Abstract
Background and aims
Ectomycorrhizal (ECM) fungal communities are affected by many abiotic and biotic factors, among which host identity is often regarded as the most significant. Although little about ECM fungal communities on endangered tree species is known, knowledge of their ECM associations could be a key to conservation given the functional importance of the symbiosis.
Methods
We collected 73 soil samples from three relict forests dominated by endangered Chinese Douglas-fir (Pseudotsuga sinensis). Identity of fungal and host species in individual ECM tips was determined by morphotyping and DNA analyses.
Results
Of the 86 ECM fungal species identified, 66 were observed on Chinese Douglas-firs. While the fungal composition did not significantly differ between coexisting trees (p = 0.843), the composition was significantly separated based on location (p = 0.021). Moreover, the observed ECM fungal communities were dissimilar to those on the Japanese Douglas-fir (p = 0.001), which is monophyletic to Chinese Douglas-fir.
Conclusions
Our results indicate that ECM fungal communities are determined more by geographical location than host monophyly on this spatial (c.770–1,600 km) and geological time scale ( c. 20–25 My). For conservation of endangered trees, it may be important to preserve local ECM fungal pools, irrespective of host species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ectomycorrhizal (ECM) associations are a prerequisite for many tree species to grow and survive in nature (Smith and Read 2008). This is mainly because host trees depend largely on colonizing ECM fungi for soil nutrients, despite their large expenditures of photosynthates to the fungi, as much as 22 % of net primary production (Hobbie 2006). ECM fungi are diverse not only in terms of species and lineage richness (Rinaldi et al. 2008; Tedersoo et al. 2010) but also in their physiological (Erland and Finlay 1992; Rineau and Courty 2011; Koide et al. 2014) and ecological traits (Nara et al. 2003; Bruns et al. 2009). In fact, host performance in the field is critically determined by colonizing ECM fungi (Nara 2006).
ECM trees dominate most boreal, temperate, and sub-tropical forests throughout the world, as well as some tropical forests (Smith and Read 2008; Tedersoo et al. 2012). In each of these forests, tens to hundreds of ECM fungal species per hectare are estimated to exist (Horton and Bruns 2001; Ishida et al. 2007; Miyamoto et al. 2014), forming diverse ECM fungal communities. Given that these ECM fungi function as a community and sustain host trees, the diversity and composition of ECM fungal communities is quite relevant to forest ecosystems. While many studies have described ECM fungal communities in this context (e.g. Tedersoo et al. 2012 and references therein), those of endangered tree species have rarely been documented (Tedersoo et al. 2007; Murata et al. 2013). To conserve an endangered tree species, it may be critically important to understand the ECM fungal communities in its remaining precious forests, which are on the verge of extinction.
While many potential determinants of ECM fungal communities have been proposed, such as climate (Bahram et al. 2012), soil (Cox et al. 2010; Jarvis et al. 2013; Roy et al. 2013), geographical position (Miyamoto et al. 2014), precipitation (Jarvis et al. 2013), nitrogen deposition (Lilleskov et al. 2002), heavy metals (Huang et al. 2014), and vegetation succession (Nara et al. 2003; Twieg et al. 2007), host effect may be the most significant factor, as has been demonstrated in many studies (Ishida et al. 2007; Tedersoo et al. 2008, 2012; Smith et al. 2009; Lang et al. 2011; Velmala et al. 2013; Põlme et al. 2013; Murata et al. 2013). If this is applicable to endangered hosts, they may be associated with unique ECM fungal communities that are different from those of coexisting and surrounding hosts.
The ECM host genus Pseudotsuga is a monophyletic conifer lineage composed of four extant species (some taxonomists accept more species; Wu and Raven 1999). It has a unique distribution pattern, that is, the intercontinental disjunction between Asia and North America. In North America, the Douglas-fir (Pseudotsuga menziesii) dominates a wide range of forests from the Pacific coast to the Rocky Mountains, representing typical forest ecosystems in these regions (Farjon 1990). This species produces good timber and may be the most economically important single tree species in the world (Eckenwalder 2009). Another North American species, Pseudotsuga macrocarpa is confined to small areas in California (Farjon 1990). In Asia, two Pseudotsuga species are accepted widely (Farjon 1990; Strauss et al. 1990): the Chinese Douglas-fir (Pseudotsuga sinensis) in mainland China and Taiwan (P. sinensis var. wilsoniana), and the Japanese Douglas-fir (Pseudotsuga japonica) in Japan. Both Asian species are designated as endangered in the International Union for Conservation of Nature (IUCN) Red List (IUCN 2014), and remaining populations are small and fragmented (Kasai and Saito 2009; Fu 1992; Fu et al. 1999). According to fossil records and molecular phylogeny, Wei et al. (2010) estimated that Pseudotsuga originated in North America in the Eocene and migrated to Asia through the Bering land bridge approximately 32 million years ago (Mya), and diversified into Chinese and Japanese lineages c. 25–20 Mya.
