Abstract
Background
Overyielding (i.e., mixtures of crops yielding higher than expected when compared with monocultures) and increased nutrient acquisition have been found in many intercropping systems. However, there are very few published studies on long-term changes in soil chemical and biological properties in intercropping systems compared to sole cropping.
Methods
A field experiment was established in 2003 in Gansu province, northwest China. The treatments comprised three intercropping systems (either continuous or rotational wheat/maize, wheat/faba bean, maize/faba bean intercropping), rotational cropping (wheat-maize, wheat-faba bean, faba bean-maize, and wheat-maize-faba bean rotations), and monocropping (sole wheat, faba bean and maize) systems. In 2011 (ninth year of the experiment) and 2012 (tenth year) the yields and some soil chemical and biological properties were examined after all crop species were harvested.
Results
There was overyielding by 6.6 % and 32.4 % in wheat/maize intercropping in 2011 and 2012, respectively. Faba bean/maize intercropping was enhanced by 34.7 % and 28.6 %, respectively but not wheat/faba bean intercropping. Soil organic matter, total nitrogen, Olsen P, exchangeable K and cation exchange capacity in all intercropping systems did not differ from the monocultures except for soil pH in wheat/maize and faba bean/maize intercropping in 2011 and soil exchangeable K and cation exchange capacity (CEC) in 2012. Soil pH in wheat/maize and faba bean/maize intercropping was significantly reduced by 3.2 % and 1.9 %, respectively. Soil exchangeable K in wheat/maize, faba bean/maize and wheat/faba bean intercropping declined markedly by 15 %, 21.7 % and 12.1 %, respectively. Soil cation exchange capacity in wheat/maize, faba bean/maize and wheat/faba bean intercropping was notably lower than the corresponding monocultures by 17.5 %, 23.3 % and 18.3 %, respectively. Soil enzyme activities after 9 and 10 years of intercropping differed little from monocultures or rotations.
Conclusions
The results indicate that intercropping overyielded compared with monocropping or rotational cropping and also maintained the stability of most of the soil chemical and enzyme activities relative to rotations and monocropping in the relatively fertile soil studied.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Intercropping has been long practiced in many parts of the world (Francis 1986). More than 28 million hectares are sown annually in China (Zou and Li 2002) and intercropping is also common in other parts of the world such as Indonesia, India, Niger, Mali, central America and western Europe (Zomer et al. 2009). More than one-third of the areas of cassava (Manihot esculenta L. var. variegata) and bananas (Musa paradisiaca L. var. sapientum O. Ktze.) grown in the Americas and Africa are intercropped (Leihner 1983; Mucheru-Muna et al. 2010). In northwest China 75,100 ha of intercropping in Ningxia province produced 43 % of regional total grain yields in 1995 and there are 200,000 ha in Gansu province with an annual grain yield of 12 t ha−1 (Li et al. 2001a).
Thus, a wide range of intercropping has been developed because of significant increases in productivity compared with monocultures (Gregorich et al. 2001; Li et al. 2007). Intercropping enhances water, nutrient and energy efficiency (Francis 1989), reduces environmental pollution (Stuelpnagel 1992), increases LER (LER is defined as the relative land area required as sole crops to produce the same yields as intercropping) (Keating and Carberry 1993; Morris and Garrity 1993a, b), reduces the risk of crop failure and increases food security (Rusinamhodzi et al. 2012).
Intercropping can provide significant overyielding (i.e., mixtures of crops performing better than expected when compared with monocultures) and nutrient acquisition advantages. Underlying mechanisms comprise border row effects and below-ground interspecific interactions and facilitation (Fortin et al. 1994; Lesoing and Francis 1999; Li et al. 2001a). Overyielding has been observed in many intercropping systems such as maize/soybean, sorghum/soybean, maize/cowpea, wheat/mungbean, wheat/chickpea and maize/faba bean. In a 4-year field experiment maize over-yielded on average by 43 % and faba bean by 26 % in a low-phosphorus calcareous soil (Li et al. 2007). A similar result was obtained in maize/faba bean intercropping in Ningxia Hui Autonomous Region, northwest China (Mei et al. 2012). Yield advantage of 21–25 % compared with monoculture has also been reported in soybean/pigeonpea intercropping in India (Ghosh et al. 2006). Martin et al. (1998) found that shoot biomass increased by 21 % in faba bean/maize intercropping. Similar results have been reported for pea/barley and maize/cowpea intercropping in Denmark and Iran, respectively (Jensen 1996; Dahmardeh et al. 2010).
There are very close relationships between yield advantage and nutrient acquisition in intercropping systems (Morris and Garrity 1993a). Observations have focused on nutrient acquisition in intercropping systems such as maize/faba bean which significantly enhanced total N and P by over 50 % compared with monoculture in Gansu (Li et al. 2003, 2011a). In a newly-reclaimed desert soil our previous study showed that in maize/faba bean intercropping P acquisition was 17.9–29.6 % greater than in corresponding monocultures (Mei et al. 2012). Similar results have been found in wheat/bean intercropping nutrient uptake compared with sole crops in Africa and N acquisition in pea/barley intercropping in Denmark (Eskandari 2011; Hauggaard-Nielsen and Jensen 2001). One question is whether increased nutrient removals from soil by intercropping leads to declining soil fertility. At present there is little information available on soil fertility in the long term in intercropping systems compared with monoculture or rotational cropping systems.
Soil fertility, including physical, chemical and biological properties, plays an important role in determining crop yields in agricultural ecosystems (Doran 2002; Pellegrino et al. 2011; Wei et al. 2006). Soil fertility has been defined as the capacity of a specific soil type to function and to sustain plant productivity and maintain or enhance water and air quality (Karlen et al. 1997). Numerous studies have been performed recently on soil fertility in relation to fertilization, irrigation, tillage or management (Mäder et al. 2002; Melero et al. 2006, 2007). Mäder et al. (2002) stated that nutrient inputs in organic systems are around 42.5 % on average lower than in conventional systems and mean crop yields were 21 % lower over a 21-year period in Switzerland with soil fertility greatly enhanced by organic farming (Mäder et al. 2002). Other studies have demonstrated that soil TOC, N, microbial biomass and soil enzyme activities in zero tillage were significantly higher than conventional or rotational systems across 5 years (Alvear et al. 2005; Madari et al. 2005; Roldan et al. 2007). As noted above, studies may suggest effects of cropping systems on soil chemical and biological properties. A perennial grassland with a high diversity of plant species stored 500 % more C and 600 % more N on average than did monocultures through greater root biomass accumulation to 60 cm soil depth by highly complementary functional groups from 1994 to 2006 in Minnesota in the US (Dybzinski et al. 2008; Fornara and Tilman 2008; Fornara et al. 2009). The yield and nutrient advantages in intercropping or mixtures started to appear even in experiments of short duration.
However, there are few studies that have focused on soil fertility in intercropping relative to monocultures and rotations at a longer time scale. The objective of the present study was therefore to further test the occurrence of grain yield and nutrient acquisition advantages in terms of changes in selected soil chemical properties and enzyme activities after 9–10 years in intercropping compared to monocultures and rotations at appropriate N and P fertilizer application rates but without application of potassium fertilizer or farmyard manure.
