Abstract
Aims
Elemental stoichiometry of soil is a useful indicator of nutrient constraints. The aim of this study is to assess the relative contributions of local-scale determinants to the soil carbon/nitrogen ratio (C:N) and nitrogen/phosphorus ratio (N:P) in a 25 ha temperate forest plot.
Methods
Leaf litter biomass at 967 soil sampling points was predicted using mapped tree distribution and leaf litter data (150 traps) for seven tree species. Boosted regression trees (BRT) were used to examine the relative contribution of local-scale variables to key elemental stoichiometry of soil. These variables included leaf litter input, neighbor species composition, soil pH and moisture, and topography.
Results
Both biotic and abiotic variables influenced soil C:N and N:P ratios, but abiotic factors had a greater influence. Soil pH was the most important predictor of soil C:N and N:P with a strong positive correlation. Topography and soil moisture explained more of the local-scale variability of soil C:N and N:P ratios than tree composition and leaf litter inputs.
Conclusions
Our results suggest soil pH was a better predictor of local-scale spatial variability of soil elemental stoichiometry than tree composition and leaf litter inputs in temperate forests. These results improve our understanding of the mechanisms underlying the heterogeneity of soil nutrients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Carbon (C), nitrogen (N) and phosphorus (P) are key elements to living organisms, and their stoichiometry (C:N and N:P ratio) is a useful indicators of nutrient limitation in both terrestrial and aquatic ecosystems (Elser et al. 2007; Gundersen et al. 2009; Bui and Henderson 2013; Cools et al. 2014), and ecosystem functioning. The influence of plant-soil feedback on soil biogeochemical heterogeneity is well documented (Finzi et al. 1998; Augusto et al. 2002; Aponte et al. 2013; Vesterdal et al. 2013). In forest ecosystems, the dominant nutrient fluxes, including litter inputs, nutrient leaching, and nutrient uptake by trees, depends largely on species composition and the size of canopy trees. These factors drive local heterogeneity of soil biogeochemistry (Finzi et al. 1998; Gomez-Aparicio and Canham 2008; Keller et al. 2013; Morhart et al. 2016). However, much of the previous research has focused on isolating effects of one or a few tree species on soil biogeochemistry properties. Consequently, the effects of plant diversity and composition on ecosystem biogeochemistry are largely unknown. This information would provide insight into potential mechanisms of species coexistence and serve as a guide for forest management in the face of climate change.
Leaf litter is a key linkage between tree diversity, forest biomass, and belowground biogeochemical processes. Differences in the quantity, quality (decomposability and nutrient content), and diversity of leaf litter inputs have shown to significantly affect the spatial variation in soil physical properties and nutrient availability (Finzi et al. 1998; Gomez-Aparicio and Canham 2008; Xu et al. 2013; Uriarte et al. 2015; Xia et al. 2015). A recent meta-analysis on 70 litter-manipulation experiments found that greater litter inputs increases soil C storage, but did not affect total N, extractable inorganic N or extractable P in mineral soil, and consequently, increases C:N ratios (Xu et al. 2013). However, field litter-manipulation experiments usually focus on very few species and little is known about how changes in aboveground litter inputs of multi-species affect soil C and nutrient heterogeneity at the local scale (20–50 ha). Together with data on inter-specific variation of leaf nutrients, plot-based studies on the distribution of leaf litterfall provide a powerful tool for understanding plant-soil feedbacks (Gomez-Aparicio and Canham 2008; Uriarte et al. 2015; Xia et al. 2015). Recently, in tropical and subtropical forests, fine-scale (<1 ha) and local-scale spatial variability of soil nutrients ratios have been found to be positively associated with leaf litter production and nutrient fluxes using inverse modeling methods (Qiao et al. 2014; Uriarte et al. 2015; Xia et al. 2015). A recent study by Uriarte et al. (2015) indicated that leaf litter inputs had varying effects soil chemical properties (e.g. soil C:N and N:P ratios) and soil physical characteristics (such as topography and rock contents). In temperate forests, lower leaf biomass allocation and higher root biomass allocation have been found compared to tropical forests (Jackson et al. 1996; Mokany et al. 2006; Poorter et al. 2012). The differences in foliar nutrient content among species in temperate forests are smaller than in tropical forests (Townsend et al. 2008; Yuan and Chen 2009). How these differences influence biogeochemical properties remain largely unknown. Therefore, there is a need to understand the importance of leaf litter inputs on the heterogeneity of soil nutrient ratios, especially in mixed temperate forests.
The heterogeneity of soil C, N and P content and their stoichiometry are also affected by abiotic factors, such as topography, soil pH, and soil moisture (Seibert et al. 2007; Wang et al. 2007; Yuan et al. 2013; Uriarte et al. 2015). Many previous studies have been demonstrated the importance of topographic position (Yoo et al. 2006; Riveros-Iregui and McGlynn 2009). For instance, soil N:P ratios were greater in steeper (slopes >15 % slope) than in flat areas (slopes <15 % slope) in a tropical forest (Uriarte et al. 2015). An additional study found that the C:N ratio decreased in upslope areas (Seibert et al. 2007). While in a temperate grassland slope-bottoms had higher soil C, and N contents than adjacent up-slope areas (Frank 2008). Soil pH and moisture have direct and indirect effects on soil C, N, P content and their stoichiometry by regulating organic matter decomposition and nutrient loss. Increasing soil pH has been shown to enhance microbial activity, soil respiration and denitrification rates, resulting in effluxes of C and N (Kemmitt et al. 2006; Pietri and Brookes 2008; Weslien et al. 2009; Cuhel et al. 2010). In contrast, at steady state, soil pH had no significant effect on total soil C and N content, and soil C:N ratios (Kemmitt et al. 2006). Increasing soil moisture has also been shown to increase C:N ratios, but not N:P ratios (Uriarte et al. 2015). There is substantial evidence suggesting that the heterogeneity of soil nutrient ratios is influenced by local abiotic and aboveground biotic factors, the relative importance of these factors (especially leaf litter inputs) is still unclear due to their complex interactions.
