 (Jouquet et al. 2016a). The ratio of active to abandoned mounds was 1.4 ha–1. The mean active mound height was 64 cm and diameter 110 cm; mean height of abandoned mound was 30 cm and diameter 69 cm. Soil samples were collected from aboveground external walls (ca. 25-cm from the base) and belowground chambers (ca. 50-cm depth to the center of the nest), using a 5-cm auger along with four corners (east, south, west, north) on the top of the mounds. This sampling location allows us to better compare the changes in P fractions of active and abandoned termite mounds (Bera et al. 2020; Jouquet et al. 2016a). Samples from the same nest and location were combined into a mixed sample, packed in a polyethylene bag, and returned immediately to the laboratory where visible stones, animals, roots, and plant material were removed manually. Totally, 16 soil samples were collected from different mounds. The sample was separated into two subsamples: one was used for aggregate analysis and the other was ground for general soil analyses.
Aggregate separation scheme
The aggregate size distribution was measured by the wet-sieving method (Jouquet et al. 2016a; Zhou et al. 2022), passing the air-dried soil through a 5-mm sieve. Air-dried soil samples (50.00 g) were placed on the top sieve of the aggregate analyzer (XY-100, Beijing Xiangyu Weiye Instrument Equipment Co., LTD., China) with mesh aperture sizes of 0.053, 0.25, 1, and 2 mm. The samples were submerged in deionized water for 10 min at room temperature by adjusting the water level of the equipment so that it just flowed over the air-dried soil. The instrument was then set to vibrate for 10 min with an amplitude of 4 cm and a frequency of 30 cycles/min. The sieve was carefully removed from the bucket and placed on the bench. The retained material was placed in the corresponding aluminum box and oven-dried at 60 ℃ until a constant weight was achieved. The mass of each oven-dried aggregate was divided by the mass of the total soil to obtain the percent mass of the aggregate fraction. The equation Eq. (1) used is as follows:
where \({M}_{i}\) is the mass percentage of the i aggregate size fraction (%), \({m}_{i}\) is the mass of the i aggregate size fraction (g), and Mt is the mass of the total soil (g).
Aggregate stability assessment
The parameters commonly used to describe aggregate stability include mean weight diameter (MWD), geometric mean diameter (GMD), fractal dimension (D), water-stable aggregate percentage content larger than 0.25 mm (R0.25), and erodibility (K). The MWD and GMD of aggregates were calculated according to Eq. (2) and Eq. (3):
where \(n\) is the number of separated aggregate classes, Xi is the mean of the i particular size of aggregate (mm), and Mi is the mass percentage of the i aggregate size fraction (%).
The fractal dimension (D) of aggregates was calculated according to the fractal model Eq. (4) (Zhou et al. 2022):
Taking the logarithm on both sides of Eq. (4) to obtain Eq. (5):
where \(\overline{{x }_{i}}\) is the average diameter of two sieving diameters \({x}_{i}\) and \({x}_{i+1}\) (mm), \(M(r<\overline{{x }_{i}})\) is the mass of aggregates with a diameter shorter than average (g), \(M\) is the mass of all aggregates (g), and \({x}_{max}\) is the largest average diameter of an aggregate (mm). Finally, the D value is calculated using Eq. (5).
The erodibility (K) was calculated using the GMD model Eq. (6) (Zhou et al. 2022).
P fractionation, relative contribution and preservation capacity
The sequential extraction procedure proposed by Hedley et al. (1982) and modified by Liu et al. (2018) and Rückamp et al. (2012) was used to differentiate P fractions in bulk soil and soil aggregate. Briefly, 3.0 g of soil aggregate sample was placed into a 50-mL centrifuge tube, and then sequentially extracted with deionized water (H2O-Pi), 0.5 M NaHCO3 (NaHCO3-Pi and NaHCO3-Po), 0.1 M NaOH (0.1 M NaOH-Pi and 0.1 M NaOH-Po), 1 M HCl (HCl-Pi), 0.5 M NaOH (0.5 M NaOH-Pi and 0.5 M NaOH-Po), and a mixture of concentrated H2SO4 (residual-Po). All extractions were carried out in a reciprocal shaker at 200 rpm for 16 h. The soil extracts were centrifuged at 10,000 rpm for 10 min and filtered through a 0.45 µm membrane (Whatman No. 42) to collect clear supernatants for later analysis. The extracted inorganic P (Pi) was determined with the molybdate colorimetric method using an inductively coupled plasma-mass spectrophotometer (Agilent 7700 × ICP‒MS, Agilent Technologies Inc., USA). The TP was measured by the same method after digestion with H2SO4 and K2S2O8 in an autoclave at 121 °C, and organic P (Po) was obtained by subtracting Pi from the TP. The residue in the centrifuge tube was moved to a crucible and burned for 1 h at 550 °C in a muffle furnace. The burned soil sample was transferred to a tube containing 1 M H2SO4, centrifuged after shaking for 24 h, and then used to determine the P concentration in the supernatant. In this procedure, Po was not measured in the water or HCl-extractable fraction because their concentrations were below the limit of detection. We classified P fractions into labile P (H2O-Pi + NaHCO3-Pi + NaHCO3-Po), moderately labile P (0.1 M NaOH-Pi + 0.1 M NaOH-Po + HCl-Pi), and non-labile P (0.5 M NaOH-Pi + 0.5 M NaOH-Po + residual-Po) according to ecological significance (Liu et al. 2018).
