Abstract
Background
Extracellular polymeric matrix (EPM) is a complex component of the organo-mineral assemblages created by biological soil crusts (BSCs). Mainly of polysaccharidic origin, it embeds soil and sediments and provides key benefits to the crust community. Services provided include: sediment cohesion and resistance to erosion, moisture provision, protection from external harmful factors, as well as support to plant establishment and growth. EPM is the product of BSC microbial community, and it is constituted by exopolysaccharides (EPS) associated to other substances, organized in a three-dimensional structure having different levels of gelation, and degrees of condensation.
Scope
This review aims at focusing scientific attention, for the first time, on the characteristics and the roles of three operationally defined EPM fractions, one water soluble, one more adherent to cells and sediments, and one firmly attached to microbial cells. The latest results obtained by analyzing EPM of natural and induced (i.e, the result of cyanobacteria inoculation) BSCs are outlined, and the optimized extraction methodology is described in details.
Conclusions
The review underlines the complexity of investigating the characteristics and the role of microbial EPS, and its supra-structure (EPM), in natural conditions (as opposed to cultures in laboratory conditions), where the matrix is subjected to continuous microbial rearrangement due to biosynthetic, self- and cross-feeding processes, and where microbial activity affected by environmental parameters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biological soil crusts (BSCs) are highly specialized complex microbial communities which are an integral component of arid and semiarid ecosystems. By colonizing the uppermost soil layers, they control the exchange of gases and nutrients (Pointing and Belnap 2012; Weber et al. 2015), play important ecological functions (Bowker et al. 2011; Maestre et al. 2011) and provide soil stability and N-enrichment, two key factors supporting vascular plants establishment (Bowker 2007; Miralles et al. 2012). They are composed of bacteria, microalgae, microfungi, green algae, lichens and mosses (Wu et al. 2013), although some of these organisms may be lacking, depending on climate and terrain age (Belnap and Lange 2001).
Microbial-produced exopolysaccharides (EPS) accumulate as a heterogeneous extracellular polymeric matrix (EPM) that is strictly associated to organisms and sediments in BSCs. Other than for exopolysaccharides, the term EPS has been used as acronym for extracellular polymeric substances to account for a number of other components that, along with the dominant polysaccharidic fraction, constitute the EPM (see section 3). EPM constitute a supra-structure of EPS, having a three-dimensional organization and varying degrees of condensation, from mucilaginous to solid gel (Fig. 1). The concept of EPM is present on a wide array of studies on microbial biofilms, although not thoroughly stressed, and addressed with slightly different terms, including extracellular polymeric substance matrix (Battiston et al. 2015; Fish et al. 2016; Gu et al. 2017) and biofilm matrix (Limoli et al. 2015).
EPM provides a wide array of services to the crust community, from conferring physical integrity and stability, to providing an optimal microenvironment with increased moisture, nutrients, and protection from harmful biological and physical agents.
Although many BSC organisms produce EPS, cyanobacteria and microalgae are known prominent contributors (Belnap and Lange 2001). Other less-acknowledged producers of EPS are microfungi (Selbmann et al. 2003), and members of proteobacteria and actinobacteria (Martínez-Cánovas et al. 2004; Suela Silva et al. 2013) that are present in crust communities (Nagy et al. 2005; Kuske et al. 2012; Rossi et al. 2012c). The excretion of EPS is an important physiological process from the first stage of BSC development, when bare soil is colonized by sheathed filamentous cyanobacteria (Belnap and Eldridge 2001). Incipient cyanobacterial crusts, representing an early stage of BSCs (Lan et al. 2013), are a nutrient-rich substrate easily colonizable by other phototrophic species and heterothrophic bacteria. Although several authors recognize the importance of EPS in BSC - water relations and survival (Mazor et al. 1996; Mager and Thomas 2011), and consider their amount an index of soil stability (Hoppert et al. 2004) and of the metabolic capacity of the community (Bu et al. 2014), there is still a lack of information concerning their contribute in increasing the resilience of BSCs to environmental constraints and in nutrient and water diffusion. In addition, information concerning their physiochemical properties and their modifications in space and time is still limited.
While the roles of cyanobacterial and microalgal EPS have been pointed out by several researchers (Mazor et al. 1996; De Philippis and Vincenzini 1998; Mager and Thomas 2010; Rossi and De Philippis 2015a), the majority of these scientific advances were attained under lab conditions, and employing strains growing in liquid culture. Studies under natural conditions (e.g., field colonies) are limited to a few cases. Some of them point out that cyanobacteria may produce compositionally different EPS depending on whether they face nutrient limitations and constraints, or optimal growth conditions (Huang et al. 1998; Brüll et al. 2000). For microalgae and cyanobacteria, several factors were demonstrated to influence EPS productivity and, in some cases, EPS characteristics. Those include light intensity and temperature; availability of carbon, nitrogen, phosphorous an sulphur; moisture level and salinity (Rossi and De Philippis 2015b).
EPM characteristics in natural microbial associations are strongly dependent on the dynamics and the activities of the microbial community (Flemming and Wingender 2010). Thus, an increasing level of complexity is expectable in moving from studies performed on laboratory isolates, to studies conducted in situ on composite microbial aggregates.
This work aims at giving an overview of the characteristics and the roles of EPS in BSCs, with a particular attention to their organization in definite stable three-dimensional structures, an aspect which has not been studied so far. In addition, we propose a clear terminology consistent with previous studies conducted on EPM in other complex microbial associations.
Cyanobacterial, microalgal, and fungal EPS: Morphological and chemical characteristics and roles
The synthesis of EPS, which is an energy-consuming process, has important ecological implications for its producers (Li et al. 2001). Several studies led to believe that EPS production is a physiological mechanism increasing organism survival, and tolerance to environmental harsh conditions. Several known roles of EPS are reported in Table 1.
It is generally believed that EPS production does not confer concrete advantages in laboratory cultures, in contrast to natural conditions, under which cells experience competitive multispecies environments and multiple stresses (Costerton et al. 1987). Cyanobacteria and microalgae have been generally considered as initiators of BSC development on bare oligothrophic soils. Being proficient EPS producers, they strongly promote the first crucial soil stabilization. Although other nonphotosynthetic bacteria were demonstrated to be significant EPS producers, and to have a role in crust formation (Wu et al. 2010), EPS produced by cyanobacteria, microalgae and filamentous fungi bear generally a higher complexity in terms of monosaccharidic composition (Osińska-Jaroszuk et al. 2015; Pereira et al. 2009). In particular, Wu et al. (2010) underlined how nonphotosynthetic bacteria in BSCs contribute compositionally simple polysaccharides, with mannose, galactose, glucose, and a glucose isomer accounting for more than 98%. Conversely, cyanobacterial and microalgal EPS may also contain high relative amounts of non neutral sugars (see below).
Cyanobacteria and microalgae produce EPS excreted as sheaths and capsules, or unevenly dispersed as mucilage (Rossi and De Philippis 2015b) depending on their chemical features, and on abiotic factors (e.g., available ions, pH). These outermost structures have also been described for other prokaryotes (e.g., Decho and Lopez 1993; Vincent et al. 1994), sometimes termed “glycocalyx” (Wingender et al. 1999). Bacterial capsules and sheaths can be attached to cells through non-covalent interactions, but also covalently to phospholipids and lipid-A molecules at the cell surface (Roberts 1996). Concerning cyanobacteria and green microalgae, the nature of these outer investments has been described in details in past publications (De Philippis and Vincenzini 1998; De Philippis et al. 2001; Pereira et al. 2009; de Paniagua-Michel et al. 2014, Rossi and De Philippis 2015b). On the other hand, regarding fungi there is a more limited amount of information, especially regarding the biosynthetic processes. In laboratory cultures, some species encompassing lower filamentous fungi and yeasts from different ecological niches produce EPS (Mahapatra and Banerjee 2013). Black yeasts such as Exophiala crusticola and Rhodotorula, often detected in BSC communities, were indicated as highly probable contributors of soil stability in desert systems owing to their EPS productivity (Bates et al. 2006). EPS excretion in fungi has been correlated with the production of sclerotia, presence of conidia, and to the phytopathogenic behavior (Selbmann et al. 2003). As for cyanobacteria and microalgae, fungal EPS production is affected by several parameters, including temperature, oxygen concentration, pH, and N source (Mahapatra and Banerjee 2013; Seviour et al. 1992).