Korean pine–broadleaf mixed forests located in northeastern China have high species richness and unique species composition among temperate forests (Yang and Xu 2003; Stone 2006; Hao et al. 2007). Understanding multi-species effects on the key belowground biogeochemical properties in this particular mixed temperate forest remains poor. In this study, we aim to investigate factors determining soil C:N and N:P ratios at a local scale using mapped trees and leaf litterfall data of seven dominant tree species, and soil chemical data in an old-growth temperate forest. We attempt to answer the following questions: 1. How does the leaf litterfall of dominant tree species vary in biomass, C, N, P content, stoichiometric ratios, and spatial deposition patterns? 2. To what extent and how is the heterogeneity of soil stoichiometry influenced by local biotic (leaf litter inputs and tree composition) and abiotic factors (topography, soil moisture and pH)?
Methods
Study site
This study was conducted in a 25 ha (500 m × 500 m) permanent forest dynamics plot in the Changbaishan (CBS) Nature Reserve in northeastern China (42°23′N, 128°05′E). The reserve is one of the largest biosphere reserves in China, located near the border with North Korea and has not experienced logging or other human disturbance for at least 300 years. The mean elevation of the CBS temperate forest plot is 801.5 m ranging from 791.8 to 809.5 m. The mean annual temperature is 3.6 °C, and the mean annual precipitation is approximately 700 mm. The forest type is a mixed broad-leaved and conifer temperate forest. The soil is classified as dark brown forest soil (Mollisol according to U.S. soil Taxonomy Series, 1999), for which the parent material is granite and basalt.
The CBS plot was established in 2004. Censuses are conducted every five years following protocols of Center for Tropical Forest Science (CTFS). Data from the 2009 tree census was used in this study. All free-standing trees at least 1 cm in diameter at breast height (DBH ≥ 1) from the ground were tagged, measured and identified to species, and their geographic coordinates were recorded following a standard field protocol (Condit 1998). The second census of 2009 recorded 34,926 individuals belonging to 51 species, 31 genera and 18 families (Wang et al. 2011). Seven dominant tree species were selected in this study: Pinus koraiensis, Tilla amurensis, Quercus mongolica, Fraxinus mandshurica, Ulmus japonica, Acer mono, and Acer pseudo-sieboldianum. These seven species accounted for nearly 88 % of adult stems (DBH ≥ 10 cm) and 91 % of leaf litter that fell into leaf litter traps.
Leaf litter collection and chemical analyses
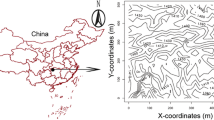
This work was conducted based on Forestry Standards “Observation Methodology for Long-term Forest Ecosystem Research” of People’s Republic of China (LY/T 1952-2011). To assess the spatial variation of leaf litterfall in the CBS plot, 150 litterfall traps were installed in June 2005 (Fig. 1). The traps were relatively evenly distributed throughout the plot. The traps were arranged using a combination of typical grids and circular plots, which limited the maximum distance (31 m) between trees and the nearest litter trap (Li et al. 2012). The average distance of adjacent traps was 38.3 m with a range of 28.3 to 44.7 m. Each trap had a surface area of 0.56 m2 (0.75 m × 0.75 m) and was constructed using fine-flexible 1 mm mesh and supported by four PVC tubes of 1 m above the ground. Litterfall was collected from the traps twice a month from May to Dec and once a month from Jan. to Apr each year. Leaf litter was sorted to species, oven dried and weighed. Leaf litter constituted 71.3 % of total litter mass. We used leaf litter collection data from 2009 for our study. From September to October in 2009, leaf litter from at least 10 traps for each of the seven study species was dried, weighed and ground using a mixer mill (Retsch MM400) for chemical analysis. For the analyses of C and N, 80 mg of the dried samples was weighed to the nearest 0.1 mg, and total C and N concentrations were determined using an elemental analyzer (vario MACRO cube, Elementar Corporation, Hanau, Germany). Total P was determined by acid digestion using HNO3-HClO4 solution and measured using an inductively coupled plasma mass spectrometer (ICP-MS) (Agilent 7500a, Agilent Technologies, Melbourne, Australia).
Distribution pattern of soil sampling points, litterfall traps and contour map of the 25 ha Changbai temperate plot. (a) Black dots represent litterfall traps; (b) red triangles represent soil sample points; green, blue and yellow colors represent low-plateau (slope < 7°, elevation <804.0 m), slope (slope < 7°), and high-plateau (slope < 7°, elevation >804.0 m), respectively
Soil sample collection and chemical analyses
From 1st to the 4th of October 2007, during a dry fall growing season, soil samples were collected based on a regular grid of points every 30 m (John et al. 2007). Two sample points were selected from 2, 5, or 15 m in a randomly assigned compass point direction from the grid point. Obstructions including fallen trees (logs) or big tree roots were avoided or discarded, resulting in 967 samples (Fig. 1) (Yuan et al. 2013). Within 0.2 m of each sample point, three topsoil (0–10 cm) subsamples were collected using a 5 cm diameter soil corer after removing large debris. The sub-samples were then mixed to give a representative sample for that grid point. Soil moisture had a clear gradient pattern with higher values occurring in the west of the CBS plot (Yuan et al. 2013). The volumetric water content (%) of the 0–20 cm soil layer was measured at sample locations using a time domain reflectometer probe (FieldScout TDR200, Spectrum Technologies, Inc. Aurora, IL). All soil samples were air-dried under shade, then ground and sieved. For each soil sample, one half was sieved through 2-mm mesh (for pH measurement) and the other half though 0.149 mm mesh (for total N, total P and total C analyses). Soil pH and total C, N and P were analyzed according to Sparks (Sparks et al. 1996). Soil pH was measured in the slurry (soil: water =2.5:1) using a pH meter (FE20-K, Mettler-Toledo Instruments, Shanghai, China). Total C was measured by the acidified dichromate (K2Cr2O7-H2SO4) oxidation method. Total N was determined by digesting samples in H2SO4-HClO4 acid and then measured using the Kjeldahl method. Total P was determined by molybdenum antimony blue colorimetry after digestion using a H2SO4-HClO4 acid solution.