Murata et al. (2013) studied ECM fungal communities in all of the major populations of Japanese Douglas-fir and found a significant difference in ECM fungal communities between co-existing hosts. However, no coevolved ECM fungal lineage has been confirmed to be associated with the Japanese Douglas-fir, except for one Rhizopogon species that was found only in soil spore banks. This is in sharp contrast to Douglas-fir ECM fungal communities in North America, where Pseudotsuga-specific lineages, such as Rhizopogon and Suillus (Molina et al. 1992; Kretzer et al. 1996; Horton and Bruns 1998; Horton et al. 2005; Twieg et al. 2007), account for a substantial portion of ECM fungal communities, as much as 25 % in relative abundance of ECM tips. Furthermore, there is little overlap of ECM fungal species between North American and Japanese Douglas-firs, indicating limited migration between the continents, as seen in Pseudotsuga (Wei et al. 2010) and many other plant lineages (Xiang et al. 1999). Considering the scarcity of coevolved fungal symbionts, host changes within the local ECM fungal pool could be important in structuring ECM fungal communities on the endangered tree species; however, further evidence is needed to confirm this. Given that the Chinese Douglas-fir is monophyletic with the Japanese Douglas-fir, and that both have been isolated for a long period on a geologic time scale, this subject is worthy of attention. Focused research will improve our understanding of how ECM fungal communities are structured with respect to local host changes, as well as host phylogeny, host biogeography, and ECM fungal biogeography.
In this study, ECM fungal communities associated with the Chinese Douglas-fir were characterized for the first time, a rare example of a study focusing on ECM fungi on an endangered tree species. We also compared the communities with those of the Japanese Douglas-fir to address the similarity of the ECM communities between the two species, which are the closest extant relatives, although they have been separated for a long geological period, possibly c. 25–20 My. Based on our results, we discuss the importance of local host sharing in structuring ECM fungal communities in endangered, small, and fragmented host populations. We also discuss the biogeography of ECM fungi, which is largely host-independent on this geologic time scale.
Materials and methods
Sampling
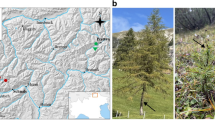
We collected samples at three sites, all of which are protected by the Chinese government for conservation of the Chinese Douglas-fir. The Sanqing Mountain site (SQM) was located in Jiangxi province, China. The other two sites were located in Sangzhi, Zhangjiajie (ZJJ1 and ZJJ2), in Hunan Province, China. Geological and climate conditions are summarized in Table 1. Generally, the climate at these three sites (annual mean temperature: 16.3–18.2 °C) is warmer than that of Japanese Douglas-fir forests (annual mean temperature: 10.6–12.5 °C), and is characterized as sub-tropical. The Chinese Douglas-fir does not form mono-specific forests and is usually accompanied by other Pinaceae trees, such as Tsuga, Pinus, and Abies. Other tree families at the three sites included Ericaceae, Fagaceae, and Lauraceae. Aquifoliaceae, Theaceae, and Rosaceae were present at the SQM site (Guo et al. 2007), while Hamamelidaceae and Pteridiaceae were frequent at the ZJJ sites. At all three sites, most Chinese Douglas-fir individuals were in mature stages (Table 1), and they were the predominant components of the forests.
At each site, 23–25 Chinese Douglas-fir trees were selected, while maintaining a distance of >10 m between the selected trees for sample independence as in previous studies (Murata et al. 2013). A soil sample (5- × 5- × 10-cm, length × width × depth) was collected within about 2 m from each selected tree. In total, 73 soil samples were collected (23, 25, and 25 from SQM, ZJJ1, and ZJJ2, respectively) and stored at 4 °C until use. All roots in each soil sample were hand-picked, washed carefully under tap water on a 1-mm sieve, and examined under a dissecting microscope to collect all roots containing ECM tips. The ECM root tips were then classified into morphotypes based on their surface color, texture, and emanating hyphae, as described in Murata et al. (2013). Three to five replicate ECM root tips (or all, if fewer than three tips were available) were selected from each morphotype in each soil sample for molecular identification. In total, 1,005 root tips (305 cumulative morphotypes) were separately placed into 2-mL test tubes and used for DNA extraction.
