Abstract
Background and aims
Crop tolerance to waterlogging depends on factors such as species sensitivity and the stage of development that waterlogging occurs. The aim of this study was to identify the critical period for waterlogging on grain yield and its components, when applied during different stages of crop development in wheat and barley.
Methods
Two experiments were carried out (E1: early sowing date, under greenhouse; E2: late sowing date, under natural conditions). Waterlogging was imposed during 15–20 days in 5 consecutive periods during the crop cycle (from Leaf 1 emergence to maturity).
Results
The greatest yield penalties occurred when waterlogging was applied from Leaf 7 appearance on the main stem to anthesis (from 34 to 92 % of losses in wheat, and from 40 to 79 % in barley for E1 and E2 respectively). Waterlogging during grain filling reduced yield to a lesser degree. In wheat, reductions in grain number were mostly explained by reduced grain number per spike while in barley, by variations in the number of spikes per plant.
Conclusions
The time around anthesis was identified as the most susceptible period to waterlogging in wheat and barley. Exposing the crop to more stressful conditions, e.g. delaying sowing date, magnified the negative responses to waterlogging, although the most sensitive stage (around anthesis) remained unchanged.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Waterlogging is a global constraint on cropping systems due to the increased frequency of extreme climate events (Wollenweber et al., 2003). Many regions of the world (e.g. Australia, Canada, US) are frequently subjected to waterlogging, because of heavy rainfall, flat topography, and/or poor soil drainage (Collaku and Harrison 2002). Furthermore, in irrigated crops, waterlogging can occur as a result of poor drainage systems (Van Ginkel et al. 1997), or as a result of subsoil compaction as in the irrigated rice-wheat rotation, where the soil preparation for rice frequently results in soil compaction (Samad et al. 2001).
Soil is considered waterlogged when excess water saturates the soil pores (with a very thin—or even without—layer of water on the soil surface), inhibiting gas exchange between the roots and the atmosphere (Setter and Waters 2003; Striker 2012). Hence, the negative effect of waterlogging on crops is caused by low oxygen concentration in soil (Armstrong 1980). These oxygen deficient conditions lead plant roots to switch from an aerobic respiration to an anaerobic fermentation, limiting energy production (Huang and Johnson 1995; Gibbs and Greenway 2003). Waterlogging also produces increases in stomatal resistance, affecting nutrients and water uptake (Sairam et al. 2008), and decreasing leaf photosynthesis rate (Malik et al. 2001); as a consequence, crop growth rate decreases, and plants wilt.
Plant tolerance to waterlogging depends on several factors, including: i) the sensitivity per se of the species or variety (Cannell et al. 1984; Setter et al. 1999), ii) the crop stage of development that waterlogging occurs (Belford 1981; Setter and Waters 2003), and iii) the waterlogging event duration (Collaku and Harrison 2002; Malik et al. 2002). From an agronomic point of view, plant tolerance to waterlogging involves the maintenance of a relatively high grain yield under these conditions.
It has been widely reported that variations in yield are mainly explained by changes in grain number with a lesser influence of grain weight (Midmore et al. 1984; Fischer 1993; Slafer and Andrade 1993; González et al. 2005 for wheat; Abeledo et al. 2003; Prystupa et al. 2004 for barley; and Peltonen-Sainio et al. 2007 for both species). Grain number per unit area can be analyzed as the product of the number of spikes per unit area and the number of grains per spike, but the importance of each sub-component to determine grain number per unit area differs between both species. In general, grain number in barley strongly depends on the number of spikes, because the structure of the spike in two rowed barley (only one grain per spikelet) limits the number of grains that each spike could bear (García del Moral and García del Moral 1995; Arisnabarreta and Miralles 2006). In wheat, spikes contribution to determine grain number per unit area is less important than in barley, due to the compensation capacity of increasing the grain number per spike (Slafer 2003). The stage of development that waterlogging occurs may determine differences in the responses of wheat and barley to waterlogging. In wheat, the critical period for grain number determination was defined between 20 days pre-anthesis and 10 days post-anthesis (Fischer 1975), while the critical period for grain number determination in barley occurs earlier than in wheat (i.e. from 40 to 10 days before heading) and it is frequently associated with the number of spikes per m2 determination (Arisnabarreta and Miralles 2008). Defining the critical period of sensitivity to waterlogging in wheat and barley is hindered by the interactions between yield components and overlap of the phases where each component is determined. An alternative approach to analyzing grain yield variations is considering its definition in terms of the total aboveground biomass produced by the crop and its partitioning to the grain (i.e. harvest index). Cossani et al. (2009) showed, for Mediterranean environments, that differences in biomass at maturity between wheat and barley were correlated to their differences in post-flowering biomass accumulation. Consequently, the timing of waterlogging events can alter grain yield definition through modifications in biomass accumulation. The few studies where partitioning was measured separately between the different plant structures showed that the harvest index of the main stem was higher than that of the tillers (Hay 1995; Peltonen-Sainio et al. 2008). Therefore, different proportions of grain yield from the main stem or the tillers due to divergence between species (or cultivars) and/or growing conditions, can affect wheat and barley susceptibility to waterlogging.
Under farm production systems, barley is considered more susceptible to waterlogging than wheat, although few studies tested this ad hoc. A screening of six wheat varieties and eight of barley grown in field conditions under natural waterlogging (4 weeks of intermittent waterlogging) reported yield reductions of 19–82 % in wheat and 51–84 % in barley, depending on the genetic variability (Setter et al. 1999). Most studies that analyzed the effect of waterlogging on wheat or barley focused on specific stages of the crop cycle such as seedling (Musgrave 1994; Huang and Johnson 1995; Robertson et al. 2009), initial vegetative phases (Malik et al. 2001, 2002; Collaku and Harrison 2002) or grain filling (Hossain et al. 2011; Li et al. 2011). However, there are no published studies that have tested the susceptibility to waterlogging at different phenological stages throughout the complete crop cycle in wheat and barley growing simultaneously in the same environment, which take into account the physiological traits that are affected by waterlogging.