Materials and methods
Site description
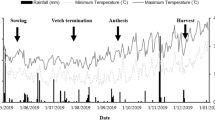
The study was conducted at the Experimental Station of the Institute of Soils, Fertilizers and Water-Saving Agriculture, Gansu Academy of Agricultural Sciences, at Baiyun (38°37’N,102°40’E) located 15 km north of Wuwei city in Gansu province, northwest China at 1504 m height above sea level. The soil is a sandy loam which contains 57 % sand, 39 % silt, and 4 % clay (International Society of Soil Science). Topsoil (0–20 cm) bulk density is 1.40 g cm−3 and the physico-chemical properties were: soil organic matter (OM) 19.1 g kg−1, total N 1.18 g kg−1, Olsen P 20.3 mg kg−1, exchangeable K 233 mg kg−1 and pH value (1:2.5 soil:DI water) 8.0 in 2003 before the start of the experiment. The soil type has been classified as an Orthic Antrosols (FAO/UNESCO 1988) and the location has a typically arid climate. Most precipitation falls between May and September and the total precipitation and potential evaporation were about 150 and 2021 mm averaged over the 13 years from 2000 to 2012, respectively, according to Wuwei Meterological Station near the experimental site. The growing season is usually from the middle of March until October. Average annual air temperature is 7.7 °C and cumulative temperatures above 0 °C and 10 °C are 3646 °C and 3149 °C, respectively. The frost-free period is 170–180 days and total solar radiation is 5988 MJ m−2 year−1.
Experimental design and management
The long-term field experiment was designed with 13 treatments and three replicates. All treatments were as shown in Table 1. The treatments comprised six intercropping systems, four rotations and three monocultures. One intercropping combination included 0.8 m faba bean or maize or wheat strip (four rows of faba bean with 0.2 m inter-row distance, two rows of maize with 0.4 m inter-row distance, or six rows of wheat with 0.133 m inter-row distance) and 0.8 m associated crop strip (six rows of wheat, two rows of maize or four rows of faba bean), so that two crop strips can be exchanged in the subsequent year for the rotational intercropping treatment. In intercropping the wheat density within each row was the same as in the monoculture wheat. Inter-row distance in monocropping was 0.20 m for faba bean, 0.40 m for maize, and 0.133 m for wheat. Inter-plant distance within the same row was 0.2 m for faba bean and 0.25 m for maize in intercropping and monocropping. These row and density arrangements made the planting density in monocropping or rotational cropping identical to intercropping on a comparable area. The same soil preparation, row spacing, fertilization, irrigation, and harvesting procedures were used for 10 years. One half of each intercropped area was occupied by wheat, maize or faba bean so that the overall proportional density of each crop species was equal in both the monoculture and intercropping treatments.
The field experiment was established in 2003 and all treatments received 225 kg N ha−1 year−1 as Urea (CO(NH2)2) and 40 kg P ha−1 year−1 as Calcium Superphosphate (Ca(H2PO4)2.H2O) in accordance with conventional agricultural practice in the region based on previous studies (Li et al. 2001a, 2003). The large amounts of N and P removed by crops were replaced by even broadcasting all P and half of the fertilizer N and incorporating into the soil before sowing. The other half of the N was top-dressed at the first irrigation (15th July, this duration was the highest water evaporation time span of the soil and the maize utilized more water than other crops, according to soil water conditions and crop utilization) or divided into two portions applied at the elongation and pre-tasselling stages of monoculture or intercropped maize. All plots were irrigated 6–7 times in the case of wheat/maize, faba bean/maize intercropping and sole cropping maize, and three to four times in the case of wheat/faba bean intercropping and sole cropping faba bean and wheat, according to the conventional farming practice in this area, to prevent water stress. Each irrigation event comprised around 75 mm.
The cultivars used in the long-term experiment were No. 2014 for wheat (Triticum aestivum L.), Zhengdan No. 958 for maize (Zea mays L.) and Lincan No. 5 for faba bean (Vicia faba L.) in both 2011 and 2012. After harvest the residues of previous crop species were ploughed into the soil and the new crop species were sown the following year. Grain yield and straw biomass of wheat, faba bean and maize were determined at maturity of the individual crop species by harvesting from a strip. Plant samples were air-dried and the grain was threshed by hand.
Soil and plant sampling and analysis
Soil samples were collected from the top 20 cm of the profile using an auger (35 mm diameter) after harvesting each crop species in both 2011 and 2012. Three soil cores were collected from each plot and combined to give one composite sample per plot for monocultures and rotations and there were two sampling sites for each crop strip per plot in intercropping. The composite samples were air-dried and sieved through a 2-mm mesh. Plant residues were removed by hand and the soil samples were placed in plastic bags for chemical analysis. Additional soil samples were collected using the same methods and stored at 4 ° C for 2 days until analysis for soil enzyme activities.
Soil organic matter (OM) was determined by wet oxidation using the acidified dichromate method (Bao 2000). Soil total N was measured after Kjeldahl digestion according to standard protocols (Bao 2000) (SKD-800, Peiou Corporation, Shanghai). Soil Olsen P was determined by colorimetry (Uvmini-1240, Shimadzu Corporation) using standard procedures (Olsen et al. 1954). Soil exchangeable K was extracted using 1 mol L−1 ammonium acetate (NH4OAc) solution buffered at pH 7 (Bao 2000) and determined by flame photometry (M410, Sherwood Corporation, UK). Soil CEC was measured by the Ammonium Acetate (1 M NH4OAc) method (Bao 2000) (M410, Sherwood Corporation, UK). Soil pH was measured in soil suspensions with deionized-distilled water (1:2.5 soil : DI water, w/v) (Rayment and Higginson 1992) (pHS-3C, SPSIC Corporation).
Soil urease activity was assayed using field-moist chloroform-fumigated soil samples (within 1 h of removal of the chloroform vapor by evacuation) and in non-fumigated subsamples in the presence and absence of toluene using the method described by Guan (1986).
Soil acid phosphatase activity was determined by the method of Tabatabai and Bremner (1969) using p-nitrophenyl phosphate disodium (PNPP) as substrate. On the basis of a modified universal buffer stock solution, the pH for the acid phosphatase analysis was adjusted to 6.5 with HCl. The pNP released by phosphatase was determined colorimetrically at 400 nm. Enzyme activity is expressed as micrograms of p-nitro phenol produced per gram of soil.
Soil nitrate reductase activity was determined by a colorimetric method (Guan 1986). Triplicate 5 g soil samples were incubated with 4 ml of 2,4-dinitrophenol solution, 1 ml potassium nitrate solution and 5 ml distilled water at 25 °C for 24 h. A similar set up was prepared for the control. The control sample was incubated at −20 °C for 24 h. After incubation, 10 ml 4 M KCl solution was added to all the soil samples including the control. This was shaken for 30 min and filtered. NH4Cl buffer (3 ml at pH 8.5) and 2 ml of color reagent were added to 5 ml of the filtrate and left for 15 min for color development. Optical density was determined in a spectrophotometer against the blank at 520 nm. The enzyme activity is expressed as micrograms of NO2 −–N per gram daily.
Soil sucrase activity was measured by the method of Guan (1986). Five grams of fresh soil were placed in a 50 mL Erlenmeyer flask together with 15 mL of 8 % sucrose solution, 5 mL phosphate buffer (pH 5.5) and 1 ml of toluene. The flask was shaken and then placed in an incubator at 37.0 ± 0.1 °C for 24 h. After incubation the sample was filtered through a quantitative filter paper. Then, 1 mL of the filtrate and 3 mL salicylic acid were taken to 50-mL in a volumetric flask and heated for 5 min at 100 °C in a water bath. After heating, the flask was cooled for 3 min with flowing tap water and deionized water was added to make up to 50 mL, and sucrase activity was measured colorimetrically at 508 nm (U-2800, Japan). Sucrase activity is expressed as mg glucose g soil−1 (24 h)−1.
