Abstract
Background and Aims
Volatile organic compound (VOC) emissions from biogenic sources are important contributors to chemical reactions in the air. Soil/forest floor VOCs contribute significantly to the ecosystem scale emissions, however, these emissions and their temporal and spatial variations are poorly characterised. The below-canopy VOC emissions have been measured mainly in campaigns; continuous measurements over the whole growing season are rare.
Methods
VOCs were measured from boreal forest floor over the snow-free season 2010 in southern Finland with automated flow-through chambers connected to proton transfer reaction-mass spectrometer (PTR-MS). We measured 10 masses in total, of which five quantitatively (M33, M45, M59, M69, M137).
Results
All of the fluxes showed clear diurnal and seasonal variation, being at their highest in early summer. Spatial variation in the fluxes was great and the lowest rates were found in chambers with dense vegetation cover. Also, VOCs deposition was observed regularly. Monoterpene (M137) emissions were one magnitude higher (up to 264 ng m−2 s−1) than other emissions. The VOC fluxes correlated positively with temperature and light, while relative humidity correlated negatively.
Conclusions
Results indicated that forest floor plays a substantial role in the boreal forest total VOC emissions. Understanding the processes controlling VOC emissions requires more detailed analysis and long-time measurements with sufficient time resolution and analytical accuracy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biogenic sources are the main contributors to global volatile organic compound (VOC) emissions into the atmosphere, where VOCs take part in numerous chemical reactions, for example, in new particle formation and particle growth (Kulmala et al. 1998, 2004). Most biogenic VOCs are emitted by plants, especially by trees (Kesselmeier and Staudt 1999 and references therein), but many sources are still unidentified on the ecosystem scale. Boreal forest ecosystems are less efficient VOC emitters compared to many warmer regions (Rinne et al. 2009), but as they are the biggest biome on Earth and consist of evergreen vegetation that remains active in the cooler months, the terpenoid emissions from boreal forests are especially substantial and climate change will even raise the importance of this role (Peñuelas and Staudt 2010). From the atmospheric chemistry point of view, the VOC fluxes from the boreal forest canopies have been the most intensively studied biogenic source, whereas forest floor VOC fluxes have mainly been measured during short campaigns or with low temporal resolution (Janson et al. 1999; Hellén et al. 2006; Aaltonen et al. 2011). VOCs commonly have a short lifetime due to their reactivity, and fluxes from the forest floor mainly influence the air chemistry below the canopy (Rinne et al. 2007). The forest floor VOC fluxes are likely to contribute to the large OH sink observed in some measurements (e.g. Sinha et al. 2010; Nölscher et al. 2012) and models (e.g. Mogensen et al. 2011).
According to the few earlier measurements, the forest floor has been shown to play an important role in boreal forest ecosystem VOC emissions, especially in the spring and in the autumn when the forest floor may contribute up to some tens of percents of forest ecosystem VOC emissions (Hellén et al. 2006; Aaltonen et al. 2011). Forest floor VOC fluxes consist of emissions from both the vegetation and soil, but their relative importance or the contributing processes in the soil are not sufficiently known. The soil VOC emissions are assumed to originate from several sources: from the degradation of organic matter, from microbes taking part in soil processes and from living roots (Hayward et al. 2001; Isidorov and Jdanova 2002; Asensio et al. 2007a, 2008a, b; Leff and Fierer 2008; Bäck et al. 2010; Insam and Seewald 2010). In addition to biological processes, physical environmental factors also have a role in VOC fluxes. Freezing and thawing cycles as well as drying and wetting affect the degradation of organic matter, releasing VOCs, while temperature increases the volatility of these compounds (Asensio et al. 2007b, 2008a; Insam and Seewald 2010). Also, environmental factors have been shown to indirectly affect soil VOC fluxes by regulating biological processes (Asensio et al. 2007a, 2008b; Insam and Seewald 2010).
Continuous, long-term measurements of VOC fluxes from the forest floor have not yet been carried out. A few earlier studies report soil VOC emissions measured with PTR-MS in laboratory conditions (e.g. Asensio et al. 2007a; Gray et al. 2010), but field studies, especially long-term studies, on soil VOCs are practically non-existent. With individual samplings and short measurement campaigns, it has been possible to estimate the overall level of VOC fluxes, but variations over longer time scales still remain unknown. Thus, to quantify the magnitude and variation of the biogenic VOC fluxes from the boreal forest floor, including the ground vegetation and soil, we conducted continuous VOC measurements during one snow-free season in a Scots pine forest. Automated on-line measurements from dynamic chambers with a proton transfer reaction mass spectrometer (PTR-MS) were performed to quantify the VOC fluxes at daily and seasonal time scales. Another objective was to analyse dependencies of VOC fluxes on environmental factors (e.g. temperature, photosynthetically active radiation (PAR), air humidity, soil water content) in the forest floor, in order to link the VOC fluxes to other ecosystem processes. These data can further be used in air chemistry models, which so far lack a comprehensive understanding of whole stand and, in particular, below-canopy VOC fluxes.