Species composition data
The size and identity of the canopy trees strongly affects ecosystem processes at the 0–25 m scale (Finzi et al. 1998; Gomez-Aparicio and Canham 2008; Xia et al. 2015). In our study, P. koraiensis (a coniferous species) was one of the seven dominant species. Thereby, we used the relative basal area (BA) of Pinus koraiensis, the total BA, stem density, and species richness of all stems ≥1 cm DBH within 25 m radius of each soil sample location to represent species composition.
Topographical data
The elevation of each 20 m × 20 m quadrat was taken as the mean of the elevation at the four corners. Convexity and slope values were calculated for each quadrat. Three habitat types were classified from this data: low plateau (slopes <7° and elevation <804.0 m), slope (slopes >7°) and high plateau (slopes <7°and elevation >804.0 m).
Data analyses
The model of Ferrari and Sugita (1996) was used to predict leaf litterfall mass (LLF) as a function of the DBH of the seven dominant tree species and their distance from the litter traps. For each the seven species, we ran pairwise correlations between the leaf litter biomass of any two years from 2008 to 2013. We found there were high correlations between any two-year combinations for all seven species (Supplementary Table 3), indicating the collection by leaf litterfall in traps was very similar each year. We used the CBS census data and litterfall data collected in 2009 to estimate parameters of leaf litter dispersal. Specifically, we used the number, tree size (DBH) and distance of all DBH equal to or exceeding 2.5 cm of source trees within a 25 m radius of a given litterfall trap to estimate species-specific litterfall mass. The LLF in traps was estimated using the following model (Ferrari and Sugita 1996):
Where LLF i (g) is the total leaf litterfall mass collected in trap i, DBH ij (cm) is the diameter at breast height of tree j associated with trap i, r ij (m) is the distance of tree j from the litterfall trap i, parameters a and b specify the increase of leaf biomass with increasing tree diameter. c0 is a parameter determining the shape of the leaf fall shadow, and η is a normalization constant.
The traps within 30 m of the plot edge were omitted to avoid edge effects and to optimize model parameters. Data from a total of 126 traps were used in the analyses. Data were divided into a calibration (n = 76) and a validation set (n = 50). Predicted leaf litterfall biomass was calculated for the validation set using parameter estimates from the calibration data only. Then, leaf litter dispersal models were optimized using the whole data set (n = 126). Parameters of the litterfall models were estimated for the seven species using the maximum likelihood method. Residual errors between observed and predicted leaf litter weights approximated a normal distribution. Interval estimation was used to measure the significance of the parameters. Model quality was evaluated by Pearson correlation coefficient (r), mean error of prediction (ME), and root mean square error of prediction (RMSE). The parametric paired t-test was used to examine whether means of measured and predicted data were significantly different. Leaf litter distribution was simulated across the whole plot for each of the seven target species based on the estimated species-specific parameters together with mapped tree data in 2009, and C, N, and P data in species-specific leaf litter. C:N and N:P ratios at a given location were calculated as the sum of C or N contributions from seven the tree species over the sum of all N or P contributions. For example N:P ratio at location i was calculated as:
Where N j and P j are the N and P content of the tree species j. leaf litter mass ij is leaf litter biomass of the species j at the location i.
All above data analyses were conducted in the R-package ‘base’ (R Development Core Team 2014).
All predictor variables were used in the boosted regression tree (BRT) models to estimate the C:N and N:P ratios in the soil (Table 1). First, we ran simple Pearson correlations to examine covariation among predictor variables (Supplementary Table 4). Variables were removed if the correlation coefficient exceeded a threshold of 0.7 (Dormann et al. 2013). Then, we used BRT models to examine the relative influence of biotic and abiotic variables on soil C:N and N:P ratios. The BRT models were built using the R- package ‘gbm’ (Version 2.1.1 Ridgeway 2010) and the custom functions developed by Elith et al. (2008). Models were fitted using the gbm. Step function and a Gaussian response type (aimed at minimizing squared error). The most effective settings for learning rate were in the range of 0.001–0.005 and the bag fraction was set at 0.75 (which specifies the proportion of data randomly drawn at each iteration). Tree complexity (the number of nodes in a tree) was set to 5, according to recommendations by Elith et al. (2008) for large datasets.
To assess the structure of spatial autocorrelation (SAC) in the data we calculated Moran’s I values of BRT model residuals. The residual correlograms of BRT models were plotted using the package spdep (Bivand 2015). Moran’s I ranges from 1 to −1, with zero indicating a random pattern with no SAC. If BRT models residuals had strong SAC, we adopted a residual autocovariate (RAC) model (Crase et al. 2012). We calculated a RAC layer for each model to get SAC for a first-order neighborhood size (3 × 3 cells). This RAC layer was then used as a predictor in the corresponding BRT model. To assess the reduction in residual SAC, the correlograms of RAC-BRT residuals were also plotted.
The spatial distribution of soil C:N, N:P ratio, soil pH and soil moisture within an area of 470 m × 470 m were interpolated using ordinary kriging in the geoR package and mapped (Ribeiro and Diggle 2001). First, the Box-Cox transformation was used to improve data normality. Second, the spatial trends of variables were checked and removed to make data stationary. Third, the semi-variogram models and the parameters of semi-variogram models were fitted. Finally, values for the grids that were constituted by the coordinates of soil sampling points were predicted for all variables using krige. Conv function. All analyses were conducted using the R statistical software version 3.1.2 (R Core Team 2014).
Results
Tree species and leaf litter properties
Population structure, the quantity and the quality of leaf litter varied substantially among the seven dominant tree species. The abundance, maximum DBH, and mean DBH of the seven target species had significant differences (Table 2). P. koraiensis had the highest proportion of adult stems in this plot. Leaf litter of each species was measured in at least 90 % of the litterfall traps, but leaf litter mass by species varied substantially among 150 litterfall traps, with coefficient of variations (CVs) ranging from 67.7 to 145.9 %. Among the seven species, the P. koraiensis leaf litter had the highest C concentration and lowest N concentration, and consequently, highest C:N and lowest N:P ratios. In contrast, T. amurensis had the lowest C:N ratios, the highest N and P concentrations (Table 2).