At maturity, the dry matter yield of grain and aboveground parts was determined on samples collected by harvesting two continuous rows maize or six continuous wheat or four continuous faba bean in both the intercropping and monoculture and rotation treatments. Aboveground parts were ground (divided into grain and straw at maturity). Plant materials were over-dried at 70 °C for 48 h and ground. Nitrogen (N), phosphorus (P) and potassium (K) concentrations in grain and straw were determined on ground sub-samples of oven-dried plant material after digestion in a mixture of concentrated H2SO4 and H2O2. Nitrogen was measured by the micro-Kjeldahl procedure with 5 ml digestion solution P by the vanadomolydate method, and K by flame photometry (Bao 2000). Aboveground nutrient acquisition (N, P and K) of each crop was calculated as the sum of grain and straw nutrient acquisition which was determined as the product of nutrient concentration and grain or straw yield based on the land area occupied by the crops.
Calculations
Grain yields, aboveground biomass, nutrient acquisition, soil chemical properties or enzymes activities by wheat/maize, faba bean/maize and wheat/faba bean in intercropping with monoculture or rotation were compared. As intercropping comprised at least two crops, the crops and soil properties by intercropping can be compared with the weighted means of the two monoculture or rotation crop species based on the land area occupied and their proportions in the intercropping system.
The weighted means of grain yields by monocultures or rotation were calculated as follows:
Where \( {\mathrm{Y}}_{monoculture\_a} \) and \( {\mathrm{Y}}_{monoculture\_b} \) are the grain yields of crops a and b in the monoculture. P a and P b are the proportions of the area occupied by the respective crops in intercropping. P a is determined by \( {\mathrm{P}}_{{}_a} = {\mathrm{W}}_a/\left({\mathrm{W}}_{{}_{{}_{\mathrm{a}}}}+{\mathrm{W}}_{{}_{{}_b}}\right) \) and \( {\mathrm{P}}_{{}_b} = {\mathrm{W}}_{{}_b}/\left({\mathrm{W}}_{{}_{\mathrm{a}}}+{\mathrm{W}}_{{}_b}\right) \), where W a and W b are the widths of crops a and b in the intercropping strips.
Similarly, the weighted means of grain yields in rotation were calculated as follows:
The above equations were also used to calculate the weighted means of soil properties in monoculture or rotation.
Statistical analysis
The experiment was a completely randomized block design with three replicates. The original treatments consisted of 13 cropping systems (as described above) and summarized four cropping systems {monoculture (M), rotation (R), continuous intercropping [Inter (C)] and rotational intercropping [Inter (R)]}. The first factors were three crop combinations [wheat + maize (W + M), wheat + faba bean (W + F) and maize + faba bean (M + F)] and secondary factors were the four cropping systems [M, R, Inter (C) and Inter (R)]. All data from the factorially designed experiment were subjected to analysis of variance using SAS version 9.1 (SAS Institute 2003) and mean values (n = 3) were compared by least significant difference (LSD) at the 5 % level.
Results
Crop productivity
There were significant differences between crop species combinations in both grain yields and aboveground biomass in both 2011 and 2012. The productivity of the wheat + maize combination was higher than that of faba bean + maize which in turn was higher than that of the wheat + faba bean combination. The average grain yields of wheat + maize, faba bean + maize and wheat + faba bean combinations were 9.6, 8.0 and 4.6 Mg ha−1 and above-ground biomass values were 19.6, 14.9, and 10.0 Mg ha−1 across both years (Fig. 1).
Grain yields and aboveground biomass as affected by monoculture, rotation, continuous or rotation intercropping in 2011 and 2012. The error bars represent the standard deviation of the means of three replicates. Different lowercase letters or no letters with the same crop combination indicate significant differences at P < 5 % or no significant diffreences at P > 5 % by LSD among different cropping systems; different capital letters indicate significant differences at P < 5 % by LSD among different crop combinations, averaged over cropping systems. M, R, Inter (C) and Inter (R) represented that monoculture, rotation, continuous intercropping and rotational intercropping, respectively; W + M, F + M and W + F represented that wheat + maize, faba bean + maize, wheat + faba bean combination, respectively
In the wheat + maize combination intercropping enhanced significantly (p < 0.05) the grain yield productivity by 32.4 % compared to the weighted means of the corresponding monocrops in 2012 but there was no significant effect in 2011. Similar trends were observed for aboveground biomass in the combinations (Fig. 1). In the faba bean + maize combination continuous intercropping overyielded by 34.8 % compared to the weighted means of the corresponding monocrops in 2011 and by 28.7 % in 2012 (Fig. 1). In the wheat + faba bean combination there were no significant increases in productivity in terms of grain yield or aboveground biomass in intercropping (C) or (R) over rotational cropping, but a decline in grain yield of intercropping over monocropping was observed (Fig. 1).
Nutrient acquisition
Generally, the N, P and K acquisition by the wheat + maize combination was significantly (p < 0.05) greater than the faba bean + maize or wheat + faba bean combinations in both years with the exception of N acquisition in 2012. The nutrient acquisition of the wheat + maize combination was significantly higher than that of faba bean + maize, which was in turn significantly greater than that of wheat + faba bean, especially in 2011. The N acquisition of wheat + maize, faba bean + maize, and wheat + faba bean combinations ranged from 201 to 276 kg ha−1, P acquisition from 23 to 45 kg ha−1 and K acquisition from 160 to 359 kg ha−1 across both years (Table 2).
In a similar fashion to productivity, in the wheat + maize intercropping combination N acquisition increased by 6.0 % in 2011 and 33.2 % in 2012 compared with the weighted means of the corresponding monocultures in the corresponding years. P acquisition in this combination was enhanced by 28.0 % compared to the weighted means of the corresponding monocultures in 2012 but not in 2011. Similarly, K acquisition increased by 27.4 % in comparison with the weighted means of the monocultures, but only in 2012 (Table 2).
N, P and K acquisition of the faba bean/maize intercropping combination either rotationally or continuously was enhanced by 20.3 %, 20.0 % and 30.2 % compared with the weighted means of the corresponding monocultures or rotational cropping in 2012, with similar results in 2011 except for K acquisition (Table 2). The N, P and K acquisition values of the wheat + faba bean combination, in contrast, were not significantly affected by cropping system (Table 2).
Soil chemical properties
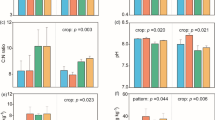
After 9 and 10 years, regardless of continuous or rotational system, the soil OM, total N, Olsen P, exchangeable K, CEC and pH values in rotational and continuous wheat/maize intercropping were 19.0 g kg−1, 1.32 g kg−1, 28.6 mg kg−1, 64.7 mg kg−1 12.1 cmol kg−1, and 8.09, respectively, which were not significantly (P > 0.05) different from the weighted means (19.6 g kg−1, 1.31 g kg−1, 34.0 mg kg−1, 68.0 mg kg−1, 12.8 cmol kg−1, and 8.22, respectively) of the corresponding monocultures and rotational cropping with the exception of soil pH in 2011 and exchangeable K and CEC in 2012 (Tables 3 and 4).