Material and methods
Measurement site
We measured VOC fluxes from a pine forest floor at SMEAR II (Station for Measuring Forest Ecosystem–Atmosphere Relations II) in southern Finland (Hari and Kulmala 2005). The forest stand at SMEAR II is 48 years old and dominated by Scots pine (Pinus sylvestris L.). Before the stand was established by sowing in 1962, it was treated by prescribed burning. The stand height is now ~18 m, and the canopy is open with an average tree density of ~1170 ha−1 (Ilvesniemi et al. 2009). The vascular plant species and mosses at the measurement points of this study were lingonberry (Vaccinium vitis-idaea L.), bilberry (Vaccinium myrtillus L.), twinflower (Linnaea borealis L.), Schreber’s big red stem moss (Pleurozium schreberi (Brid.) Mitt.), a dicranum moss (Dicranum Hedw. sp.), hair moss (Polytrichum Hedw. sp.) and stairstep moss (Hylocomium splendens Hedw.). The vascular plant species and their coverage (%) inside the collars were determined visually from photographs taken in May and September (Table 1). The soil above the homogeneous bedrock is Haplic podzol formed in a glacial till, with an average depth of 0.5–0.7 m. The long-term (1971–2000) annual average precipitation and the annual mean temperature at the SMEAR II station is 713 mm and 3.3 °C, and those of May to November 494 mm and 8.8 °C, respectively (Drebs et al. 2002). During the sampling period, from May to November 2010, the weather at the station was warmer and slightly drier than average, the cumulative precipitation being 463 mm and the mean temperature 9.9 °C (Fig. 1).
Chamber VOC measurements
The VOCs were measured between 6 May and 15 November 2010 with three permanently installed flow-through chambers. The chambers were placed 10 to 30 m apart, and the forest cover over the chambers was rather homogeneous with almost closed canopy layer. Stainless-steel collars (80 cm × 40 cm × 10 cm) for mounting the chambers on the soil had previously been installed at the site in September 2009 by pushing the collar edge through the moss and litter layers. The chambers (80 cm × 40 cm × 25 cm) were made of an aluminium frame whit a transparent fluorinated ethylene—propylene (FEP) film (0.05 mm) covering the sides and top from the inside. The chambers opened towards the north, leaving the south-facing side free of any barriers. When the chamber was open, it covered approximately 10 % of the collar area, allowing quite undisturbed litter input from trees. Two small fans continuously mixed the air inside the chambers. The operation of the chambers was automated; each chamber was pneumatically closed for 15 min once every three hours, i.e. eight times per day, and the first 7 min non-steady-state part of the closures was used for quantifying the fluxes. Sample air was drawn from the chambers at a rate of 1.1 dm3 min−1 in a 64-m long heated FEP tube with an internal diameter of 4 mm. A smaller air sample (flow 0.1 dm3 min−1) was taken from the main sample line and diverted into a PTR-MS (Ionicon Analytik, Innsbruck, Austria) with polytetrafluoroethylene (PTFE) tubing. Compressed ambient air, a mixture of air drawn from six levels (4.2, 8.4, 16.8, 33.6, 50.4 and 67.2 m above ground) of measurements in mast located at the site (Vesala et al. 1998), was supplied into the chamber at the same flow rate as the sample air was drawn out to replace the sampled air volume in the chamber.
PTR-MS
Proton transfer reaction-mass spectrometry (PTR-MS) is a relatively new technique for analysing VOCs without any sample preparation (Lindinger et al. 1998). Since PTR-MS has a fast response time and high sensitivity (Taipale et al. 2008), it is optimal for process studies, where concentration changes are commonly fast but low. For flux measurements, when flux is calculated from a concentration change inside a closure, concentration screening online with PTR-MS is more convenient. In addition to the laboratory experiments, PTR-MS also performs well for campaign-related measurements and monitoring and has been used especially in atmospheric studies (de Gouw and Warneke 2007 and references therein). This method is also suitable for oxygenated VOCs, which are difficult to detect with other methods.
A PTR-MS was used to measure concentrations of the selected VOCs (Table 2) and calibrated biweekly according to Taipale et al. (2008). The concentrations of all the compounds in the calibration gas (Apel Riemer Environmental Inc., USA) were approximately 1 ppm, and for calibration they were diluted to 5 ppb to reach the level of linear response of the PTR-MS. The results from the non-calibrated masses are discussed only as qualitative values. However, the relative flux over the whole measurement period is reliable with the non-calibrated masses as well. A detailed description of the PTR-MS technique is given by Lindinger et al. (1998) and De Gouw and Warneke (2007). The volume mixing ratio calculations are explained in Taipale et al. (2008). The results from the PTR-MS analyses are expressed with the measured protonated mass symbol (amu+1, e.g., M33= methanol, M69= isoprene, etc.). As PTR-MS equipped with quadrupole MS measures with accuracy of one mass unit, it is unable to identify compounds due to the low mass resolution. Identification of compounds can be helped by using calibration standards and screening the samples with other techniques.