Prediction and spatial distribution patterns of leaf litter and soil
The leaf litterfall model parameters derived from our plot were species dependent (Supplementary Table 5). P. koraiensis and A. pseudo-sieboldianum have the highest c0, indicating that dispersal of leaf litter away from source trees is not far for these two species. For each of the seven species, the leaf litter dispersal models exhibited a good fit to the data. The model predictions of leaf litter are highly correlated with actual leaf litterfall measured in the validation set data (Supplementary Fig. 4, r = 0.79–0.89). The best predictions were for P. koraiensis accounting for up to 79 % of the observed variation in leaf litter. The least accurate predictions were for A. mono, which accounted for 62 % of the observed variation. RMSE and ME were low for all species models, and measured and predicted total LLF were not significantly different (P < 0.05) (Supplementary Table 5). Composite C:N ratios of leaf litter inputs across the plot ranged from 31.4 to 59.4 (mean = 40.3 ± 5.3 SD) and N:P ratios from 15.1 to 29.5 (mean = 20.7 ± 2.6 SD).
The nugget/sill (C0/C) for C:N ratio, N:P ratio, pH values and moisture in soil were 43.5 %, 68.7 %, 61.9 % and 62.8 %, respectively (Supplementary Fig. 5a-d). The kriged maps of C:N ratio, N:P ratio, pH values and moisture in soil across the whole plot are given in Fig. 2. Predicted C:N ratios (Fig. 2a) and N:P ratios (Fig. 2b) in leaf litter were higher than soil C:N ratios (Fig. 2c) and N:P ratios (Fig. 2d), respectively, and their spatial distributions were non-synchronous. The maps of soil pH had a similar distribution to soil N:P ratios. Soil C:N and N:P ratios exhibited smaller variation than leaf litter, with an averages of 14.8 ± 2.1 and 5.1 ± 0.7, respectively (Fig. 2).
Predicted spatial distribution of (a) leaf litter C:N ratio, (b) soil C:N ratio, (c) leaf litter N:P ratio, (d) soil N:P ratio, (e) soil pH and (f) soil moisture in the 25 ha CBS plot based on the 2009 tree census data and estimated dispersal kernels (Supplementary Table 5). White indicates values higher than the median, and green color means values less than the median
Spatial variation associations in soil C:N and N:P ratios
The residual correlograms from the BRT models and the RAC-BRT models showed that inclusion of RAC as a predictor in the model substantially reduced first-order spatial autocorrelation in the model residuals (Supplementary Fig. 6). All of the predictor variables in our BRT models explained 18.1 % and 23.5 % of the spatial variation in soil C:N (Fig. 3a) and N:P ratios (Fig. 3b) respectively, after accounting for spatial autocorrelation effects. The relative contribution of biotic and abiotic factors to soil C:N ratio was 8.4 % and 9.7 %, respectively. The relative contribution of biotic and abiotic factors to soil N:P ratio was 4.7 % and 18.8 %, respectively. Among all predictors, soil pH was the most important predictor, explaining 4.4 % and 12.1 % of the variation in soil C:N ratio and N:P ratios, respectively. Positive relationship between C:N and N:P ratios and soil pH were observed when pH ranged from 5.5 to 6.5. For soil C:N ratios, topography was the second most important predictor, explaining about 4 % of the variation in soil C:N ratios. Soil C:N ratios were high in low plateau areas, intermediate on slopes, and low soil C:N ratios in high plateau areas. For soil N:P ratios, soil moisture was the second most important explanatory factor. Soil moisture had a negative effect on soil N:P ratio and contributed to 6.6 % of the variation. The soil C:N ratio increased with increasing Pinus BA ratio, but there were only weak relationships between soil C:N and N:P ratios and variables related to leaf litter inputs and species composition within a radius of 25 m of the soil sampling locations.
Partial dependence plots of the influential variables in the BRT model of (a) soil C:N and (b) N:P ratio. RAC = residuals autocovariate. For other explanation of variables and their units see Table 1. Y axes are on a logarithmic scale and are centred to have zero mean over the data distribution. The values (%) in brackets are the relative contribution of each predictor variable. Rug plots at inside bottom of plots show the distribution of a value from a sample site across that variable, in deciles
Discussion
Variations in the stoichiometry of leaf litter
The seven dominant tree species varied substantially in population structure and the quantity and quality of leaf litter (Table 1). The spatial distribution of dominant species varied with different DBH and height classes (Hao et al. 2007), which contributed to spatial heterogeneity of leaf litter biomass inputs across the 150 leaf litter collection traps (Table 1). Consistent with the findings in previous studies (Augusto et al. 2002; Yang and Luo 2011; Cools et al. 2014), we found that leaf litter of P. koraiensis (the coniferous species) had higher C:N and lower N:P ratios than other broadleaf species. As coniferous P. koraiensis was the dominant species in this study, the average values of leaf litter C:N (40.3) and N:P ratios (20.7) in our study area were lower and higher, respectively, compared to C:N (55.9) and N:P (17.6) ratios of leaf litter in temperate forests from a global analysis (Yuan and Chen 2009). Leaf litter C:N and N:P ratios exhibited little difference among broadleaf species.
Spatial patterns of stoichiometry of leaf litter and soil
Nutrient ratios maps of soil and leaf litter are used to visualize nutrient distribution and limitations. As leaf litter is one of most important source of soil C and nutrients, soil nutrient ratios maps should correspond with leaf litter maps. However, the C:N:P stoichiometric ratios in leaf litter and soil showed little correspondence in our study (Fig. 2). This could be because litter C:N:P ratios converged towards relatively stable stoichiometric ratios during decomposition (Manzoni et al. 2010). This suggests that local environmental factors (e.g. soil pH and moisture) that determine organic matter decomposition rates may contribute to little correspondence of nutrient ratios between soil and leaf litter. Although Ferrari and Sugita’s model was powerful in the CBS temperate forest (Supplementary Fig. 4), and has been used successfully in other temperate forests (Ferrari and Sugita 1996; Staelens et al. 2004), the leaf litter dispersal model could not reproduce observed leaf litter deposition patterns (Staelens et al. 2004; Jonard et al. 2006). This may also lead to little correspondence in stoichiometric ratios between soil and leaf litter.
