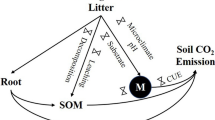
Abstract
Aims
This study investigated the effects of changes in litter quantity and quality on litter decomposition, soil respiration, and soil organic carbon (SOC) in subtropical forests.
Methods
The experiment had a nested factorial design with three factors: (1) successional stage with three levels (early, mid and mature), (2) litter type with two levels (Schima superba Gardn. et Champ. and Ormosia pinnata (Lour.) Merr.), and (3) litter addition with five levels (0, 218, 436, 654 and 873 g·m−2·yr−1, respectively).
Results
In all forests, an increase in litter input increased litter decomposition, litter carbon (C) loss and soil respiration but did not alter SOC content after 2.5 years. The increases in litter decomposition, litter C loss, and soil respiration in response to increased litter input were greater with the lower quality Schima superba litter than with the higher quality Ormosia pinnata litter. Litter quality did not affect SOC content at any of the three forest sites. The responses of litter decomposition and soil respiration to increasing litter input differed depending on forest successional stage.
Conclusions
In subtropical forests, increases in litter production under climate change may accelerate C cycling. Net soil C storage in subtropical forests, however, may not change over short time scales in response to increased litter input.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Litter quantity and quality in forests are likely to change as a consequence of climate change. Many studies have shown that litter quantity and quality are altered by elevated atmospheric carbon dioxide (CO2) concentration (Norby et al. 2005; Liu et al. 2005; Hickler et al. 2008; Clark et al. 2010; Ellsworth et al. 2012), changes in rainfall distribution patterns and rising temperature (Martínez-Vilalta et al.; 2012; Doughty et al.; 2014; Raich et al. 2006; Zhou et al. 2013). Because litter represents a major pathway for C cycling between vegetation and soil in forest ecosystems, changes in aboveground litter quantity and quality could have important consequences for C cycling.
Relatively short-term experiments have shown that both rising atmospheric CO2 concentrations and warming increase net primary production (Norby et al. 2005; Liu et al. 2005; Hickler et al. 2008; Clark et al. 2010; Ellsworth et al. 2012), which in turn may increase soil C stocks via enhanced litter inputs. Over longer time scales, however, the effects of elevated CO2 on belowground C cycling are less clear; some research suggests that elevated CO2 will increase soil C content (Jastrow et al. 2005) but other research indicates no significant change (Lichter et al. 2005; Hoosbeek and Scarascia-Mugnozza 2009). Elevated CO2 may also alter the concentrations of nitrogen (N) and phosphorus (P) concentrations in litter (Norby et al. 2001; King et al. 2005; Liu et al. 2013). Because soil microbes preferentially utilize high quality litter, Sylvia et al. (1998) hypothesized that increased inputs of litter with higher C/N under elevated CO2 may decrease the litter decomposition rate. Several studies have shown, however, that small reductions in litter N under elevated CO2 will have little effect on litter mass loss and C storage (Booker et al. 2005; Liu et al. 2009). Overall, these studies suggest that changes in litter quality combined with increasing litter inputs in the context of climate change could result in potential changes in C cycling. Most of these studies have been conducted in temperate ecosystems, and the possible effects of changes in litter inputs caused by climate change on belowground C cycling in subtropical forests are less known.
Plant-soil feedbacks play a decisive role in determining whether tropical and subtropical forest soils act as sources or sinks of atmospheric CO2 (Sayer 2006). In general, greater litter input is expected to increase C sequestration in soil. However, reports on the effects of increased litter input on soil C storage have been inconsistent because the relationships among net primary production, increased litter production, and net soil C storage are complex (Sayer 2006; Crow et al. 2009). For example, CO2 fluxes often increase disproportionately with litter addition, suggesting that increased C inputs may accelerate decomposition of extant soil C via priming effects (Kuzyakov et al. 2000; Fontaine et al. 2004; Schaefer et al. 2009; Chemidlin Prévost-Bouré et al. 2010; Sayer et al. 2011), and ultimately lead to net losses of soil C. Although priming may mineralize some recalcitrant soil C, some of the increased C input may move into stabilized pools of soil C (Hyvönen et al. 2007; Hoosbeek et al. 2007) and result in a net soil C storage (Crow et al. 2009). In Costa Rica, Leff et al. (2012) found that litter addition increased both soil respiration and total C pools, and that priming did not occur; the lack of priming was attributed to low soil fertility. However, results from other tropical rain forests suggested that soil C content does not change with elevated litter input (Wood and Lawrence 2008; Vincent et al. 2010; Sayer et al. 2012).
Many tropical and subtropical forests grow on highly weathered soils that contain low levels of P and many other nutrients (Vitousek and Sanford 1986; Kaspari et al. 2008; Huang et al. 2013). Nutrient availability in the surface soils may also affect C cycling (Prescott et al. 1993; Hobbie and Vitousek 2000; Cleveland and Townsend 2006). Based on the results of a fertilization experiment with combinations of N, P, K, or micronutrients in a lowland forest in Panama, Kaspari et al. (2008) proposed that deficiencies in multiple nutrients limited litter decomposition in a tropical forest. Cleveland et al. (2006) showed that although nutrient availability in the surface soil may not affect litter mass loss during decomposition in nutrient-poor ecosystems, nutrient availability may ultimately regulate CO2 losses (and hence C storage) by limiting microbial mineralization of dissolved organic carbon (DOC) leached from the litter layer. Other studies showed that the priming effect was limited by nutrient availability in the surface soil, which also could influence soil C storage (Fontaine et al. 2004; Nottingham et al. 2012). The wide range of soil age and weathering status could also result in differences in nutrient availability among tropical forest types (Walker and Syers 1976; Hedin et al. 2003; Huang et al. 2013). Therefore, variations in litter input may have different effects on litter decomposition, soil respiration and hence soil C storage in different types of subtropical forest.