Maize/faba bean intercropping after 9 and 10 years did not alter significantly (p > 0.05) any of the soil chemical properties examined except exchangeable K, CEC and pH in 2012 compared to the corresponding monocrops and rotational systems (Tables 3 and 4). Soil chemical properties of rotational and continuous maize/faba bean intercropping vs rotational and monocultured cropping were 18.9 g kg−1 vs 19.3 g kg−1 for soil organic matter, 1.30 g kg−1 vs 1.29 g kg−1 for soil total N, 27.9 vs 31.8 mg kg−1 for Olsen-P, 68.3 mg kg−1 vs 73.4 mg kg−1 for exchangeable K, 11.6 cmol kg−1 vs 12.5 cmol kg−1 for CEC, and 8.02 vs 8.23 for soil pH (Tables 3 and 4).
Wheat/faba bean intercropping after 9 and 10 years did not change significantly (p > 0.05) any soil chemical properties determined in 2011 compared with the weighted means of the corresponding monocultures. Olsen P, exchangeable K, CEC and pH of soils in continuous or rotational intercropping decreased slightly compared to the weighted means of the corresponding monocultures or rotations in 2012 with the exception of OM and total N (Tables 3 and 4). Soil chemical properties of rotational and continuous maize/faba bean intercropping vs rotational and monoculture cropping were 19.0 g kg−1 vs 19.2 g kg−1 for soil organic matter, 1.31 g kg−1 vs 1.32 g kg−1 for soil total N, 33.7 vs 30.3 mg kg−1 for Olsen-P, 66.9 mg kg−1 vs 68.3 mg kg−1 for exchangeable K, 12.2 cmol kg−1 vs 13.1 cmol kg−1 for CEC, and 8.07 vs 8.10 for soil pH (Tables 3 and 4).
There were different trends in soil pH in 2011 and 2012 with intercropping both rotationally and continuously lowering soil pH to 8.33 in 2011 compared to 8.48 for rotational and monoculture cropping systems, and rotational and continuous intercropping reducing soil pH to 7.79 in 2012 compared to 7.92 for rotational cropping (Tables 3 and 4).
Soil enzyme activities
Wheat/maize intercropping after 9 and 10 years enhanced or did not change soil urease activity (1.76 mg NH4 +-N g−1 soil (24 h)−1 for continuous intercropping in 2011 and 2.43 NH4 +-N g−1 soil (24 h)−1 for both intercropping in 2012) compared with the weighted means (1.53 NH4 +-N g−1 soil (24 h)−1 in 2011 and 2.40 NH4 +-N g−1 soil (24 h)−1) of corresponding monocultures or rotations. Soil urease activity, ranging from 1.43 to 2.37 NH4 +-N g−1 soil (24 h)−1, did not differ significantly (p < 0.05) among intercropping, monoculture and rotational cropping systems for maize and faba bean combination in either year. Soil urease activity for wheat and faba bean combination, ranging from 1.48 to 2.97 NH4 +-N g−1 soil (24 h)−1, also did not differ significantly (p > 0.05) among intercropping, monoculture and rotational cropping systems in either year (Table 5).
Soil acid phosphatase activity for all of three crop combinations and all four cropping systems, ranging from 67 to 124 μg p-nitrophenol g−1 soil h−1, did not differ significantly (p > 0.05) among wheat/maize, faba bean/maize or wheat/faba bean intercropping and their corresponding monocultures or rotational cropping systems in either 2011 or 2012 with the exception of faba bean/maize intercropping in 2012 (Table 5). In 2012 soil acid phosphatase activity after rotational maize/faba bean intercropping was 95 μg p-nitrophenol g−1 soil h−1, which was significantly (p < 0.05) higher than in rotational cropping systems (Table 5).
Soil nitrate reductase activity, ranging from 3.83 to 7.79 μg NO2 −-N g−1 soil (24 h)−1 in wheat/maize intercropping, did not differ significantly (p > 0.05) from the weighted means (5.87 μg NO2 −-N g−1 soil (24 h)−1) of wheat and maize monocultures or rotations in 2011 or 2012. Faba bean/maize intercropping significantly reduced (p < 0.05) nitrate reductase activity to 3.69 μg NO2 −-N g−1 soil (24 h)−1 from 5.61 μg NO2 −-N g−1 soil (24 h)−1 for the weighted means of the rotation, but not significantly from 3.52 μg NO2 −-N g−1 soil (24 h)−1 for that of the monoculture in 2011 and 2012. Nitrate reductase actitity (5.32 μg NO2 −-N g−1 soil (24 h)−1) in wheat/faba bean intercropping was not significantly (p > 0.05) different from rotational and monoculture cropping systems (5.63 μg NO2 −-N g−1 soil (24 h)−1) in 2011 and 2012, with one exception in comparison with monoculture (3.36 μg NO2 −-N g−1 soil (24 h)−1) in 2012 (Table 6). Furthermore, after 9 and 10 years there was no significant difference in soil sucrase activity among wheat/maize, faba bean/maize and wheat/faba bean intercropping in 2011 or 2012 (Table 6).
Discussion
Continuous intercropping for 9–10 years continued to show overyielding and nutrient acquisition advantages. Irrespective of continuous or rotational cropping systems, productivities of wheat/maize and faba bean/maize intercropping were significantly higher than those of monocultures or rotations in 2012 but not in 2011. The results are consistent with our previous studies of wheat/maize (Li et al. 2001a) and maize/faba bean (Li et al. 2007) intercropping in terms of grain yield and biomass of aboveground parts. However, there was no yield advantage in wheat/faba bean intercropping. Yield advantage of wheat/maize intercropping is derived mainly from the “competition-recovery principle” where an earlier mature crop species acquires more resources and overyields and thereby suppresses the growth of the associated later mature crop species which has a compensated or recovery growth and finally overyielding after harvest of the early mature crop species (Li et al. 2001a, b). The advantage of faba bean/maize intercropping results mainly from interspecific facilitation between the two intercropped species (Li et al. 2007). Faba bean and wheat were sown at roughly the same time and were mature at about the same time (faba bean maturity 15–20 days later than wheat), thus there are neither interspecific facilitation nor competition-recovery processes in the wheat/faba bean intercropping. Other intercropping systems in other regions have also shown yield advantage of intercropping over monocultures (de Carvalho, Nunes, and de Oliveira 2009; Andrade et al. 2012; Rusinamhodzi et al. 2012). For instance, maize-based intercropping with ecological intensification increased productivity by 42.1 and 88.9 % in central Mozambique compared with monoculture (Rusinamhodzi et al. 2012). Tomato fruit yields increased by about 26 % in tomato/ryegrass (Lolium perenne L.) intercropping compared with sole crops (Carvalho et al. 2009) and there was higher productivity in sunflower/soybean intercropping than in sole cropping (Andrade et al. 2012).
As a consequence, intercropping removes more nutrients from the soil than corresponding monocrops and this has been observed in the present study and also in previous studies (Xia et al. 2013; Yang et al. 2013; Zhang and Li 2003). Faba bean/maize intercropping N acquisition enhancement ranged from 9 to 32 % compared with monocultures at different N levels (Li et al. 2011b). P acquisition of maize/faba bean intercropping increased by 29 and 28 % in a relatively fertile soil (Li et al. 2007) and by 23.5 % over several P application rates in comparison with monocultures in a reclaimed desert soil in northwest China (Mei et al. 2012). As observed in some studies, legume/cassava intercropping increased K acquisition by 44 % compared with cassava monoculture across several years (Qi et al. 2004). The question thus arises as to how soil fertility is influenced when nutrient removal from the soil is increased in intercropping systems.