Effects of biotic and abiotic factors on soil C:N and N:P ratios
Soil pH is an important determinant of microbial community composition and activity in forest ecosystems (Hogberg et al. 2007; Li et al. 2014; Shen et al. 2014; Tripathi et al. 2015), which in turn affects many ecological processes such as microbial carbon-use efficiency, soil respiration and N mineralization (Curtin et al. 1998; Kemmitt et al. 2006; Weslien et al. 2009; Cuhel et al. 2010; Manzoni et al. 2012). Among all the variables examined, soil pH was strongly positively correlated with and was the most important predictor of soil C:N and N:P ratios (Fig. 3). This result is different from what has been found in agriculture soils (Kemmitt et al. 2006), soil pH had no significant effect on soil C:N ratios. Previous studies in the CBS temperate forest have indicated that soil pH was the main factor shaping microbial composition and activity and was positively correlated with soil organic C content (Yuan et al. 2013; Li et al. 2014; Shen et al. 2014), which is in support of our results. Our results also suggest that the spatial variation of soil C:N and N:P ratios may be controlled directly and indirectly by soil pH influencing belowground processes. Soil pH can be affected by plant types. Generally, coniferous litter tend to lower pH, because they generate chemical compounds that contain polyphenols and carboxylic acids during their decomposition (Zinke 1962; Augusto et al. 2002). P. koraiensis was abundant in our study plot, however, Pinus BA ratio was not correlated with soil pH (Supplementary, Table 4), suggesting that in addition to leaf litter input other factors such as topography and soil moisture may also influence the soil pH. A previous study in a tropical rainforest found that soil pH was strongly correlated with topography at the scale of 25 m (Xia et al. 2015). Though variation in topography was small in our study plot, microhabitats also were important for nutrient cycling (Fig. 3a). The topography and special plant may influence the spatial pattern of soil acidity and nutrient cycling.
Litter inputs are important pathways of C and N to the soil. In this study litter inputs were less important to soil C:N and N:P ratio than soil pH at the local-scale. No correlation between soil C:N ratios and leaf litter C:N ratios was found (Fig. 3). This is in contrast to previous studies in tropical forests (Xu et al. 2013; Uriarte et al. 2015), where soil C:N ratios were positively correlated with litter C:N ratios and contributed significantly to the heterogeneity of soil. Limited leaf litter effects on soil C:N and N:P ratio may be attributed to several biotic factors (Powers et al. 2004). First, boreal and temperate forests have higher root biomass allocation than tropical forests because plant functions are constrained by low temperature (photosynthesis, nutrient and water uptake, growth) (Jackson et al. 1996; Mokany et al. 2006; Poorter et al. 2012). In CBS and other temperate regions, the contribution of root litter to the soil C and nutrients pools can be equal to or more than that of leaf litter (Vogt et al. 1986; Finer et al. 2007; Wang et al. 2015). Preferential retention of root-derived carbon can make up a larger fraction of new carbon in soil organic matter than leaf litter inputs (Schmidt et al. 2011). Second, variability of litter nutrient fluxes among tree species have been shown to be larger in a given tropical forest site than across all temperate forests (Townsend et al. 2008; Mueller et al. 2015). An increasing proportion of depolymerized litter that is subsequently mineralized or leached from the soil could also explain the lack of correlation (Mueller et al. 2015). Additionally, soil freeze-thaw events may increase rates of N and P loss (Fitzhugh et al. 2001; Campbell et al. 2014). In the CBS region, the climate is characterized by a mountain climate with a short and rainy summer, and a long harsh winter. There are many freeze-thaw cycles in late autumn and early spring (Zhou et al. 2011), resulting in higher loss through leaching and denitrification for N and surface runoff for P in this region. Finally, a change in the strength of priming effects is another possible reason. High litter inputs could cause greater priming effects, and the increase in new soil C derived from the litter may be offset by the decomposition of older soil C (Sayer et al. 2011; Cheng et al. 2014).
In addition to soil pH and leaf litter inputs effects, topography and soil moisture also influenced the spatial variations of soil C:N and N:P ratios. We observed that spatial variation of soil C:N ratio was strongly affected by topography relative to soil moisture (Fig. 3a). Generally, soils in topographic trough locations trend to have higher soil moisture. Soil C, N and nitrate leaching are positively associated with soil moisture (Johnson et al. 2000; Seibert et al. 2007; Yuan et al.2013), possibly accounting for the topographic effects on soil C:N ratio. In contrast, spatial variation of soil N:P was more strongly affected by soil moisture than by topography (Fig. 3b). Two reasons may contribute to this phenomenon. First, there is higher risk of nitrate leaching due to higher mobility of nitrate (NO3 −) than phosphate (HPO4 2−, H2PO4 −). Second, where sites tend to be N- and P-rich, topography has a similar impact on N and P concentrations (Güsewell 2004).
The above-ground species composition plays a less important role in determining the variation of soil C:N and N:P ratios compared to abiotic factors (Fig. 3a, b). Many previous studies suggest that a single plant can modify soil nutrients (Finzi et al. 1998; Keller et al. 2013; Qiao et al. 2014). Indeed, we found that the Pinus BA ratio was positively correlated with soil C:N ratio and explained more variation of soil C:N ratio than other biotic factors (leaf litter inputs, stem density, species richness and total BA) (Fig. 3a). This is due to three mechanisms: first, leaf litter of coniferous species have higher C:N ratios compared with broadleaf species (Augusto et al. 2002; Yang and Luo 2011); second, coniferous species suppress biomass and activity of soil microflora and soil fauna (Augusto et al. 2002; Li et al. 2014), resulting in lower soil organic matter decomposition rate and higher C:N ratio; third, the canopies of coniferous species can intercept more elements (eg. sulphur and nitrogen) from the atmosphere compared to broadleaf species, decreasing nitrogen inputs (Augusto et al. 2002). Overall, our results demonstrated that P. koraiensis could be considered as a useful indicator of soil nutrient status.