EPS produced by cyanobacteria and microalgae can be compositionally complex. In cyanobacteria, they can contain up to 15 sugar moieties (Pereira et al. 2009), organized in complex repeating units and often having a molecular weight (MW) up to 1–2 MDa. Non saccharidic components like peptides, lipids and nucleic acids are also present. Glucose, galactose, arabinose, xylose and uronic acids have been frequently detected in major amounts. Methyl, pyruvyl, succinyl and sulphate groups were also detected in some cases. The presence of hydrophilic moieties on one side (sulphated sugars, uronic acids and ketal-linked pyruvyl groups, among others), and hydrophobic on the other (acetyl groups, dehoxysugars and peptides) confers an amphiphilic character to the macromolecules and hence provide greater plasticity in organisms’ response to surrounding environment (Rossi and De Philippis 2015b). While hydrophobic EPS fractions are more involved in the adhesion to solid surfaces, hydrophilic fractions are more involved in binding minerals, nutrients and water molecules (Rossi et al. 2012a). Some cyanobacteria are also reported to excrete cellulose (de Winder et al. 1990), which is often localized in the sheath (Stuart et al. 2016).
Fungal EPS can consist of polymers with a high MW (around 2 MDa), and be organized in very diverse repeating units with a complexity similar to that observed for EPS produced by cyanobacteria, although not always displaying a similar high compositional complexity (Seviour et al. 1992). In some cases, glucose, mannose and galactose are the only components, whereas in other cases uronic acids, aminosugars and rhamnose may be present (Mahapatra and Banerjee 2013).
The EPM of complex natural microbial communities
EPM of BSCs is similar to those already described for other typologies of bacterial aggregates (Wingender et al. 1999). It is an ordered, hydrated, semi-solid state structure organized in a polymeric three-dimensional network embedding organisms and soil sediments. Its physical state is governed, to a large extent, by environmental parameters. It provides structural and functional integrity to BSCs.
Being the result of microbial synthesis and demolition, EPM of natural communities is in continuous modification and rearrangement, with its composition and distribution varying spatially and temporary according to the prevailing activities of the biofilm (Wingender et al. 1999). The presence of a so structured matrix allows consortial activities, needed by microorganisms to maximize their fitness through cooperative interactions, while synergistic activities enhance the resilience to stress factors and oligotrophic conditions (Wingender et al. 1999). Within the EPM, the spatial arrangement creates gradients of oxygen (determining aerobic and anaerobic habitats), other electron acceptors, as well as organic substrates (e.g., proteins, peptides, aminoacids and colloids), and pH value (Costerton et al. 1987; Kepkay 1994; Mayer et al. 1995; Wingender et al. 1999). Some processes such as the accumulation of nutrients and other substances from the bulk soil water, gene exchange, and quorum sensing are favored.
Beside exopolysaccharides, EPM is constituted by other components that are the result of secretion processes or are released after cell lysis (Gu et al. 2017; Wingender et al. 1999), although the polysaccharidic component is often strongly dominant (Al-Thani 2015). Proteins, nucleic acids, and amphiphilic substances as (phospho)-lipids can be often detected in varying amounts as part of the matrix in the extracellular space. Extracellular proteins may establish hydrogen bonds within the EPM structure (Dignac et al. 1998); some can be glycosylated to create glycoproteins, or substituted with fatty acids to form lipoproteins. One main function of extracellular proteins is to act as enzymes for the digestion of exogenous macromolecules (Wingender et al. 1999).
Several authors have attempted to define the different EPM fractions observed in complex biofilms. Some used the term “slime” to indicate EPM fractions that are loosely bound to cells (and soil sediments) and less condensed, although not dissolved. Dissolved EPM fractions are referred to as “colloidal” (Nielsen and Jahn 1999). A generic distinction used by some authors is between “bound” EPS (sheaths, capsules, condensed gels, loosely bound polymers, attached organic material) and “soluble” EPS (soluble polymers, colloids, slimes) (Nielsen et al. 1997). Operationally, the more easily recoverable fraction is that which is less condensed, and weakly attached to cells and sediments (loosely bound EPS, LB-EPS) (Fig. 2). A second fraction consists in molecules with a higher level of gelification and thus thickened, having strong bonds with cells and sediments (tightly bound EPS, TB-EPS). This fraction may include more sub-levels of gelification and the extent of recovery is strictly determined by the extraction methodology (see section 4). In addition to LB-EPS and TB-EPS fractions, we identify a third “glycocalix” fraction (G-EPS) (Wingender et al. 1999), which is firmly attached to the cells (capsules or sheaths). G-EPS may be either containing filaments or hollow, following filament migration.
Extraction and analysis of EPS from complex microbial associations: The case of BSCs
In most studies present in the literature, the selected extraction procedure defines the typology of recovered fractions (Nielsen and Jahn 1999). Most of the extraction procedures rely on the fact that EPS fractions have varying levels of solubility. The immediately soluble fractions can generally be removed with a washing with H2O, whereas more hydrophobic fractions are not expected to be recovered in this way. Less soluble fractions can be recovered by applying proper extraction methods. For the majority of studies on complex microbial biofilms, extraction methodologies can be encompassed in the following procedural scheme (Table 2).
Any extraction method must be developed and optimized according to the type and structure the biofilm and the type of environment (soil or aquatic environment) which it belongs to.
There is not an universally followed extraction method to recover EPS from BSCs. The existing extraction procedures that appeared recently in literature were adapted from methods previously applied to other types of biofilms, or to cyanobacterial strains grown in liquid cultures.
It has to be stressed that if the analysis is aimed only at the EPS, the maintenance of cell integrity is a prerequisite. In this case, the extraction procedure must be selected in a way to not cause cell leakage, or the EPS will be contaminated with intracellular material. When the extraction causes cell lysis, the subsequent analysis, whether simple quantification or macromolecular characterization, is performed on “total polysaccharides” or “total carbohydrates” and not on EPS. The determination of cell lysis after extraction is not straightforward, since some intracellular substances, e.g. proteins and nucleic acids, may be naturally present in the extracellular environment (see section 2). The use of truly intracellular compounds as markers for cell lysis is suggested. One compound is ATP, although in past studies some concerns regarded the level of accuracy in measuring this molecule (Grotenhuis et al. 1991). Another suggested marker is the enzyme glucose-6-phosphate dehydrogenase (G6PDH) (Platt et al. 1985). Stuart et al. (2016) used this enzyme to evaluate the extent of cell lysis after EPS extraction from cyanobacterial mats. Another parameter supporting the loss/maintenance of cell integrity is the content of chlorophyll in the extracts, although the reliability is related to the abundance of the phototrophic fraction of the community.
Extraction procedure is generally a combination of chemical and physical approaches, based on considering the major type of interactions that keep EPS together in EPM, namely van der Waals forces, electrostatic interactions, hydrogen bonds, hydrophobic interactions and covalent bonds (Christensen 1999). The application of only physical methods (addition of water followed by centrifugation, mixing, shaking or sonication) gives lower EPS yields than combining with chemical methods (Nielsen and Jahn 1999). Physical methods alone result in a minimal, if not null, release of bound EPS.