Molecular analysis
Internal transcribed spacer (ITS) regions of fungal rDNA were amplified by polymerase chain reaction (PCR) using the ITS 1 F (Gardes and Bruns 1993) and LR 21 or LR 22 primer pairs (http://biology.duke.edu/fungi/mycolab/primers.htm), depending on the success of amplification. For host identification, the plastid trnL intron was amplified with the primers trnE (5′—GGTTCAAGTCCCTCTATCCC—3′) and trnF (5′—ATTTGAACTGGTGACACGAG—3′). We used AmpliTaq Gold 360 Master Mix (Applied Biosystems, CA, USA) for PCRs, however, when PCR products were faint or absent, the Multiplex PCR kit (Qiagen, GmbH, Hilden, Germany) was also used. Amplified PCR products were verified on 1.2 % agarose gels with Gel Red (Biotium, CA, USA) under UV light (Benchtop 2UV Transilluminator, UVP, UK) and then purified using a PCR product pre-sequencing kit (Exonuclease I, Shrimp Alkaline Phospatase, GE Healthcare, UK). Sequencing reactions were carried out using the BigDye Terminator version 3.1 Cycle Sequencing kit (Applied Biosystems, Foster City, CA, USA), and sequencing was performed with a 3,130 Genetic Analyzer (Applied Biosystems, Foster City, CA, USA). All sequences were manually cleaned by checking the original chromatograms and eliminating ambiguous base-calls. The cleaned sequences were then grouped into operational taxonomic units (OTUs) based on a 97 % identity threshold in ITS regions (including the ITS1, 5.8S, and ITS2 regions) using ATGC (version 7, GENETYX, JAPAN). Representative sequences from each OTU were compared with known sequences in the international nucleotide sequence database (INSD: DDBJ/EMBL/NCBI) using the Megablast algorithm on the NCBI website. We only retained those OTUs that belong to ECM phylogroups (Tedersoo et al. 2010) by confirming phylogenetic relationships with available sequences in INSD, especially for genera containing both ECM and other trophic types such as Entoloma (Kinoshita et al. 2012) and Sebacina (Selosse et al. 2007). Theoretically, each OTU represents a different ECM fungal species; hereafter we use the term “species” unless otherwise specified (Murata et al. 2013). ECM fungal species names were assigned as the genus name followed by sequential numbers (e.g., Amanita sp.1) when ITS similarities were >95 % to known sequences in the Blast results. Family or order level classification was applied to those species showing the highest similarity of 90–95 % or <90 %, respectively, to known species in the INSD database. The identified sequences were deposited to DDBJ under the accession numbers AB981985- AB982069.
Analyses
The frequency of a fungus was defined as the number of soil samples in which that species occurred. The relative abundance of an ECM fungus was the percentage of ECM tips colonized by that fungus out of the total number of ECM tips observed. Soil samples containing no ECM fungi were excluded from the following analyses.
To estimate ECM fungal richness for Chinese Douglas-firs, for all hosts, and for each sampling site, we calculated Chao2 and Jackknife2 (Jack2) estimators using EstimateS version 9.0 (http://viceroy.eeb.uconn.edu/EstimateS), with 1,000 randomizations without sample replacement. The Sørensen similarity index value was used to calculate the similarity of the ECM fungal communities between Chinese Douglas-fir trees and the other hosts, based on frequency data.
To unravel the effects of host identity on ECM fungal community structure in our data set, we employed nonmetric multidimensional scaling (NMS) implemented in PC-ORD ver. 6 (McCune and Mefford 2011). Each host genus within a sampling site was regarded as a sampling unit in the ordination analyses, in which frequency data were used. Hosts represented by less than 10 % of the total soil samples were excluded from the analysis. We used the relative Sørensen distance measure for NMS analysis. To test the effect of hosts and geographical positions (i.e., sampling sites) on fungal community composition, we used the Adonis function in the Vegan package of R (Oksanen et al. 2011) with the following options: Hellinger-transformed species frequency, Bray–Curtis distance measure, and 999 permutations.
We also combined the present results with Japanese Douglas-fir data published previously (Murata et al. 2013), where the sampling and identification methods were basically the same as in the present study, to examine the effects of host monophyly and long-time host isolation on ECM fungal communities. NMS and Adonis analyses for the combined data set were performed as described above.
Results
Of the 73 soil samples, 65 contained ECM roots and 54 included ECM roots from Chinese Douglas-firs. The other dominant host genera were Pinus (Pinus densata and Pinus massoniana) and Quercus (Quercus multinervis and Quercus phillyraeoides), which were observed in 15 and 9 soil samples, respectively. Tsuga diversifolia was confirmed in 5 soil samples, while Abies farges and Fraxinus americana were each detected only once (Table 2). Eight soil samples did not contain ECM root-tips. In terms of ECM root abundance, the Chinese Douglas-fir was dominant, accounting for 61 % of the identified ECM root tips.
In total, we identified 86 ECM fungal species, of which 66 species were found on Chinese Douglas-fir roots. Pinus spp. and Quercus spp. were associated with 17 and 14 ECM fungi, respectively. Of the 66 species identified on Chinese Douglas-firs, 39 species appeared only on Chinese Douglas-firs, while 11 and 9 species were shared with Pinus and Quercus, respectively. These shared species were relatively frequent in the forests (Table 2). An average of 2.8 ECM fungal species were detected per soil sample, with a maximum of 7 species.