We conducted a litter-manipulation experiment to examine the effects of changes in litter production and changes in litter quality on litter decomposition, soil respiration and SOC in three subtropical forests in southern China. We hypothesized that: (1) An increase in litter addition would accelerate litter decomposition, soil respiration, and reduce C storage; (2) These responses to litter input would be greater with high quality than with low quality litter; and (3) The variations in litter decomposition, soil respiration, and SOC in response to litter input would be related to nutrient availability in the surface soil.
Materials and methods
Study sites
This study was conducted in three subtropical forests: a monsoon evergreen broadleaved forest (BF), a mixed pine and broadleaved forest (MF), and a plantation forest (PF). The BF and MF are located at the Dinghushan Biosphere Reserve (23°09′ N-23°11′ N, 112°30′ E-112°33′ E, DBR) in southern China, ca. 90 km west of Guangzhou city. DBR covers an area of 1155 ha and was accepted as the first National Natural Reserve in China in 1956 (Zhou et al. 2006). The MF and BF represent the middle and climax vegetation in this area, respectively. The PF, which represents an early stage of succession, is located at the South China Botanical Garden in Guangzhou City (23°10′ N, 113°21′ E). All sites are characterized by a typical subtropical monsoon humid climate. Annual precipitation is ca. 1700 mm, and >80 % of the rain falls in the wet season (April–September). Annual mean relative humidity and temperature are ca. 78 % and 21.7 °C. The bedrock of all three forest types is sandstone and shale. Soils are all highly weathered and classified in the ultisol group according to the USDA soil classification system (Buol et al. 2003). Site characteristics of the three forest types are listed in Table 1. Soil organic C and nutrient content vary with forest type. Soil organic C content is higher in the BF than MF and PF. Soil total N is the highest in the BF, intermediate in the MF, and lowest in the PF. Soil P concentration is much lower in the PF than in the BF and the MF. Additionally, soil water content is highest in the BF, intermediate in the MF, and lowest in the PF.
The BF has not been disturbed for more than 400 years (Wang and Ma 1982; Zhou et al. 2006) and is located in the core area of the reserve. As the climax vegetation of this area, the BF has a complex species composition. The upper canopy is dominated by a small number of individuals, including Castanopsis chinensis Hance, Schima superba Chardn. & Champ., Cryptocarya chinensis (Hance) Hemsl., Cryptocarya concinna Hance, Machilus chinensis (Champ. Ex Benth.) Hemsl., and Syzygium rehderianum Merr. & Perry (Wang and Ma 1982). The MF, which is ca. 80 years old, is located between the core area and the periphery of the DBR. The upper canopy is dominated by Pinus massoniana Lamb, Schima superba Chardn. & Champ., Castanopsis chinensis Hance, and Craibiodendron kwangtungense S. Y. Hu. Pinus massoniana accounts for ca. 35 % of the biomass of the MF community. The PF was planted in 1980s with a pure stand of Schima superba (S. superba) and a pure stand of Acacia mangium (A. mangium). Mean annual leaf litter production was 386, 589, and 482 g m2 for the PF, MF, and BF, respectively (Zhou et al. 2007).
Experimental design
Naturally senesced leaf litter (S. superba and Ormosia pinnata (O. pinnata) was collected and used for this experiment. O. pinnata is an N2 fixer, while S. superba is not. Leaf litter of S. superba (47.3 % C, 1.3 % N, 0.05 % P, 0.06 % potassium, 0.88 % calcium) and O. pinnata (49.4 % C, 2.6 % N, 0.1 % P, 0.07 % potassium, 0.63 % calcium) was oven-dried for 48 h at 70 °C. In July 2010, we prepared 900 PVC cylinders, with 10.8 cm inner diameter and 10.0 cm high. The experiment had a nested factorial design with three factors: (1) successional stage with three levels (BF, MF, and PF), (2) litter type with two levels (low and high quality, i.e., S. superba and O. pinnata), and (3) litter addition rate with five levels (0, 2, 4, 6 and 8 g per cylinder; the highest rate represented 225, 150, and 180 % of the annual litter input in early, mid, and mature forests, respectively). This resulted in 30 treatment combinations with 30 replicates per combination (30 replicates × 5 levels of litter addition × 2 litter types × 3 forest types = 900 PVC cylinders). Forest floor materials were removed before the experiment was begun, and all cylinders were sunk into the soil with 5 cm of the cylinder above the ground and 5 cm below the ground. Then, appropriate quantities of each litter species were added to the PVC cylinders in a random manner. Three hundred cylinders were placed within a 30 × 30 m plot in the BF and in a similar plot in the MF. The study site in the PF was divided into two subplots; S. superba litter was added at the pure S. superba stand, and O. pinnata litter was added at the pure A. mangium stand. The distance between the two subplots was <200 m. To prevent other kinds of litter falling into the cylinders but to expose the litter to the natural environment and most organisms, we covered each cylinder with a 2-mm-mesh Dacron cloth; we used fine wire to fasten the cloth to the cylinders. Although the mesh would prevent passage of some larger soil fauna (earthworms, isopods, ants, ect.), large soil organisms and fine roots could access the decomposing litter through the bottom of the PVC cylinders. The experimental setup was completed in September 2010.