Chemical methods include the use of a wide array of substances that are meant to facilitate the release of TB-EPS. Extractants include pyridine acetate, used for Escherichia coli (Pelkonen et al. 1988), NaOH (Sato and Ose 1980) and NaCl used for Pseudomonas aeruginosa (May and Chakrabarty 1994). The use of alkali (e.g., NaOH) leads to the ionization of charged groups in EPS, due to their isoelectric point that is generally below pH 4–6. The result is a strong repulsion within the EPM and the increase of water solubility of more condensed fractions, although this process seldom leads to the removal of G-EPS. The structural order of EPM can be shifted to disorder on heating or removal of ions (Sutherland 1999). For example, the removal of cations such as Ca2+ and Mg2+ using complexant agents such as ethylenediaminetetraacetic acid (EDTA) and ethylene glycol-bis(ß-aminoethyl-ether)-N, N, N′, N′ – tetraacetic acid (EGTA) strongly compromises the stability of the EPS strands, and EPM tends to fall apart. In a study dedicated to the comparison of several methods for the extraction of EPS from soil biofilms, Redmile-Gordon et al. (2014) suggested that the best method is based on the use of cation exchange resins as they are capable of maintaining the integrity of the cells thus preventing the contamination of the extracted EPS with humified soil organic matter.
The extraction procedure can be aimed at a general quantification of EPS in a BSC sample, or at recovering specific fractions for separate quantifications. A method to extract and quantify EPS from intertidal sediments proposed by Underwood et al. (1995) was recently successfully applied to quantify EPS in BSCs (Rossi et al. 2012c; Colica et al. 2014). It consists of extractions in 0.1 M Na2EDTA of small amounts (~100 mg) of homogenized BSC for 15′ at room temperature. The extracts can be assayed for total extracellular carbohydrate amount by applying phenol-sulfuric acid assay (Dubois et al. 1956). To quantify actual EPS (the fraction with a MW ≥ 100 k Da), the quantification must be performed after treating the extract with ethanol (70% final concentration) (Decho and Lopez 1993). The extraction efficiency of the method in analysis preliminary to those published in Rossi et al. (2012c) was evaluated by extracting EPS from BSCs collected in North American deserts (F. Rossi, personal communication). The analysis was conducted employing two BSC typologies (described in Rossi et al. 2012c), one collected in the Chihuauan Desert, and one in the Mohave Desert, that were different for relative abundances of species (significant differences in cyanobacterial and proteobacterial relative abundances), and in the percents of sand and silt contents.
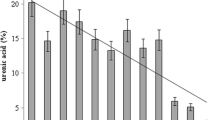
These results pointed out that repeated extractions may increase extraction yields. At least five extractions were needed to remove all the extracellular carbohydrates, although the carbohydrates removed with the first two extractions represented over 60% of the total amount recovered with the sum of all the extractions (Fig. 3) (F. Rossi, personal communication). Although the needed number of extractions may depend on the typology of crust and soil texture, the method is relatively rapid, and allows to process multiple samples simultaneously.
Repeated extracellular carbohydrate extractions from two different typologies of BSCs from North American deserts, utilizing the method of Underwood et al. (1995) for the first time on BSCs. Dark brown bars, Chihuahuan Desert crust; light brown bars, Mohave Desert crust. Yields were expressed as mg of extracellular carbohydrates (g soil)−1 for each extraction on each sample. Each sample type was extracted in triplicate and results were expressed as value ±SD (F. Rossi, personal communication)
According to some authors, treatment with EDTA may cause cell wall destabilization due to divalent cations removal causing cell leakage (Nielsen and Jahn 1999). Nonetheless, Underwood et al. (1995) observed low, if not null, intracellular contamination extracting intertidal sediments. In using EDTA extraction on cyanobacterial biofilms, Stuart et al. (2016) ruled out cell lysis.
We recommend the procedure described in Fig. 4 to recover LB-EPS, TB-EPS and G-EPS from BSCs. Most of the hereby proposed methods have been published in several previous papers (Rossi et al. 2012c; Colica et al. 2014; Chen et al. 2014; Colica et al. 2015).
The procedure includes recovering LB-EPS by water extraction and then recovering TB-EPS by extracting with 0.1 M Na2EDTA. These two treatments in sequence will leave a pellet of sediments, cells and G-EPS. Several methods have been proposed for the removal of sheaths and capsules for microalgae and cyanobacteria. Those include sucrose gradients, acidic treatment and heat treatment (Rossi and De Philippis 2015b). To remove G-EPS, we suggest treating the pellet resulting from the removal of LB- and TB-EPS with hot water (80 °C) for 1 h after washing with 1,5% NaCl (Mugnai et al. 2017).
Following the recovery of the three fractions, the phenol-sulfuric acid assay can be used to quantify them. It is possible to further purify the fractions, or treat them for further analytical analysis. The purification processes often require precipitation in alcohol/acetone (we recommend isopropyl alcohol or ethanol) of the extract. Although the process removes impurities, it also removes low MW polymers that will not be detected in further macromolecular analysis (e.g., gel permeation chromatography). The level of purification strictly depends on the aim(s) of the investigation and may involve further purification steps depending on the hydrophobicity/hydrophilicity of the macromolecules to be removed. When not of interest, proteins may be removed by using proteases, phenol extraction or gel filtration chromatography. DNA or RNA may be removed by using nucleases.
The EPM of BSCs: Morphological and chemical characteristics of EPS and their role
Morphology of EPM of BSCs
Mager and Thomas (2011) reported the presence of EPS-containing structures in BSCs from the Kalahari Desert, and they categorized them as “capsules”, “granules” and “slime”, basing on their morphology. To our knowledge, it was the first time that a tentative morphological description of EPM was attempted. Arid soil pioneers like the non-heterocystous cyanobacterium Microcoleus vaginatus, members of the genera Schizothrix or Hydrocoleum forms filaments constituted of rope-like bundles of trichomes encased in tubular exopolysaccharidic sheaths (Garcia Pichel and Wojciechowski 2009), resulting from one or more secretion events. This is recognized as the first step in BSC formation. Cyanobacterial sheaths can be considered a central element in the formation of EPM, as they constitute the first accumulation of EPS material on which early stages of BSCs are structured (Rajeev et al. 2013). Organized in “large bodies” (~100 μm), sheaths bind soil particles stabilizing soil against erosion by wind or water (Belnap and Büdel 2016; Belnap and Gardner 1993). In studying early-stage BSCs from Gurbantunggut Desert, China, Zhang (2005) observed that cyanobacterial sheaths either contained filaments, or were leftover material, after filament migration or death. Empty sheaths remain solidly attached to the organo-mineral material as a cement stabilizing the crust structure. Unless crashed by compressional disturbances, a primitive discernible organization of the EPM is visible from the first stages of development of BSCs by microscopical observations. Three month-old artificial cyanobacterial crusts obtained by inoculating the cyanobacterium Schizothrix delicatissima AMPL0116 on bare sand in microcosms were constituted by an EPM distinctly organized in a LB-EPS and a TB-EPS fraction (Fig. 5) (Mugnai et al. 2017).
Microscopical images of the operationally-defined EPM fractions in a 15 day-old induced cyanobacterial crusts obtained by inoculating the cyanobacterial strain Schizothrix cf. delicatissima AMPL0116 in microcosms. G-EPS, glycocalix EPS (encompassing sheaths and capsules); LB-EPS, loosely bound EPS; TB-EPS, tightly bound EPS. a) Filaments and sheaths of S. delicatissima gluing two adjacent sand particles. The picture underline the prominent role of EPS in sediment cohesion. b) TB-EPS covering a sand grain surface. Pictures by GM
Chemical and macromolecular characteristics of EPM in BSCs
The chemical and macromolecular characteristics of EPM of BSCs have been studied in only a limited number of cases. Recently, the monosaccharidic composition and MW distribution of EPS extracted from BSCs of known age, collected in the Hopq Desert, Inner Mongolia, China, was carried out. The area is hyper-arid, with a climate classified as semiarid, temperate and continental monsoon. These BSCs were the result of an inoculation-based treatment carried out in different years (Chen et al. 2006; Li et al. 2014; Wang et al. 2009). Notwithstanding the stressful environmental conditions which would suggest a compositional simplicity (Mager and Thomas 2010), the extracted EPS showed instead a certain complexity, unrelated to the age of the crusts. Up to 13 different types of sugars were identified. They included the hexoses galactose, fructose and glucose, which had the highest relative abundance, the dehoxy-sugars fucose and rhamnose, the amino-sugars galactosamine and glucosamine, and the pentose ribose. In addition, uronic acids, namely galacturonic and glucuronic acids, were also detected (Chen et al. 2014; Colica et al. 2015). Extracted EPS showed mainly MWs comprised between 2 M and 485 kDa (in the range characterizing EPS produced by cyanobacteria) and between 72.6 k Da and 0.34 k Da, comprised of small MW saccharides, dimers and monomers. In a further study, the composition and MW distribution of LB-EPS and TB-EPS were determined and compared. While the two fractions had a similar compositional pattern, they appeared to be different in MW distribution. The 90% of TB-EPS had a MW between 2 M and 0.76 M Da, while the less condensed fractions were polymers in the lower MW ranges (Chen et al. 2014). In Arctic BSCs, collected around Ny-Ålesund, Svalbard archipelago, the cyanobacterial relative abundance was very low in favor of the relative abundances of Proteobacteria, Actinobacteria and Acidobacteria (Mugnai et al. 2015; Rossi et al. 2012b). Notwithstanding this, and the obvious different environmental characteristics, monosaccharidic composition was of a similar complexity between the two types of crusts. On the contrary, the MW distribution profile was significantly different, with high relative percentages of small MW carbohydrates. The comparison of these first surveys suggests that MW distribution profile may be more distinctive among crusts with different characteristics, and among different environments.