Continuous intercropping maintains soil fertility in terms of chemical properties
The present study evaluated changes in soil chemical and biological properties after 9–10 years of intercropping in comparison with monoculture or rotation. In previous studies different results were obtained from different soil fertility conditions. For example, Thierfelder and Wall (2012) found that intercropping gave a 31 % increase in soil carbon content compared with conventional practice in a sandy soil with a low organic matter content (11.2 g kg−1). Our results are not consistent with these findings. The initial soil organic matter content was 19.1 g kg−1 before the start of the experiment and the average soil organic matter content across all of the intercropping treatments over 2 years was 18.9 g kg−1 after 9–10 years of intercropping and was almost stable. Cong et al. (2014) found that soil organic carbon content increased by 4 % by intercropping over monocropping using a C/N analyser after the soil was treated with 0.5 M HCl. The present study presents soil organic matter content using conventional chemical procedures, which likely derived from the difference between the methods.
Although soil total N content did not differ in intercropping from monocropping or rotational cropping, soil total N (1.20–1.42 g kg−1 soil) after 9 or 10 years increased in comparison with the initial value (1.18 g kg−1 soil) before the start of the experiment (2003). Higher plant diversity enhanced soil N availability and retention in natural grass systems (Dybzinski et al. 2008). Yong et al. (2012) also found that wheat/maize/soybean intercropping increased soil total N by 9.4–38.6 % but wheat/maize/sweet potato intercropping reduced soil total N by 1.8–14.0 % (Wang et al. 2012; Yong et al. 2012a, b). Our present results do not confirm the diversity effect on soil total N accumulation in the relatively fertile soil studied. However, our present results show that all cropping systems enhanced soil total N content compared to the initial value in 2003 before the start of the field experiment. This indicates that a fertilizer N application rate of 225 kg N ha−1 is appropriate for these intercropping systems and is able to maintain soil N fertility over a longer time scale. Cong et al. (2014) found in the same experiment that soil organic N content in intercropping was higher than that in monocropping through removal of inorganic N by pre-treatment with 0.5 M HCl. In the present study soil total N did not show similar trends to organic N (Cong et al. 2014). This may be due to differences in soil organic and total N, suggesting that intercropping changes the composition of soil N, and this requires further study. Recent work has shown 40 kg ha−1 N driven from belowground and rhizodeposition to soil available N to transform the immobilization state (Jensen and Hauggaard-Nielsen 2003; Köpke and Nemecek 2010; López-Bellido et al. 2006). In addition, the lack of consistency may be due to different crop species, soil types, irrigation and management practices in different studies (Berthrong et al. 2009).
When soil N is not limiting soil P is often the most important major growth-limiting nutrient for crops (Ritter 2007). In our present study soil Olsen P did not differ in wheat/maize intercropping from monocropping or rotational cropping. However, when faba bean was involved in intercropping Olsen P showed a decreasing trend in faba bean/maize and wheat/faba bean intercropping in 2011 and wheat/maize intercropping in 2012 compared with the corresponding monoculture or rotation. Faba bean/maize intercropping involved a decline in Olsen P by 25.3 % in comparison with the corresponding monoculture, which resulted from the rhizosphere effect of faba bean via rhizosphere acidification and exudation of organic acids and protons from roots and mobilization of insoluble inorganics (Li et al. 2007). Similar studies have shown that pigeon pea roots secret piscidic acid and promote P release from FePO4 by chelating iron (Ae et al. 1990). Lupine roots secrete citric and malic acids and increase P uptake in P deficient soils (Dakora and Phillips 2002) and enhanced acid phosphatase activity in chickpea/maize intercropping mobilizes P from organic to soluble forms (Li et al. 2004). These underlying mechanisms can benefit crops themselves and also neighboring plants whose roots intermingle (Li et al. 2007). Regardless of continuous or rotational intercropping, faba bean/maize intercropping removed 32–44 kg P ha−1 year−1 from the soil, 3.4–7.1 P kg ha−1 more than was removed by the corresponding monocultures. This will contribute to the decline in soil Olsen-P in intercropping compared to monocropping or rotational cropping. However, the soil Olsen-P concentrations (23.5–38.8 mg kg−1) after 9 or 10 years of intercropping were higher than the initial value (20.3 mg kg−1) in 2003 before the start of the experiment. These results indicate that 40 kg P ha−1 was sufficient in all wheat/maize, faba bean/maize, wheat/faba bean intercropping treatments. Similar results were obtained in soybean/wheat intercropping which required 60 kg ha−1 P to both soybean and wheat to meet their P requirements in a sandy loam soil with very low available P (Aulakh et al. 2003). Our previous study found that faba bean/maize intercropping and monocultures averaged P acquisition of about 37 and 30 kg ha−1 at different P application rates on a newly reclaimed P-deficient soil across 2 years (Mei et al. 2012). This suggests that adequate P inputs can sustain soil P fertility in these intercropping systems.
In the present study there were no fertilizer K or manure applications due to the relatively high K content of the soil at the start of the experiment (233 g kg−1) in 2003. This led to intercropping, monoculture and rotational cropping systems showing substantially lower soil exchangeable K by as much as 66.1–76.8 % after 9 or 10 years of cropping compared to the start of the experiment (233 mg K kg−1). The crops removed about 200 to 300 kg K ha−1 year−1 without any return of K to the soil. Furthermore, intercropping removes more K due to overyielding, which leads a greater decrease in soil exchangeable K after 10 years of faba bean/maize intercropping than monoculture faba bean and maize. Mondal et al. (2004) found that in soybean/sesame intercropping the maximum K acquisition was 121 to 133 kg ha−1 at an application rate of 66 kg K ha−1, suggesting that soil exchangeable K was depleted even with crop fertilization. Thus, soil exchangeable K in wheat/maize, faba bean/maize and wheat/faba bean intercropping was significantly lower than the corresponding monocultures and decreased by about 13.7 % especially in 2012. Furthermore, K acquisition averaged over the intercropping systems was 299 kg ha−1 and 45.5 kg ha−1 higher than by the monocultures in 2012. Our results are consistent with several published studies (Blaise et al. 2005; Mondal et al. 2004). This implies that K fertilizer or manure applications are potentially important in both intercropping and rotational cropping systems.
Soil CEC is related to SOC, pH and texture to some extent (Morari et al. 2008; Ross et al. 2008; Makinde et al. 2006). In comparison with monoculture, soil CEC significantly declined by 16.1 % in intercropping in 2012. However, Makinde et al. (2006) found that soil CEC was not influenced by intercropping because lower calcium (Ca2+) and magnesium (Mg2+) concentrations reduced the exchangeable base saturation (Saikh et al. 1998). Thus, intercropping significantly decreased soil CEC and contributed to the removal of more cations from the soil.
Soil pH in all treatments was lower in 2012 than in 2011, possibly due to systematic errors in soil pH measurement. Averaged over the soil pH results in faba bean/maize intercropping we observed a decrease of 0.16 pH units compared to the corresponding monoculture. In previous studies we also observed that faba bean acidified its rhizosphere notably, with the pH declining by around 2 units in agar gel in 6 h, due to organic acids or H+ which were exuded from faba bean roots (Li et al. 2007). Similarly, we found that chickpea released acids from roots and led to a decline in soil pH (Li et al. 2004). However, we observed that wheat/maize intercropping also reduced the soil pH compared to monocropping in 2011. Soil acidification results mainly from an imbalance of cation removal from soil (Tang et al. 1997). Therefore, the reduction in soil pH was likely due to both the presence of legumes in the cropping systems and enhanced cation removal from the soil due to overyielding in the intercropping systems.