Flux calculations
The VOC flux was calculated from the development of measured VOC concentration during the chamber closure. The change in VOC concentration C inside the chamber during the closure is derived from the mass balance equation (Hari et al. 1999)
where V is the volume of the chamber, E the rate of emission (positive) or uptake (negative) of VOC inside the chamber, F the volumetric flow rate through the chamber, and C i the measured VOC concentration in the replacement air that enters the chamber to compensate for the sample flow. Solving Eq. 1 for concentration C in the chamber as a function of time t yields solution
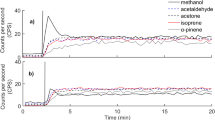
where C 0 is the VOC concentration at the moment that the chamber closes. VOC emission E was calculated by fitting Eq. 2 to the development of measured concentration during chamber closure (Fig. 2). Ordinary least-squares methods were employed in the flux calculation. The ambient concentration C 0 was calculated as the average of PTR-MS readings taken from the open chamber for half a minute before closing the chamber.
Examples of the fitting of the mass balance equation to the development of measured concentrations of masses 33 (methanol), 59 (acetone) and 137 (monoterpenes) in chamber 3. The closure started at 16:14 on the 1st of June 2010. The first two grey dots represent the ambient concentrations of these masses
Supporting data
In addition to the VOCs, forest floor CO2 and H2O fluxes were also measured from the same chambers with infrared light absorption analysers (URAS 4, Hartmann & Braun, Frankfurt am Main, Germany), and the chamber air temperatures were measured with thermocouples. Below canopy photosynthetically active radiation (PAR) was measured at a height of 60 cm from four points (not in the same locations as the chambers) with an LI-190SZ quantum sensor (Li-Cor, Lincoln, NE, USA). Soil temperature (Philips KTY81-110, Philips Semiconductors, Eindhoven, the Netherlands) and soil water content (TDR 100, Campbell Scientific Inc., Logan, USA) were measured continuously from seven points and at five depths at the SMEAR II stand. Precipitation was measured with an FD12P weather sensor (Vaisala Oyj, Helsinki, Finland).
Statistical analyses
In order to test the spatial variation of the VOC fluxes and the environmental variables between the measurement chambers (n = 3) and to analyse the correlations between the VOC fluxes and the environmental variables, the Kruskal-Wallis non-parametric analysis of variance and the Pearson correlation analysis, respectively, were run by PASW Statistics 18 (SPSS Inc., Quarry Bay, Hong Kong). The non-parametric test was chosen after the data was checked for normal distribution using the Kolmogorov-Smirnov test of normality and for homogeneity of variances using Levene’s test of equality of variances. The significance level chosen was <0.05, i.e. lower p-values than 0.05 indicated statistical significance. For the correlation analysis the dataset was modified in two ways: 1) it was divided into monthly periods to eliminate the interfering effects of seasonal changes in environmental parameters and the phenology of the leaves of the ground vegetation and 2) the night-time values (three of the eight closures per day) were omitted as the VOC fluxes were always close to zero at night.
Results
The overall level of the VOC fluxes from the forest floor
Fluxes of all the masses that we selected to measure with the PTR-MS (Table 3) were detected from the forest floor, albeit statistically significant spatial variability (i.e. differences between the three chambers) was seen in the measured fluxes. The fluxes were mostly positive (showing emissions from forest floor to the atmosphere), but with some masses the average flux over the whole measurement period remained slightly negative. M33 (methanol) showed clearly positive fluxes in two chambers, whereas in one chamber (chamber 2) the average flux was negative. The maximum fluxes of M137 (monoterpenes) were one magnitude higher than those of the other masses (Fig. 3), the highest peak value of M137 being 264 ng m−2 s−1. In contrast, the flux of M69 (isoprene) was always below 10 ng m−2 s−1. The differences could be owing to the vegetation characteristics inside the chambers: the higher the vascular plant coverage, the lower the VOC fluxes were (Fig. 4).
The seasonal variation of the fluxes of the masses 33 (methanol) and 137 (monoterpenes), measured between 6 May and 16 November 2010. The highest fluxes of M33 and M137 were measured in chamber 1 (panels a) and c)), and the lowest with chamber 2 (panels b) and d)), respectively. Note the different y-axis scale in panel c)
Uncalibrated masses M99, M101 and M153 showed similar flux levels as the calibrated ones, excluding M137. However, the flux levels of these masses should be treated only as indicative. The major fragmentation product of monoterpenes was measured in M81 (Taipale et al. 2008), which showed clearly higher fluxes (~4 x higher) than M137. This indicated the presence of interfering compounds in M81, and thus, only the values from M137 were used to represent the monoterpene fluxes.