Structural role of EPM in BSCs
EPM is essential for crust physical structure, prompting the cohesion between the biotic and the a-biotic crust components, and the adhesion to solid substrates (Rossi and De Philippis 2015a).
Due to the stability that they confer to the BSC structure, and their capability to counteract erodibility, EPS are considered by some authors as an index of soil aggregation and BSC development (Bowker et al. 2008).
The sediment-cementing nature of EPS has been often observed and acknowledged (Zhang 2005; Malam-Issa et al. 2007). This structuring role is important for the incipient cyanobacterial colonization of bare soils. Some BSC-dwelling cyanobacteria and microalgae, tested for soil stabilizing capability, provided evidence that EPS productivity is in agreement with their capability of stabilizing fine sand grains (Hu et al. 2003) and crust resistance to wind erosion (Hu et al. 2002). EPS strands bind fine soil particles (< 65 μm) enforcing the stabilizing action of bacterial filaments, mosses and lichens (Bowker et al. 2008). In addition, Malam Issa et al. (2009) provided evidence that EPS, due to their amphiphilic properties, confer to bacterial filaments the capability to drive the formation of additional soil pores (“microbial pores”), affecting their geometry, and determining soil spatial organization within the BSC thickness.
By studying the EPM of three to eight year-old induced BSCs in a semiarid environment, it was observed that TB-EPS is the fraction more involved in providing a structural role, appearing more “preserved” from enzymatic activity, and being more condensed than LB-EPS (Chen et al. 2014).
EPS are also involved in the formation of induced sedimentary structures, which include roll-ups, folds, desiccation polygons, especially on sandy soils (Garcia Pichel et al. 2016) and in hot arid conditions. The formation of these structures may have an ecological meaning for crust community. In natural dry hot environments the formation of cracks may provide open pathways to increase soil permeability and aeration under crust layers (Williams et al. 2012), leading to new colonizable niches with enhanced moisture regimes (Danin et al. 1998).
Role of the EPM in BSC-water relations
One of the first reported effects of the presence of EPS in BSCs was that their swelling, and pore-clogging effect upon re-wetting, leading to run-off during rain events (Kidron et al. 1999; Fischer et al. 2012). The role of EPM of BSCs in affecting soil infiltrability was investigated in two different studies (Rossi et al. 2012c; Colica et al. 2014), considering soil textures ranging from silt loam to sandy, according to the USDA classification (e.g., Groenendyk et al. 2015). Some differences related to soil texture appeared evident: loamy sand and silt loam soils showed a high positive correlation of sorptivity values with sand content and negative with silt and clay. At the same time, no correlation was found between sorptivity and EPS content (Rossi et al. 2012c). Nonetheless, the application of a simple nondestructive extraction of EPS (detailed in Rossi et al. 2012c) resulted in a more compacted crust structure that lost the ability to absorb water. This strongly supported the idea of a contribution of the EPM in structuring BSC waterways. This aspect was further investigated by Felde et al. (2016) that examined the effects of this extraction procedure by using X-ray computerized tomography. This analysis ruled out any “artificial” modification of the crust structure, apart from EPS extraction, limiting the cause of sorptivity change only to their removal. Studies conducted with BSCs growing on sandy soils in the Hopq Desert, Inner Mongolia, China, where water infiltration velocity is high, depicted a different scenario. A significant correlation between EPS content and soil sorptivity was found. In this context, the presence of BSCs diminishes remarkably water infiltrability (up to 90% detected reduction). The correlation observed supports the notion that EPS swell following water contact, reducing the volume of soil pores (Fischer et al. 2010a), and promoting the maintenance of the moisture in the very first soil layers which are the most biologically active.
The presence of an EPM is very important for cells to counterbalance water-deficient conditions. Owing to their amphiphilic character, EPS accumulate water, and regulate water loss (Pereira et al. 2009), maintaining hydration (Mazor et al. 2009) and counteracting evapotraspiration. The presence of a moistened environment at the soil surface is very important for an optimal physiological activity of the crust community. In addition, EPS are involved in dew formation at the surface of BSCs (Fischer et al. 2012).
In one study conducted on BSCs grown on sandy soil in the same Hopq Desert, it was demonstrated that i) EPS are fundamental to retain humidity and ii) EPS are fundamental for water uptake from non-rainfall water sources (i.e., dew, fog and plant guttation) (Colica et al. 2014). The Hopq Desert site is characterized by an average yearly evapotranspiration that broadly exceeds that of precipitation. The capability of BSCs to retain water against evapotranspiration was correlated to the presence of EPS, especially those having a high MW. After removing 90% of EPS from BSC samples, utilizing the previously mentioned nondestructive method, water uptake capability decreased sensibly, to being not statistically different from that of bare sand. These studies expanded the knowledge on the important roles of EPM in: i) delaying water movement through the soil when sorptivity is high, and contributing to the creation of viable waterways when sorptivity is low, ii) regulating water uptake and water loss from the cells (Pereira et al. 2009), and iii) reducing evaporation loss and increasing soil water-holding capacity (Mager and Thomas 2011).
EPM in BSCs as a nutriment source and plant fertilizer
EPM is a medium constantly subjected to enzymatic and abiotic degradation, to modification and condensation processes. For example, humic substances are thought to have a chemical structure which is the result of partial enzymatic degradation and condensation (Hedges 1988). Enzymes mostly involved include hydrolases, (less frequently) lyases, invertases, and sucrases (Wingender et al. 1999; Miralles et al. 2012). The activity of hydrolases can provide correlative information on microbial activity and microbial population and in some cases it is related to soil organic content (Chen et al. 2014). The hydrolytic enzymatic action in BSCs increases according to the level of development (LOD), from cyanobacteria-dominated crusts to lichen-dominated crusts, primed by an increase in C and N (Miralles et al. 2012). In one study, it was observed that the activity of hydrolases and sucrases is correlated not only to LOD, but also to the age of BSCs (Chen et al. 2014).
EPM promotes the association in consortia that depolymerize complex compounds to simple molecules easily assimilable even by community members having a smaller genome, a less specialized lifestyle, and not possessing a full range of degradative enzymes (Mba Medie et al. 2012). Some microorganisms are able to degrade and feed on their own EPS. Although some authors deem it limited to a few cases, the “reuse” theory has been demonstrated for fungi and cyanobacteria. Fungi possess glucanases, which degrade (1–3)-ß- and (1–6)-ß-glucans with several mechanisms of action and functions (Seviour et al. 1992). Stuart et al. (2016) demonstrated that in induced cyanobacterial microbial mats, cyanobacteria can degrade oligosaccharides, proteins and nucleic acids. In addition, they found that cyanobacteria are able, in a short time, to feed on the C from their own EPS, both in the light and in the dark. This information may be important for fully describing C cycle within these communities.