At the family level, the relative abundances of Russulaceae and Sebacinaceae were 33 and 32 %, respectively. Russulaceae (35 spp.) was the most species-rich ECM fungal lineage, followed by Sebacinaceae (12 spp.), Thelephoraceae (12 spp.), and Clavulinaceae (5 spp.) (Table 2). At the species level, Sebacina sp.1, Sebacinaceae sp.3, and C. geophilum were dominant on Chinese Douglas-firs, and were found in 16, 9, and 8 soil samples, respectively. Only eight ECM fungal species were found in more than five soil samples. In contrast, 49 species were singletons, represented by a single soil sample (Table 2, Supplemental Fig. S1).
The richness estimator chao2 (±SD) indicated that at least 164 ± 35 ECM fungal species are expected to inhabit these forests, while the estimator for Chinese Douglas-fir alone was 154 ± 40. The observed ECM fungal richnesses (and Chao2 estimates) at the ZJJ1, ZJJ2, and SQM sites were 30 (67 ± 43), 38 (111 ± 46), and 39 (83 ± 40), respectively. Species accumulation curves of ECM fungi for all hosts and for Chinese Douglas-fir alone did not approach asymptote at our maximum sampling effort (Fig. 1), indicating that additional species would be found with greater sampling effort. The curves for Chao2 became asymptotic at around 15 samples for all hosts and for Chinese Douglas-fir alone, but then increased linearly because of the species-area relationship, that is, with greater sampling area, more species are found, as is the general ecological principle (MacArthur and Wilson 1967).
Species accumulation curves for ectomycorrhizal (ECM) fungi found in Chinese Douglas-fir forests. Filled and open triangles represent observed species richness of ECM fungi from all host species, and Chinese Douglas-fir alone, respectively, with 95 % confidence intervals. Jackknife2 (squares) and Chao2 (circles) minimum species richness estimates of ECM fungi are shown for all host species and Chinese Douglas-fir and indicated by filled and open symbols, respectively
Nonmetric multidimensional scaling (NMS) plot for ECM fungal communities in Chinese Douglas-fir and Japanese Douglas-fir forests. Frequency data for ECM fungi on individual host genera at each research site were used for NMS ordination with the relative Sørensen distance measure. Open symbols and filled symbols represent ECM fungal communities in China and Japan, respectively. Circles, triangles, inverted triangles, diamonds and squares represent ECM fungal communities on Pseudotsuga, Pinus, Abies, Tsuga and Quercus, respectively
The Sørensen similarity index value indicated that the ECM fungal community of Chinese Douglas-fir was most similar to that of Pinus (0.27), followed by Quercus (0.21). The similarity value was 0.30 for the Quercus–Pinus pair. NMS ordination did not clearly separate ECM communities between host genera (Supplemental Fig. S2). The Adonis test also revealed that host identity had no significant effect on ECM fungal community composition (F2,5 = 0.838, p = 0.843). In contrast, the communities were significantly different between sampling sites (F2,5 = 1.417, p = 0.021).
Only eight ECM fungal species (Amanita sp.1, Ceratobasidium sp.1, Entoloma sp.2, Russula sp.2, Russula sp.9, Russulaceae sp.5, Thelephoraceae sp.4, Thelephoraceae sp.5) were shared between Chinese and Japanese Douglas-fir forests, after applying the 97 % ITS identity threshold to the combined data set. Of the eight species, Amanita sp.1, Ceratobasidium sp.1, and Entoloma sp.2 were not Pseudotsuga specific, and were found also in association with other coexisting hosts (Table 2). Although the other five species were found only on Chinese Douglas-fir in our study sites, their frequency was not sufficient to examine the biased occurrence on Chinese Douglas-firs. In addition, we found no Pseudotsuga-specific fungal lineages in our phylogenetic analyses using sequences found only on Chinese Douglas-fir and their related sequences in the INSD, with the exception of one Rhizopogon sp.1 (Supplemental Figs. S3, S4). The NMS plot for the combined data set clearly demonstrates that ECM fungal communities found in Chinese Douglas-fir forests are dissimilar to those of Japanese Douglas-fir forests (Fig. 2). A significant regional effect (China vs. Japan) was also confirmed by the Adonis test (F1,18 = 3.851, p = 0.001), while the effect of host was not significant in the combined data set (F2,17 = 1.027, p = 0.359).