Litter decomposition sampling
The litter decomposition experiment lasted for ca. 2.5 years in all forests. At ca. 0.5, 1.0, and 1.5 years after the start of the experiment, we sampled six replicate cylinders for each combination of litter addition rate (excluding the rate of 0 g/cylinder), litter type, and forest type, giving a total of 144 litter samples (3 forest types × 2 litter types × 4 litter addition rates × 6 replicates). Litter was not collected after 1.5 years because, in most treatments, > 80 % of the litter had already decomposed at that time and the remaining litter was too difficult to collect. Tweezers were used to collect all litter (>2 mm in either length or width) in the designated cylinders, litter samples were then transported to laboratory, washed softly and quickly (same treatment with the same washing standard) to remove foreign materials, weighed for mass loss after drying for 48 h at 70 °C, and then finely ground for C concentration analysis. C concentrations in the initial litter and in the litter collected from cylinders were determined with Walkley-Black’s wet digestion method (Nelson and Sommers 1982).
Soil respiration measurements
Soil respiration was measured in six undisturbed replicate cylinders for each treatment combination. The Dacron cloth that covered each cylinder was removed from the collar prior to all measurements and was returned once the measurements were completed. Soil respiration was measured in the collar of each replicate cylinder when the experiment began and every 3 months thereafter using a Li-Cor 6400 infrared gas analyzer (Li-COR, Inc., Lincoln, Nebraska, USA) connected to a Li-Cor 6400-09 soil respiration chamber (Li-COR, Inc., Lincoln, Nebraska, USA). At each sampling date, soil respiration was measured five times for each cylinder, and the five values were averaged to give one value per cylinder and per date. All the measurements were made between 8:30 am and 12:00 pm local time on sunny days. The ambient CO2 concentration was automatically determined for each site.
Soil sampling and measurements
Soil samples were collected by removing the 5 cm of soil within the designated cylinders. Six replicates of soil samples were collected for each treatment combination at 0.5, 1.0, 1.5, 2.0, and 2.5 years after the start of the experiment. For the first three soil collection dates, we used cylinders from which the litter had just been removed as described earlier. For the fourth and fifth collection dates, we carefully removed all of the litter before collecting the soil samples. After soil samples were passed through a 2-mm sieve and visible plant material was removed, each soil sample was divided into two parts. One part was stored at 4 °C and used for determination of soil water content and soil microbial biomass carbon (SMBC). The other part was air-dried and used for determination of SOC content.
SOC was determined by the Walkley-Black’s wet digestion method (Nelson and Sommers 1982). SMBC was determined by subjecting fresh soil samples to the chloroform fumigation-extraction method (Brookes et al. 1985; Martens 1995). Briefly, for each sample, soil microbial biomass was measured as the difference in 0.5 M K2SO4 extractable C between fumigated and unfumigated samples. Organic C in the extracts was measured with a TOC analyzer (TOC-5050A; Shimadzu Corporation, Kyoto, Japan), and SMBC was calculated as the difference in extractable C multiplied by the conversion factor of 0.45 (Brookes et al. 1985; Martens 1995). The unfumigated samples were used to estimate background DOC values.
Data analysis
SAS software (Statistical Analysis System, version 9.2, SAS Institute, Inc.) was used for all statistical analysis. Data were transformed to meet the assumptions of normality and homogeneity of variances when necessary. Litter mass loss and litter C loss were expressed as ratios of the initial oven-dry weight and initial C weight, respectively (Mo et al. 2006; Fang et al. 2007). Because samples were collected and analyzed continuously throughout the study, repeated ANOVAs with Tukey’s HSD test were used to estimate the effects of changes in litter input on litter decomposition, litter C loss, soil respiration, SMBC, soil DOC, SOC and soil water content. Statistical significance was determined at P <0.05 unless otherwise stated.
Results
Litter decomposition
Litter mass loss differed depending on the quantity of litter added, litter type, and forest type (Fig. 1; Table 2). The temporal patterns of litter decomposition were generally similar regardless of litter quantity, litter type, or forest type. Decomposition was faster during the first year than during the second year (Fig. 1). More than 70 % of total litter was lost within 1.5 years. Averaged across litter type and forest type, repeated ANOVA with Tukey’s HSD test showed that increasing the quantity of litter added significantly accelerated litter mass loss (P < 0.001) and litter C loss (Fig. 1; Tables 2 and 3). Although N and P contents were lower in the undecomposed S. superba litter than in the undecomposed O. pinnata litter, litter decomposition rates and litter C loss rates were higher (P < 0.01) for S. superba litter than for O. pinnata litter (Fig. 1; Tables 2 and 3).
Litter mass loss as affected by the quantity of litter added and litter type in three forest types, including monsoon evergreen broadleaved forest (BF), mixed pine and broadleaved forest (MF), and plantation forest (PF). Values are means ± SD (n = 6). * and ** indicate significant differences at 5 and 1 % among litter addition rates
Forest type also significantly (P < 0.01) affected litter mass loss and litter C loss in the following order: MF>BF>PF (Fig. 1; Table 2). In the BF, increasing the litter input resulted in significantly greater (P < 0.05) litter mass loss and litter C loss; averaged across both types of litter, litter mass loss and litter C loss rates tended to increase as the quantity of litter added increased (Fig 1; Table 3). In the MF and PF, litter mass loss and litter C loss for both litter types also tended to increase with the quantity of litter added but the effect was not statistically significant (P > 0.05; Fig 1; Table 3). In addition, litter mass loss and litter C loss were significantly greater (P < 0.05) for S. superba litter than for O. pinnata litter in the BF and MF and were slightly but not significantly greater for S. superba litter than for O.pinnata in the MF.
Soil respiration
Soil respiration rates of all samples in the three forest types showed a similar seasonal pattern (Fig. 2), with higher rates in the warm-wet seasons and lower rates in the cool-dry ones. Overall, repeated ANOVA showed that increasing the quantity of litter added resulted in significantly higher (P < 0.0001) respiration throughout the study period (Fig. 2; Table 2). Litter type also significantly affected soil respiration (Table 2). Soil respiration was significantly higher for S. superb litter than for O. pinnata in the BF (P < 0.05) and MF (P < 0.001), but the opposite was true in the PF (P < 0.0001).