The relative contributions of P fractions within aggregates to their corresponding concentrations in the soil were calculated by the Eq. (7):
where \({C}_{P fraction}\) is the relative contribution of the P fraction within aggregates (%), \({M}_{a}\) is the P fraction content in the aggregates of a particular size fraction (mg kg–1), \({M}_{i}\) is the mass percentage of a particular aggregate size fraction (%), and \({M}_{p}\) presents the P fraction in the bulk soil (mg kg–1).
The P preservation capacity (PPC) of soil aggregates was calculated by Eq. (8):
where \({M}_{a}\) is the P fraction concentration in the aggregates of a particular size fraction (mg kg–1) and \({m}_{i}\) is the mass of the aggregates of a particular size fraction (g).
General soil chemical analyses
For soil aggregate and bulk soil, pH value was measured in a 1:2.5 soil‒water suspension with a pH meter (FE28-Standard, Mettler Toledo, Germany). TC and TN were analyzed using an elemental analyzer (MAX CNS Elemental Analyzer, Elementar, Germany). The detailed results are shown in Table S1.
Statistical analysis
The single and interactive effects of termite mounds of different types (active vs. abandoned) and locations (aboveground vs. belowground) on the mass percentage and stability of aggregates and various P fractions were tested using two-way ANOVA. We then used one-way ANOVA to analyze differences in the mass percentage and stability of aggregates, P fractions and their related parameters, and chemical properties among mounds for each aggregate size, followed by multiple comparison with Tukey’s post hoc test at the P < 0.05 level. Moreover, relationships between the P fractions and other soil properties were examined using Pearson’s correlation analysis. Data processing was performed using the SPSS 21.0 software package.
Results
Aggregate mass percentage and stability
The type of termite mound affected the mass percentage of > 2 mm and 0.053–0.25 mm aggregates, and the location mainly had a significant effect on microaggregates (< 0.25 mm) (P < 0.05; Table S2). However, their interaction highly affected the mass percentage of each aggregate size (P < 0.001). Macroaggregates (> 0.25 mm) were plentiful in both active and abandoned mounds, and the 0.25–1 mm size was the most abundant (Fig. 1). In active mounds, aggregates > 1 mm were more abundant belowground than aboveground, but < 1 mm aggregates were more abundant aboveground, which showed an opposite trend in abandoned mounds. At the same location, the mass percentages of > 1 mm and < 1 mm aggregates in active mounds were contrary to abandoned ones, respectively.
Aggregate stability indices were affected by mound type, location, and their interactions (P < 0.05), except for the effect of type on GMD (P = 0.30) and D (P = 0.91; Table S2). In active mounds, MWD, GMD, and R0.25 values of the belowground aggregates were higher than those of aboveground (Fig. 2a, b and c), whereas K and D values presented an opposite trend (Fig. 2d and e), which was contrary to abandoned mounds. For aboveground, MWD, GMD, and R0.25 values of active mounds were lower than those of abandoned ones, whereas the opposite pattern was observed for K and D values, which was in contrast to belowground chambers.
Aggregate stability in relation to location from active and abandoned termite mounds. Mean weight diameter (MWD), geometric mean diameter (GMD), > 0.25 mm water-stable aggregate content (R0.25), erodibility (K), and fractal dimension (D) are used to evaluate aggregate stability of mound soils. Histograms with the different letters denote significant differences among mound types (P < 0.05, Tukey's test). Bars are standard errors, n = 4
P fractions in aggregates and bulk soils
Labile P (7.3–10.4% of TP) in > 2 mm aggregates was affected by mound type and in 0.25–1 mm aggregates by location (P < 0.05; Table S3). Its concentration in aggregates > 2 mm was greater in active than abandoned mounds, especially aboveground (Fig. 3a). Specifically, H2O-Pi, NaHCO3-Pi, and NaHCO3-Po in microaggregates were affected by the interaction between mound type and location (P < 0.051; Table S3). H2O-Pi concentration in aggregates and bulk soils of active mounds was generally higher belowground than aboveground, whereas the opposite pattern was observed in abandoned mounds (Table 1). In active mounds, NaHCO3-Pi concentration in aggregates (except those > 2 mm) was higher belowground than aboveground, but NaHCO3-Po exhibited an opposite trend. There was no location effect on NaHCO3-extracable P of abandoned mounds. Compared with active belowground chambers, NaHCO3-Pi in > 1 mm aggregates was lower, while NaHCO3-Po in microaggregates was greater in abandoned ones.