Since arid and semiarid soils contain low amounts of C, the emission of EPS is of vital importance. According to Chenu (1993), EPS may represent up to 500% of cellular biomass, and were hypothesized to be the primary substrate respired after heavy rainfall events (Thomas et al. 2008; Fischer 2009; Thomas and Hoon 2010). A study on the enzymatic activity in induced BSCs of different years, was coupled with a characterization of the EPM fractions (Chen et al. 2014). It was observed that the enzymatic activity was mostly directed towards the more soluble LB-EPS, which displayed substantial variation in MW distribution comparing BSCs of different ages. A study conducted by Decho and Lopez (1993) on the digestibility of EPS-producing bacterial cells, demonstrated an higher resilience to digestion of G-EPS, which has more stable, ordered and definite secondary structures, in comparison with less condensed EPS fractions. This supports the idea that slime diffusing in the soil is the principal more easily degradable EPS fraction, while TB-EPS and G-EPS, due to their lower degradability, constitute more a “structural skeleton” of BSCs.
EPM may also act as a nutrient accumulator, concentrating dissolved organic matter (DOM) with compounds containing C, N, P and trace metals which are essential for cell metabolism (Wolfaardt et al. 1999; Flemming and Wingender 2010). In this respect, such a nutrient source may be of importance for vegetation establishment, determining an increase of essential nutrients in plant tissues (Zhang et al. 2016).
Studies addressing the effects of BSCs in plant establishment are often contradictory (Zhang et al. 2016). It is nonetheless obvious that, since EPM conditions soil cohesion, soil properties, soil porosity and the formation of microbial induced sedimentary structures (e.g., roll-ups, folds, desiccation polygons) (Garcia Pichel et al. 2016) BSCs indirectly affects seeds entrapment and subsequent emerging. For example, BSCs enhance germination and emergence of seeds in cool desert (Belnap 2003a, b) where morphology is characterized as rolling or pinnacled (Belnap 2001).
Whether EPS are directly effective and benign to plant establishment and growth is still unknown. One study (Xu et al. 2012) seems to suggest so. The treatment of the seeds of Caragana koshinskii, a desert-dwelling shrub belonging to Leguminosae, with the solubilized EPS produced by Phormidium tenue (Oscillatoriales), a cyanobacterial species commonly found in BSC communities (e.g., Hu et al. 2002), promoted germination (germination index, and germination energy) as well as root growth, nutrient and water uptake, photosynthetic efficiency, and defense from oxidative damages. Although these data are the result of a single study, they point out the need of a more in-depth exploration the bioactive characteristics of EPS.
Conclusions
In spite of the unanimously recognized importance of the EPM in the formation and stabilization of BSCs, and compelling SEM imagery illustrating this importance (Belnap 1993; Zhang et al. 2006; Fischer et al. 2010b; Mager and Thomas 2011), an increasing number of reports on the characteristics of the most important constituents of EPM, namely EPS, has appeared only recently. The papers reviewed here clearly show the complexity of the functions of EPM. Indeed, one of the most striking recent results obtained is that EPS are constituted by more than one fraction, with each fraction having different chemical properties and possibly playing different roles in BSCs. This may contribute to the amazing capability of these complex structures to withstand very harsh environmental conditions. Moreover, the presence of such complex EPS structures points out the need of applying specifically developed methods for the extraction of the different fractions in order to obtain a sound characterization of their chemical and macromolecular features.
However, many important aspects still have to be investigated and clarified in order to fully understand the role of EPS in BSCs. In our opinion, future studies should be focused in particular on investigating:
-
i)
the complex interplay between EPS producers and consumers in BSCs, defining the specific role of the microorganisms;
-
ii)
how the EPS affect the flux of nutrients (C, N, P and inorganic ions) in the EPM;
-
iii)
how the different EPS fractions interact with water molecules, affecting their movement within the crusts;
-
iv)
if and how the characteristics and the role of the EPS are affected by the different typologies of soils where BSC developed.
Answering these questions will provide a fundamental contribution to the understanding of the role of the EPS in the dynamic and complex habitat constituted by BSC but we are sure that at the same time it will also open new questions to be addresses in further studies.
References
Al-Thani RF (2015) Cyanomatrix and Cyanofilm. J Res Dev 3:2
Bates ST, Reddy GSN, Garcia Pichel F (2006) Exophiala crusticola anam. nov. (affinity Herpotrichiellaceae), a novel black yeast from biological soil crusts in the Western United States. Int J Syst Evol Microbiol 56:2697–2702. https://doi.org/10.1099/ijs.0.64332-0
Battiston KG, Labow RS, Simmons CA, Santerre JP (2015) Immunomodulatory polymeric scaffold enhances extracellular matrix production in cell co-cultures under dynamic mechanical stimulation. Acta Biomater 24:74–86. https://doi.org/10.1016/j.actbio.2015.05.038
Belnap J (2001) Biological soil crusts: function and management. US Department of Interior Technical Reference 1730–2
Belnap J (2003a) Biological soil crusts in deserts: a short review of their role in soil fertility, stabilization, and water relations. Algol Stud 109:113–126. https://doi.org/10.1127/1864-1318/2003/0109-0113
Belnap J (2003b) The world at your feet: desert biological soil crusts. Front Ecol Environ 1:181–189
Belnap J (1993) Recovery rates of cryptobiotic crusts: inoculant use and assessment methods. Great Basin Naturalist 53(1):89–95
Belnap J, Büdel B (2016) Biological soil crusts as soil stabilizers. In: Weber B, Büdel B, Belnap J (eds) Biological soil crusts: an organizing principle in drylands. Springer International Publishing, Cham, pp 305–320
Belnap J, Eldridge D (2001) Disturbance and recovery of biological soil crusts. In: Belnap J, Lange OL (eds) Biological soil crusts: structure, function, and management. Springer Verlag, Berlin, pp 363–383
Belnap J, Gardner JS (1993) Soil microstructure in soils of the Colorado Plateau: the role of the cyanobacterium Microcoleus vaginatus. West North Am Nat 53:40–47
Belnap J, Lange OL (eds) (2001) Biological soil crusts: structure, function, and management. Springer Verlag, Berlin, 503 pp
Bowker MA (2007) Biological soil crust rehabilitation in theory and practice: an underexploited opportunity. Restor Ecol 15(1):13–23
Bowker MA, Belnap J, Bala Chaudhary V, Johnson NC (2008) Revisiting classic water erosion models in drylands: The strong impact of biological soil crusts. Soil Biol Biochem 40:2309–2316. https://doi.org/10.1016/j.soilbio.2008.05.008
Bowker MA, Mau RL, Maestre FT et al (2011) Functional profiles reveal unique ecological roles of various biological soil crust organisms. Funct Ecol 25:787–795. https://doi.org/10.1111/j.1365-2435.2011.01835.x
Brüll LP, Huang Z, Thomas-Oates JE et al (2000) Studies of polysaccharides from three edible species of Nostoc (cyanobacteria) with different colony morphologies: structural characterization and effect on the complement system of polysaccharides from N. commune. J Phycol 36:871–881