The aim of this study was to analyze the effect of waterlogging, applied during different growth stages throughout the entire crop cycle of wheat and barley, on grain yield and its components to identify the most sensitive period in both species.
Material and methods
Growing conditions
Two experiments were carried out at the School of Agronomy, University of Buenos Aires (34° 35′ S, 58° 29′ W) during 2010. Experiment 1 (E1) was carried out in a greenhouse and included Klein Chajá and Scarlett as wheat and barley cultivars, respectively. Experiment 2 (E2) was carried out under natural conditions of radiation and temperature and Baguette 13 and Scarlett were used as commercial cultivars of wheat and barley respectively. The barley cultivar chosen represents more than 95 % of the total barley area in Argentina (Miralles 2013) and wheat cultivars were chosen because they are similar to Scarlett in grain yield potential and phenology (similar time to anthesis). Sowing date was 2nd July for E1 and 6th September for E2. Both experiments were carried out using 12 L pots filled with clay loam soil (E1) or a mixture of sand and clay loam soil in a ratio 3:1 (E2) with a density of six plants per pot. In Experiment 2, carried out under field conditions, a sandy mixture was used in order to keep the control pots well drained in case of prolonged or heavy rainfall. In both experiments a layer of gravel (5 cm) was placed on the bottom of the pots to improve drainage conditions.
Experimental design was completely randomized with eight replicates for E1 and three replicates for E2. Both experiments were conducted without biotic stresses by applications of insecticides and fungicides. Weeds were removed by hand. Plants were fertilized with 2.5 g of Triple 15 (Yara Company) (15 % N:15 % P2O5:15 % K2O) per pot.
Waterlogging treatments
Six waterlogging treatments were imposed: one control well drained during all the crop cycle, and five waterlogging treatments applied at different stages from appearance of the first leaf (L1) and physiological maturity viz: (i) from leaf 1 to leaf 4 stages (L1-4), (ii) from leaf 4 to leaf 7 (L4-7), (iii) from leaf 7 to leaf 10 (L7-10), (iv) from leaf 10 to anthesis (L10-At), and (v) from anthesis to physiological maturity (At-PM). The leaf number at which waterlogging was applied was always measured on the main stem. Waterlogging duration was 20 (E1) or 15 (E2) days, in order to ensure the phenological stages at the beginning and ending of waterlogging treatments were consistent between the experiments. Higher temperatures and longer day durations in E2 compared to E1 caused more rapid development. Crop phenology was determined following the decimal code of Zadoks et al. (1974). Anthesis in barley was determined by opening the spikelets and visualizing pollen release.
In order to impose waterlogging treatments, pots were placed into containers (1 m × 1 m × 0.5 m) with 1 cm layer of free water above the surface of the pots during the whole period of each waterlogging treatment. At the end of each waterlogging treatment, pots were taken out of the containers and remained without irrigation during 10 days, allowing to drain freely, and after that they were re-watered normally. Control pots, from sowing to physiological maturity, together with the waterlogging pots before and after treatments were maintained at 80 % of field capacity through irrigation. Volumetric humidity content on the top and bottom of the pots was continuously monitored (AT Theta Kit HH2 Moisture content, Delta Devices, UK), and irrigation were applied when necessary (approximately every 3 days in the control situation).
Measurements
At maturity, total above-ground biomass was harvested in each pot and separated into main stem and tillers, and within each category, biomass was divided into vegetative biomass (shoots plus sheaths and leaves) and spikes. Then, material was oven- dried at 65 °C until constant weight and dry weight was measured. The number of spikes from the main stem and from tillers was counted and threshed. Harvest index (HI) was calculated as the ratio between grain yield and total above-ground biomass. Average individual grain weight of the main stem and tillers was measured in two sub-samples of 50 grains per category and used to calculate grain number per spike in both strata. Meteorological data (air minimum and maximum temperature, photosynthetically active radiation, and relative humidity) were recorded every hour throughout the crop cycle in both experiments by an automatic meteorological station (Davis Vantage Pro2, USA) placed in the same site where each experiment was carried out. The vapor pressure deficit (VPD, kPa) was estimated as the difference between the actual air vapor pressure (ea) and the saturated vapor pressure (es), following the Clausius-Clayperon equation:
where es (i) is the saturated vapor pressure (es, kPa) of the day i, e is the natural number, Tm(i) is the mean temperature of the day (i) (°C).
where, for the day i, ea (i) is the air vapor pressure (kPa), es (i) is the saturated vapor pressure (kPa), and HR (i) is the mean daily relative humidity (%).
Statistical analysis
Statistical differences between treatments were tested through analyses of variance (ANOVA) using InfoStat Professional v.1.1 (Di Rienzo et al. 2010). The mean treatment values were compared using Tukey test with significance level of 0.05. The degree of association between different variables was made using linear regression models.