Discussion
In total, we identified 66 ECM fungal species associated with the Chinese Douglas-fir, while at least 154 ± 40 species were estimated to exist. The observed and estimated species richness values were similar to those obtained for other abundant host species using the same sampling method and analyses (Ishida et al. 2007; Miyamoto et al. 2014). Although ECM fungal richness in endangered tree species has rarely been reported, Tedersoo et al. (2007) found only 15 ECM fungal species in 16 samples collected from a small remaining population of Vateriopsis seychellarum (Dipterocarpaceae) in Seychelles, and estimated a fungal richness of 29.5 species. This remaining V. seychellarum population may be too small (constituting ~15 trees) and too distant from other ECM forests to sustain rich ECM communities. Although individual Chinese Douglas-fir populations were comparably small, they were mixed with other common ECM trees that have wider distributions. Therefore, the Chinese Douglas-fir would be able to maintain rich ECM fungal communities by utilizing a larger regional ECM fungal pool.
Accordingly, Chinese Douglas-fir shared many ECM fungi with coexisting trees (e.g., Pinus and Quercus), and ECM fungal communities did not differ significantly between coexisting hosts. A similar pattern was also demonstrated in a North American mixed Douglas-fir forest (Horton and Bruns 1998), where 12 of 16 ECM fungal species were shared between P. menziesii and Pinus muricata. Molina et al. (1992) estimated that Douglas-fir and pines have a 72 % (or 1,800 species) overlap of compatible fungi. In addition, most of the ECM fungi found only in Chinese Douglas-fir were not significantly biased toward this host (i.e., exclusive observations occurred by chance) and did not belong to Pseudotsuga-specific lineages (i.e., no host conservatism during long evolutional periods). Because of the existence of multi-host fungi and the prevailing host changes, an endangered tree species could acquire compatible ECM fungal symbionts if surrounded by other ECM host trees that have evolved in the same region.
There is sharp contrast between ECM fungal compositions in North American and Asian Douglas-fir forests. In North American Douglas-fir forests, co-evolved and specific ECM fungi, such as some Rhizopogon and Suillus species, often appear as major components in the communities (Horton et al. 2005; Luoma et al. 2006; Twieg et al. 2007). In Chinese Douglas-fir forests, no ECM fungi were confirmed to be phylogenetically specific to Pseudotsuga, except for one rare Rhizopogon sp.1 (Table 2, Supplemental Figs. S4). This is in agreement with ECM fungal communities in Japanese Douglas-fir forests, where no co-evolved ECM fungal lineages were found on mature trees (Murata et al. 2013). Although the contrast in ECM fungal communities between North American and Asian Douglas-fir forests may be related to differences in climate conditions, we think the small and isolated populations of Asian Douglas-firs, as indicated by their endangered status, could also account for the rarity of host specialists. As in all plant and animal species, each ECM fungal species exists as assemblages of populations. Small populations are vulnerable to extinction because of demographic fluctuation, limited genetic diversity and inbreeding depression, and random catastrophes (Lande 1993). In association with small and isolated host populations, host-specific ECM fungi would have difficulty in maintaining effective population sizes over a long geological time period so as to avoid extinction. Therefore, the richness and relative abundance of host-specific ECM fungi may be determined by the effective population sizes of hosts.
Co-evolved specialists are less represented in mature forests than in soil spore banks, as seen in Suilloid fungi in pine forests (Taylor and Bruns 1999). The spores of these specialists can survive disturbances and play important roles in seedling establishment after disturbances for pines (Baar et al. 1999; Izzo et al. 2006; Peay et al. 2009), and likely also for Douglas-fir (Cline et al. 2005). Indeed, while Rhizopogon species were a minor population on mature tree roots in Chinese and Japanese Douglas-fir forests, they were relatively frequent in spore banks in both forests (unpublished data). Thus, we should not underestimate the function of minor specialists in forest dynamics or regeneration. For conservation of endangered Asian Pseudotsuga, further research is necessary to clarify ECM fungal symbionts at all host life stages, especially in the seedling stage.
ECM fungal communities in Chinese Douglas-fir forests differed significantly among three sites, which were separated by a maximum of 770 km. The effect of geographical location on ECM fungal communities was more pronounced between the Chinese and Japanese Douglas-fir forests, which were >1,610 km apart, with a sea barrier. These results indicate that ECM fungal migration is infrequent on these spatial scales, even over long geological periods. In fact, Southern China and the main island of Japan have been separated for over 1.7 My by Watase’s Line (Tokara Strait, maximum depth >1,000 m), which constrains the distribution of many plant and animal species (Hotta 1974; Ono 1989; Hiramatsu et al. 2001). Indeed, Chinese and Japanese Douglas-fir lineages have been separated without genetic exchange for c. 20–25 My (Wei et al. 2010). Similarly, many ECM fungal components in Chinese and Japanese Douglas-fir forests may have been separated for a comparable geological period; this was confirmed by the sequence divergence between the Chinese and Japanese Rhizopogon species specific to Pseudotsuga (Supplement Fig. S4).