Temporal variation of VOC fluxes
All of the masses showed regular diurnal variations consistently in all chambers; the forest floor was a VOC source during the day, while at night the emissions decreased to close to zero, and deposition was frequently observed (Fig. 3). Irrespective of the day-time flux level, the night-time fluxes were always very low or negative.
In addition to the diurnal variation, the fluxes of almost all of the masses showed a large seasonal fluctuation during the measurement period (Fig. 4). The fluxes were clearly highest during the early summer and late autumn. The time of the highest VOC fluxes was the period between the end of May and mid July, especially for monoterpenes (M137). Another period with fluxes above average was in November, after a long stable period of quite low fluxes. This increase in fluxes in late autumn was observable only with heavier masses (bigger than M69); however, it was not seen in monoterpenes (M137).
Relationships of fluxes with environmental factors
The differences in the environmental factors between the enclosures were statistically significant, even though in most cases the values were rather close to each other (Table 1). For example, the differences in the mean temperatures between the chambers over the whole period were less than 2° (9.8–10.7 °C), and the differences in mean relative humidity (RH) were only seven percentages (82–89 %). Chamber 1 was the warmest and driest, and had the smallest CO2 flux as well as the smallest vascular plant coverage. On the other hand, chamber 3 had abundant plant cover, a two times higher CO2 flux, as well as the highest RH and lowest average temperature.
To analyse the different drivers for VOC fluxes under the field conditions, the full dataset was divided into monthly periods, and the night-time values (with small fluxes and frequent periods of deposition) were omitted from the correlation analysis. Most of the environmental factors (Table 1) correlated significantly (p < 0.05) with the VOC fluxes, at least during some part of the measurement period (Table 4). Only in the late fall were correlations between the environmental factors and VOC fluxes weak. Even though the chambers differed significantly in terms of vegetation, the correlations between the VOC fluxes and most of the environmental variables were similar for all the chambers. Figure 5 shows the correlations of the fluxes of M33 (methanol) and M137 (monoterpenes) between the chamber temperature, RH, PAR and CO2 flux. The strongest positive correlations were observed between the VOC fluxes and chamber temperature, the below-canopy PAR, and the water flux (not shown), while RH was clearly negatively correlated with the VOC fluxes. Soil temperature (correlations not shown) influenced the VOC fluxes less than the chamber temperature. Soil water content correlated negatively with the VOC fluxes mainly during the first month of measurements (correlations not shown). Most of the time the CO2 flux did not correlate with the VOC fluxes (Fig. 5). Overall, the correlations between the VOC fluxes and environmental factors were strongest at the time when the VOC fluxes as well as the temperature and PAR were high, i.e. in June. Towards the autumn most of the correlations clearly declined, and after August the correlations with temperature and PAR especially were very small or disappeared completely.
The correlations of the day-time fluxes of M33 (methanol, upper row) and M137 (monoterpenes, lower row) with air temperature, relative humidity (RH), photosynthetic photon flux density (PPFD) and CO2 flux during June 2010. All of the variables except PPFD were measured inside the chambers. Fluxes of M137 from chamber 1 have a right side axis with a wider scale
The influence of humidity on the VOC fluxes in chamber 2 differed from that in the other chambers. During the first 3 months, the correlation between the VOC fluxes and RH in chamber 2 was similar to that in the other chambers (clearly negative), but after mid summer this relationship changed, and the correlation with humidity disappeared completely.
The fluxes of all the measured masses correlated rather similarly with environmental factors in the forest floor. An exception was the only weakly negative correlation between M69 and RH in all chambers, while it was strongly negative with all the other VOCs. The relationship between M137 and temperature was strongly positive in the summer, but was reversed to clearly negative during the last month of measurements in all chambers.
Discussion
The quality and sources of VOC emissions from the forest floor
We identified a large number of different VOCs from the below-canopy air. The boreal Scots pine forest floor was shown to be a source of all of the identified compounds at some points of the year, although the average fluxes remained quite low for most of the masses. The largest emissions were observed with masses 81 and 137, i.e. the monoterpene fragment and total monoterpene masses. Interpretation of the field measurements is challenging, because several sources can contribute to the flux simultaneously.
One way to evaluate the potential sources under field conditions is to compare the observed emission blend to those measured in experimental studies from known sources. Several studies report VOC emissions of litter from tree species. The Scots pine needle litter (Isidorov et al. 2003) emissions consisted mainly of monoterpenes, but emissions of acetaldehyde, acetone and benzene were also measured. Oxygenated compounds may be originating from litter decomposed by fungi (Isidorov et al. 2003) or from root-associated fungal isolates (Bäck et al. 2010). Gray et al. (2010) observed emissions of M33, M45, M59, M69, M81 and M137 from the litter of two Pinus species in the laboratory. They also noticed that although the emissions were higher with an active decomposer community, M33 (methanol) was the dominating mass in emissions from both sterile and non-sterile litter (Gray et al. 2010). We saw weak M79 (presumably benzene) emissions, which have also been detected in several other studies from soil and litter, for example, by Leff and Fierer (2008) and Asensio et al. (2007a). Several masses common in our measurements (M33, M45, M59, M137) were also measured from soil and litter samples by Asensio et al. (2007b) in a broadleaf temperate forest. The similarity of results by Isidorov et al. (2003), Bäck et al. (2010) and this study, indicate that the decomposers are contributing at least partly to the measured forest floor VOC fluxes. However, in contrast to those controlled experiments, the measured flux in our study was dominated by monoterpenes, which indicates that in the field there are also other sources to the fluxes. Most likely the monoterpene fluxes in this study were originating from forest floor vegetation, Scots pine roots and decomposing needle litter.