Bu C, Wu S, Yang Y, Zheng M (2014) Identification of factors influencing the restoration of cyanobacteria-dominated biological soil crusts. PLoS One 9:e90049. https://doi.org/10.1371/journal.pone.0090049
Chen L, Xie Z, Hu C et al (2006) Man-made desert algal crusts as affected by environmental factors in Inner Mongolia, China. J Arid Environ 67:521–527. https://doi.org/10.1016/j.jaridenv.2006.02.018
Chen L, Rossi F, Deng S, Liu Y, Wang G, Adessi A, De Philippis R (2014) Macromolecular and chemical features of the excreted extracellular polysaccharides in induced biological soil crusts of different ages. Soil Biol Biochem 78:1–9. https://doi.org/10.1016/j.soilbio.2014.07.004
Chen B, Li F, Liu N et al (2015) Role of extracellular polymeric substances from Chlorella vulgaris in the removal of ammonium and orthophosphate under the stress of cadmium. Bioresour Technol 190:299–306. https://doi.org/10.1016/j.biortech.2015.04.080
Chenu C (1993) Clay- or sand-polysaccharide associations as models for the interface between micro-organisms and soil: water related properties and microstructure. Geoderma 56:143–156. https://doi.org/10.1016/0016-7061(93)90106-U
Christensen BE (1999) Physical and chemical properties of extracellular polysaccharides associated with biofilms and related systems. In: Wingender DJ, Neu DTR, Flemming PDH-C (eds) Microbial Extracellular Polymeric Substances. Springer Verlag, Berlin, pp 143–154
Colica G, Li H, Rossi F, Li D, Liu Y, De Philippis R (2014) Microbial secreted exopolysaccharides affect the hydrological behavior of induced biological soil crusts in desert sandy soils. Soil Biol Biochem 68:62–70. https://doi.org/10.1016/j.soilbio.2013.09.017
Colica G, Li H, Rossi F, De Philippis R, Liu Y (2015) Differentiation of the characteristics of excreted extracellular polysaccharides reveals the heterogeneous primary succession of induced biological soil crusts. J Appl Phycol 27:1935–1944. https://doi.org/10.1007/s10811-015-0532-6
Costerton JW, Cheng KJ, Geesey GG et al (1987) Bacterial biofilms in nature and disease. Annu Rev Microbiol 41:435–464. https://doi.org/10.1146/annurev.mi.41.100187.002251
Danin A, Dor I, Sandler A, Amit R (1998) Desert crust morphology and its relations to microbiotic succession at Mt. Sedom, Israel. J Arid Environ 38:161–174
de Alexandre SF, Pilarski F, Lemos MVF (2013) Composition of Extracellular Polymeric Substances (EPS) produced by Flavobacterium columnare isolated from tropical fish in Brazil. Braz J Microbiol 44:861–864. https://doi.org/10.1590/S1517-83822013005000058
de Paniagua-Michel JJ, Olmos-Soto J, Morales-Guerrero ER (2014) Chapter eleven-algal and microbial exopolysaccharides: new insights as biosurfactants and bioemulsifiers. In: Kim S-K (ed) Advances in food and nutrition research, vol 73, Academic Press, pp 221–257. https://doi.org/10.1016/B978-0-12-800268-1.00011-1
De Philippis R, Vincenzini M (1998) Exocellular polysaccharides from cyanobacteria and their possible applications. FEMS Microbiol Rev 22:151–175
De Philippis R, Sili C, Paperi R, Vincenzini M (2001) Exopolysaccharide-producing cyanobacteria and their possible exploitation: A review. J Appl Phycol 13:293–299. https://doi.org/10.1023/A:1017590425924
de Winder B, Stal LJ, Mur LR (1990) Crinalium epipsammum sp. nov.: a filamentous cyanobacterium with trichomes composed of elliptical cells and containing poly-β-(1,4) glucan (cellulose). Microbiology 136:1645–1653. https://doi.org/10.1099/00221287-136-8-1645
Decho AW, Lopez GR (1993) Exopolymer microenvironments of microbial flora: multiple and interactive effects on trophic relationships. Limnol Oceanogr 38:1633–1645
Dignac M-F, Urbain V, Rybacki D et al (1998) Chemical description of extracellular polymers: Implication on activated sludge floc structure. Water Sci Technol 38:45–53. https://doi.org/10.1016/S0273-1223(98)00676-3
Dubois M, Gilles KA, Hamilton JK et al (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Felde VJMNL, Rossi F, Colesie C et al (2016) Pore characteristics in biological soil crusts are independent of extracellular polymeric substances. Soil Biol Biochem 103:294–299. https://doi.org/10.1016/j.soilbio.2016.08.029
Fischer T (2009) Substantial rewetting phenomena on soil respiration can be observed at low water availability. Soil Biol Biochem 41:1577–1579. https://doi.org/10.1016/j.soilbio.2009.04.009
Fischer T, Veste M, Wiehe W, Lange P (2010) Water repellency and pore clogging at early successional stages of microbiotic crusts on inland dunes, Brandenburg, NE Germany. Catena 80:47–52. https://doi.org/10.1016/j.catena.2009.08.009
Fischer T, Veste M, Bens O, Hüttl RF (2012) Dew formation on the surface of biological soil crusts in central European sand ecosystems. Biogeosciences 9:4621–4628. https://doi.org/10.5194/bg-9-4621-2012
Fish KE, Osborn AM, Boxall J (2016) Characterising and understanding the impact of microbial biofilms and the extracellular polymeric substance (EPS) matrix in drinking water distribution systems. Environ Sci : Water Res Technol 2:614–630. https://doi.org/10.1039/C6EW00039H
Flemming H-C, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8:623–633. https://doi.org/10.1038/nrmicro2415
Garcia Pichel F, Castenholz RW (1991) Characterization and biological implications of Scytonemin, a cyanobacterial sheath pigment. J Phycol 27:395–409. https://doi.org/10.1111/j.0022-3646.1991.00395.x
Garcia Pichel F, Castenholz RW (1993) Occurrence of UV-Absorbing, Mycosporine-like compounds among cyanobacterial isolates and an estimate of their screening capacity. Appl Environ Microbiol 59:163–169
Garcia Pichel F, Wojciechowski MF (2009) The evolution of a capacity to build supra-cellular ropes enabled filamentous cyanobacteria to colonize highly erodible substrates. PLoS One 4:e7801. https://doi.org/10.1371/journal.pone.0007801
Garcia Pichel F, Felde VJMNL, Drahorad SL, Weber B (2016) Microstructure and weathering processes within biological soil crusts. In: Weber B, Büdel B, Belnap J (eds) Biological Soil Crusts: An Organizing Principle in Drylands. Springer International Publishing, Cham, pp 237–255
Groenendyk DG, Ferré TPA, Thorp KR, Rice AK, Singer AC (2015) Hydrologic-Process-Based Soil Texture Classifications for Improved visualization of landscape function. PLOS ONE 10(6):e0131299
Grotenhuis JTC, Smit M, van Lammeren AAM et al (1991) Localization and quantification of extracellular polymers in methanogenic granular sludge. Appl Microbiol Biotechnol 36:115–119. https://doi.org/10.1007/BF00164710
Gu C, Gao P, Yang F, An D, Munir M, Jia H, Xue G, Ma C (2017) Characterization of extracellular polymeric substances in biofilms under long-term exposure to ciprofloxacin antibiotic using fluorescence excitation-emission matrix and parallel factor analys. Environ Sci Pollut Res 24:13536–13545. https://doi.org/10.1007/s11356-017-8986-5
Harel Y (2004) Activation of photosynthesis and resistance to photoinhibition in Cyanobacteria within Biological Desert Crust. Plant Physiol 136:3070–3079. https://doi.org/10.1104/pp.104.047712
Hedges JI (1988) Polymerization of humic substances in natural environments. In: Frimmel FH, Christman FH (eds) Humic substances and their role in the environment. John Wiley & Sons, Chichester, pp 45-58
Hoiczyk E (1998) Structural and biochemical analysis of the sheath of Phormidium uncinatum. J Bacteriol 180:3923–3932