Limitations of the analysis
Our study presented comprehensive analyses on the relative effects of soil properties, topography, aboveground litter inputs, and species composition on soil C:N and N:P ratios in a relatively diverse temperate forest. Multiple factors collectively determine soil C, N and P stoichiometry. However, the factors included in our study only explained 18.1 % and 23.5 % of the spatial variation in soil C:N and N:P ratios, respectively, which are insufficient to fully understand and interpret the variability of soil stoichiometry. Several limitations may affect our results. First, some potentially important factors were not considered here, such as belowground litter inputs (e.g. fine roots) (Freschet et al. 2013), soil biota (Bever et al. 2013), and other soil physical and chemical properties (Manzoni et al. 2012; Plaza et al. 2013) and their interactions (Mueller et al. 2015). Second, the litterfall traps had lower spatial resolution than the soil samples. Individual-tree leaf litter dispersal models were powerful and were successfully applied to predict leaf litter mass at soil sampling sites. However, there were still some sources of variances, which may contribute to inaccurate predictions: (i) the location of the tree crowns did not necessarily correspond to map coordinate of the focal trees. (ii) Wind or tree height influences on dispersal were not considered, although the latter would correlate with DBH. To better understand the underlying mechanisms of soil nutrient heterogeneity, future works need to focus on the influence of root litter and soil microorganisms.
Conclusions
Our results suggest that biotic and abiotic determinants contribute to the spatial variation of soil elemental stoichiometry, but abiotic factors tended to be more important. Specifically, soil pH rather than leaf litter inputs and species composition was the major factor explaining the spatial variation of soil C:N and N:P stoichiometry. This implies that belowground processes in this temperate forest drive heterogeneity of soil elemental stoichiometry. This result offers important insights into nutrient cycling under climate change. Among the included biotic variables, the dominant coniferous species, Pinus koraiensis, played an important role and had a positive effect on the variability of soil C:N ratio. Thus, management decisions should be designed with particular species distribution in mind.
Abbreviations
- BA:
-
Basal area
- BRT:
-
Boosted Regression Tree analysis
- CBS:
-
Changbaishan (Forest area)
- DBH:
-
Diameter at breast height
- LLF:
-
Leaf litterfall mass
- RAC:
-
Residual autocovariate
- SAC:
-
Spatial autocorrelation
References
Aponte C, Garcia LV, Maranon T (2013) Tree species effects on nutrient cycling and soil biota: a feedback mechanism favouring species coexistence. For Ecol Manag 309:36–46. doi:10.1016/j.foreco.2013.05.035
Augusto L, Ranger J, Binkley D, Rothe A (2002) Impact of several common tree species of European temperate forests on soil fertility. Ann For Sci 59:233–253. doi:10.1051/forest:2002020
Bever JD, Broadhurst LM, Thrall PH (2013) Microbial phylotype composition and diversity predicts plant productivity and plant-soil feedbacks. Ecol Lett 16:167–174. doi:10.1111/ele.12024
Bivand R (2015) Spdep: spatial dependence: weighting schemes, statistics and models. R package version 0.6-4. Available online at https://cran.r-project.org/web/packages/spdep/index.html
Bui EN, Henderson BL (2013) C:N:P stoichiometry in Australian soils with respect to vegetation and environmental factors. Plant Soil 373:553–568. doi:10.1007/s11104-013-1823-9
Campbell JL, Socci AM, Templer PH (2014) Increased nitrogen leaching following soil freezing is due to decreased root uptake in a northern hardwood forest. Glob Chang Biol 20:2663–2673. doi:10.1111/gcb.12532
Cheng WX et al. (2014) Synthesis and modeling perspectives of rhizosphere priming. New Phytol 201:31–44. doi:10.1111/nph.12440
Condit R (1998) Tropical forest census plots. Springer Science, Berlin
Cools N, Vesterdal L, De Vos B, Vanguelova E, Hansen K (2014) Tree species is the major factor explaining C:N ratios in European forest soils. For Ecol Manag 311:3–16. doi:10.1016/j.foreco.2013.06.047
Crase B, Liedloff AC, Wintle BA (2012) A new method for dealing with residual spatial autocorrelation in species distribution models. Ecography 35:879–888. doi:10.1111/j.1600-0587.2011.07138.x
Cuhel J, Simek M, Laughlin RJ, Bru D, Cheneby D, Watson CJ, Philippot L (2010) Insights into the effect of soil pH on N2O and N2 emissions and denitrifier community size and activity. Appl Environ Microbiol 76:1870–1878. doi:10.1128/aem.02484-09
Curtin D, Campbell CA, Jalil A (1998) Effects of acidity on mineralization: pH-dependence of organic matter mineralization in weakly acidic soils. Soil Biol Biochem 30:57–64. doi:10.1016/s0038-0717(97)00094-1
Dormann CF et al. (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46. doi:10.1111/j.1600-0587.2012.07348.x
Elith J, Leathwick JR, Hastie T (2008) A working guide to boosted regression trees. J Anim Ecol 77:802–813. doi:10.1111/j.1365-2656.2008.01390.x
Elser JJ et al. (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142. doi:10.1111/j.1461-0248.2007.01113.x
Ferrari JB, Sugita S (1996) A spatially explicit model of leaf litter fall in hemlock-hardwood forests. Can J For Res 26:1905–1913. doi:10.1139/x26-215
Finer L et al. (2007) Variation in fine root biomass of three European tree species: beech (Fagus sylvatica L.), Norway spruce (Picea abies L. Karst.), and scots pine (Pinus sylvestris L.). Plant Biol 141:394–405. doi:10.1080/11263500701625897