Soil P fractions within different aggregate sizes and bulk soils in relation to location from active and abandoned termite mounds. TPi, total inorganic P; TPo, total organic P. Labile P is the sum of H2O-Pi + NaHCO3-Pi + NaHCO3-Po, moderately labile P is the sum of 0.1 M NaOH-Pi + 0.1 M NaOH-Po + HCl-Pi, non-labile P is the sum of 0.5 M NaOH-Pi + 0.5 M NaOH-Po + residual-Po. Histograms with the different letters denote significant differences among mound types for each aggregate size (P < 0.05, Tukey's test). Bars are standard errors, n = 4
Moderately labile P (3.9–8.9% of TP) in 0.25–1 mm aggregates was affected by mound location and in bulk soil by type (P < 0.05; Table S3), and its concentration in active mounds was higher aboveground than belowground (Fig. 3b). However, 0.1 M NaOH-Pi, 0.1 M NaOH-Po, and HCl-Pi in aggregates responded differently to mound type and location (Table S3). In active mounds, 0.1 M NaOH-Pi concentration in > 2 mm and 0.053–0.25 mm aggregates were lower belowground than aboveground, whereas 0.1 M NaOH-Po presented an opposite trend (Table 1). Moreover, HCl-Pi concentrations in aggregates > 2 mm and bulk soils were higher in active aboveground mounds than belowground, but the contrary pattern was seen in aggregates < 0.053 mm. Location had no effect on moderately labile P of abandoned mounds. Compared with same location of active mounds, 0.1 M NaOH-Pi in aggregates was greater in abandoned belowground chambers, but HCl-Pi in > 1 mm aggregates was lower in abandoned aboveground.
Non-labile P (83.8–88.1% of TP) and residual-Po in 0.053–1 mm aggregates were affected by mound type, location, and their interactions, except for the type effect on residual-Po in 1–0.25 mm aggregates and in bulk soil by type (P < 0.05; Table S3). Their concentrations in 0.053–1 mm aggregates and bulk soils were highest in active aboveground mounds than others (Fig. 3c and Table 1). In active mounds, 0.5 M NaOH-Pi concentration in 0.053–0.25 mm aggregates was greater aboveground than belowground, which was also true for 0.5 M NaOH-Po in abandoned mounds (Table 1).
TPi (14.1–19.8% of TP) was affected by mound location in all aggregates (P < 0.01), except for 0.053–1 mm (P = 0.25; Table S3). In active mounds, its concentration in aggregates > 2 mm was higher aboveground than belowground, whereas the opposite pattern was found in 1–2 mm and < 0.053 mm aggregates (Fig. 3d). TPo (80.2–85.9% of TP) and TP in 0.053 mm–1 aggregates were affected by mound type and location and in bulk soil by type (P < 0.01; Table S3), and their concentrations were highest in active aboveground mounds than others (Fig. 3e).
Aggregate-associated P contribution and preservation
The relative contributions of various P fractions showed the same changes as the mass percentage in each aggregate size, of which the contribution rate of 0.25–1 mm aggregate to P was the highest (19.9–32.1% from labile to stable P) (Fig. S1). The P preservation capacity of aggregates varied greatly in different termite mounds (Fig. 4). In active mounds, P storage capacity in macroaggregates was higher belowground than aboveground, whereas the opposite pattern was found in microaggregates. In abandoned mounds, P storage capacity in aggregates > 2 mm was lower belowground than aboveground, but microaggregates showed an opposite trend. For aboveground, P storing capacity in macroaggregates was lower in active than abandoned mounds, whereas microaggregates showed an opposite trend, which was contrary to belowground chambers. Moreover, 0.25–2 mm aggregates stored more P and had the highest P preservation capacity.
Soil P preservation capacity within different aggregate sizes in relation to location from active and abandoned termite mounds. IPPC, inorganic P preservation capacity; OPPC, organic P preservation capacity; TPPC, total P preservation capacity. Histograms with the different letters denote significant differences among mound types for each aggregate size (P < 0.05, Tukey's test). Bars are standard errors, n = 4
Correlation between P fractions and other chemical properties
The specific P fractions were related to the other chemical properties in different termite mounds, especially underground, as indicated by significant linear correlations for 34 (P < 0.001), 32 (P < 0.01) and 62 (P < 0.05) of the pairs studied (Fig. 5). In active aboveground mounds, labile P was negatively correlated with pH, TC, TN, and the ratios of C:P and N:P (Table S4). Labile P, moderately labile P, and TPi of active belowground mounds were negatively correlated with chemical properties, except for moderately labile P with TN (r = − 0.29, P = 0.17). In contrast, there was only a significant correlation between P fractions and C:N ratio of abandoned aboveground mounds, except for moderately labile P (r = − 0.10, P = 0.66). In abandoned belowground mounds, labile P and TPi were positively correlated with pH but negatively correlated with nutrient variables, which was contrary to non-labile P and TPo. More specifically, P availability of both active and abandoned mounds was strongly related to pH, while it also had a negative correlation with C and N belowground chambers (Fig. 5).