In addition to oxygenated short-chained volatiles and monoterpenes, we also frequently measured the emission of isoprene (M69). Emissions of isoprene from the boreal soil or forest floor have been so far reported only by Hellén et al. (2006). In addition to clear isoprene fluxes from a boreal fen, Hellén et al. (2006) measured forest floor isoprene fluxes which were slightly higher than in this study (up to 0.53 ng m−2 s−1). Small isoprene emissions were measured from root-associated soil fungi (Bäck et al. 2010) and from needle litter (Gray et al. 2010) in laboratory conditions. Since forest floor vegetation was abundant inside our chambers, it is possible that the forest floor vegetation contributed to the isoprene fluxes in addition to the soil and litter. Taking into consideration the reported strong light dependency of isoprene synthesis (Guenther et al. 1993; Guenther 1997), it was surprising that the correlation between M69 fluxes and light, and also temperature, was in most cases the lowest of all the masses. The solar irradiance levels in the forest floor are much lower than those above the canopy, and the diurnal variation in irradiance is also lower compared to the canopy level. However, the spatial variation in irradiance is higher below the canopy compared to above-canopy irradiance, and since we did not have the PAR measurements on the chambers, this can partly explain the lower correlation between PAR and M69 fluxes. Further, since compound identification with PTR-MS is not conclusive, M69 may include several other compounds, for example, a fragment of methyl butenol (Fall et al. 2001; Greenberg et al. 2012).
Temporal variation in VOC fluxes
This is the first long-term study to reveal seasonal patterns in VOC emissions from the forest floor in high temporal resolution. The biologically most active part of the growing season in our site, from the beginning of May until mid July, was also the time of the highest VOC fluxes. The monoterpene fluxes were highest in the early summer period; approximately 260 ng m−2 s−1 at the maximum. The mid and late summer was characterized by low emissions from all the masses. Hellén et al. (2006) and Aaltonen et al. (2011) measured similar monoterpene fluxes with manually operated chambers at SMEAR II during the springs of 2004, 2005 and 2008, but in these studies the highest emission peaks were observed slightly earlier, in April. In this dataset, another period with fluxes above average was the late autumn in November, but only with heavier compounds (M69, M79, M99, M101 and M153). A period of high late autumn monoterpene fluxes was measured in 2008 (Aaltonen et al. 2011), although then the fluxes peaked already in October. Since in this study the high autumnal emissions consisted only of heavier compounds, it suggests that the peak we measured may not originate from soil fungi, which emit mainly low molecular weight oxygenated compounds (Bäck et al. 2010). However, the biomasses of soil microbes and animals peak in the autumn, active fungal biomass peaking especially in late autumn and early spring (Persson et al. 1980; Wallander et al. 2001). Also environmental conditions may have had an influence on this: the low molecular weight compounds such as methanol and acetone are highly water soluble, which may lead to low fluxes from the soil to the atmosphere irrespective of the source strength during the fall season with lower temperatures and higher soil moisture content. An important result of this study is that by long term measurements the temporal dynamics occurring in natural ecosystems can be analysed in detail, and that this analysis will provide an insight to the dynamic responses varying significantly in time.
In addition to the seasonal fluctuation, clear diurnal patterns were seen in the forest floor VOC fluxes. Thus, clear positive correlations between the VOC fluxes and the parameters following the diurnal cycles (temperature, PAR) were observed. However, they did not completely explain the diurnal and seasonal cycle in the fluxes. The VOC fluxes had already declined in July, although the clear positive correlations between fluxes and temperature and PAR remained until August. The temperature and PAR values started to decline already in August, but did not clearly drop until the end of September.
Varying responses to environmental factors
Long-term measurements are necessary in capturing the potential changes in source strengths or their dependencies on environmental factors, which both are impossible to see in short-term and campaign-related measurements. Correlation between the VOC fluxes and RH inside chamber 2 provided an example of how factors affecting the fluxes can change sharply; during the first 3 months RH clearly correlated negatively with the VOC fluxes, and from August onwards there was no correlation. Other parameters lost their correlations with the VOC fluxes more slowly, but none of them were a major factor driving the autumnal VOC fluxes. Obviously, one reason for the decreasing correlations was the markedly decreased VOC fluxes themselves, however, this does not fully explain this phenomenon, since many of the environmental parameters remained constant for several following weeks. So it seems that some of the processes behind the VOC fluxes changed during mid summer. Based on these measurements, we are not able to say whether these processes are related to the soil processes under the chamber or the metabolism of the forest floor vegetation. However, the seasonal maximum in the photosynthesis of Vaccinium vitis-idaea and V. myrtillus in our study site is usually reached in early July, when the development of the leaves of the deciduous V. myrtillus also ends (Kulmala et al. 2011). Also, the leaf senescence of these dwarf shrubs occurring in early autumn (Kulmala et al. 2008; Kulmala et al. 2011) may have affected the VOC emissions and their composition.