Finzi AC, Van Breemen N, Canham CD (1998) Canopy tree soil interactions within temperate forests: species effects on soil carbon and nitrogen. Ecol Appl 8:440–446. doi:10.2307/2641083
Fitzhugh RD, Driscoll CT, Groffman PM, Tierney GL, Fahey TJ, Hardy JP (2001) Effects of soil freezing disturbance on soil solution nitrogen, phosphorus, and carbon chemistry in a northern hardwood ecosystem. Biogeochemistry 56:215–238. doi:10.1023/a:1013076609950
Frank DA (2008) Ungulate and topographic control of nitrogen: phosphorus stoichiometry in a temperate grassland; soils, plants and mineralization rates. Oikos 117:591–601. doi:10.1111/j.2007.0030-1299.16220.x
Freschet GT et al. (2013) Linking litter decomposition of above- and below-ground organs to plant-soil feedbacks worldwide. J Ecol 101:943–952. doi:10.1111/1365-2745.12092
Gomez-Aparicio L, Canham CD (2008) Neighborhood models of the effects of invasive tree species on ecosystem processes. Ecol Monogr 78:69–86. doi:10.1890/06-2036.1
Gundersen P, Sevel L, Christiansen JR, Vesterdal L, Hansen K, Bastrup-Birk A (2009) Do indicators of nitrogen retention and leaching differ between coniferous and broadleaved forests in Denmark? For Ecol Manag 258:1137–1146. doi:10.1016/j.foreco.2009.06.007
Güsewell S (2004) N: P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266. doi:10.1111/j.1469-8137.2004.01192.x
Hao Z, Zhang J, Song B, Ye J, Li B (2007) Vertical structure and spatial associations of dominant tree species in an old-growth temperate forest. For Ecol Manag 252:1–11. doi:10.1016/j.foreco.2007.06.026
Hogberg MN, Hogberg P, Myrold DD (2007) Is microbial community composition in boreal forest soils determined by pH, C-to-N ratio, the trees, or all three? Oecologia 150:590–601. doi:10.1007/s00442-006-0562-5
Jackson RB, Canadell J, Ehleringer JR, Mooney HA, Sala OE, Schulze ED (1996) A global analysis of root distributions for terrestrial biomes. Oecologia 108:389–411. doi:10.1007/bf00333714
John R et al. (2007) Soil nutrients influence spatial distributions of tropical tree species. Proc Natl Acad Sci U S A 104:864–869. doi:10.1073/pnas.0604666104
Johnson CE, Ruiz-Mendez JJ, Lawrence GB (2000) Forest soil chemistry and terrain attributes in a Catskills watershed. Soil Sci Soc Am J 64:1804–1814
Jonard M, Andre F, Ponette Q (2006) Modeling leaf dispersal in mixed hardwood forests using a ballistic approach. Ecology 87:2306–2318. doi:10.1890/0012-9658(2006)87[2306:mldimh]2.0.co;2
Keller AB, Reed SC, Townsend AR, Cleveland CC (2013) Effects of canopy tree species on belowground biogeochemistry in a lowland wet tropical forest. Soil Biol Biochem 58:61–69. doi:10.1016/j.soilbio.2012.10.041
Kemmitt SJ, Wright D, Goulding KWT, Jones DL (2006) pH regulation of carbon and nitrogen dynamics in two agricultural soils. Soil Biol Biochem 38:898–911. doi:10.1016/j.soilbio.2005.08.006
Li BH, Hao ZQ, Bin Y, Zhang J, Wang M (2012) Seed rain dynamics reveals strong dispersal limitation, different reproductive strategies and responses to climate in a temperate forest in Northeast China. J Veg Sci 23:271–279. doi:10.1111/j.1654-1103.2011.01344.x
Li H et al. (2014) Soil bacterial communities of different natural forest types in Northeast China. Plant Soil 383:203–216. doi:10.1007/s11104-014-2165-y
Manzoni S, Trofymow JA, Jackson RB, Porporato A (2010) Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol Monogr 80:89–106. doi:10.1890/09-0179.1
Manzoni S, Taylor P, Richter A, Porporato A, Agren GI (2012) Environmental and stoichiometric controls on microbial carbon-use efficiency in soils. New Phytol 196:79–91. doi:10.1111/j.1469-8137.2012.04225.x
Mokany K, Raison RJ, Prokushkin AS (2006) Critical analysis of root: shoot ratios in terrestrial biomes. Glob Chang Biol 12:84–96. doi:10.1111/j.1365-2486.2005.001043.x
Morhart C, Sheppard JP, Schuler JK, Spiecker H (2016) Above-ground woody biomass allocation and within tree carbon and nutrient distribution of wild cherry (Prunus avium L.) – a case study. Forest Ecosystems 3. doi:10.1186/s40663-016-0063-x
Mueller KE et al. (2015) Effects of litter traits, soil biota, and soil chemistry on soil carbon stocks at a common garden with 14 tree species. Biogeochemistry 123:313–327. doi:10.1007/s10533-015-0083-6
Pietri JCA, Brookes PC (2008) Relationships between soil pH and microbial properties in a UK arable soil. Soil Biol Biochem 40:1856–1861. doi:10.1016/j.soilbio.2008.03.020
Plaza C, Courtier-Murias D, Fernandez JM, Polo A, Simpson AJ (2013) Physical, chemical, and biochemical mechanisms of soil organic matter stabilization under conservation tillage systems: a central role for microbes and microbial by-products in C sequestration. Soil Biol Biochem 57:124–134. doi:10.1016/j.soilbio.2012.07.026
Poorter H, Niklas KJ, Reich PB, Oleksyn J, Poot P, Mommer L (2012) Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol 193:30–50. doi:10.1111/j.1469-8137.2011.03952.x
Powers JS, Kalicin MH, Newman ME (2004) Tree species do not influence local soil chemistry in a species-rich Costa Rica rain forest. J Trop Ecol 20:587–590. doi:10.1017/s0266467404001877
Qiao L, Schaefer DA, Zou X (2014) Variations in net litter nutrient input associated with tree species influence on soil nutrient contents in a subtropical evergreen broad-leaved forest. Chin Sci Bull 59:46–53. doi:10.1007/s11434-013-0019-2
R Core Team (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Ribeiro PJ Jr, Diggle PJ (2001) geoR: a package for geostatistical analysis. R news 1:14–18
Ridgeway G (2010) Gbm: generalized boosted regression models. R Package Version 1:6–3.1
Riveros-Iregui DA, McGlynn BL (2009) Landscape structure control on soil CO2 efflux variability in complex terrain: Scaling from point observations to watershed scale fluxes. J Geophys Res Biogeosci 114. doi:10.1029/2008jg000885