Continuous intercropping maintains soil biological properties
Soil urease catalyzes the transformation of urea to release NH4 + and this increases the risk of gaseous NH3 loss from soils (Haynes and Williams 1999; Singh and Kumar 2008). Baligar et al. reported that urease activity was positively correlated with total C and N due to higher microbial biomass and greater stabilization via humic substances (Baligar et al. 1991, 2005; Burns 1978). Moreover, sampling date and soil moisture content also influence soil urease activity (Baligar et al. 1991). Regardless of crop combinations, soil urease activities in faba bean/maize, wheat/maize, and wheat/faba bean intercropping were similar to those found in the corresponding monocultures in both 2011 and 2012, indicating that intercropping does not influence soil urease activity over a timescale of 10 years.
Soil acid phosphatase activity is associated with mobilization of soil organic P sources (Conn and Dighton 2000; Dick et al. 2000). Phosphatase activity was positively affected by soil organic C and N and total P and negatively with soil available P, pH and soil texture (Sarapatka and Krskova 1997). In previous pot experiments we found that chickpea/maize intercropping enhanced rhizosphere phosphatase activity with organic P sources and root barriers (Li et al. 2004). The results show that soil phosphatase activities in wheat/maize, faba bean/maize and wheat/faba bean intercropping did not differ from monoculture or rotation.
Soil nitrate reductase activity indicates anaerobic nitrate reduction, including denitrification and dissimilatory processes. The first step is NO3 − reduction to NO2 − and NO2 − is further reduced to N2O by nitrate reductase. Furthermore, the N2O to N2 pathway is catalyzed by nitrous oxide reductase (Singh and Kumar 2008) which can indicate soil available nitrate concentration (Högberg et al. 1986). Most studies have focused on N accumulation, availability and effects of plant metabolism on nitrate reductase in soil (Chen et al. 2004; Eilrich and Hageman 1973; Hageman et al. 1961). Nitrification and denitrification are determined by soil nitrate reductase activity via soil physico-chemical properties. High temperatures and moisture, neutral pH, bulk density, texture, soil structure and plants accelerate or favor the reaction (Fu and Tabatabai 1989; Ma 2000; Šimek et al. 2002; Venterea and Rolston 2000). In the present study there were no significant differences in nitrate reductase activities between intercropping and monoculture but faba bean-based intercropping reduced nitrate reductase and likely increased N fertility because stimulation of nitrate reductase activity occurred under low N fertility conditions. The inconsistency between the 2 years in the present study may be due to the lower soil moisture content in 2011 because low moisture availability inhibits nitrate reductase activity.
Sucrase activity is responsible for the breakdown of the water soluble plant material in soils (Frankenberger and Johanson 1983; Ross 1983). Soil pH and temperature stability and kinetic properties may explain the differences in soil enzyme activities among the cropping systems in both years (Frankenberger and Johanson 1983).
Conclusions
Continuous faba bean/maize and wheat/maize intercropping for nine to 10 years still resulted in overyielding and provided nutrient acquisition advantage compared with the corresponding monocultures or rotations. Intercropping maintained or enhanced the major soil chemical properties and enzyme activities studied. However, soil Olsen-P and exchangeable K contents declined in intercropping to some extent, an effect that can be ameliorated by the application of manures and/or inorganic fertilizer K. Thus, continuous intercropping enhanced productivity and sustained soil chemical properties and enzyme activities over a period of at least one decade. Further observations on a longer timescale are needed to investigate the overyielding mechanisms involved in intercropping together with all soil properties contributing to the maintenance of soil quality in the long term.
Abbreviations
- OM:
-
organic matter
- TN:
-
total nitrogen
- CEC:
-
cation exchange capacity
- M:
-
monoculture or monocropping
- R:
-
rotation
- Inter (C):
-
continuous intercropping
- Inter (R):
-
rotational intercropping
- W + M:
-
wheat and maize combination
- F + M:
-
faba bean and maize combination
- W + F:
-
wheat and faba bean combination
- LER:
-
land equivalent ratio
References
Ae N, Arihara J, Okada K, Yoshihara T, Johansen C (1990) Phosphorus uptake by pigeonpea and its role in cropping systems of the Indian subcontinent. Science 248:477–480
Alvear M, Rosas A, Rouanet JL, Borie F (2005) Effects of three soil tillage systems on some biological activities in an Ultisol from southern Chile. Soil Till Res 82:195–202
Andrade JF, Cerrudo A, Rizzalli RH, Monzon JP (2012) Sunflower-soybean Intercrop productivity under different water conditions and sowing managements. Agron J 104:1049–1055
Aulakh MS, Pasricha NS, Bahl GS (2003) Phosphorus fertilizer response in an irrigated soybean-wheat production system on a subtropical, semiarid soil. Field Crops Res 80:99–109
Baligar V, Staley T, Wright R (1991) Enzyme activities in Appalachian soils: 2. Urease. Commun Soil Sci Plant Anal 22:315–322
Baligar VC, Wright RJ, Hern JL (2005) Enzyme activities in soil influenced by levels of applied sulfur and phosphorus. Commun Soil Sci Plant Anal 36:1727–1735
Bao SD (2000) Analysis on soil and agricultural chemistry (in Chinese). China Agricultural Press, Beijing
Berthrong ST, Jobbágy EG, Jackson RB (2009) A global meta-analysis of soil exchangeable cations, pH, carbon, and nitrogen with afforestation. Ecol Appl 19:2228–2241
Blaise D, Bonde AN, Chaudhary RS (2005) Nutrient uptake and balance of cotton + pigeonpea strip intercropping on rainfed Vertisols of central India. Nutr Cycl Agroecosys 73:135–145
Burns RG (1978) Enzyme activity in soil: some theoretical and practical considerations. Soil enzymes Academic Press, New York, pp 295–340
Chen BM, Wang ZH, Li SX, Wang GX, Song HX, Wang XN (2004) Effects of nitrate supply on plant growth, nitrate accumulation, metabolic nitrate concentration and nitrate reductase activity in three leafy vegetables. Plant Sci 167:635–643
Cong WF, Hoffland E, Li L, Six J, Sun JH, Bao XG, Zhang FS, Van Der Werf W (2014) Intercropping enhances soil carbon and nitrogen. Global Change Biol doi: 10.1111/gcb.12738
Conn C, Dighton J (2000) Litter quality influences on decomposition, ectomycorrhizal community structure and mycorrhizal root surface acid phosphatase activity. Soil Biol Biochem 32:489–496
Dahmardeh M, Ghanbari A, Syahsar B, Ramrodi M (2010) The role of intercropping maize (Zea mays L.) and Cowpea (Vigna unguiculata L.) on yield and soil chemical properties. Afr J Agr Res 5:631–636
Dakora FD, Phillips DA (2002) Root exudates as mediators of mineral acquisition in low-nutrient environments. Plant Soil 245:35–47