Results
Environmental conditions and phenology
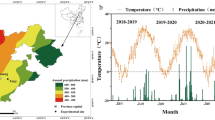
Mean values of temperature, radiation, photoperiod, and vapor pressure deficit (VPD) during different phenological stages and experiments are shown in Fig. 1. The average temperature during the crop cycle (i.e. between the appearance of L1 and physiological maturity) in E2 was on average 4.3 °C higher than in E1. This difference was a consequence of increases in minimum (5.3 °C higher in E2 than in E1) rather than in maximum temperature (1.1 °C higher in E2 than in E1). Accumulated global radiation during crop cycle in E1 was 935 MJ m−2 while in E2 was 1,759 MJ m−2. The greater accumulated radiation in E2 than in E1 (despite the shorter cycle) was due to E1 being conducted in a greenhouse where incident radiation was reduced in ca. 30 % compared to field conditions. Average photoperiod was almost 2 h longer in E2 than in E1 and average VPD in E2 was ca. 36 % higher than in E1.
Meteorological conditions during the crop cycle in the two experiments: E1 (early sowing date in greenhouse; upper panel) and E2 (late sowing date under natural conditions; bottom panel). Values are daily means of medium (Tm), maximum (Tmax) and minimum (Tmin) temperature, daily photosynthetic incident radiation (Rad), daily photoperiod (Photop), and air vapor pressure deficit (VPD). Bars into the graphs indicate the different timing waterlogging treatments, where L with their numbers refers to the number of leaves on the main stem when treatments were applied, At stands for anthesis and PM stands for physiological maturity
Within each experiment, phenology of the controls without waterlogging was similar between wheat and barley, but the duration of the cycle from emergence to physiological maturity was on average 22 days longer in E1 than in E2 (111 days in wheat and barley in E1 vs. 93 days in wheat and 85 days in barley in E2) as a consequence of warmer temperatures and longer photoperiods in E2. In E1, plants emerged on July 9th for both species and in E2 seedling emergence of both species was on September 12th. The duration of the controls pre-anthesis phase was 83 days in E1 and 62 days in E2, without significant differences between species (p > 0.10), while the post-anthesis phase lasted 28 and 27 days in E1 and E2, respectively. Waterlogging did not affect the duration of the phases, except when treatments were applied around the beginning of tillering (L4-7) where a significant delay (p < 0.05) in the time to anthesis of wheat and barley (of 7 and 13 days respectively, in E1 and E2) was observed.
Yield differences between wheat and barley under control conditions
Grain yield and its components for wheat and barley under no waterlogging conditions (control treatments) are described in Table 1. Barley grain yield was significantly higher (p < 0.05) than that of wheat (4.09 g pl−1 for barley vs. 2.73 g pl−1 for wheat, average of the two experiments) with no interaction between experiments (p > 0.10). Total above-ground biomass at maturity and harvest index were also higher (p < 0.05) for barley than for wheat. Differences between species in grain number per plant were not significant; however, grain weight was higher in barley than in wheat in both experiments (p < 0.05). Grain yield and above-ground biomass per plant in control treatments were significantly lower in E2 than in E1 in both species, while HI was not modified by the delay in sowing date (Table 1).
The contribution to the total grain yield from main stem or tillers was different between species and experiments (Table 1). In wheat, the contribution to grain yield or total above-ground biomass under control conditions in E1 was similar between main stem and tillers (the main stem accounted for ca. 55 % of total grain yield and ca. 50 % of total biomass per plant), while in barley grain yield contribution was mainly from tillers in both experiments as main stem contribution to total grain yield and total biomass was only 20 %. In E2 the differences between species followed the same trend (i.e. 48 % contribution of main stem in wheat and 34 % in barley). The absolute values of crop growth presented in Table 1 provide opportunities to calculate the impacts of waterlogging treatments on growth which for convenience are expressed in following sections on a percentage control basis.
Yield reduction by waterlogging
Yield, biomass and partitioning (HI)
There was a greater impact of waterlogging effects on E2 than on E1, while the magnitude of the reduction in grain yield due to waterlogging in both experiments varied according to crop stage (Fig. 2a). In both species, the greatest losses in yield due to waterlogging were in the treatments where waterlogging occurred immediately prior to anthesis (L7-10 and L10-At) and to a lesser degree during grain filling (At-PM) (p < 0.05) (Fig. 2a). Yield losses (relative to the control) were ca. 34 to 92 % in wheat and ca. 40 to 79 % in barley, for E1 and E2 respectively. Waterlogging events during early vegetative stages (L1-4) did not generate significant losses in grain yield for the early sowing in E1, although in the later sowing in E2, the losses were ca. 40 % for both species (p < 0.05). In general, the yield loss was greater as the crop cycle progressed to anthesis (Fig. 2a).
Variation in grain yield (a–c), total above-ground biomass at maturity (d–f) and harvest index (g–i) measured in the total plant (left panels) and in main stems (central panels) and tillers (right panels) relative to the control for different waterlogging treatments applied at different stages throughout the crop cycle. Each point is indicated as the midpoint of waterlogging period (days from anthesis) in wheat and barley in two experiments (E1: early sowing date, under greenhouse, and E2: late sowing date, under natural conditions). Duration of the waterlogging events was 20 days in E1 and 15 days in E2. Vertical bars indicate ± standard error
Waterlogging affected the contribution to grain yield from main stem and tillers differently, depending on the species and sowing date. In wheat, waterlogging produced yield reductions in main stem as well as in tillers in both experiments (Fig. 2b–c). In barley, waterlogging strongly reduced the yield contribution from tillers more than from main stems (p < 0.05), with maximum losses in tillers from treatments applied around anthesis (ca. 45 % in E1 and 89 % in E2) (Fig. 2b–c). Only the treatments L10-At and At-PM significantly reduced the barley yield from main stem with losses of around 25 % in E1 and 58 % in E2 (Fig. 2b–c).