Pearson's correlations between specific P fractions in all aggregates and bulk soils and other soil variables for different termite mounds. TC, total carbon; TN, total nitrogen; C:N, the ratio of C:N; TPi, total inorganic P; TPo, total organic P. *: 0.01 < P < 0.05; **: 0.001 < P < 0.01; ***: P < 0.001
Discussion
Effects of termite mounds on mass percentage and stability of aggregates
The fate of aggregates rearranged by termites varies at different stages of mound construction (Fall et al. 2001). We found that active belowground chambers of fungus-growing termites contained more > 1 mm aggregates than aboveground, but the opposite occurred in abandoned mounds (Fig. 1). These changes correspond to the two developments of mound formation: i) termites pile up many basic aggregates (average size is 0.61 mm) from the argic horizon before delivering them to the surface as compound aggregates (average size is 1.00 mm), which is the preferential size for termites, and ii) once mounds are abandoned, colluvial aggregates (average size is 1.12 mm) are mainly produced by splash and runoff during rain at the external wall (Ayuke et al. 2011; Jungerius et al. 1999). The sequence is also the main reason for the difference in mass percentage between active and abandoned at the same location. The alternating drying and wetting cycles can also promote the formation of new water-stable structures (Bera et al. 2020; Chen et al. 2019), thereby leading to greater > 1 mm aggregates in abandoned aboveground mounds. In addition, 0.25–1 mm aggregates were found to be the most abundant size, similar to 50% of aggregates > 0.2 mm encountered in mound walls (Contour-Ansel et al. 2000), but in contrast with soil-feeding termite mounds that are constituted by > 2 mm aggregates (Sarcinelli et al. 2009), indicating that the basic aggregates are preserved in mound matrix throughout (Jungerius et al. 1999). Soil organic C, as an important binding agent for soil aggregation, was positively correlated with the mass distribution of > 1 mm aggregates (r > 0.61, P < 0.05) and negatively correlated with those smaller than 1 mm (r < − 0.78, P < 0.001). Therefore, 1 mm is a key aggregate size criterion for evaluating the soil structure of fungus-growing termite mounds.
Weaker soil aggregate stability and hence less erosion resistance were observed in active aboveground mounds than belowground, as revealed by decreased MWD, GMD, and R0.25 values and increased K and D values (Fig. 2). Similarly, most studies have reported that the low aggregate stability of termite-worked mounds compared to their surrounding soils (Harit et al. 2017; Jouquet et al. 2004, 2016a, b; Tilahun et al. 2021). While a few showed more stable aggregates from aboveground mounds relative to the lower chambers and adjacent soils (Bera et al. 2020; Contour-Ansel et al. 2000; de Oliveira et al. 2012) or no further influence (Fall et al. 2001; Paul et al. 2015). Van Thuyne and Verrecchia (2021) reviewed that no definitive results can be proposed regarding the effects of termite activity on mound stability. The discrepancies highly depend on termite impacts on mound soil properties (e.g., nutrients, pH, oxides, and cations), among which high organic C (Table S1) or clay content are important agents contributing to nest structural stability (Jouquet et al. 2007, 2018; Tuma et al. 2022), rather than their feeding and mound-building strategies (Jouquet et al. 2022). Moreover, the reduced aggregate of aboveground mounds may be related to the absence of plant root fragments and mycorrhizal hyphae that are major binding agents for larger soil aggregates (personal observation as Fig. 6). As our first hypothesis, abandoned aboveground mounds exhibited stronger stability and less erodibility than belowground chambers. This is because unstable aggregates have been intermittently eroded and leached downwards by rainfall over time (Chen et al. 2018; Menichetti et al. 2014), as well as increased mound densification due to gravity and repeated wetting and drying cycles (i.e., soil stress history) (Van Thuyne and Verrecchia 2021). It is also reflected by the morphological characteristics of abandoned mounds with a hard outer wall but a cavity inside. The same reasoning can explain the location variations in aggregate stability and erosion resistance of the chronological development of termite mounds. These results suggest a specific role of termite activity and environmental change in the formation and disintegration of soil particles at different mound stages.
Schematic diagram of the P nutrient recycle processes mediated by termites in active and abandoned termite mounds. H2O-Pi and NaHCO3-Po indicate deionized water- and NaHCO3-extractable organic P form. Labile P is the sum of H2O-Pi + NaHCO3-Pi + NaHCO3-Po. In active mounds (left), P nutrient inputs due to termite grazing belowground, transfers to the aboveground by termite nest building, and finally accumulates in the mound walls, which may improve P cycling. In abandoned mounds (right), P nutrient losses from mound walls aboveground due to erosion and leaching as well as increased microbial mineralization
Effects of termite mounds on P fractions
Labile P in > 2 mm aggregates was higher in active mounds than abandoned, especially aboveground, attributed to NaHCO3-Po accumulation (Fig. 3a), which is similar to previous studies reporting a gross enrichment of labile P fractions in termite-colonized mounds compared to the below chambers and adjacent soils of various tropical ecosystems (Erens et al. 2015; López‐Hernández et al. 1989, 2001; Rückamp et al. 2010, 2012). This result indicates that large aggregates tend to store active nutrients from plant residue translocation by termites. Specifically, H2O-Pi and NaHCO3-Pi in most aggregates and bulk soils were greater in active belowground chambers than aboveground, with an inverse trend for NaHCO3-Po (Table 1). This is attributed to rapid NaHCO3-Po microbial mineralization induced by termite activity (Menichetti et al. 2014) and its desorption with decreasing Fe/Al oxides under a weakly acidic (pH = 6.0) nest environment (López‐Hernández et al. 2006; Mamo and Wortmann 2009), which is further confirmed by an obvious negative correlation between labile P forms and chemical properties (Fig. 5). Unexpectedly, more H2O-Pi was found in abandoned aboveground mounds, although these mounds normally experienced runoff and erosion, leading to low pH values. This is the cause of P deposition by scattered plant material growing on outer wall after mound abandonment, where plants are hardly enriched in P (Seymour et al. 2014). This finding contradicts that the pattern of resin-Pi is a pH-dependent behavior (Erens et al. 2015; Van Thuyne and Verrecchia 2021). Moreover, labile Pi from > 1 mm aggregates of active belowground chambers was transformed into the Po form and temporarily stored in microaggregates after abandonment.