de Carvalho LM, Nunes MUC, de Oliveira IR (2009) Yield of tomato in monocrop and intercropping with aromatics plants. Hortic Bras 27:458–464
Dick W, Cheng L, Wang P (2000) Soil acid and alkaline phosphatase activity as pH adjustment indicators. Soil Biol Biochem 32:1915–1919
Doran JW (2002) Soil health and global sustainability: translating science into practice. Agr Ecosyst Environ 88:119–127
Dybzinski R, Fargione JE, Zak DR, Fornara D, Tilman D (2008) Soil fertility increases with plant species diversity in a long-term biodiversity experiment. Oecologia 158:85–93
Eskandari H (2011) Intercropping of wheat (Triticum aestivum) and bean (Vicia faba): effects of complementarity and competition of intercrop components in resource consumption on dry matter production and weed growth. Afr J Biotechnol 10:17755–17762
FAO/UNESCO. (1988) FAO/UNESCO Soil map of the world, 1:5,000,000 Vol. 1 Paris: UNESCO
Fornara DA, Tilman D (2008) Plant functional composition influences rates of soil carbon and nitrogen accumulation. J Ecol 96:314–322
Fornara DA, Tilman D, Hobbie SE (2009) Linkages between plant functional composition, fine root processes and potential soil N mineralization rates. J Ecol 97:48–56
Fortin MC, Culley J, Edwards M (1994) Soil water, plant growth, and yield of strip-intercropped corn. J Sci Food Agr 7:63–69
Francis CA (1986) Multiple cropping systems. Macmillan Publishing Company pp 383
Francis CA (1989) Biological efficiencies in multiple-cropping systems. Adv Agron 42:42
Frankenberger WT Jr, Johanson JB (1983) Factors affecting invertase activity in soils. Plant Soil 74:313–323
Fu MH, Tabatabai MA (1989) Nitrate reductase activity in soils: effects of trace elements. Soil Biol Biochem 21:943–946
Ghosh PK, Mohanty M, Bandyopadhyay KK, Painuli DK, Misra AK (2006) Growth, competition, yields advantage and economics in soybean/pigeonpea intercropping system in semi-arid tropics of India. Field Crop Res 96:90–97
Gregorich EG, Drury CF, Baldock JA (2001) Changes in soil carbon under long-term maize in monoculture and legume-based rotation. Can J Soil Sci 81:21–31
Gt E, Hageman R (1973) Nitrate reductase activity and its relationship to accumulation of vegetative and grain nitrogen in wheat (Triticum aestivum L.). Crop Sci 13:59–66
Guan SY (1986) Soil enzyme and research method (in Chinese). China Agricultural Press, Beijing
Hageman RH, Flesher D, Gitter A (1961) Diurnal variation and other light effects influencing the activity of nitrate reductase and nitrogen metabolism in corn. Crop Sci 1:201–204
Hauggaard-Nielsen H, Jensen ES (2001) Evaluating pea and barley cultivars for complementarity in intercropping at different levels of soil N availability. Field Crop Res 72:185–196
Haynes RJ, Williams PH (1999) Influence of stock camping behaviour on the soil microbiological and biochemical properties of grazed pastoral soils. Biol Fert Soils 28:253–258
Högberg P, Granström A, Johansson T, Lundmark-Thelin A, Näsholm T (1986) Plant nitrate reductase activity as an indicator of availability of nitrate in forest soils. Can J For Res 16:1165–1169
SAS Institute (2003) SAS. 9.1 ed. Cary, NC: SAS Institute, Inc
Jensen ES (1996) Grain yield, symbiotic N2 fixation and interspecific competition for inorganic N in pea-barley intercrops. Plant Soil 182:25–38
Jensen ES, Hauggaard-Nielsen H (2003) How can increased use of biological N2 fixation in agriculture benefit the environment? Plant Soil 252:177–186
Karlen DL, Mausbach MJ, Doran JW, Cline RG, Harris RF, Schuman GE (1997) Soil quality: a concept, definition, and framework for evaluation (a guest editorial). Soil Sci Soc Am J 61:4–10
Keating BA, Carberry PS (1993) Resource capture and use in intercropping: solar radiation. Field Crops Res 34:273–301
Köpke U, Nemecek T (2010) Ecological services of faba bean. Field Crops Res 115:217–233
Leihner DE (1983) Management and evaluation of intercropping systems with cassava. Centro International of Agricultura Tropical, Cali
Lesoing GW, Francis CA (1999) Strip intercropping of corn-soybean in irrigated and rainfed environments. J Sci Food Agr 12:187–192
Li L, Sun J, Zhang F, Li X, Yang S, Rengel Z (2001a) Wheat/maize or wheat/soybean strip intercropping: I. Yield advantage and interspecific interactions on nutrients. Field Crops Res 71:123–137
Li L, Sun J, Zhang F, Li X, Rengel Z, Yang S (2001b) Wheat/maize or wheat/soybean strip intercropping: II. Recovery or compensation of maize and soybean after wheat harvesting. Field Crop Res 71:173–181
Li WX, Li L, Sun JH, Zhang FS, Christie P (2003) Effects of nitrogen and phosphorus fertilizers and intercropping on uptake of nitrogen and phosphorus by wheat, maize, and faba bean. J Plant Nutr 26:629–642
Li S, Li L, Zhang F, Tang C (2004) Acid phosphatase role in chickpea/maize intercropping. Ann Bot 94:297–303
Li L, Sun JH, Zhang FS, Guo TW, Bao XG, Smith FA, Smith SE (2006) Root distribution and interactions between intercropped species. Oecologia 147:280–290
Li L, Li SM, Sun JH, Zhou LL, Bao XG, Zhang HG, Zhang FS (2007) Diversity enhances agricultural productivity via rhizosphere phosphorus facilitation on phosphorus-deficient soils. Proc Natl Acad Sci U S A 104:11192–11196
Li CJ, Li YY, Yu CB, Sun JH, Christie P, An M, Zhang FS, Li L (2011a) Crop nitrogen use and soil mineral nitrogen accumulation under different crop combinations and patterns of strip intercropping in northwest China. Plant Soil 342:221–231
Li QZ, Sun JH, Wei XJ, Christie P, Zhang FS, Li L (2011b) Overyielding and interspecific interactions mediated by nitrogen fertilization in strip intercropping of maize with faba bean, wheat and barley. Plant Soil 339:147–161
López-Bellido L, López-Bellido RJ, Redondo R, Benítez J (2006) Faba bean nitrogen fixation in a wheat-based rotation under rainfed Mediterranean conditions: effect of tillage system. Field Crops Res 98:253–260
Ma RX (2000) Effects of allelochemicals on activity of nitrate reductase. J Environ Sci 2:125–128
Madari B, Machado PLOA, Torres E, de Andrade AG, Valencia LIO (2005) No tillage and crop rotation effects on soil aggregation and organic carbon in a Rhodic Ferralsol from southern Brazil. Soil Till Res 80:185–200
Mäder P, Fliessbach A, Dubois D, Gunst L, Fried P, Niggli U (2002) Soil fertility and biodiversity in organic farming. Science 296:1694–1697
Makinde E, Oluwatoyinbo F, Ayoola O (2006) Intercropping and crop residue incorporation: effects on soil nutrient status. J Plant Nutri 29:235–244
Martin RC, Astatkie T, Cooper JM (1998) The effect of soybean variety on corn-soybean intercrop biomass and protein yields. Can J Plant Sci 78:289–294
Mei PP, Gui LG, Wang P, Huang JC, Long HY, Christie P, Li L (2012) Maize/faba bean intercropping with rhizobia inoculation enhances productivity and recovery of fertilizer P in a reclaimed desert soil. Field Crops Res 130:19–27
Melero S, Porras JCR, Herencia JF, Madejon E (2006) Chemical and biochemical properties in a silty loam soil under conventional and organic management. Soil Till Res 90:162–170