Waterlogging reduced total above-ground biomass at maturity in a similar way to that observed for grain yield: treatments immediately prior to anthesis significantly reduced total biomass at maturity (p < 0.05) (Fig. 2d). In fact, the greatest reductions of biomass at maturity due to waterlogging occurred in treatments L7-10 and L10-At, and the magnitude of biomass reductions were from 24 to 66 % for wheat and from 34 to 60 % for barley. Biomass losses in wheat were slightly higher in tillers than in main stems, especially in E2 (Fig. 2e–f), reaching losses of 60 % in main stem and 70 % in tillers (L7-10). In the case of barley, tiller biomass was more affected than that of main stems, with maximum reduction of 37 % in E1 and 72 % in E2 for tillers and 20 % in E1 and 30 % in E2 for main stems (Fig. 2e–f). Variations in yield due to waterlogging were strongly explained by changes in biomass for both species and experiments (r 2 = 0.95; p < 0.001 for all data set) (Fig. 3a).
Relationship between grain yield and a total above-ground biomass at maturity and b harvest index for wheat and barley cultivars exposed to waterlogging through the crop cycle in two experiments (E1: early sowing date, under greenhouse, and E2: late sowing date, under natural conditions). Solid lines represent the linear regressions for the different data sets
Waterlogging not only reduced the biomass at maturity, but also the partitioning to the grain. Events of waterlogging from L7 to physiological maturity significantly reduced wheat HI in both experiments (Fig. 2g), with a maximum reduction of ca. 27 % in E1 (treatment At-PM) and ca. 84 % in E2 (treatment L10-At). Those reductions occurred as a consequence of decreases in HI of main stem as well as in tillers, and with similar magnitude (Fig. 2g–i). The decrease in HI due to waterlogging was lower in barley than in wheat (Fig. 2g–i). Variations in yield were explained by changes in HI but not with a single regression for both experiments due to the large difference in biomass between E1 and E2. Thus, for the relationship between grain yield and harvest index, two linear regressions were fitted separately for each experiment (Fig. 3b). E1 showed higher HI than E2, while E2 showed a wider range of variation.
Yield components
Yield components were also affected differently by waterlogging (Fig. 4). In wheat, the greatest reductions in grain number per plant due to waterlogging were during L7-10 treatment in both experiments, with losses of 27 % and 74 % in E1 and E2 respectively (p < 0.05) (Fig. 4a). In E1, wheat grain number contribution from the main stem and tillers was similarly reduced by waterlogging (25 and 28 % reduction respectively), but in E2, grain number from tillers showed higher reductions than those from main stems (64 % reduction in main stem vs. 82 % in tillers relative to control) (Fig. 4b–c). Reductions in grain number per plant in wheat were consistent with decreases in the grain number per spike in main stems and tillers, especially in E2, where reductions in grains per spike were ca. 65 % with respect to the control in both strata, compared to ca. 20 % of reduction in E1 (Fig. 4g–i). In both species, the number of spikes from main stems were not affected by waterlogging (p > 0.10) as no plant mortality was evident in any experiment (Fig. 4e).
Variation in grain number (a–c), spikes per plant (d–f) and grain number per spike (g–i) measured in the total plant (left panels), and in the main stems (central panels) and tillers (right panels) relative to the control for different waterlogging treatments applied at different stages throughout the crop cycle. Each point is indicated as the midpoint of waterlogging period (days from anthesis) in wheat and barley in two experiments (E1: early sowing date, under greenhouse, and E2: late sowing date, under natural conditions). Duration of the waterlogging events was 20 days in E1 and 15 days in E2. Vertical bars indicate ± standard error
In barley, the reductions in grain number per plant reached 35 % in E1 and 60 % in E2, due to a large reduction in grain number from tillers (ca. 40 % in E1 and 75 % in E2), and less impact on main stem (ca. 10 % in E1 and 30 % in E2) (Fig. 4b–c). Conversely to wheat, the spike number per plant was the main sub-component affected by waterlogging in barley, with losses of ca. 40 % in E1 and 63 % in E2 (Fig. 4d–f). Thus, the spike number per plant was the main component that explained the variations in the number of grains per plant, since the grain number per spike was slightly reduced only in the main stem (10 % in E1, treatment L10-At and 30 % in E2, treatment L4-7) (Fig. 4d–i).
Grain weight was reduced by waterlogging treatments applied prior to anthesis (L7-10 and L10-At), as well as during grain filling period (p < 0.05), and main stem and tillers were affected similarly in both species (Fig. 5). The greatest reductions in grain weight were observed in waterlogging treatments At-PM and L10-At in the E1 and E2 experiments, respectively; and in general, wheat was more affected than barley. In E2, barley grain weight was 15 mg per grain (55 % reduction with respect to control) while wheat grains did not complete their filling and were shriveled, with weights of 3.6 mg per grain (losses of 83 % with respect to control).
Variation in average individual grain weight (a–c) relative to the control measured in the total plant (left panels), in main stems (central panels) and tillers (right panels) for different waterlogging treatments applied at different stages throughout the crop cycle. Each point is indicated as the midpoint of waterlogging period (days from anthesis) in wheat and barley in two experiments (E1: early sowing date, under greenhouse, and E2: late sowing date, under natural conditions). Duration of the waterlogging events was 20 days in E1 and 15 days in E2. Vertical bars indicate ± standard error
Variations in yield due to waterlogging were largely explained by changes in grain number per plant in both species and experiments and all data were fitted by a common linear regression (r 2 = 0.89; p < 0.001; Fig. 6a). However, as waterlogging reduced grain weight, variations in grain yield were also explained by changes in grain weight, especially in E2 (r 2 = 0.66, p < 0.001 for E1 and r 2 = 0.76, p < 0.001 for E2; Fig. 6b). In spite of the fact that waterlogging produced similar reductions on grain number in both species, the grain number sub-components were affected differently depending on the species. In wheat, 99 % of reductions in grain number were explained by variations in grain number per spike (Fig. 6d), and there was no relation with the number of spikes per plant (Fig. 6c). Conversely, barley grain number reductions by waterlogging were explained mainly by variations in the number of spikes per plant (r 2 = 0.96; p < 0.001; Fig. 6c) and to a minor extent by the number of grains per spike (r 2 = 0.76; p < 0.001; Fig. 6d).