Moderately labile P, dominated by 0.1 M NaOH-Pi form, was concentrated in 0.25–1 mm aggregates and bulk soils of active aboveground mounds than belowground (Fig. 2b), owing to a larger specific surface area of this size. In active mounds, however, more 0.1 M NaOH-Pi was distributed in > 2 mm and 0.053–0.25 mm aggregates aboveground than belowground, but the opposite trend was observed for 0.1 M NaOH-Po (Table 1), which is due to the higher P adsorption capacity (i.e., more exposed Fe/Al sorption sites) (Li et al. 2016). This fraction also represents partially hydrolyzed monoester-P having a vigorous capacity for complexing with soil particles which reduces microbial accessibility (Liu et al. 2018). Hence, incorporation of organic residues by termite nesting facilitates the formation of monoester P, resulting in 0.1 M NaOH-Pi accumulation at outer wall (López‐Hernández et al. 2001). In most cases, 0.1 M NaOH-Pi was enriched in termite aboveground mounds relative to the surrounding soils, but was depleted in nests of some Nasutitermes and Cornitermes with a high proportion of monoester-P (Rückamp et al. 2010). When mounds were abandoned, 0.1 M NaOH-Pi accumulated belowground relative to active mounds. Meanwhile, HCl-Pi was greater in > 2 mm aggregates and bulk soils of active aboveground mounds than belowground, whereas < 0.053 mm size showed a contrary pattern, primarily because of Ca2+ enrichment (Seymour et al. 2014) or depletion (Ackerman et al. 2007). This is also true for aboveground when comparing active and abandoned mounds. In the lower locations of active and abandoned mounds, the high pH could promote Ca-bound P (HCl-Pi) precipitation with increasing depth (Erens et al. 2015), despite no variations in HCl-Pi across the mound profile (Rückamp et al. 2010, 2012).
Non-labile P was accumulated in 0.053–1 mm aggregates and bulk soils of active aboveground mounds relative to others due to a considerable contribution of residual-P (Fig. 2c), which is similar to the finding that a great proportion of residual-P accumulated in most nest walls (López‐Hernández et al. 2001; Rückamp et al. 2010). Residual-P is considered to comprise inositol phosphates and is highly stable against microbial and enzymatic attacks (Liu et al. 2018), therefore, its accumulation on the mound surface can compensate for labile Pi leaching downwards and Po mineralization loss. This is reinforced by the negative relationship between residual-P and C:N:P ratios (Fig. 5), indicating the potential function of SOM in maintaining P reserves. Thus, even if there is a greater P concentration in active aboveground mounds, it does not necessarily mean that P is more bioavailable (Edosomwan et al. 2012). There were no effects of type and location on 0.5 M NaOH-extractable P and this seems not to participate in P cycling of termite mounds.
Overall, the above changes in specific P forms lead to enrichment of TPi in aggregates > 2 mm, as well as TPo and TP in 0.053–1 mm aggregates and bulk soils of active aboveground mounds relative to others (Fig. 2d and e), rejecting our second hypothesis. This highlights the importance of Po storage in microaggregates induced by termite foraging activity involved in long-term P transformation and cycling when Pi is deficient.
Implications of termite management for agroforestry
Termites historically have a dual role of “soil engineers” vs. “pests” in different agroforestry ecosystems around the world (Paul et al. 2015). However, their positive impacts on several key ecological functions from the soil profile to landscape scale, such as C sequestration, nutrient cycling, aggregate modification, and hydraulic regulation, are often overshadowed by their pest status threatening agroforestry in the tropics, especially for fungus-growing termite (Jouquet et al. 2011; Seetapong et al. 2021; Tuma et al. 2022). Thus, two main agroforestry practices are proposed for improving soil quality and crop yield while reducing the negative effects of termite foraging activity: i) less intensive management providing litter, predators, and pathogens, and ii) considering the heterogeneity and complexity of mound landscapes (Jouquet et al. 2018). Intensive rubber monocultures can dramatically decrease termite populations, even far worse in mature plantations, resulting in their negative services and lower stand resistance (Arifin et al. 2016; Hidayat et al. 2018). The establishment of rubber-cover crop agroforestry can provide a palatable and abundant food resource (e.g., legume mulch and litter) for termites, while reduce the diversity and abundance of arthropods and pathogens (Liu et al. 2021a). However, this system will deplete more P to assure symbiotic N2 fixation for productivity (Liu et al. 2018; 2021b). Termite mounds are “hot-spots” of P nutrient, as we observed, which is a feasible source of available P for cash crops if smallholders can effectively use abandoned mound soils and meanwhile conserve active mounds without destruction of termite habitats (Apori et al. 2020; Van Thuyne and Verrecchia 2021). The spatial arrangement of complex agroforestry around termite mounds according to the soil fertility gradients is important for their nutrient management (Tilahun et al. 2021). Exploring the microscale variability of P concentration is therefore useful in determining the potential of termite mounds as fertility amendments in highly weathered tropical soils.