Temperature controls the volatility and biological production processes of VOCs in leaves and most likely also in soil. However, at the very end of the measurement period, we found negative correlations between the fluxes of M137 and temperature. One potential reason for this is a strong new source for VOCs in the fall, namely the breakdown of fresh needle litter. Scots pines drop off the oldest needles in the autumn. Needles consist of over 90 % of the autumnal litterfall at SMEAR II, litterfall peaking in October (Aaltonen et al. 2011), which may decouple the emissions from temperature. Potential frost damage due to sub-zero temperatures may also cause the release of stored monoterpenes in the litter and thus increase the M81 and M137 fluxes. This assumption is supported by measurements made during the winter by Aaltonen et al. (2012), which show that physically damaged Scots pines can release substantial amounts of monoterpenes even in freezing temperatures.
Deposition
In addition to the microbial consumption of VOCs (Owen et al. 2007), another explanatory factor for the negative VOC fluxes may be related to physical conditions in the measurement chamber. The negative flux values observed frequently at night coincided with the increased air humidity inside the chambers, which caused the negative correlations between the VOC fluxes and RH. Oxygenated compounds in particular stick easily to the moist inner surfaces of the chamber as well as to plant and moss surfaces. Sumner et al. (2004) found that FEP does not adsorb water as efficiently as many other materials (e.g. glass, quartz, and wax), but like all surfaces, a water film on FEP thickens rapidly when 80 % RH is exceeded. This phenomenon was clearly visible in our measurements. Below ~50 %, the increase in RH decreased the VOC fluxes sharply (Fig. 5). When higher RH values were obtained, the decreasing effect on VOC fluxes was less significant. At 80 % RH, the VOC fluxes were already at about zero, as all the inner surfaces were covered by water film (Sumner et al. 2004), dissolving or sticking to VOCs. An increase in RH from 80 to 100 % had little effect on the fluxes; it only made the water film thicker, which may have had an effect on the most water-soluble compounds. The effect of the vegetation cover in the chambers on the VOC fluxes can also be mediated by humidity in two ways: 1) the average RH was ~10 % higher in the chambers with more dense vegetation, and 2) more vegetation offers more wet surfaces of which VOCs can stick. Also, the high soil water content (0.3–0.45 m3 m−3) after the snowmelt in the spring correlated negatively with the VOC fluxes; however, at this time the lowered diffusion of the VOCs from the soil might have been the major reason for very low and even negative VOC fluxes, not the wet surfaces inside the chamber.
The night-time VOC fluxes were always close to zero and often even negative. This was also the case during the periods of maximum daily fluxes, implying that the high average fluxes were the outcome of high mid-day values. The factors differing in the chambers between day and night are naturally related to variations in light and air temperature, whereas the soil temperature changes very little between night and day. The correlation between the VOC fluxes and soil temperature was weaker than with the chamber air temperature, indicating that the temperature experienced by plant foliage and the processes right at the soil surface were critical for the VOC emissions. That is probably one of the key reasons why the night-time VOC fluxes were very low. Moreover, the humidity issues discussed above are closely linked to this temperature effect.
Spatial heterogeneity
The monoterpene emissions (M137) in two out of the three chambers (2 and 3) were comparable to those measured with manual chambers and GC-MS in 2008 (Aaltonen et al. 2011). However, the monoterpene emissions in chamber 1 were one magnitude higher than in the other two chambers, which is an indication of high spatial heterogeneity in the fluxes. The measurements of Aaltonen et al. (2011) were conducted with five manually operated chambers, and the spatial heterogeneity was also substantial in these results. The manual measurements were performed with adsorbent tube sampling and a GC-MS analysis, targeted for measuring only certain monoterpenes, whereas the PTR-MS measures all the compounds occurring in mass 137, and therefore these results are not fully comparable with Aaltonen et al. (2011).
Even though the emission levels varied between the chambers, the diurnal and seasonal variations as well as the responses to environmental factors were fairly similar between the three chambers. Although the stand is rather homogeneous and all our measurement points were clearly within the central areas, on top of solid bedrock, the forest soil has huge heterogeneity (Pihlatie et al. 2007) on a scale of tens of centimetres, and the distances between the chambers (in this study ~15–30 m) may already imply clear differences in soil conditions. The most likely difference influencing the VOC fluxes in the chambers was the quantity and species diversity of the plant coverage. The ground vegetation at the site consisted of typical plant species for a Vaccinium type (Cajander 1926) pine forest, the classification of this stand. Our measurement places represented the variety of ground vegetation coverage well; from very sparse vegetation in chamber 1 to dense vegetation in chamber 2. Thus, our measurements were quite representative for boreal forests in northern Europe. We suggest that with our measurements we were able to capture the effects of the special characteristics of these measurement places, including the effects of varying plant coverage and variations in soil characteristics.