Grain yield plotted against a grain number and b grain weight; and relationship between grain number and c spike number per plant and d grains per spike for wheat and barley cultivars exposed to waterlogging during the crop cycle in two experiments (E1: early sowing date, under greenhouse, and E2: late sowing date, under natural conditions). Solid lines represent the linear regressions for the different data sets
Discussion
One of the previous main attempts to identify the critical periods of sensitivity to waterlogging in small grain crops was by Setter and Waters (2003), who reviewed several studies about the effects of waterlogging in wheat and barley, including information on different varieties, timings and duration of waterlogging. That review suggested that reproductive stages were more adversely affected than the vegetative stages of growth. However, in the experiments included in that revision, there were no studies where wheat and barley were exposed to waterlogging either during different stages of development, or both species simultaneously in the same environment. In the present study, the effect of waterlogging on grain yield and its components was tested during the whole cycle of wheat and barley in experiments in which both species were exposed to the same environmental conditions. Our results demonstrate that the period from the beginning of stem elongation to anthesis (treatments L7-10 and L10-At) in wheat, as well as in barley, were the most sensitive to waterlogging in terms of yield penalties.
Yield reductions were the consequence of decreases in the total above-ground biomass produced at maturity, as well reductions in its partitioning to reproductive organs, especially in E2, where the HI was strongly depressed. The greatest reductions in biomass at maturity occurred when the waterlogging treatment was applied during stem elongation, which coincides with the period of highest crop growth rate under ideal conditions (Miralles and Slafer 2007; Arisnabarreta and Miralles 2008). Thus, biomass reductions when crops are waterlogged during the stem elongation phase can result from reductions in the plant growth rate, as well as increases in tiller mortality. Waterlogging after stem elongation reduced maturity biomass contribution from tillers more strongly than biomass from main stem.
The difference in the magnitude of yield reductions between the experiments (higher in E2 than in E1) could be associated with the differences in the environment experienced by the crops during the cycle, as plants in E2 had more stressful conditions than in E1. In the present study, incident radiation was 30 % lower in E1 than in E2, while temperatures and VPD were higher in E2 than in E1, which increased the atmospheric transpiration demand in E2 and presumably increased plant stress. The higher temperature in E2 could also raise the rate of oxygen depletion of soil water by increasing the biological oxygen demand from roots and microorganism (Collaku and Harrison 2002). In sunflower, it was shown that reductions in the environmental demand (by shading), when crop faced waterlogging during post-anthesis, decreased the negative effect on grain yield (Grassini et al. 2007). Moreover, the shorter crop cycle duration in E2 due to a long photoperiod and high temperatures resulted in smaller and weaker plants, thereby amplifying the waterlogging stress effects in E2 more than in E1.
In the present study, waterlogging events during the initial phases of the crop cycle did not produce significant reductions in grain yield in wheat or barley, which was consistent with the results shown by Cannell et al. (1980) with early waterlogging in wheat. Nevertheless, several studies showed a decrease in biomass production during vegetative stages with early waterlogging events (Malik et al. 2001, 2002; Pang et al. 2004). On the one hand, it is possible to speculate that waterlogging occurring early in the crop cycle allows plants to recover from stress through different mechanisms along the crop cycle once the stress has been removed. In barley, waterlogging during initial phases could reduce tiller appearance similarly to what happens with nutrients deficiencies (Alzueta et al. 2012) and water restrictions (Cossani et al. 2009). However, the final number of fertile spikes, the principal yield sub component in this species, depends on tiller appearance rate (Alzueta et al. 2012) and the percentage of tiller mortality (Baethgen et al. 1995; García del Moral and García del Moral 1995). Thus, waterlogging occurring early during the crop cycle could affect tiller appearance rate, but tiller mortality could have been reduced, without remarkable reductions in the final spike number per plant. In wheat, Robertson et al. (2009) showed that waterlogging during the early stages of the crop delayed the tillering period promoting the appearance of higher order tillers and thereby delaying the time to anthesis. In our experiment, a similar effect, as previously reported by Robertson et al. (2009) was observed, since anthesis date was delayed when plants were under waterlogging during early stages of development, probably associated with tiller appearance of higher order. Those higher order tillers could compensate the early tiller mortality and thereby the final spikes number per plant was equal, or even slightly higher than in controls. On the other hand, it is possible to speculate that oxygen depletion rate in the pots is lower during waterlogging applied during early developmental stages (when the root system is smaller and the oxygen consumption is lower) than those during more advanced stages. Consequently, waterlogging events at the beginning of the crop cycle would be less severe than waterlogging events of similar duration applied near anthesis, where the root system has a larger development and the oxygen in the soil solution could be quickly exhausted compared to the early stages during the crop cycle. However, many studies reported that in submerged soils, oxygen in the soil solution is practically exhausted within few hours of beginning of the waterlogging event (Ponnamperuma 1972), and during the next few days after waterlogging was applied (i.e. between 5 and 10 days), the soil redox potential decreased to levels associated with anoxia (Striker et al. 2005; Setter et al. 2009).