Conclusion
The present study shows that the presence or absence of fungus-growing termite nesting exhibited contrasting effects on soil aggregate stability and distribution of aggregate-associated P fractions in a mature rubber plantation. Active belowground chambers exhibited stronger stability and less erodibility due to more aggregates > 1 mm size than aboveground mounds. In active aboveground mounds, the concentrations of labile P (in > 2 mm aggregate size), moderately labile P (0.25–1 mm), and non-labile P (0.053–1 mm) were highest than other types. However, H2O-Pi and NaHCO3-Pi in most aggregates were enriched in active belowground chambers relative to aboveground due to rapid NaHCO3-Po transformation. The changes in various P fractions induced accumulation of TPi in aggregates > 2 mm and TPo in 0.053–1 mm size of active aboveground mounds relative to others. This indicates the importance of Po storage in microaggregates induced by termite activity involved in long-term P turnover. More P was sequestered in 0.25–2 mm aggregates, particularly in active belowground chambers. Our results suggest that although termite activity reduced aggregate stability aboveground, the effect of promoting P availability is beneficial. Further study should be conducted to explore the potential regulatory mechanisms of microbes involved in P cycling across the chronological development of termite mounds.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding authors on reasonable request.
References
Ackerman IL, Teixeira WG, Riha SJ, Lehmann J, Fernandes EC (2007) The impact of mound-building termites on surface soil properties in a secondary forest of Central Amazonia. Appl Soil Ecol 37:267–276
Aguero CM, Eyer PA, Crippen TL, Vargo EL (2021) Reduced environmental microbial diversity on the cuticle and in the galleries of a subterranean termite compared to surrounding soil. Microb Ecol 81:1054–1063
Aleixo S, Gama-Rodrigues AC, Gama-Rodrigues EF, Campello EFC, Silva EC, Schripsema J (2020) Can soil phosphorus availability in tropical forest systems be increased by nitrogen-fixing leguminous trees? Sci Total Environ 712:136405
Apori SO, Murongo M, Hanyabui E, Atiah K, Byalebeka J (2020) Potential of termite mounds and its surrounding soils as soil amendments in smallholder farms in central Uganda. BMC Res Notes 13:397
Arifin Z, Dahlan Z, Irsan C, Hartono Y (2016) Impact of the presence of subterranean termites Macrotermes gilvus (Termitidae) to physico-chemical soil modification on the rubber plantation land. J Ecol Nat Environ 8:13–19
Ayuke FO, Brussaard L, Vanlauwe B, Six J, Lelei DK, Kibunja CN, Pulleman MM (2011) Soil fertility management: Impacts on soil macrofauna, soil aggregation and soil organic matter allocation. Appl Soil Ecol 48:53–62
Bera D, Bera S, Das Chatterjee N (2020) Termite mound soil properties in West Bengal India. Geoderma Reg 22:e00293
Chen C, Wu J, Zhu X, Jiang X, Liu W, Zeng H, Meng FR (2019) Hydrological characteristics and functions of termite mounds in areas with clear dry and rainy seasons. Agric Ecosyst Environ 277:25–35
Chen QL, Hu HW, Yan ZZ, Li CY, Nguyen BAT, Zheng Y, Zhu YG, He JZ (2021) Termite mounds reduce soil microbial diversity by filtering rare microbial taxa. Environ Microb 23:2659–2668
Chisanga K, Mbega ER, Ndakidemi PA (2020) Prospects of using termite mound soil organic amendment for enhancing soil nutrition in southern Africa. Plants 9:649
Cui H, Ou Y, Wang L, Wu L, Li Y (2019) Distribution and release of phosphorus fractions associated with soil aggregate structure in restored wetlands. Chemosphere 223:319–329
Contour-Ansel D, Garnier-Sillam E, Lachaux M, Croci V (2000) High performance liquid chromatography studies on the polysaccharides in the walls of the mounds of two species of termite in Senegal, Cubitermes oculatus and Macrotermes subhyalinus: their origin and contribution to structural stability. Biol Fertil Soils 31:508–516
Davies AB, Brodrick PG, Parr CL, Asner GP (2020) Resistance of mound-building termites to anthropogenic land-use change. Environ Res Lett 15:094038
de Lima SS, Pereira MG, Pereira RN, de Pontes RM, Rossi CQ (2018) Termite mounds effects on soil properties in the Atlantic forest biome. Rev Bras Ciênc Solo 42:e0160564
de Oliveira LBT, dos Santos AC, da Silva Neto SP, da Silva JEC, de Paiva JA (2012) Physical and chemical changes in soil under termite hills of Tocantins. Eng Agricul 20:118–130
Edosomwan NL, Edosomwan EU, Oke O (2012) Physical and chemical characteristics of termite mounds and associated soils in tropical forest-savanna transitional area of southern Nigeria. Indian J Agric Res 46:338–343
Erens H, Mujinya BB, Mees F, Baert G, Boeckx P, Malaisse F, Van Ranst E (2015) The origin and implications of variations in soil-related properties within Macrotermes falciger mounds. Geoderma 249:40–50
Fall S, Brauman A, Chotte J-L (2001) Comparative distribution of organic matter in particle and aggregate size fractions in the mounds of termites with different feeding habits in Senegal: Cubitermes niokoloensis and Macrotermes bellicosus. Appl Soil Ecol 17:131–140
Garnier-Sillam E, Harry M (1995) Distribution of humic compounds in mounds of some soil-feeding termite species of tropical rainforests: its influence on soil structure stability. Insectes Soc 42:167–185
Griffiths HM, Ashton LA, Evans TA, Parr CL, Eggleton P (2019) Termites can decompose more than half of deadwood in tropical rainforest. Curr Biol 29:R118–R119
Harit A, Moger H, Duprey JL, Gajalakshmi S, Abbasi SA, Subramanian S, Jouquet P (2017) Termites can have greater influence on soil properties through the construction of soil sheetings than the production of above-ground mounds. Insect Soc 64:247–253
Haydu-Houdeshell CA, Graham RC, Hendrix PF, Peterson AC (2018) Soil aggregate stability under chaparral species in southern California. Geoderma 310:201–208
Hedley MJ, Stewart JWB, Chauhan BS (1982) Changes in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci Soc Am J 46:970–976
Hidayat MR, Endris WM, Dwiyanti Y (2018) Effect of a rubber plantation on termite diversity in Melawi, West Kalimantan Indonesia. Agri Nat Res 52:439–444
Holt, J.A., Lepage, M., 2000. Termites and soil properties. Termites: evolution, sociality, symbioses, ecology, 389–407.