Taken together, our results suggest that, except for rare specialists, most ECM fungi may have been able to migrate independently of the biogeography of endangered hosts, as far and as long as contiguous ECM forests of other species existed. In fact, most ECM fungal genera or families have global distributions while the distribution of hosts is not global (Tedersoo and Nara 2010). Thus, one might conclude that the lack of compatible ECM fungi is not problematic to endangered host trees. On the other hand, while artificially introduced trees suffer from a lack of compatible ECM fungi (Mikola 1973 and references therein, Pringle et al. 2009), some specificity likely exists after long periods of allopatric evolution in different regions. Therefore, with the aim of conserving endangered trees, we should take into consideration regional ECM fungal pools that share long histories of evolutional interactions.
References
Baar J, Horton TR, Kretzer AM, Brun TD (1999) Mycorrhizal colonization of Pinus muricata from resistant propagules after stand-replacing wildfire. New Phytol 143:409–418
Bahram M, Põlme S, Kõljalg U, Zarre S, Tedersoo L (2012) Regional and local patterns of ectomycorrhizal fungal diversity and community structure along an altitudinal gradient in the Hyrcanian forests of northern Iran. New Phytol 193:465–473
Bruns TD, Peay KG, Boynton PJ, Grubisha LC, Hynson NA, Nguyen NH, Rosenstock NP (2009) Inoculum potential of Rhizopogon spores increases with time over the first 4-yr of a 99-yr spore burial experiment. New Phytol 181(2):463–470
Cline ET, Ammirati JF, Edmonds RL (2005) Does proximity to mature trees influence ectomycorrhizal fungus communities of Douglas-fir seedlings? New Phytol 166(3):993–1009
Cox F, Barsoum N, Lilleskov EA, Bidartondo MI (2010) Nitrogen availability is a primary determinant of conifer mycorrhizas across complex environmental gradients. Ecol Lett 13:1103–1113
Eckenwalder JE (2009) Conifers of the world. Timber Press, Portland
Erland S, Finlay R (1992) Effects of temperature and incubation time on the ability of three ectomycorrhizal fungi to colonize Pinus sylvestris roots. Mycol Res 96:270–272
Farjon A (1990) Pinaceae: drawings and descriptions of the Genera Abies, Cedrus, Pseudolarix, Keteleeria, Nothotsuga, Tsuga, Cathaya, Pseudotsuga. Koeltz Scientific Books, Königstein
Fu LK (1992) China plant red data book: rare and endangered plants: volume 1. Science Press, Beijng
Fu LK, Li N, Mill RR (1999) Pseudotsuga. In: Wu ZY, Raven PH (eds) Flora of China, vol 4. Science Press and Missouri Botanical Garden, Beijing, pp 33–37
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Guo W, Shen RJ, Wu JH, Liao WB, Peng SL, Chen H (2007) Analysis on community composition and structure of Pseudotsuga gauseni in Sanqing Mountain of Jiangxi Province (in Chinese). J Plant Resour Environ 16(3):46–52
Hiramatsu M, Ii K, Okubo H, Huang KL, Huang CW (2001) Biogeography and origin of Liliumlongiflorum and L-formosanum (Liliaceae) endemic to the Ryukyu Archipelago and Taiwan as determined by allozyme diversity. Am J Bot 88(7):1230–1239
Hobbie EA (2006) Carbon allocation to ectomycrrhizal fungi correlates with belowground allocation in culture studies. Ecology 87:563–569
Horton TR, Bruns TD (1998) Multiple-host fungi are the most frequent and abundant ectomycorrhizal types in a mixed stand of Douglas-fir (Pseudotsuga menziesii D. Don) and bishop pine (Pinus muricata. D. Don). New Phytol 139:331–339
Horton TR, Bruns TD (2001) The molecular revolution in ectomycorrhizal ecology: peeking into the black-box. Mol Ecol 10:1855–1871
Horton TR, Molina R, Hood K (2005) Douglas-fir ectomycorrhizae in 40- and 400-year-old stands: mycobiont availability to late successional western hemlock. Mycorrhiza 15(6):393–403
Hotta M (1974) Evolutionary biology in plants. III. History and geography of plants (in Japanese). Sanseido Co, Tokyo
Huang J, Nara K, Zong K, Wang J, Xue S, Peng K, Shen Z, Lian C (2014) Ectomycorrhizal fungal communities associated with Masson pine (Pinus massoniana) and white oak (Quercus fabri) in a manganese mining region in Hunan Province, China. Fungal Ecol 9:1–10
Ishida T, Nara K, Hogetsu T (2007) Host effects on ectomycorrhizal fungal communities: insight from eight host species in mixed conifer–broadleaf forests. New Phytol 147:430–440