Annual soil respiration rates were significantly influenced by forest type, and were lowest in the BF (2.07 μmol CO2 m−2 s−1), intermediate in the MF (2.76 μmol CO2 m−2 s−1), and highest in the PF (3.15 μmol CO2 m−2 s−1). In all forest types, an increase in litter input led to significantly higher (P < 0.001) soil respiration for both litter types (Fig. 2). Averaged across both litter types and compared to the 0 g of litter treatment, addition of 8 g of litter significantly increased (P < 0.001) the annual average soil respiration by 33.8, 18.4, and 11.6 % in the PF, MF, and BF, respectively. Compared to the 0 g of litter treatment, addition of 8 g of S. superba litter increased annual average soil respiration by 41.5, 25.5, and 7.5 % in the PF, MF, and BF, respectively. Compared to the 0 g of litter treatment, addition of 8 g of O. pinnata litter increased annual averaged soil respiration by 27.7, 18.4, and 16.8 % in PF, MF, and BF, respectively.
Soil organic C, SMBC, and DOC
SOC content was measured periodically during the experiment. An overall statistical analysis showed that SOC was not significantly affected by the quantity of litter added during the whole experimental period (Fig 3; Table 2). The quantity of litter added also did not affect DOC, soil water content, and soil temperature in any of the forests (Tables 2 and 3). However, repeated ANOVAs with Tukey’s HSD tests showed that SMBC significantly increased (P < 0.05) as the quantity of litter added increased for both litter types in the PF and for O. pinnata litter in the BF (Fig. 4). In the MF, SMBC for both litter types tended to increase with the quantity of litter added but the effect was not significant.
Although forest type did not significantly affect SOC content, forest type did affect SMBC (Table 2), which was obviously higher in the BF than in the MF or PF (Fig. 4). Soil DOC content was also affected by forest type (Table 2) and was higher in the BF and PF than in the MF (Table 3). There were also no obvious effects of litter quantity on SOC at two species levels in three forests (Fig. 3). Litter quality also did not significantly influence SOC and DOC in all forests (Fig. 3 and Table 3).
Soil temperature and water content
Averaged across litter type and forest type the litter quantity of litter added did not significantly affects soil water content or soil temperature. Annual soil water content was highest in the BF (26.4 %), intermediate in the MF (23.2 %), and lowest in the PF (15.5 %; P < 0.0001). Soil temperature was significantly higher (P < 0.0001) in the PF (21.2 °C) than in the MF (20.5 °C) or BF (20.2 °C). Litter type also did not affect soil water content or temperature in the BF and MF. In the PF, however, soil temperature was significantly higher (P < 0.0001) in the A. mangium stand (21.8 °C) than in the S. superba stand (20.7 °C), and soil water content was significantly lower (P < 0.001) in the pure A. mangium stand (13.0 %) than in S. superba stand (20.7 %).
Discussion
Effects of changes in litter input on litter decomposition
In a litter manipulation experiment, Sayer (2006) showed that litter addition elicited increases in litter decomposition in the early stages, although not always significant. Our result also showed that increased litter input resulted in increased litter mass loss and litter C loss. This result is also consistent with Ostertag et al. (2003), who found that the large input of litter following a hurricane accelerated decomposition. That increases in litter input enhance decomposition might be explained in part by the nutrients in the litter. Tropical forests are often limited by P and other nutrients (Vitousek and Sanford 1986; Kaspari et al. 2008; Huang et al. 2013). Higher litter addition corresponded to greater inputs of labile C components and available nutrients (Wood et al. 2009; Liu et al. 2009), which could stimulate microbial activity and thus increases the decomposition rate (Sayer 2006). We found that increases in litter input led to increased SMBC content in our experiment, which suggests that an increase in litter input resulted in increased microbial activity and thus in higher rates of decomposition. Soil respiration was enhanced by increased litter input in all forests, also indicating that microbial activity may increase in response to greater inputs of labile C components and nutrients.
Many studies have reported that decomposition rates are positively correlated with litter quality (Prescott et al. 1993; Hobbie and Vitousek 2000). In our experiment, however, the higher quality O. pinnata litter when compared to the lower quality litter S. superba did not significantly increase litter decomposition rates. This is probably because soil N availability is not a limiting factor in these forests and, especially in the BF (Mo et al. 2006). In addition, decomposer communities are often adapted to degrade the type of leaf litter that they encounter most often, which typically comes from the plant species growing above them (Ayres et al. 2009; Strickland et al.; 2009). This results in litter decomposing more rapidly in its ‘home’ environment than in an ‘away’ environment (Hunt et al.; 1988), that had been called the ‘home-field advantage’ (Gholz et al.; 2000). Unlike S. superba, O. pinnata does not grow in the three forests used in our study, and the soil microorganisms in these forests therefore have been less well adapted to metabolizing O.pinnata litter than S.superba litter. S. superba litter also contains more calcium than O. pinnata litter, and a higher content of calcium could enhance decomposition in this area (Waring 2012).
Nutrient availability in the surface soil may also affect litter decomposition (Prescott et al. 1993; Hobbie and Vitousek 2000; Cleveland and Townsend 2006). Kaspari et al. (2008) proposed that multiple nutrients limited litter decomposition in a tropical forest. At our study site, the BF is in a late stage of succession, and the nutrient availability in surface soil is higher in the BF than in the other two forests (Mo et al. 2006; Fang et al. 2009). The higher nutrient availability may help explain why the rate of decomposition was highest in the BF, because higher nutrient availability in the BF may stimulate soil microbial productivity and multiple enzyme activities to decompose organic matter (Allison and Vitousek 2005; Kaspari et al. 2008). In addition, our results indicated that soil water content significantly increases from early to late successional forests. Increased water availability should promote soil microbial processes such as litter decomposition and nutrient mineralization (Niklaus et al. 1998; Cusack et al. 2009). These results suggest that nutrient and water availability in the surface soil may help explain why the increase in litter input enhanced litter decomposition more in the BF than in the MF and PF.