Hoiczyk E, Baumeister W (1998) The junctional pore complex, a prokaryotic secretion organelle, is the molecular motor underlying gliding motility in cyanobacteria. Curr Biol 8:1161–1168. https://doi.org/10.1016/S0960-9822(07)00487-3
Hoppert M, Reimer R, Kemmling A et al (2004) Structure and reactivity of a biological soil crust from a xeric sandy soil in Central Europe. Geomicrobiol J 21:183–191. https://doi.org/10.1080/01490450490275433
Hu C, Liu Y, Song L, Zhang D (2002) Effect of desert soil algae on the stabilization of fine sands. J Appl Phycol 14:281–292
Hu C, Liu Y, Paulsen BS et al (2003) Extracellular carbohydrate polymers from five desert soil algae with different cohesion in the stabilization of fine sand grain. Carbohydr Polym 54:33–42
Huang Z, Liu Y, Paulsen BS, Klaveness D (1998) Studies on polysaccharides from three edible species of Nostoc (Cyanobacteria) with different colony morphologies: comparison of monosaccharide compositions and viscosities of polysaccharides from field colonies and suspension cultures. J Phycol 34:962–968. https://doi.org/10.1046/j.1529-8817.1998.340962.x
Kepkay AFD (1994) Particle aggregation and biological reactivity of colloids. Mar Ecol Prog Ser 109:293–304
Kidron GJ, Yaalon DH, Vonshak A (1999) Two causes for runoff initiation on microbiotic crusts: hydrophobicity and pore clogging. Soil Sci 164(1):18–27
Kuske CR, Yeager CM, Johnson S, Ticknor LO, Belnap J (2012) Response and resilience of soil biocrust bacterial communities to chronic physical disturbance in arid shrublands. ISME J 6(4):886–897. https://doi.org/10.1038/ismej.2011.153
Lan S, Wu L, Zhang D, Hu C (2013) Assessing level of development and successional stages in Biological Soil Crusts with biological indicators. Microb Ecol 66:394–403. https://doi.org/10.1007/s00248-013-0191-6
Li P, Harding SE, Liu Z (2001) Cyanobacterial exopolysaccharides: their nature and potential biotechnological applications. Biotechnol Genet Eng Rev 18:375–404
Li H, Rao B, Wang G, Shen S, Li D, Hu C, Liu Y (2014) Spatial heterogeneity of cyanobacteria-inoculated sand dunes significantly influences artificial biological soil crusts in the Hopq Desert (China). Environ Earth Sci 71:245–253. https://doi.org/10.1007/s12665-013-2428-6
Limoli DH, Jones CJ, Wozniak DJ (2015) Bacterial Extracellular Polysaccharides in Biofilm Formation and Function. Microbiol Spectr 3(3):https://doi.org/10.1128/microbiolspec.MB-0011-2014. doi:https://doi.org/10.1128/microbiolspec.MB-0011-2014
Maestre FT, Bowker MA, Cantón Y et al (2011) Ecology and functional roles of biological soil crusts in semi-arid ecosystems of Spain. J Arid Environ 75:1282–1291. https://doi.org/10.1016/j.jaridenv.2010.12.008
Mager DM, Thomas AD (2010) Carbohydrates in cyanobacterial soil crusts as a source of carbon in the southwest Kalahari, Botswana. Soil Biol Biochem 42:313–318. https://doi.org/10.1016/j.soilbio.2009.11.009
Mager DM, Thomas AD (2011) Extracellular polysaccharides from cyanobacterial soil crusts: A review of their role in dryland soil processes. J Arid Environ 75:91–97. https://doi.org/10.1016/j.jaridenv.2010.10.001
Mahapatra S, Banerjee D (2013) Fungal Exopolysaccharide: production, composition and applications. Microbiol Insights 6:1–16. https://doi.org/10.4137/MBI.S10957
Malam Issa O, Défarge C, Trichet J et al (2009) Microbiotic soil crusts in the Sahel of Western Niger and their influence on soil porosity and water dynamics. Catena 77:48–55. https://doi.org/10.1016/j.catena.2008.12.013
Malam-Issa O, Défarge C, Le Bissonnais Y et al (2007) Effects of the inoculation of cyanobacteria on the microstructure and the structural stability of a tropical soil. Plant Soil 290:209–219
Martínez-Cánovas MJ, Quesada E, Martínez-Checa F et al (2004) Salipiger mucescens gen. nov., sp. nov., a moderately halophilic, exopolysaccharide-producing bacterium isolated from hypersaline soil, belonging to the alpha-Proteobacteria. Int J Syst Evol Microbiol 54:1735–1740. https://doi.org/10.1099/ijs.0.63166-0
May TB, Chakrabarty AM (1994) Isolation and assay of Pseudomonas aeruginosa alginate. Methods Enzymol 235:295–304
Mayer LM, Linda L. S, Sawyer T, et al (1995) Bioavailable amino acids in sediments: A biomimetic, kinetics based approach. Limnol Oceanogr 40:511–520. doi: https://doi.org/10.4319/lo.1995.40.3.0511
Mazor G, Kidron GJ, Vonshak A, Abeliovich A (1996) The role of cyanobacterial exopolysaccharides in structuring desert microbial crusts. FEMS Microbiol Ecol 21:121–130. https://doi.org/10.1111/j.1574-6941.1996.tb00339.x
Mba Medie F, Davies GJ, Drancourt M, Henrissat B (2012) Genome analyses highlight the different biological roles of cellulases. Nat Rev Microbiol 10:227–234. https://doi.org/10.1038/nrmicro2729
Miralles I, Domingo F, Cantón Y et al (2012) Hydrolase enzyme activities in a successional gradient of biological soil crusts in arid and semi-arid zones. Soil Biol Biochem 53:124–132. https://doi.org/10.1016/j.soilbio.2012.05.016
Mugnai G, Ventura S, Mascalchi C, Rossi F, Adessi A, De Philippis R (2015) Biological soil crusts from Arctic environments: characterization of the prokaryotic community and exopolymeric matrix analysis. European Geoscience Union 2015 (EGU-2015). doi: 10.13140/RG.2.2.33276.74888
Mugnai G, Rossi F, Felde VJMNL, Colesie C, Büdel B, Peth S, Kaplan A, De Philippis R (2017) Development of the polysaccharidic matrix in biocrusts induced by a cyanobacterium inoculated in sand microcosm. Biol Fert Soils (in press). https://doi.org/10.1007/s00374-017-1234-9
Nagy ML, Pérez A, Garcia Pichel F (2005) The prokaryotic diversity of biological soil crusts in the Sonoran Desert (Organ Pipe Cactus National Monument, AZ). FEMS Microbiol Ecol 54:233–245. https://doi.org/10.1016/j.femsec.2005.03.011
Nielsen PH, Jahn A (1999) Extraction of EPS. In: Wingender DJ, Neu DTR, Flemming PDH-C (eds) Microbial Extracellular Polymeric Substances. Springer, Berlin Heidelberg, pp 49–72
Nielsen PH, Jahn A, Palmgren R (1997) Conceptual model for production and composition of exopolymers in biofilms. Water Sci Technol 36:11–19
Osińska-Jaroszuk M, Jarosz-Wilkołazka A, Jaroszuk-Ściseł J et al (2015) Extracellular polysaccharides from Ascomycota and Basidiomycota: production conditions, biochemical characteristics, and biological properties. World J Microbiol Biotechnol 31:1823–1844. https://doi.org/10.1007/s11274-015-1937-8
Pelkonen S, Häyrinen J, Finne J (1988) Polyacrylamide gel electrophoresis of the capsular polysaccharides of Escherichia coli K1 and other bacteria. J Bacteriol 170:2646–2653
Pereira S, Zille A, Micheletti E et al (2009) Complexity of cyanobacterial exopolysaccharides: composition, structures, inducing factors and putative genes involved in their biosynthesis and assembly. FEMS Microbiol Rev 33:917–941. https://doi.org/10.1111/j.1574-6976.2009.00183.x
Platt RM, Geesey GG, Davis JD, White DC (1985) Isolation and partial chemical analysis of firmly bound exopolysaccharide from adherent cells of a freshwater sediment bacterium. Can J Microbiol 31:675–680
Pointing SB, Belnap J (2012) Microbial colonization and controls in dryland systems. Nat Rev Microbiol 10:551–562. https://doi.org/10.1038/nrmicro2831
Rajeev L, Da Rocha UN, Klitgord N et al (2013) Dynamic cyanobacterial response to hydration and dehydration in a desert biological soil crust. ISME J 7:2178–2191