Izzo A, Canright M, Bruns TD (2006) The effects of heat treatments on ectomycorrhizal resistant propagules and their ability to colonize bioassay seedlings. Mycol Res 110:196–202
Jarvis S, Woodward S, Alexander IJ, Taylor AFS (2013) Regional scale gradients of climate and nitrogen deposition drive variation in ectomycorrhizal fungal communities associated with native Scots pine. Glob Chang Biol 19:1688–1696
Kasai S, Saito M (2009) Distribution and suitable site of Pseudotsuga japonica in Mie prefecture (in Japanese). Chubu For Res 57:55–56
Kinoshita A, Sasaki H, Nara K (2012) Multiple origins of sequestrate basidiomes within Entoloma inferred from molecular phylogenetic analyses. Fungal Biol 116:1250–1262
Koide RT, Fernandez C, Malcolm G (2014) Determining place and process: functional traits of ectomycorrhizal fungi that affect both community structure and ecosystem function. New Phytol 201:433–439
Kretzer A, Li YN, Szaro T, Bruns TD (1996) Internal transcribed spacer sequences from 38 recognized species of Suillus sensulato: phylogenetic and taxonomic implications. Mycologia 88:776–785
Lande (1993) Risk of population extinction from demographic and environmental stochasticity and random catastrophes. Am Nat 142(6):911–927
Lang C, Seven J, Polle A (2011) Host preferences and differential contributions of deciduous tree species shape mycorrhizal species richness in a mixed Central European forest. Mycorrhiza 21(4):297–308
Lilleskov EA, Fahey TJ, Horton TR, Lovett GM (2002) Belowground ectomycorrhizal fungal community change over a nitrogen deposition gradient in Alaska. Ecology 83(1):104–115
Luoma DL, Stockdale CA, Molina R, Eberhart JL (2006) The spatial influence of Douglas-fir retention trees on ectomycorrhiza diversity. Can J Forest Res 36:2561–2573
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, New Jersey
McCune B, Mefford MJ (2011) PC-ORD. Multivariate analysis of ecological data. Version 6. MjM Software, Gleneden Beach, Oregon
Mikola P (1973) Application of mycorrhizal symbiosis in forestry practice. In Ectomycorrhizae-Their ecology and Pgysiology. Academic Press, New York, pp 383–411
Miyamoto Y, Nakano T, Hattori M, Nara K (2014) The mid-domain effect in ectomycorrhizal fungi: range overlap along an elevation gradient on Mount Fuji, Japan. ISME J 8:1739–1746
Molina R, Massicotte H, Trappe JM (1992) Specificity phenomena in mycorrhizal symbioses: community-ecological consequences and practical implications. In: Allen MF (ed) Mycorrhizal functioning: an integrative plant-fungal process. Chapman and Hall, New York, pp 357–423
Murata M, Kinoshita A, Nara K (2013) Revisiting the host effect on ectomycorrhizal fungal communities: implications from host–fungal associations in relict Pseudotsuga japonica forests. Mycorrhiza 23:641–653
Nara K (2006) Ectomycorrhizal networks and seedling establishment during early primary succession. New Phytol 169:169–178
Nara K, Nakaya H, Wu B, Zhou Z, Hogetsu T (2003) Underground primary succession of ectomycorrhizal fungi in a volcanic desert on Mount Fuji. New Phytol 159:743–756
Oksanen J, Blanchet FG, Kindt R, Legendre P, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2011) vegan: community ecology package. R package version 2.0-10 Available at http://CRAN.R-project.org/package=vegan
Ono M (1989) Phytogeography in Ogasawara and Ryukyu (Nansei) Islands (in Japanese). In: Miyawaki A (ed) Vegetation of Japan, vol. 10, Okinawa and Ogasawara. Shibundo Co., Tokyo, pp 127–137
Peay KG, Garbelotto M, Bruns TD (2009) Spore heat resistance plays an important role in disturbance-mediated assemblage shift of ectomycorrhizal fungi colonizing Pinus muricata seedlings. J Ecol 97(3):537–547
Põlme S, Bahram M, Yamanaka T, Nara K, Dai YC, Grebenc T, Kraigher H, Toivonen M, Wang P, Matsuda Y, Naadel T, Kennedy PG, Kõljalg U, Tedersoo L (2013) Biogeography of ectomycorrhizal fungi associated with alders (Alnus spp.) in relation to biotic and abiotic variables at the global scale. New Phytol 198:1239–1249
Pringle A, Bever JD, Gardes M, Parrent JL, Rillig MC, Klironomos JN (2009) Mycorrhizal symbioses and plant invasions. Annu Rev Ecol Evol Syst 40:699–715
Rinaldi AC, Comadini O, Kuyper TW (2008) Ectomycorrhizal fungal diversity: separating the wheat from the chaff. Fungal Divers 33:1–45