Effects of changes in litter input on soil respiration
According to previous research, an increased in litter input leads to an increased labile organic matter in the surface soil, which enhances microbial activity and generally increases soil respiration (Cleveland et al. 2007; Liu et al. 2009; Crow et al. 2009; Chemidlin Prévost-Bouré et al. 2010; Sayer et al. 2011; Leff et al. 2012). Consistent with these earlier results and with our hypothesis, our study showed that soil respiration significantly increased as litter input increased regardless of litter type or forest type (Fig. 2). The increase in soil respiration with an increase in litter input might be at least partially explained by the associated increases in SMBC and decomposition rates. Ngao et al. (2012) found that aboveground organic residue was the main contributor to soil CO2 efflux in a tropical Eucalyptus forest. The litter layer is thought to greatly affect soil CO2 efflux in tropical climates because of high decomposition rates in that layer (Aerts 1997; Couteaux et al. 1995). Previous studies have noted that increases in litter inputs may cause priming (Kuzyakov et al. 2000), which could accelerate the decomposition of older organic matter and result in increases in soil respiration (Fontaine et al. 2004; Sayer et al. 2011). Thus, the increase in soil respiration with an increase in litter input in our study may also be explained by priming effects (Fontaine et al. 2007; Sayer et al. 2007, 2011; Schaefer et al. 2009).
In the BF and MF, soil respiration rates were higher with S. superba litter than with O.pinnata litter. These higher respiration rates are likely associated with the more rapid decomposition of S. superba litter than O. pinnata litter in these two forests (Aerts 1997; Couteaux et al. 1995; Ngao et al. 2012). In the PF, however, soil respiration rates were higher in the A. mangium stand with O. pinnata litter input than in the S. superba stand with S. superba litter input. Relative to the 0 g-litter treatment, addition of 8 g of litter to the cylinders increased the annual average soil respiration by 41.5 % in the stand treated with O. pinnata litter but only by 27.7 % in the stand treated with S. superba litter. The higher soil respiration with input of O. pinnata litter at the PF could be explained by the higher background soil respiration in the A. mangium stand than in the S. superba stand. This result is also consistent with our findings that addition of 8 g of litter per cylinder at the PF increased annual SMBC by 48.3 % in the S. superba stand but only 28.4 % in the A. mangium stand. Overall, soil respiration was increased more by an increase in the input of S. superba litter than O. pinnata litter.
Effects of changes in litter input on soil organic C storage
We found that increases in litter input did not significantly affect surface SOC content (Table 2; Fig 3). Our results agree with earlier studies which found that increased litter input caused non-significant or only subtle effects on surface SOC content (Nadelhoffer et al. 2004; Hoosbeek and Scarascia-Mugnozza 2009; Crow et al. 2009; Lajtha et al. 2013). Other studies, in contrast, reported that litter addition either increased or decreased SOC pools (Liu et al. 2009; Leff et al. 2012; Fekete et al. 2014; Fontaine et al. 2007; Schaefer et al. 2009). These discrepancies can be explained by the complexity of the underlying biological processes and interactions and also by differences in study duration, forest type, soil depth, and the quantity and type of litter added (Yano et al. 2005; Sayer 2006; Liu et al. 2009; Lajtha et al. 2014). Contrary to our expectations, litter quality did not influence SOC content. In a microcosm study, Liu et al. (2009) also found that changes in litter chemistry did not alter SOC concentrations. The results of our study suggest that surface C pools are not very sensitive to changes in leaf litter inputs over short timescales.
Litter contains a highly soluble fraction that may rapidly move into soils with rainfall and may be retained in mineral soils (McDowell and Likens 1988; Cleveland et al. 2004). Although earlier studies reported that litter addition increased DOC concentration in mineral soils (Liu et al. 2009; Leff et al. 2012), we found that DOC content in the soil was unaffected by litter quantity or quality in any of the forest types. This lack of effect can be explained in several ways. First, soil DOC content varies greatly with season (Cleveland et al. 2004; Crow et al. 2009), and we may have missed differences in DOC content because our sampling interval have been too long. Second, soil DOC content can vary greatly with depth and we sampled only the surface soil (Kalbitz et al. 2005; Lajtha et al. 2005). In a long-term litter manipulation experiment, Crow et al. (2009) showed that DOC concentrations in soil solutions collected at 30 cm depth were greater in control plots than in double litter plots, it was suggested that long-term priming reduced mineralizeable C, thus reducing DOC losses from the double litter plots. In this short-term study, priming was also likely to occur, thus led to reduced DOC content in the higher litter addition treatments. Because the leaching of DOC from the litter layer and its retention in the mineral soil is a complex process (McDowell and Likens 1988; Cleveland et al. 2004; Kalbitz et al. 2005; Lajtha et al. 2005; Crow et al. 2009), understanding the changes in soil DOC following litter enrichment at our study site will require additional research.
Whether climate change will lead to an increase or decrease in the soil C pool will depend on the balance between inputs of above-and belowground litter and outputs from decomposition (Scholes et al. 1997). In our study, SOC content did not change with increasing litter input, suggesting that the increased C input was balanced by losses to soil respiration/leaching over short timescales. Although increased litter input may lead to accelerated decomposition of some stored SOC due to priming (Fontaine et al. 2004; Sayer et al. 2011), some of the increased C input may move into stabilized pools of soil C (Hyvönen et al. 2007; Hoosbeek et al. 2007) and result in a net soil C storage (Crow et al. 2009). However, in a 50-year litter manipulation experiment, Lajtha et al. (2014) found slow rates of C accumulation in response to litter additions, which was due to the increases in the light density fraction C pool. These results suggest that that a better understanding of how different litter-derived C sources are moved, mineralized, and stabilized in soil in the long-term may provide insight into how climate change, by altering litter quality and quantity, affects soil C storage.