Waterlogging during the critical period (i.e. from stem elongation to anthesis) reduced yield in both wheat and barley cultivars as a consequence of reductions in grain number and grain weight (although in lesser magnitude for the latter). In our study, the critical period for reduced grain number as a consequence of waterlogging was L7-10 and L10-At, coinciding with the critical periods for grain number determination identified previously in wheat (Fischer 1975) and barley (Arisnabarreta and Miralles 2008) by reductions in the incident solar radiation. In barley, yield reductions due to waterlogging were mainly due to a diminished grain number contribution from tillers, and spike number per plant. Different evidence demonstrated that in barley, rather than in wheat, the number of spikes per plant is the main component that explains the variations in grain number (Abeledo et al. 2003; García del Moral et al. 2003; Alzueta et al. 2012). In the present study, although spike number per plant explained most of variation in grain number (96 %), grain number per spike also explained an important proportion (79 %) of the variation in grain number per plant due to waterlogging. However, the variation in grains per spike in barley due to different waterlogging treatments was narrow (from 10 to 20 grains per spike) compared to wheat (from 10 to 45 grains per spike), confirming the low plasticity to modify this component for abiotic stress tolerance, compared to wheat (Arisnabarreta and Miralles, 2006). Thus, the main yield component that was reduced by waterlogging in wheat was the number of grains per spike and it was affected equally in main stems and tillers (ca. 70 %). That sub-component explained most of the variation in the number of grains per plant (99 %), as spikes per plant did not show significant correlation with grains per plant.
In E1, waterlogging occurring during grain filling (treatment At-PM) also produced reductions in grain yield of wheat and barley of similar magnitude to those caused by treatments prior to anthesis. This occurred as a consequence of reductions in grain weight in both species, because grain number was not reduced by waterlogging during grain filling in any species. The reduction in grain weight in E1 was ca. 30 %, (similarly to that reported by Hossain et al. 2011 when waterlogging was applied during grain filling) which was a relatively low value considering that waterlogging occurred when endospermatic cells are being defined (i.e. around 2 weeks after anthesis; Blocklehurst 1977). It is possible to speculate that most of the source to complete grain weight came from carbohydrates accumulated in the stems and translocated to the grains as photosynthetic active leaf area was severely affected (Serrago et al. 2011). However, the reductions in grain weight suggest that translocation was not enough to fulfill the grains previously established. In this sense, it was shown that reductions in grain growth due to waterlogging during grain filling were attributable to decreased current assimilation and poor remobilization of water soluble carbohydrates from the stem to the grains (Jiang et al. 2008; Hossain et al. 2011), suggesting that carbohydrate translocation is also negatively affected by waterlogging, probably through an effect on the roots functionality.
The fact that in E2 the greatest reductions in grain weight occurred during waterlogging treatments prior to anthesis (L7-10 and L10-At) (84 % in wheat and 55 % in barley), suggests that grain size potential could be affected by waterlogging. In wheat, Calderini et al. (2001) demonstrated that reductions in carpel size determined reductions in potential grain weight. Similarly in barley, the importance of pre-anthesis environment to determine potential grain weight was also demonstrated (Scott et al. 1983; Bingham et al. 2007; Ugarte et al. 2007). Thus, waterlogging around anthesis in E2 was likely to reduce grain size potential through reductions in carpel size, as well as in the number of endospermatic cells determining lighter grain weight, while in the previous treatment (L7-10) there is an additional effect on the photosynthetically active leaf area.
Despite the fact that waterlogging conditions affect grain yield through different pathways in both species, wheat and barley cultivars used in our experiments (i.e. Scarlett - barley - and Klein Chajá and Baguette 13 -wheat -) did not show differences in their sensitivity when waterlogging treatments were applied at different stages of the crop cycle. Although barley appeared to be more chlorotic and wilt than wheat during the early waterlogging treatments (data not shown), those effects were not correlated later with greater reductions in grain yield. It is known that genotypes differ in sensitivity (Setter and Waters 2003). However, little information is known about the mechanisms of tolerance in wheat (Musgrave 1994; Musgrave and Ding 1998; Setter et al. 1999; Collaku and Harrison 2002), and especially of barley (Setter et al. 1999) to waterlogging.
In summary, the time around anthesis was identified as the most susceptible period to waterlogging in wheat, as well as in barley, while treatments during initial stages of the crop cycle did not produce significant reductions in grain yield. Waterlogging reduced grain yield in both wheat and barley cultivars to the same magnitude, but the yield components were affected differently between the species. The environment experienced during specific stages of growth modified the magnitude of wheat and barley responses to waterlogging. Exposing the crop to a more stressful environment by delayed sowing magnified the negative responses, although it did not modify the most sensitive period to waterlogging.