Jouquet P, Bottinelli N, Lata JC, Mora P, Caquineau S (2007) Role of the fungus-growing termite Pseudacanthotermes spiniger (Isoptera, Macrotermitinae) in the dynamic of clay and soil organic matter content An experimental analysis. Geoderma 139:127–133
Jouquet P, Chaudhary E, Kumar ARV (2018) Sustainable use of termite activity in agro-ecosystems with reference to earthworms A review. Agrono Sustain Dev 38:3
Jouquet P, Chintakunta S, Bottinelli N, Subramanian S, Caner L (2016a) The influence of fungus-growing termites on soil macro and micro-aggregates stability varies with soil type. Appl Soil Ecol 101:117–123
Jouquet P, Guilleux N, Caner L, Chintakunta S, Ameline M, Shanbhag RR (2016b) Influence of soil pedological properties on termite mound stability. Geoderma 262:45–51
Jouquet P, Guilleux N, Chintakunta S, Mendez M, Subramanian S, Shanbhag RR (2015) The influence of termites on soil sheeting properties varies depending on the materials on which they feed. Eur J Soil Biol 69:74–78
Jouquet P, Harit A, Bottinelli N, Eldridge DJ (2023) Termite bioturbation: Fungal versus non-fungal building strategies lead to different soil sheeting stability. Soil Biol Biochem 176:108868
Jouquet P, Harit A, Hervé V, Moger H, Carrijo T, Donoso DA, Eldridge D et al (2022) The impact of termites on soil sheeting properties is better explained by environmental factors than by their feeding and building strategies. Geoderma 412:115706
Jouquet P, Ranjard L, Lepage M, Lata JC (2005) Incidence of fungus-growing termites (Isoptera, Macrotermitinae) on the structure of soil microbial communities. Soil Biol Biochem 37:1852–1859
Jouquet P, Traore S, Choosai C, Hartmann C, Bignell D (2011) Influence of termites on ecosystem functioning. Ecosystem services provided by termites. Eur J Soil Biol 47:215–222
Jungerius PD, Van Den Ancker JAM, Mücher HJ (1999) The contribution of termites to the microgranular structure of soils on the Uasin Gishu Plateau, Kenya. CATENA 34:349–363
Lejoly J, Cornelis JT, Van Ranst E, Jansegers E, Tarpin C, Degré A, Colinet G, Malaisse F (2019) Effects of termite sheetings on soil properties under two contrasting soil management practices. Pedobiologia 76:150573
Li B, Ge T, Xiao H, Zhu Z, Li Y, Shibistova O, Liu S, Wu J, Inubushi K, Guggenberger G (2016) Phosphorus content as a function of soil aggregate size and paddy cultivation in highly weathered soils. Environ Sci Pollut Res 23:7494–7503
Liu C, Jin Y, Hu Y, Tang J, Xiong Q, Xu M, Bibi F, Beng KC (2019a) Drivers of soil bacterial community structure and diversity in tropical agroforestry systems. Agr Ecosys Environ 278:24–34
Liu C, Jin Y, Liu C, Tang J, Wang Q, Xu M (2018) Phosphorous fractions in soils of rubber-based agroforestry systems: Influence of season, management and stand age. Sci Total Environ 616:1576–1588
Liu C, Liang M, Tang J, Jin Y, Guo Z, Siddique KH (2021a) Challenges of the establishment of rubber-based agroforestry systems: Decreases in the diversity and abundance of ground arthropods. J Environ Manage 292:112747
Liu C, Wang QW, Jin Y, Tang J, Lin F, Olatunji OA (2021b) Perennial cover crop biomass contributes to regulating soil P availability more than rhizosphere P-mobilizing capacity in rubber-based agroforestry systems. Geoderma 401:115218
López-Hernández D (2001) Nutrient dynamics (C, N and P) in termite mounds of Nasutitermes ephratae from savannas of the Orinoco Llanos (Venezuela). Soil Biol Biochem 33:747–753
López-Hernández D, Brossard M, Fardeau JC, Lepage M (2006) Effect of different termite feeding groups on P sorption and P availability in African and South American savannas. Biol Fertil Soils 42:207–214
López-Hernández D, Fardeau JC, Nino M, Nannipieri P, Chacón P (1989) Phosphorus accumulation in savanna termite mound in Venezuela. Soil Sci 40:635–640
Liu S, Lin X, Behm JE, Yuan H, Stiblik P, Šobotník J, Gan J, Xia S, Yang X (2019b) Comparative responses of termite functional and taxonomic diversity to land-use change. Ecol Entomol 44:762–770
Mamo M, Wortmann C (2009) Phosphorus sorption as affected by soil properties and termite activity in eastern and southern Africa. Soil Sci Soc Am J 73:2170–2176
Menichetti L, Landi L, Nannipieri P, Katterer T, Kirchmann H, Renella G (2014) Chemical properties and biochemical activity of colonized and abandoned litter-feeding termite (Macrotermes spp.) mounds in chromic Cambisol Area on the Borana Plateau Ethiopia. Pedosphere 24:399–407