In conclusion, we found that an increase in the quantity of litter added to soil resulted in increased rates of litter decomposition, litter C loss, and soil respiration in three subtropical forests representing different stages of succession. The effect was not always significant, but an increase in litter addition also resulted in higher SMBC content. Although the S. superba litter contained less N and P than the O.pinnata litter, the increases in decomposition, litter C loss, and soil respiration in response to increases in litter input were greater with S. superba litter than with O. pinnata litter, perhaps because the soil microorganisms at our study sites were better adapted to metabolizing S. superba litter (Gholz et al. 2000). Our results also suggest that the responses of litter decomposition, soil respiration, and SMBC to increases in litter input differ among forest successional stages. When measured 2.5 years after the start of the experiment, however, SOC content was not affected by the quantity of litter added regardless of litter type or forest type. Additional research is needed to determine whether these findings can be extrapolated to longer time scales.
References
Aerts R (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449
Allison SD, Vitousek PM (2005) Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol Biochem 37:937–944
Ayres E, Steltzer H, Berg S, Wall DH (2009) Soil biota accelerate decomposition in high-elevation forests by specializing in the breakdown of litter produced by the plant species above them. J Ecol 97:901–912
Booker FL, Prior SA, Torbert HA, Fiscus EL, Pursley WA, Hu SJ (2005) Decomposition of soybean grown under elevated concentrations of CO2 and O3. Glob Chang Biol 11:685–698
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil-nitrogen - a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842
Buol SW, Southard RJ, Graham RC, McDaniel PA (2003) Soil genesis and classification, 5th edn. Iowa State Press, Iowa
Chemidlin Prévost-Bouré N, Soudani K, Damesin C, Berveiller D, Lata JC, Dufrêne E (2010) Increase in aboveground fresh litter quantity over-stimulates soil respiration in a temperate deciduous forest. Appl Soil Ecol 46:26–34
Clark DB, Clark DA, Oberbauer SF (2010) Annual wood production in a tropical rain forest in NE Costa Rica linked to climatic variation but not to increasing CO2. Glob Chang Biol 16:747–759
Cleveland CC, Townsend AR (2006) Nutrient additions to a tropical rain forest drive substantial soil carbon dioxide losses to the atmosphere. PANS 103:10316–10321
Cleveland CC, Neff JC, Townsend AR, Hood E (2004) Composition, dynamics, and fate of leached dissolved organic matter in terrestrial ecosystems: results from a decomposition experiment. Ecosystems 7:175–285
Cleveland CC, Reed SC, Townsend AR (2006) Nutrient regulation of organic matter decomposition in a tropical rain forest. Ecology 87:492–503
Cleveland CC, Nemergut DR, Schmidt SK, Townsend AR (2007) Increases in soil respiration following labile carbon additions linked to rapid shifts in soil microbial community composition. Biogeochemistry 82:229–240
Couteaux M, Bottner P, Berg B (1995) Litter decomposition, climate and litter quality. Trends Ecol Evol 10:63–66
Crow SE, Lajtha K, Bowden RD, Yano Y, Brant JB, Caldwell BA, Sulzman EW (2009) Increased coniferous needle inputs accelerate decomposition of soil carbon in an old-growth forest. For Ecol Manag 258:2224–2232
Cusack DF, Chou WW, Yang WH, Harmon ME, Silver WL, Team TL (2009) Controls on long-term root and leaf litter decomposition in neotropical forests. Glob Chang Biol 15:1339–1355
Doughty CE, Malhi Y, Araujo-Murakami A, Metcalfe DB, Silva-Espejo JE, Arroyo L et al (2014) Allocation trade-offs dominate the response of tropical forest growth to seasonal and interannual drought. Ecology 95:2192–2201
Ellsworth DS, Thomas R, Crous KY, Palmroth S, Ward E, Maier C, Delucia E, Oren R (2012) Elevated CO2 affects photosynthetic responses in canopy pine and subcanopy deciduous trees over 10 years: a synthesis from Duke FACE. Glob Chang Biol 18:223–242
Fang H, Mo JM, Peng SL, Li Z, Wang H (2007) Cumulative effects of nitrogen additions on litter decomposition in three tropical forests in southern China. Plant Soil 293:233–242
Fekete I, Kotroczo Z, Varga C, Nagy PT, Varbiro G, Bowden RD, Toth JA, Lajtha K (2014) Alterations in forest detritus inputs influence soil carbon concentration and soil respiration in a Central-European deciduous forest. Soil Biol Biochem 74:106–114
Fontaine S, Bardoux G, Abbadie L, Mariotti A (2004) Carbon input to soil may decrease soil carbon content. Ecol Lett 7:314–320
Fontaine S, Barot S, Barre P, Bdioui N, Mary B, Rumpel C (2007) Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 450:277–280
Gholz HL, Wedin DA, Smitherman SM, Harmon ME, Parton WJ (2000) Long term dynamics of pine and hardwood litter in contrasting environments: toward a global model of decomposition. Glob Chang Biol 6:751–765
Hedin LO, Vitousek PM, Matson PA (2003) Nutrient losses over four million years of tropical forest development. Ecology 84:2231–2255
Hickler T, Smith B, Prentice IC, Mjofors K, Miller P, Arneth A, Sykes MT (2008) CO2 fertilization in temperate FACE experiments not representative of boreal and tropical forests. Glob Chang Biol 14:1531–1542