Rineau F, Courty PE (2011) Secreted enzymatic activities of ectomycorrhizal fungi as a case study of functional diversity and functional redundancy. Ann Forest Sci 68:69–80
Roy M, Rochet J, Manzi S, Jargeat P, Gryta H, Moreau PA, Gardes M (2013) What determines Alnus-associated ectomycorrhizal community diversity and specificity? A comparison of host and habitat effects at a regional scale. New Phytol 198:1228–1238
Selosse MA, Setaro S, Glatard F, Richard F, Urcelay C, Weiss M (2007) Sebacinales are common mycorrhizal associates of Ericaceae. New Phytol 174:864–878
Smith ME, Douhan GW, Fremier AK, Rizzo DM (2009) Are true multihost fungi the exception or the rule? Dominant ectomycorrhizal fungi on Pinus sabiniana differ from those on co-occurring Quercus species. New Phytol 182:295–299
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Academic, Cambridge
Strauss SH, Doerksen AH, Byrne JR (1990) Evolutionary relationships of Doughlas-fir and its relatives (genus Pseudotsuga) from DNA restriction fragment analysis. Can J Bot 68:1502–1510
Taylor DL, Bruns TD (1999) Community structure of ectomycorrhizal fungi in a Pinus muricata forest: Minimal overlap between the mature forest and resistant propagule communities. Mol Ecol 8:1837–1850
Tedersoo L, Bahram M, Toots M, Diédhiou AG, Henkel TW, Kjøller R, Morris M, Nara K, Nouhra E, Peay KG, Põlme S, Ryberg M, Smith ME, Kõljalg U (2012) Towards global patterns in the diversity and community structure of ectomycorrhizal fungi. Mol Ecol 21:4160–4170
Tedersoo L, Jairus T, Horton BM, Abarenkov K, Suvi T, Saar I, Kõljalg U (2008) Strong host preference of ectomycorrhizal fungi in a Tasmanian wet sclerophyll forest as revealed by DNA barcoding and taxon-specific primers. New Phytol 180:479–490
Tedersoo L, May TW, Smith ME (2010) Ectomycorrhizal lifestyle in fungi: global diversity, distribution, and evolution of phylogenetic lineages. Mycorrhiza 20:217–263
Tedersoo L, Nara K (2010) General latitudinal gradient of biodiversity is reversed in ectomycorrhizal fungi. New Phytol 185(2):351–354
Tedersoo L, Suvi T, Beaver K, Kõljalg U (2007) Ectomycorrhizal fungi of the Seychelles: diversity patterns and host shifts from the native Vateriopsis seychellarum (Dipterocarpaceae) and Intsia bijuga (Caesalpiniaceae) to the introduced Eucalyptus robusta (Myrtaceae), but not Pinus caribea (Pinaceae). New Phytol 175:321–333
Twieg BD, Durall DM, Simard SW (2007) Ectomycorrhizal fungal succession in mixed temperate forests. New Phytol 176(2):437–447
Velmala SM, Rajala T, Haapanen M, Taylor AFS, Pennanen T (2013) Genetic host-tree effects on the ectomycorrhizal community and root characteristics of Norway spruce. Mycorrhiza 23:21–33
Wei XX, Yang ZY, Li Y, Wang XQ (2010) Molecular phylogeny and biogeography of Pseudotsuga (Pinaceae): insights into the floristic relationship between Taiwan and its adjacent areas. Mol Phylogenet Evol 55(3):776–785
Wu ZY, Raven PH (1999) Flora of China, volume 4, Cycadaceae through Fagaceae. Science Press and Missouri Botanical Garden, Beijing
Xiang QY, Soltis DE, Soltis PS (1999) The eastern Asian and eastern and western North American floristic disjunction: congruent phylogenetic patterns in seven diverse genera. Mol Phyl Evol 10(2):178–190
Acknowledgments
This research was funded by an award from the China Scholarship Council to ZW, by Japan Society for the Promotion of Science KAKENHI Grants to KN (25257411, 25660115, 25304026), and by an award from the National Natural Science Foundation of China-Guangdong Province Natural Science Foundation Joint Research Fund to YC (U1133004). We also thank Liang Shi, Haolin Zhu, and Qizhi Zha for their support in field sampling.
Conflict of Interest
The authors declare no conflict of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Duncan D. Cameron.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig. S1
(DOCX 571 kb)
Supplemental Fig. S2
(DOCX 987 kb)
Supplemental Fig. S3
(DOCX 6799 kb)
Supplemental Fig. S4
(DOCX 1243 kb)
Rights and permissions
About this article
Cite this article
Wen, Z., Murata, M., Xu, Z. et al. Ectomycorrhizal fungal communities on the endangered Chinese Douglas-fir (Pseudotsuga sinensis) indicating regional fungal sharing overrides host conservatism across geographical regions. Plant Soil 387, 189–199 (2015). https://doi.org/10.1007/s11104-014-2278-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2278-3