Muvengwi J, Witkowski ETF (2020) Cascading effects of termite mounds in African savannas. New Zeal J Bot 58:167–193
Muvengwi J, Witkowski ETF, Davies AB, Parrini F (2017) Termite mounds vary in their importance as sources of vegetation heterogeneity across savanna landscapes. J Veg Sci 28:1008–1017
Paul BK, Vanlauwe B, Hoogmoed M, Hurisso TT, Ndabamenye T, Terano Y, Six J, Ayuke FO, Pulleman MM (2015) Exclusion of soil macrofauna did not affect soil quality but increased crop yields in a sub-humid tropical maize-based system. Agric Ecosyst Environ 208:75–85
Rückamp D, Amelung W, Theisz N, Bandeira AG, Martius C (2010) Phosphorus forms in Brazilian termite nests and soils: Relevance of feeding guild and ecosystems. Geoderma 155:269–279
Rückamp D, Martius C, Bornemann L, Kurzatkowski D, Naval LP, Amelung W (2012) Soil genesis and heterogeneity of phosphorus forms and carbon below mounds inhabited by primary and secondary termites. Geoderma 170:239–250
Sarcinelli TS, Schaefer CEG, de Souza Lynch L, Arato HD, Viana JHM, de Albuquerque Filho MR, Gonçalves TT (2009) Chemical, physical and micromorphological properties of termite mounds and adjacent soils along a toposequence in Zona da Mata, Minas Gerais State, Brazil. CATENA 76:107–113
Seetapong N, Chulok S, Thongkhong V (2021) Physical properties of termite mound soil in para rubber plantation of southern border provinces. J Phys Conf Ser 1719:012038
Seymour CL, Milewski AV, Mills AJ, Joseph GS, Cumming GS, Cumming DHM, Mahlangu Z (2014) Do the large termite mounds of Macrotermes concentrate micronutrients in addition to macronutrients in nutrient-poor African savannas? Soil Biol. Biochem 68:95–105
Tilahun A, Cornelis W, Sleutel S, Nigussie A, Dume B, Van Ranst E (2021) The potential of termite mound spreading for soil fertility management under low input subsistence agriculture. Agriculture 11:1002
Traore S, Bottinelli N, Aroui H, Harit A, Jouquet P (2019) Termite mounds impact soil hydrostructural properties in southern Indian tropical forests. Pedobiologia 74:1–6
Tuma J, Frouz J, Veselá H, Křivohlavý F, Fayle TM (2022) The impacts of tropical mound-building social insects on soil properties vary between taxa and with anthropogenic habitat change. Appl Soil Ecol 179:104576
Van Thuyne J, Verrecchia EP (2021) Impacts of fungus-growing termites on surficial geology parameters: A review. Earth-Sci Rev 223:103862
Wang J, Zou Y, Di Gioia D, Singh B, Li Q (2020) Conversion to agroforestry and monoculture plantation is detrimental to the soil carbon and nitrogen cycles and microbial communities of a rainforest. Soil Biol Biochem 147:107849
Zanne A, Flores-Moreno H, Powell J, Cornwell W, Dalling J, Austin A, Classen A (2022) Temperature sensitivity of termites determines global wood decay rates. Science 377:1440–1444
Zhou Y, Ma H, Xie Y, Lu Q, Shen Y, Ma J (2022) Response of soil aggregate stability and erodibility to different treatments on typical steppe in the Loess Plateau. China Restor Ecol 30:e13593
Acknowledgements
We acknowledge the Institutional Center for Shared Technologies and Facilities of Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences (CAS) for chemical analysis. We thank Shengjia Li from Pu'er University for her contributions to the fieldwork.
Funding
This work was jointly supported by the National Natural Science Foundation of China (32071744, 32271854, 31901303), the Yunnan Fundamental Research Projects of China (202201AT070123), the ‘Light of West China Program’ of CAS, the "Yunnan Revitalization Talent Support Program" in Yunnan Province and the Youth Innovation Promotion Association CAS (2019388).
Author information
Authors and Affiliations
Contributions
FL, CJ and YR conduct the experiment. CL and YJ conceive the design. All the authors take part in analyzing, writing, and revising the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible Editor: Leo M. Condron.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, F., Jin, Y., Liu, C. et al. Termite mounds affect soil aggregate stability and aggregate-associated phosphorus forms in a tropical rubber plantation. Plant Soil 498, 93–109 (2024). https://doi.org/10.1007/s11104-023-05880-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-05880-4