Hobbie SE, Vitousek PM (2000) Nutrient limitation of decomposition in Hawaiian forests. Ecology 81:1867–1877
Hoosbeek MR, Scarascia-Mugnozza GE (2009) Increased litter build up and soil organic matter stabilization in a poplar plantation after 6 years of atmospheric CO2 enrichment (FACE): final results of POP-EuroFACE compared to other forest FACE experiments. Ecosystems 12:220–239
Hoosbeek MR, Vos JM, Meinders MBJ, Velthorst EJ, Scarascia-Mugnozza GE (2007) Free atmospheric CO2 enrichment (FACE) increased respiration and humification in the mineral soil of a poplar plantation. Geoderma 138:204–212
Huang WJ, Liu JX, Wang YP, Zhou GY, Han TF, Li Y (2013) Increasing phosphorus limitation along three successional forests in southern China. Plant Soil 362:181–191
Hunt HW, Ingham ER, Coleman DC, Elliott ET, Reid CPP (1988) Nitrogen limitation of production and decomposition in prairie, mountain meadow, and pine forest. Ecology 69:1009–1016
Hyvönen R, Agren GI, Linder S, Persson T, Cotrufo MF, Ekblad A, Freeman M, Grelle A, Janssens IA et al (2007) The likely impact of elevated CO2, nitrogen deposition, increased temperature and management on carbon sequestration in temperate and boreal forest ecosystems: a literature review. New Phytol 173:463–480
Jastrow JD, Michael MR, Matamala R, Norby RJ, Boutton TW, Rice CW, Owensby CE (2005) Elevated atmospheric carbon dioxide increases soil carbon. Glob Chang Biol 11:2057–2064
Kalbitz K, Schwesig D, Rethemeyer J, Matzner E (2005) Stabilization of dissolved organic matter by sorption to the mineral soil. Soil Biol Biochem 37:1319–1331
Kaspari M, Garcia MN, Harms KE, Santana M, Wright SJ, Yavitt JB (2008) Multiple nutrients limit litterfall and decomposition in a tropical forest. Ecol Lett 11:35–43
King JS, Pregitzer KS, Zak DR, Holmes WE, Schmidt K (2005) Fine root chemistry and decomposition in model communities of north-temperate tree species show little response to elevated atmospheric CO2 and varying soil resource availability. Oecologia 146:318–328
Kuzyakov Y, Friedel JK, Stahr K (2000) Review of mechanisms and quantification of priming effects. Soil Biol Biochem 32:1485–1498
Lajtha K, Crow SE, Yano Y, Kaushal SS, Sulzman E, Sollins P, Spears JDH (2005) Detrital controls on soil solution N and dissolved organic matter in soils: a field experiment. Biogeochemistry 76:261–281
Lajtha K, Peterson F, Nadelhoffer K, Bowden R (2013) Twenty years of litter and root manipulations: insights into multidecadal som dynamics. Soil Sci Soc Am J 67:1418–1427
Lajtha K, Townsend KL, Kramer MG, Swanston C, Bowden RD, Nadelhoffer K (2014) Changes to particulate versus mineral-associated soil carbon after 50 years of litter manipulation in forest and prairie experimental ecosystems. Biogeochemistry 119:341–360
Leff JW, Wieder WR, Taylor PG, Townsend AR, Nemergut DR, Grandy AS, Cleveland CC (2012) Experimental litterfall manipulation drives large and rapid changes in soil carbon cycling in a wet tropical forest. Glob Chang Biol 18:2969–2979
Lichter J, Barron SH, Bevacqua CE, Finzi AC, Irving KF, Stemmler EA, Schlesinger WH (2005) Soil carbon sequestration and turnover in a pine forest after six years of atmospheric CO2 enrichment. Ecology 86:1835–1847
Liu LL, King JS, Giardina CP (2005) Effects of elevated concentrations of atmospheric CO2 and tropospheric O3 on leaf litter production and chemistry in trembling aspen and paper birch communities. Tree Physiol 25:1511–1522
Liu LL, King JS, Booker FL (2009) Enhanced litter input rather than changes in litter chemistry drive soil carbon and nitrogen cycles under elevated CO2: a microcosm study. Glob Chang Biol 15:441–453
Liu JX, Huang WJ, Zhou GY, Zhang DQ, Liu SZ, Li YL (2013) Nitrogen to phosphorus ratios of tree species in response to elevated carbon dioxide and nitrogen addition in subtropical forests. Glob Chang Biol 19:208–216
Martens R (1995) Current methods for measuring microbial biomass C in soil: potentials and limitations. Biol Fertil Soils 19:87–99
Martínez-Vilalta J, Lloret F, Breshears DD (2012) Drought-induced forest decline: causes, scope and implications. Biol Lett 8:689–691
McDowell WH, Likens GE (1988) Origin, composition, and flux of dissolved organic carbon in the Hubbard Brook Valley. Ecol Monogr 58:177–195
Mo JM, Brown S, Xue JH, Fang YT, Li ZA (2006) Response of litter decomposition to simulated N deposition in disturbed, rehabilitated and mature forests in subtropical China. Plant Soil 282:135–151
Nadelhoffer K, Boone R, Bowden RD et al (2004) The dirt experiment: litter and root influences on forest soil organic matter stocks and function. In: Foster DR, Aber JD (eds) Forests in time: the environmental consequences of 1,000 years of change in New England. Yale University Press, New Haven, pp 300–315
Nelson DW, Sommers LE (1982) Carbon and organic matter. In: Page, Mille AL, Keeney RH (eds) Methods of soil analysis-part 2: chemical and microbiological properties. American Society of Agronomy, Madison, pp 561–579
Ngao J, Epron D, Delpierre N, Bréda N, Granier A, Longdoz B (2012) Spatial variability of soil CO2 efflux linked to soil parameters and ecosystem characteristics in a temperate beech forest. Agric For Meteorol 154:136–146
Niklaus PA, Spinnler D, Kornerb C (1998) Soil moisture dynamics of calcareous grassland under elevated CO2. Oecologia 117:201–208