Redmile-Gordon MA, Brookes PC, Evershed RP, Goulding KWT, Hirsch PR (2014) Measuring the soil-microbial interface: extraction of extracellular polymeric substances (EPS) from soil biofilms. Soil Biol Biochem 72:163–171
Roberts IS (1996) The biochemistry and genetics of capsular polysaccharide production in bacteria. Annu Rev Microbiol 50:285–315. https://doi.org/10.1146/annurev.micro.50.1.285
Rossi F, De Philippis R (2015a) Role of cyanobacterial Exopolysaccharides in phototrophic biofilms and in complex microbial mats. Life 5:1218–1238. https://doi.org/10.3390/life5021218
Rossi F, De Philippis R (2015b) Exocellular polysaccharides in microalgae and cyanobacteria: chemical features, roles and enzymes and genes involved in their biosynthesis. In: Borowitzka M, Beardall J, Raven JA (eds) The Physiology of microalgae. Springer, Berlin, pp 565–590
Rossi F, Micheletti E, Bruno L et al (2012a) Characteristics and role of the exocellular polysaccharides produced by five cyanobacteria isolated from phototrophic biofilms growing on stone monuments. Biofouling 28:215–224
Rossi F, Mugnai G, Colica G, Ventura S, Sili C, Mascalchi C, De Philippis R (2012b) Exopolysaccharidic matrix of biological soil crusts from arctic environments. Environ Engin Manag J 11:S162
Rossi F, Potrafka RM, Garcia Pichel F, De Philippis R (2012c) The role of the exopolysaccharides in enhancing hydraulic conductivity of biological soil crusts. Soil Biol Biochem 46:33–40. https://doi.org/10.1016/j.soilbio.2011.10.016
Sabra W, Zeng AP, Lunsdorf H, Deckwer WD (2000) Effect of oxygen on formation and structure of Azotobacter vinelandii and its role in protecting nitrogenase. Appl Environ Microbiol 66:4037–4044
Sato T, Ose Y (1980) Floc-forming substances extracted from activated sludge by sodium hydroxide solution. Water Res 14:333–338. https://doi.org/10.1016/0043-1354(80)90080-9
Selbmann L, Stingele F, Petruccioli M (2003) Exopolysaccharide production by filamentous fungi: the example of Botryosphaeria rhodina. Antonie Van Leeuwenhoek 84:135–145. https://doi.org/10.1023/A:1025421401536
Seviour RJ, Stasinopoulos SJ, Auer DPF, Gibbs PA (1992) Production of Pullulan and other exopolysaccharides by filamentous fungi. Crit Rev Biotechnol 12:279–298. https://doi.org/10.3109/07388559209069196
Stuart RK, Mayali X, Lee JZ et al (2016) Cyanobacterial reuse of extracellular organic carbon in microbial mats. ISME J 10:1240–1251. https://doi.org/10.1038/ismej.2015.180
Suela Silva M, Naves Sales A, Teixeira Magalhães-Guedes K et al (2013) Brazilian Cerrado soil Actinobacteria ecology. Biomed Res Int 2013:1–10. https://doi.org/10.1155/2013/503805
Sutherland IW (1994) Structure-function relationships in microbial exopolysaccharides. Biotechnol Adv 12:393–448
Sutherland IW (1999) Biofilm Exopolysaccharides. In: Wingender DJ, Neu DTR, Flemming PDH-C (eds) Microbial Extracellular Polymeric Substances. Springer, Berlin Heidelberg, pp 73–92
Tamaru Y, Takani Y, Yoshida T, Sakamoto T (2005) Crucial role of extracellular polysaccharides in desiccation and freezing tolerance in the terrestrial cyanobacterium Nostoc commune. Appl Environ Microbiol 71:7327–7333. https://doi.org/10.1128/AEM.71.11.7327-7333.2005
Thomas AD, Hoon SR (2010) Carbon dioxide fluxes from biologically-crusted Kalahari Sands after simulated wetting. J Arid Environ 74:131–139. https://doi.org/10.1016/j.jaridenv.2009.07.005
Thomas AD, Hoon SR, Linton PE (2008) Carbon dioxide fluxes from cyanobacteria crusted soils in the Kalahari. Appl Soil Ecol 39:254–263
Underwood GJC, Paterson DM, Parkes RJ (1995) The measurement of microbial carbohydrate exopolymers from intertidal sediments. Limnol Oceanogr 40:1243–1253
Varin T, Lovejoy C, Jungblut AD et al (2012) Metagenomic analysis of stress genes in microbial mat communities from Antarctica and the High Arctic. Appl Environ Microbiol 78:549–559. https://doi.org/10.1128/AEM.06354-11
Vincent P, Pignet P, Talmont F, Bozzi L, Fournet B, Guezennec J, Jeanthon C, Prieur D (1994) Production and characterization of an exopolysaccharide excreted by a deep-sea hydrothermal vent bacterium isolated from the polychaete annelid Alvinella pompejana. Appl Environ Microbiol 60(11):4134–4141
Wang W, Liu Y, Li D et al (2009) Feasibility of cyanobacterial inoculation for biological soil crusts formation in desert area. Soil Biol Biochem 41:926–929. https://doi.org/10.1016/j.soilbio.2008.07.001
Weber B, Wu D, Tamm A et al (2015) Biological soil crusts accelerate the nitrogen cycle through large NO and HONO emissions in drylands. Proc Natl Acad Sci 112:15384–15389. https://doi.org/10.1073/pnas.1515818112
Welch SA, Vandevivere P (1994) Effect of microbial and other naturally occurring polymers on mineral dissolution. Geomicrobiol J 12:227–238. https://doi.org/10.1080/01490459409377991
Williams AJ, Buck BJ, Beyene MA (2012) Biological soil crusts in the Mojave Desert, USA: micromorphology and pedogenesis. Soil Sci Soc Am J 76(5):1685
Wingender J, Neu TR, Flemming H-C (1999) What are bacterial extracellular polymeric substances? In: Wingender DJ, Neu DTR, Flemming PDH-C (eds) Microbial Extracellular Polymeric Substances. Springer, Berlin Heidelberg, pp 1–19
Wolfaardt GM, Lawrence JR, Korber DR (1999) Function of EPS. In: Wingender DJ, Neu DTR, Flemming PDH-C (eds) Microbial Extracellular Polymeric Substances. Springer, Berlin Heidelberg, pp 171–200
Wu Y, Rao B, Wu P et al (2013) Development of artificially induced biological soil crusts in fields and their effects on top soil. Plant Soil 370:115–124. https://doi.org/10.1007/s11104-013-1611-6
Wu N, Zhang YM, Pan HX, Zhang J (2010) The role of nonphotosynthetic microbes in the recovery of biological soil crusts in the gurbantunggut desert, Northwestern China. Arid Land Res Manag 24(1):42–56
Xiao R, Zheng Y (2016) Overview of microalgal extracellular polymeric substances (EPS) and their applications. Biotechnol Adv 34:1225–1244. https://doi.org/10.1016/j.biotechadv.2016.08.004
Xu Y, Rossi F, Colica G et al (2012) Use of cyanobacterial polysaccharides to promote shrub performances in desert soils: a potential approach for the restoration of desertified areas. Biol Fertil Soils 49:143–152. https://doi.org/10.1007/s00374-012-0707-0
Zhang Y (2005) The microstructure and formation of biological soil crusts in their early developmental stage. Chin Sci Bull 50:117. https://doi.org/10.1360/982004-559
Zhang Y, Wang HL, Wang XD, Yang WK, Zhang DY (2006) The microstructure of microbiotic crust and its influence on wind erosion for a sandy soil surface in the Gurbantunggut Desert of Northwestern China. Geoderma 132:441–449. https://doi.org/10.1016/j.geoderma.2005.06.008
Zhang Y, Aradottir AL, Serpe M, Boeken B (2016) Interactions of Biological Soil Crusts with vascular plants. In: Weber B, Büdel B, Belnap J (eds) Biological soil crusts: an organizing principle in drylands. Springer International Publishing, Cham, pp 385–406. https://doi.org/10.1007/978-3-319-30214-0_19
Acknowledgements
The authors wish to acknowledge the Editor Matthew Bowker and the two anonymous Reviewers for their helpful comments to the manuscript, which contributed to largely improve its final version.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Matthew A. Bowker.
Rights and permissions
About this article
Cite this article
Rossi, F., Mugnai, G. & De Philippis, R. Complex role of the polymeric matrix in biological soil crusts. Plant Soil 429, 19–34 (2018). https://doi.org/10.1007/s11104-017-3441-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3441-4