Significance of the below-canopy layer to ecosystem-scale emissions
Ecosystem-scale VOC fluxes in the forest are dominated by emissions from foliage (Guenther et al. 2006; Tarvainen et al. 2007; Noe et al. 2012), but the stems and branches of trees also emit VOCs, in addition to the forest floor. Ecosystem-scale VOC emissions have been measured in different types of forests with gradient methods and with disjunct eddy covariance (e.g. Rinne et al. 2000, 2007; Spanke et al. 2001; Räisänen et al. 2009). Measurements conducted above the forest canopy are not optimal for describing the actual emission strength or the emission sources, but they are suitable for evaluating the impacts of VOC emissions on atmospheric chemistry and hence on climate. Due to the diurnal cycle of the air mixing and atmospheric chemistry, the above-canopy terpenoid concentrations are higher at night, even though the emissions are higher during the day (Hakola et al. 2000; Rinne et al. 2007). Canopy-scale emissions are often also up-scaled from branch enclosure measurements. These measurements describing VOC emissions at the ecosystem scale provide an overview of the climatic effects of the volatiles emitted from boreal forests; however, the measured emission levels are not very consistent between studies. The measurements and air chemistry models imply a missing OH sink in the canopy layer (Di Carlo et al. 2004; Sinha et al. 2010; Nölscher et al. 2012; Mogensen et al. 2011), suggesting incompletely determined sources of VOCs. Models show that the largest OH sink is located near the ground level, and thus the results of this study may improve the models considerably.
Logically, our results on boreal forest floor VOC fluxes show clearly lower values than Rinne et al. (2007) obtained for the whole forest ecosystem with measurements conducted in July at the SMEAR II stand with a disjunct eddy covariance technique coupled with PTR-MS. Their daily average flux values for M33, M45, M59 and M137 were similar to our highest early June values from chamber 1. It means that the ecosystem scale fluxes were one or two orders of magnitude higher than our daily average fluxes from forest floor. Overall, it seems that the forest floor accounts for several per cents to tens of per cents, depending on the season, of the total ecosystem VOC emissions in this kind of boreal coniferous forest. Ecosystem-scale VOC emissions measured in European boreal forests were collected by Rinne et al. (2009); all of them show similar monoterpene flux levels as Rinne et al. (2007), i.e. approximately one order of magnitude higher than those we observed. In contrast, some studies report boreal forest canopy or forest ecosystem monoterpene emissions as so low that the share of forest floor emissions becomes very important. For example, Ruuskanen et al. (2005) measured late summer monoterpene emissions from Scots pine branches, the values which were even lower than our results showed. The day-time emissions of α–pinene, the most emitted monoterpene, from the SMEAR II stand according to Hakola et al. (2009) were at the same level as our M137 results, the total monoterpene emissions from forest ecosystem thus being at most two to three times higher than we measured from the forest floor.
Even though we also measured low isoprene fluxes from the forest floor, the European boreal forest site studies in Rinne et al. (2009) did not report isoprene emissions at all. The isoprene emissions from fen sites were however at a similar level as the monoterpene emissions from forest sites (Rinne et al. 2009), thus remarkably higher than those we measured.
Reliability of the measurements
In the case of the isoprene fluxes, both the manual GC-MS (Aaltonen et al. 2011) and the automated PTR-MS measurements were at a similar level. The overall agreement between the flux levels measured in this study and by Aaltonen et al. (2011) gives reliability to both datasets and both measurement techniques. In addition to the similar overall emission levels, the timing of the high fluxes was similar in both studies. High monoterpene fluxes were measured during the first half of June, decreasing towards the last half of the summer. The results presented in Aaltonen et al. (2011) gave a detailed outlook of the terpenoid species found in the atmosphere close to the forest floor, but automated continuous long-term measurements are needed in order to resolve the temporal patterns of the VOC fluxes and the correlations with the driving factors.
The whole VOC measurement system, starting from the chamber and ending at the PTR-MS, contained several parts, which may have caused errors in the results. The PTR-MS measures with accuracy one mass unit only (devices equipped with quadrupole MS) and is potentially unable to identify the compounds in air samples due to this low mass resolution. Screening of the air samples with other techniques and using calibration standards for target compounds helps to identify the compounds; although the problem of compound identification still remains (all compounds with identical masses are detected in 1 m/z value). In dynamic chamber measurements, the fast response time of the PTR-MS enables short closure times, consequently reducing the effects of changing environmental variables (most importantly temperature and humidity) on the measured fluxes during the closure. Adsorbent sampling and later analysis with GC-MS usually implies closure times from 30 min to several hours, and thus e.g. irradiation as well as air temperature and humidity inside the chamber change greatly during the measurement.