References
Abeledo LG, Calderini DF, Slafer GA (2003) Genetic improvement of barley yield potential and its physiological determinants in Argentina (1944–1998). Euphytica 130(3):325–334
Alzueta I, Abeledo LG, Mignone CM, Miralles DJ (2012) Differences between wheat and barley in leaf and tillering coordination under contrasting nitrogen and sulfur conditions. Eur J Agron 41(1):92–102. doi:10.1016/j.eja.2012.04.002
Arisnabarreta S, Miralles DJ (2006) Yield responsiveness in two- and six-rowed barley grown in contrasting nitrogen environments. J Agron Crop Sci 192(3):178–185
Arisnabarreta S, Miralles DJ (2008) Critical period for grain number establishment of near isogenic lines of two- and six-rowed barley. Field Crop Res 107(3):196–202
Armstrong W (1980) Aeration in higher plants. In: Woolhouse HW (ed) Advances in botanical research, vol 7. London, pp 225–332
Baethgen WE, Christianson CB, Lamothe AG (1995) Nitrogen fertilizer effects on growth, grain yield, and yield components of malting barley. Field Crop Res 43(2–3):87–99. doi:10.1016/0378-4290(95)00034-N
Belford RK (1981) Response of winter wheat to prolonged waterlogging under outdoor conditions. J Agric Sci 97(3):557–568. doi:10.1017/S0021859600036881
Bingham IJ, Blake J, Foulkes MJ, Spink J (2007) Is barley yield in the UK sink limited?: II. Factors affecting potential grain size. Field Crop Res 101(2):212–220
Blocklehurst PA (1977) Factors controlling grain weight in wheat. Nature 266:348–349
Calderini DF, Savin R, Abeledo LG, Reynolds MP, Slafer GA (2001) The importance of the period immediately preceding anthesis for grain weight determination in wheat. Euphytica 119(1):199–204
Cannell RQ, Belford RK, Gales K, Dennis CW, Prew RD (1980) Effects of waterlogging at different stages of development on the growth and yield of winter wheat. J Sci Food Agric 31 (2):117–132. doi:10.1002/jsfa.2740310203
Cannell R, Belford R, Gales K, Thomson R, Webster C (1984) Effects of waterlogging and drought on winter wheat and winter barley grown on a clay and a sandy loam soil. Plant Soil 80(1):53–66. doi:10.1007/bf02232939
Collaku A, Harrison SA (2002) Losses in wheat due to waterlogging. Crop Sci 42(2):444–450. doi:10.2135/cropsci2002.4440
Cossani CM, Slafer GA, Savin R (2009) Yield and biomass in wheat and barley under a range of conditions in a Mediterranean site. Field Crop Res 112(2–3):205–213
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2010) InfoStat Profesional. 2010 edn. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Córdoba, Argentina
Fischer RA (1975) Yield potential of dwarf spring wheat and the effect of shading. Crop Sci 15:607–613
Fischer RA (1993) Irrigated spring wheat and timing and amount of nitrogen fertilizer. II. Physiology of grain yield response. Field Crop Res 33(1–2):57–80
García del Moral MB, García del Moral LF (1995) Tiller production and survival in relation to grain yield in winter and spring barley. Field Crop Res 44(2–3):85–93. doi:10.1016/0378-4290(95)00072-0
García del Moral LF, García B, del Moral M, Molina-Cano JL, Slafer GA (2003) Yield stability and development in two- and six-rowed winter barleys under Mediterranean conditions. Field Crop Res 81(2–3):109–119
Gibbs J, Greenway H (2003) Mechanisms of anoxia tolerance in plants. I. Growth, survival and anaerobic catabolism. Funct Plant Biol 30(1):1–47
González FG, Slafer GA, Miralles DJ (2005) Photoperiod during stem elongation in wheat: is its impact on fertile floret and grain number determination similar to that of radiation? Funct Plant Biol 32(3):181–188
Grassini P, Indaco GV, Pereira ML, Hall AJ, Trápani N (2007) Responses to short-term waterlogging during grain filling in sunflower. Field Crop Res 101(3):352–363
Hay RKM (1995) Harvest index: a review of its use in plant breeding and crop physiology. Ann Appl Biol 126(1):197–216
Hossain MA, Araki H, Takahashi T (2011) Poor grain filling induced by waterlogging is similar to that in abnormal early ripening in wheat in Western Japan. Field Crop Res 123(2):100–108. doi:10.1016/j.fcr.2011.05.005
Huang B, Johnson JW (1995) Root respiration and carbohydrate status of two wheat genotypes in response to hypoxia. Ann Bot 75(4):427–432. doi:10.1006/anbo.1995.1041
Jiang D, Fan X, Dai T, Cao W (2008) Nitrogen fertiliser rate and post-anthesis waterlogging effects on carbohydrate and nitrogen dynamics in wheat. Plant Soil 304(1–2):301–314
Li C, Jiang D, Wollenweber B, Li Y, Dai T, Cao W (2011) Waterlogging pretreatment during vegetative growth improves tolerance to waterlogging after anthesis in wheat. Plant Sci 180(5):672–678. doi:10.1016/j.plantsci.2011.01.009
Malik AI, Colmer TD, Lambers H, Schortemeyer M (2001) Changes in physiological and morphological traits of roots and shoots of wheat in response to different depths of waterlogging. Aust J Plant Physiol 28(11):1121–1131
Malik AI, Colmer TD, Lambers H, Setter TL, Schortemeyer M (2002) Short-term waterlogging has long-term effects on the growth and physiology of wheat. New Phytol 153(2):225–236. doi:10.1046/j.0028-646X.2001.00318.x
Midmore PM, Cartwright PM, Fischer RA (1984) Wheat in tropical environments. II. Crop growth and grain yield. Field Crop Res 8:207–227
Miralles D (2013) Agronomic and varietal characteristics driving the growth of the Argentinian Barley industry. In: The 16th Australian Barley technical symposium: the Australian Barley industry goes global, Australia, p 53