Melero S, Madejon E, Ruiz JC, Herencia JF (2007) Chemical and biochemical properties of a clay soil under dryland agriculture system as affected by organic fertilization. Eur J Agron 26:327–334
Mondal SS, Ghosh A, Acharya D, Mait D (2004) Production potential and economics of different rainfed rice (Oryza sativa)-based utera cropping systems and its effect on fertility build up of soil. Indian J Agron 49:6–9
Morari F, Lugato E, Giardini L (2008) Olsen phosphorus, exchangeable cations and salinity in two long-term experiments of north-eastern Italy and assessment of soil quality evolution. Agr Ecosyst Environ 124:85–96
Morris RA, Garrity DP (1993a) Resource capture and utilization in intercropping non-nitrogen nutrients. Field Crops Res 34:319–334
Morris RA, Garrity DP (1993b) Resource capture and utilization in intercropping: water. Field Crops Res 34:303–317
Mucheru-Muna M, Pypers P, Mugendi D, Kung’u J, Mugwe J, Merckx R, Vanlauwe B (2010) A staggered maize–legume intercrop arrangement robustly increases crop yields and economic returns in the highlands of Central Kenya. Field Crops Res 115:132–139
Olsen SR, Cole CV, Watanabe FS, Dean LA (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate, USDA. Circular939. US Government Printing Office, Washington
Pellegrino E, Di Bene C, Tozzini C, Bonari E (2011) Impact on soil quality of a 10-year-old short-rotation coppice poplar stand compared with intensive agricultural and uncultivated systems in a Mediterranean area. Agr Ecosyst Environ 140:245–254
Qi ZP, Rao IM, Ricaurte J, Amézquita E, Sanz JI, Kerridge PC (2004) Root distribution and nutrient uptake in crop-forage systems on Andean hillsides. J Sustain Agr 23:39–50
Rayment GE, Higginson FR (1992) Australian laboratory handbook of soil and water chemical methods. Inkata Press Pty Ltd, Australia, pp 89–92
Ritter E (2007) Carbon, nitrogen and phosphorus in volcanic soils following afforestation with native birch (Betula pubescens) and introduced larch (Larix sibirica) in Iceland. Plant Soil 295:239–251
Roldan A, Salinas-Garcia JR, Alguacil MM, Caravaca F (2007) Soil sustainability indicators following conservation tillage practices under subtropical maize and bean crops. Soil Till Res 93:273–282
Ross DJ (1983) Invertase and amylase activities as influenced by clay minerals, soil-clay fractions and topsoils under grassland. Soil Biol Biochem 15:287–293
Ross DS, Matschonat G, Skyllberg U (2008) Cation exchange in forest soils: the need for a new perspective. Eur J Soil Sci 59:1141–1159
Rusinamhodzi L, Corbeels M, Nyamangara J, Giller KE (2012) Maize-grain legume intercropping is an attractive option for ecological intensification that reduces climatic risk for smallholder farmers in central Mozambique. Field Crops Res 136:12–22
Saikh H, Varadachari C, Ghosh K (1998) Effects of deforestation and cultivation on soil CEC and contents of exchangeable bases: a case study in Simlipal national park, India. Plant Soil 204:175–181
Sarapatka B, Krskova M (1997) Interactions between phosphatase activity and soil characteristics from some locations in the Czech Republic. Rostlinna Vyroba-UZPI 43
Šimek M, Jíšová L, Hopkins DW (2002) What is the so-called optimum pH for denitrification in soil? Soil Biol Biochem 34:1227–1234
Singh DK, Kumar S (2008) Nitrate reductase, arginine deaminase, urease and dehydrogenase activities in natural soil (ridges with forest) and in cotton soil after acetamiprid treatments. Chemosphere 71:412–418
Stuelpnagel R (1992) Intercropping of faba beans (Vicia faba L.) with oats or spring wheat. In: Proceedings of the International Crop Science Congress, p 44
Tabatabai MA, Bremner JM (1969) Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307
Tang C, Barton L, McLay CDA (1997) A comparison of proton excretion of twelve pasture legumes grown in nutrient solution. Anim Prod Sci 37:563–570
Thierfelder C, Wall PC (2012) Effects of conservation agriculture on soil quality and productivity in contrasting agro-ecological environments of Zimbabwe. Soil Use Manage 28:209–220
Venterea RT, Rolston DE (2000) Mechanisms and kinetics of nitric and nitrous oxide production during nitrification in agricultural soil. Global Change Biol 6:303–316
Wang XC, Yang WY, Ren WJ, Deng XY, Zhang Q, Xiang DB, Yong TW (2012) Study on the differences in yield and nutrient absorption of maize in wheat/maize/soybean and wheat/maize/sweet potato relay intercropping systems. Plant Nutr Fert Sci 18:803–812
Wei X, Hao M, Shao M, Gale WJ (2006) Changes in soil properties and the availability of soil micronutrients after 18 years of cropping and fertilization. Soil Till Res 91:120–130
Xia HY, Zhao JH, Sun JH, Xue YF, Eagling T, Bao XG, Zhang FS, Li L (2013) Maize grain concentrations and above-ground shoot acquisition of micronutrients as affected by intercropping with turnip, faba bean, chickpea, and soybean. Science China-Life Sciences 56:823–834
Yang WT, Li Z, Wang JW, Wu P, Zhang Y (2013) Crop yield, nitrogen acquisition and sugarcane quality as affected by interspecific competition and nitrogen application. Field Crops Res 146:44–50
Yong TW, Yang WY, Xiang DB, Zhu ZY (2012a) Effect of wheat/maize/soybean and wheat/maize/sweet potato relay strip intercropping on bacterial community diversity of rhizosphere soil and nitrogen uptake of crops. Acta Agron Sin 38:333–343
Yong TW, Yang WY, Xiang DB, Chen XR, Wan Y (2012b) Production and N nutrient performance of wheat-maize-soybean relay strip intercropping system and evaluation of interspecies competition. Acta Prataculturae Sinica 21:50–58
Zhang FS, Li L (2003) Using competitive and facilitative interactions in intercropping systems enhances crop productivity and nutrient-use efficiency. Plant Soil 248:305–312
Zomer RJ, Trabucco A, Coe R, Place F (2009) Trees on farm: analysis of global extent and geographical patterns of agroforestry. ICRAF Working Paper-World Agroforestry Centre,p 89
Zou CY, Li ZJ (2002) Intercropping and relay intercropping. In: Shi YC (ed) Chinese academic canon in the 20th century: agriculture (in Chinese). Fujian Education Press, Fuzhou
Acknowledgments
The study was financially supported by the National Basic Research Program of China (Project No. 2011CB100405) and the Chinese Ministry of Science and Technology (Project Nos. 2012BAD14B04 and 2012BAD14B10). We thank Messrs, Wen-yu Yang and Quan-fu Li for their assistance with fieldwork. The authors also thank two anonymous reviewers for their helpful suggestions and comments toward improving an earlier version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Martin Weih.
Rights and permissions
About this article
Cite this article
Wang, Zg., Bao, Xg., Li, Xf. et al. Intercropping maintains soil fertility in terms of chemical properties and enzyme activities on a timescale of one decade. Plant Soil 391, 265–282 (2015). https://doi.org/10.1007/s11104-015-2428-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2428-2