Norby RJ, Cotrufo MF, Ineson P, O’eill EG, Canadell JG (2001) Elevated CO2, litter chemistry, and decomposition: a synthesis. Oecologia 127:153–165
Norby RJ, DeLucia EH, Gielen B, Calfapietra C et al (2005) Forest response to elevated CO2 is conserved across a broad range of productivity. PANS 102:18052–18056
Nottingham AT, Turner BL, Chamberlain PM, Stott AW, Tanner EVJ (2012) Priming and microbial nutrient limitation in lowland tropical forest soils of contrasting fertility. Biogeochemistry 111:219–237
Ostertag R, Scatena FN, Silver WL (2003) Forest floor decomposition following hurricane litter inputs in several Puerto Rican forests. Ecosystems 6:261–273
Prescott CE, Taylor BR, Parsons WFJ, Durall DM, Parkinson D (1993) Nutrient release from decomposing litter in Rocky Mountain coniferous forests: influence of nutrient availability. Can J For Res 23:1576–1586
Raich JW, Russell AE, Kitayama K, Parton WJ, Vitousek PM (2006) Temperature influences carbon accumulation in moist tropical forests. Ecology 87:76–87
Sayer EJ (2006) Using experimental manipulation to assess the roles of leaf litter in the functioning of forest ecosystems. Biol Rev 81:1–31
Sayer EJ, Powers JS, Tanner EVJ (2007) Increased litterfall in tropical forests boosts the transfer of soil CO2 to the atmosphere. PLoS One 2:e1299
Sayer EJ, Heard MS, Grant HK, Marthews TR, Tanner EVJ (2011) Soil carbon release enhanced by increased tropical forest litterfall. Nat Clim Chang 1:304–307
Sayer EJ, Joseph Wright S, Tanner E et al (2012) Variable responses of lowland tropical forest nutrient status to fertilization and litter manipulation. Ecosystems 15:387–400
Schaefer DA, Feng WT, Zou XM (2009) Plant carbon inputs and environmental factors strongly affect soil respiration in a subtropical forest of southwestern China. Soil Biol Biochem 41:1000–1007
Scholes MC, Powlson D, Tian GL (1997) Input control of organic matter dynamics. Geoderma 79:25–47
Strickland MS, Osburn E, Lauber C, Fierer N, Bradford MA (2009) Litter quality is in the eye of the beholder: initial decomposition rates as a function of inoculum characteristics. Funct Ecol 23:627–636
Sylvia DM, Fuhrman JJ, Hartel PG, Zuberer DA (1998) Principles and applications of soil microbiology. Prentice-Hall, Englewood Cliffs
Vincent AG, Turner BL, Tanner EVJ (2010) Soil organic phosphorus dynamics following perturbation of litter cycling in a tropical moist forest. Eur J Soil Sci 61:48–57
Vitousek PM, Sanford RL (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Syst 17:137–167
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19
Wang BS, Ma MJ (1982) The successions of the forest community in Dinghushan. Trop Subtrop For Ecosyst Res 1:142–156 (in Chinese)
Waring BG (2012) A meta-analysis of climatic and chemical controls on leaf litter decay rates in tropical forests. Ecosystems 15:999–1009
Wood TE, Lawrence D (2008) No short-term change in soil properties following four-fold litter addition in a Costa Rican rain forest. Plant Soil 307:113–122
Wood TE, Lawrence D, Clark DA, Chazdon RL (2009) Rain forest nutrient cycling and productivity in response to large-scale litter manipulation. Ecology 90:109–121
Yano Y, Lajtha K, Sollins P et al (2005) Chemistry and dynamics of dissolved organic matter in a temperate coniferous forest on Andic soils: effects of litter quality. Ecosystems 8:286–300
Zhou GY, Liu SG, Li Z, Zhang DQ, Tang XL, Zhou CY, Yan JH, Mo JM (2006) Old-growth forests can accumulate carbon in soils. Science 314:1417–1417
Zhou GY, Guan LL, Wei XH, Zhang DQ, Zhang QM, Yan JH, Wen DZ, Liu JX, Liu SG, Huang ZL (2007) Litterfall production along successional and altitudinal gradients of subtropical monsoon evergreen broadleaved forests in Guangdong, China. Plant Ecol 188:77–89
Zhou GY, Peng CH, Li YL, Liu SZ, Zhang QM, Tang XL, Liu JX, Yan JH, Zhang DQ (2013) A climate change-induced threat to the ecological resilience of a subtropical monsoon evergreen broad-leaved forest in Southern China. Glob Chang Biol 19:1197–1210
Acknowledgments
This study was jointly funded by the National Natural Science Foundation of China (Grant Nos.31370530 and 31070439), the Science and Technology Innovation Project of Guangdong Province Forestry (Grant No.2012KJCX019-02) and the South China Botanical Garden-Shanghai Institute of Plant Physiology & Ecology Joint Fund.
Ethical Statement
The authors declare that this manuscript is an original paper, has not been published, and is not currently submitted to any other journals for review. This manuscript is a single study, and is not split up into several parts to increase the quantity of submissions and submitted to various journals. No data have been fabricated to support my conclusions. The submission for publication has been approved by all relevant authors. The authors also declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Kees Jan van Groenigen.
Rights and permissions
About this article
Cite this article
Fang, X., Zhao, L., Zhou, G. et al. Increased litter input increases litter decomposition and soil respiration but has minor effects on soil organic carbon in subtropical forests. Plant Soil 392, 139–153 (2015). https://doi.org/10.1007/s11104-015-2450-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2450-4