Sayer EJ, Heard MS, Grant HK, Marthews TR, Tanner EVJ (2011) Soil carbon release enhanced by increased tropical forest litterfall. Nat Clim Chang 1:304–307. doi:10.1038/nclimate1190
Schmidt MWI et al. (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:49–56. doi:10.1038/nature10386
Seibert J, Stendahl J, Sorensen R (2007) Topographical influences on soil properties in boreal forests. Geoderma 141:139–148. doi:10.1016/j.geoderma.2007.05.013
Shen CC et al. (2014) Contrasting elevational diversity patterns between eukaryotic soil microbes and plants. Ecology 95:3190–3202
Sparks DL et al. (1996) Methods of soil analysis. Part 3-Chemical methods. Soil Science Society of America Inc., Madison, Wisconsin
Staelens J, Nachtergale L, Luyssaert S (2004) Predicting the spatial distribution of leaf litterfall in a mixed deciduous forest. For Sci 50:836–847
Stone R (2006) Ecology - a threatened nature reserve breaks down Asian borders. Science 313:1379–1380. doi:10.1126/science.313.5792.1379
Townsend AR, Asner GP, Cleveland CC (2008) The biogeochemical heterogeneity of tropical forests. Trends Ecol Evol 23:424–431. doi:10.1016/j.tree.2008.04.009
Tripathi BM et al. (2015) Soil pH and biome are both key determinants of soil archaeal community structure. Soil Biol Biochem 88:1–8. doi:10.1016/j.soilbio.2015.05.004
Uriarte M, Turner BL, Thompson J, Zimmerman JK (2015) Linking spatial patterns of leaf litterfall and soil nutrients in a tropical forest: a neighborhood approach. Ecol Appl 25:2022–2034
Vesterdal L, Clarke N, Sigurdsson BD, Gundersen P (2013) Do tree species influence soil carbon stocks in temperate and boreal forests? For Ecol Manag 309:4–18. doi:10.1016/j.foreco.2013.01.017
Vogt KA, Grier CC, Vogt D (1986) Production, turnover, and nutrient dynamics of above-and belowground detritus of world forests. Adv Ecol Res 15:3–377
Wang L, Mou PP, Huang J, Wang J (2007) Spatial heterogeneity of soil nitrogen in a subtropical forest in China. Plant Soil 295:137–150. doi:10.1007/s11104-007-9271-z
Wang L et al. (2011) Dynamics of short-term tree mortality in broad-leaved Korean pine (Pinus koraiensis) mixed forest in the Changbai Mountains. Biodivers Sci 19:260–270
Wang C, Han S, Zhou Y, Zhang J, Zheng X, Dai G, Li M-H (2015) Fine root growth and contribution to soil carbon in a mixed mature Pinus koraiensis forest. Plant Soil. doi:10.1007/s11104-015-2724-x
Weslien P, Klemedtsson AK, Borjesson G, Klemedtsson L (2009) Strong pH influence on N2O and CH4 fluxes from forested organic soils. Eur J Soil Sci 60:311–320. doi:10.1111/j.1365-2389.2009.01123.x
Xia S-W, Chen J, Schaefer D, Detto M (2015) Scale-dependent soil macronutrient heterogeneity reveals effects of litterfall in a tropical rainforest. Plant Soil. doi:10.1007/s11104-015-2402-z
Xu S, Liu LL, Sayer EJ (2013) Variability of above-ground litter inputs alters soil physicochemical and biological processes: a meta-analysis of litterfall-manipulation experiments. BGeo 10:7423–7433. doi:10.5194/bg-10-7423-2013
Yang Y, Luo Y (2011) Carbon:nitrogen stoichiometry in forest ecosystems during stand development. Glob Ecol Biogeogr 20:354–361. doi:10.1111/j.1466-8238.2010.00602.x
Yang X, Xu M (2003) Biodiversity conservation in Changbai Mountain biosphere reserve, northeastern China: status, problem, and strategy. Biodivers Conserv 12:883–903. doi:10.1023/a:1022841107685
Yoo K, Amundson R, Heimsath AM, Dietrich WE (2006) Spatial patterns of soil organic carbon on hillslopes: integrating geomorphic processes and the biological C cycle. Geoderma 130:47–65. doi:10.1016/j.geoderma.2005.01.008
Yuan ZY, Chen HYH (2009) Global trends in senesced-leaf nitrogen and phosphorus. Glob Ecol Biogeogr 18:532–542. doi:10.1111/j.1466-8238.2009.00474.x
Yuan Z et al. (2013) Soil organic carbon in an old-growth temperate forest: spatial pattern, determinants and bias in its quantification. Geoderma 195:48–55. doi:10.1016/j.geoderma.2012.11.008
Zhou WM et al. (2011) Effect of freezing-thawing on nitrogen mineralization in vegetation soils of four landscape zones of Changbai Mountain. Ann For Sci 68:943–951. doi:10.1007/s13595-011-0100-4
Zinke PJ (1962) The pattern of influence of individual forest trees on soil properties. Ecology 43:130–133. doi:10.2307/1932049
Acknowledgments
This study was funded by the National Natural Science Foundation of China (31570432 and 41301057), Program of International Co-operation, Chinese Academy of Sciences (151221KYSB20130003), Institute of Applied Ecology, Chinese Academy of Sciences (Y5YZX151YD), and CFERN & GENE Award Funds on Ecological Paper. We would like to thank Tom McRae, Xingguo Han and Smithsonian CTFS - ForestGEO Analytical Workshop V participants for their valuable suggestions to improve the manuscript. We would like to thank Mathieu Jonard and Qingqing Zhao for sharing R codes of the leaf dispersal model. We would also like to thank Alison Beamish at the University of British Columbia for her assistance with English language and grammatical editing of the manuscript. We would also like to thank the CBS plot census and data management teams, and especially thank Yuqiang Zhao, Xu Kuang, Guodong Liu, Baizhang Song and Xichang He for their assistance with the field work and data collection.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Harry Olde Venterink.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(RAR 1 mb)
Rights and permissions
About this article
Cite this article
Suo, Y., Yuan, Z., Lin, F. et al. Local-scale determinants of elemental stoichiometry of soil in an old-growth temperate forest. Plant Soil 408, 401–414 (2016). https://doi.org/10.1007/s11104-016-2939-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-2939-5