Miralles DJ, Slafer GA (2007) Sink limitations to yield in wheat: how could it be reduced? J Agric Sci 145:139–149
Musgrave ME (1994) Waterlogging effects on yield and photosynthesis in eight winter wheat cultivars. Crop Sci 34(5):1314–1318. doi:10.2135/cropsci1994.0011183X003400050032x
Musgrave ME, Ding N (1998) Evaluating wheat cultivars for waterlogging tolerance. Crop Sci 38(1):90–97. doi:10.2135/cropsci1998.0011183X003800010016x
Pang J, Zhou M, Mendham N, Shabala S (2004) Growth and physiological responses of six barley genotypes to waterlogging and subsequent recovery. Aust J Agric Res 55(8):895–906. doi:10.1071/AR03097
Peltonen-Sainio P, Kangas A, Salo Y, Jauhiainen L (2007) Grain number dominates grain weight in temperate cereal yield determination: evidence based on 30 years of multi-location trials. Field Crop Res 100(2–3):179–188. doi:10.1016/j.fcr.2006.07.002
Peltonen-Sainio P, Muurinen S, Rajala A, Jauhiainen L (2008) Variation in harvest index of modern spring barley, oat and wheat cultivars adapted to northern growing conditions. J Agric Sci 146:35–47
Ponnamperuma FN (1972) The chemistry of submerged soils. In: Sparks DL (ed) Advances in Agronomy, vol 24. Academic Press, pp 29–96
Prystupa P, Savin R, Slafer GA (2004) Grain number and its relationship with dry matter, N and P in the spikes at heading in response to N × P fertilization in barley. Field Crop Res 90(2–3):245–254
Robertson D, Zhang H, Palta JA, Colmer T, Turner NC (2009) Waterlogging affects the growth, development of tillers, and yield of wheat through a severe, but transient, N deficiency. Crop Pasture Sci 60(6):578–586. doi:10.1071/CP08440
Sairam R, Kumutha D, Ezhilmathi K, Deshmukh P, Srivastava G (2008) Physiology and biochemistry of waterlogging tolerance in plants. Biol Plant 52(3):401–412. doi:10.1007/s10535-008-0084-6
Samad A, Meisner CA, Saifuzzaman M, Van Ginkel M (2001) Waterlogging tolerance. In: Reynolds MP, Ortiz-Monasterio JI, McNab A (eds) Application of physiology in wheat breeding. CIMMYT, Mexico, pp 135–144
Scott WR, Appleyard M, Fellowes G, Kirby EJM (1983) Effect of genotype and position in the ear on carpel and grain growth and mature grain weight of spring barley. J Agric Sci 100(2):383–391. doi:10.1017/S0021859600033530
Serrago RA, Carretero R, Bancal MO, Miralles DJ (2011) Grain weight response to foliar diseases control in wheat (Triticum aestivum L.). Field Crop Res 120(3):352–359. doi:10.1016/j.fcr.2010.11.004
Setter TL, Waters I (2003) Review of prospects for germplasm improvement for waterlogging tolerance in wheat, barley and oats. Plant Soil 253(1):1–34. doi:10.1023/a:1024573305997
Setter TL, Burguess P, Waters I, Kuo J (1999) Genetic diversity of barley and wheat for waterlogging tolerance in Western Australia. In: 9th Australian Barley technical symposium, Melbourne, 1999. Australian Barley Technical Symposium
Setter TL, Waters I, Sharma SK, Singh KN, Kulshreshtha N, Yaduvanshi NPS, Ram PC, Singh BN, Rane J, McDonald G, Khabaz-Saberi H, Biddulph TB, Wilson R, Barclay I, McLean R, Cakir M (2009) Review of wheat improvement for waterlogging tolerance in Australia and India: the importance of anaerobiosis and element toxicities associated with different soils. Ann Bot 103(2):221–235. doi:10.1093/aob/mcn137
Slafer GA (2003) Genetic basis of yield as viewed from a crop physiologist’s perspective. Ann Appl Biol 142(2):117–128
Slafer GA, Andrade FH (1993) Physiological attributes related to the generation of grain yield in bread wheat cultivars released at different eras. Field Crop Res 31(3–4):351–367
Striker GG (2012) Flooding stress on plants: anatomical, morphological and physiological responses. In: Mworia JK (ed) Botany. InTech, pp 3–28. doi:10.5772/2245
Striker G, Insausti P, Grimoldi A, Ploschuk E, Vasellati V (2005) Physiological and anatomical basis of differential tolerance to soil flooding of Lotus corniculatus L. and Lotus glaber mill. Plant Soil 276(1–2):301–311. doi:10.1007/s11104-005-5084-0
Ugarte C, Calderini DF, Slafer GA (2007) Grain weight and grain number responsiveness to pre-anthesis temperature in wheat, barley and triticale. Field Crop Res 100(2–3):240–248
Van Ginkel M, Sayre K, Boru G (1997) La tolerancia al anegamiento en el trigo: problemas relacionados con el fitomejoramiento. In: Mohan Kohli M, Martino DL (eds) Explorando altos rendimientos en trigo. CIMMYT, INIA, Colonia, pp 193–208
Wollenweber B, Porter JR, Schellberg J (2003) Lack of Interaction between Extreme High-Temperature Events at Vegetative and Reproductive Growth Stages in Wheat. J Agron Crop Sci 189(3):142–150
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res (Oxf) 14(6):415–421
Acknowledgments
The authors gratefully acknowledge P. J. Lo Valvo, J. M. Brihet and A. V. Seco for their excellent technical assistance. RdSC currently holds a postgraduate scholarship from CONICET. This research was funded by UBACyT and PIP (CONICET) grants.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: John A. Kirkegaard.
Rights and permissions
About this article
Cite this article
de San Celedonio, R.P., Abeledo, L.G. & Miralles, D.J. Identifying the critical period for waterlogging on yield and its components in wheat and barley. Plant Soil 378, 265–277 (2014). https://doi.org/10.1007/s11104-014-2028-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2028-6