Despite the large spatial variation, the clear diurnal and seasonal variations in emissions and their responses to environmental factors indicate that the chamber VOC measurements were reliable and can be used for estimating the source strength of the forest soil and floor. However, due to several disturbances that the enclosure causes to the environmental conditions at the measurement point (Ortega and Helmig 2008; Ortega et al. 2008; Schaub et al. 2010; Niinemets et al. 2011; Kolari et al. 2012), the measured flux levels may be over- or underestimations of the actual fluxes, depending on the conditions. Commonly, the temperature inside the chamber is slightly higher than the ambient temperature, increasing the flux of volatiles. Increased humidity inside the chamber has the opposite effect. As well as increasing the temperature, enclosure techniques often also increase air humidity and the amount of moist surfaces inside the chamber, potentially leading to compound losses due to adsorption (Ortega and Helmig 2008; Niinemets et al. 2011; Kolari et al. 2012). Based on our measurements, the increase in humidity seemed to be the most important environmental factor changing during the closures, and thus these flux values may be more under- than overestimated. To assess and minimize the errors in the results caused by the measurement techniques, further studies both in the field and in the laboratory on the surface reactions occurring on the chamber walls and inside the sample lines are needed.
Conclusions
This long-term field study revealed that the pine forest floor emits several VOC compounds, including oxygenated compounds and terpenoids. Monoterpene fluxes were clearly the highest; approximately one magnitude higher than the other compounds. Forest floor emissions were generally lower, but occasionally, such as in the autumn, even comparable with those measured from Scots pine branches. All of the measured compounds showed diurnal variation with a midday maxima, as well as larger scale variation, with the highest fluxes during early summer. The spatial variation of the fluxes was remarkable, and the dense forest floor vegetation had a negative effect on the fluxes.
The most important environmental variables driving the VOC fluxes in the spring and the first half of the summer were light, temperature and RH. The effect of RH was most probably connected to reactions on plant surfaces and chamber walls, and thus complicates the analysis of the results. Due to this, the presented results represent the lower estimates of the actual fluxes. Later during the measurement period, none of the measured environmental parameters seemed to be a main driver for the VOC fluxes, and thus also other unaccounted factors such as soil biological and physical processes may influence the fluxes at field conditions. Most probably the autumnal VOC fluxes were closely connected to the high needle litter input on the ground from Scots pines. In order to clarify the importance of the contributing sources, more detailed, controlled field and laboratory experiments would be needed.
The below-canopy VOC sources need to be studied more carefully in the future, but this study already provides new data on boreal forest floor VOC fluxes and their seasonality, which can be used for air chemistry and climate models. Since significant VOC fluxes have been found from the forest floor, other ecosystem components should also be taken into account when determining or modelling ecosystem-scale fluxes. Forest floor VOCs may also partially explain the observed missing OH reactivity, since the forest floor compartment is commonly omitted from air chemistry models and measurements.
References
Aaltonen H, Pumpanen J, Pihlatie M, Hakola H, Hellén H, Kulmala L, Vesala T, Bäck J (2011) Boreal pine forest floor biogenic volatile organic compound emissions peak in early summer and autumn. Agr Forest Meteorol 151:682–691
Aaltonen H, Pumpanen J, Hakola H, Vesala T, Rasmus S, Bäck J (2012) Snowpack concentrations and estimated fluxes of volatile organic compounds in a boreal forest. Biogeosciences Discuss 9:527–555
Asensio D, Peñuelas J, Filella I, Llusià J (2007a) On-line screening of soil VOCs exchange responses to moisture, temperature and root presence. Plant Soil 291:249–261
Asensio D, Peñuelas J, Llusià J, Ogaya R, Filella I (2007b) Interannual and interseasonal soil CO2 efflux and VOC exchange rates in a Mediterranean holm oak forest in response to experimental drought. Soil Biol Biochem 39:2471–2484
Asensio D, Peñuelas J, Prieto P, Estiarte M, Filella I, Llusià J (2008a) Interannual and seasonal changes in the soil exchange rates of monoterpenes and other VOCs in a Mediterranean shrubland. Eur J Soil Sci 59:878–891
Asensio D, Owen SM, Llusià J, Peñuelas J (2008b) The distribution of volatile isoprenoids in the soil horizons around Pinus halepensis trees. Soil Biol Biochem 40:2937–2947
Bäck J, Aaltonen H, Hellén H, Kajos MK, Patokoski J, Taipale R, Pumpanen J, Heinonsalo J (2010) Variable emissions of microbial volatile organic compounds (MVOCs) from root-associated fungi isolated from Scots pine. Atmos Environ 44:3651–3659
Cajander AK (1926) The theory of forest types. Acta Forestalia Fennica 29:1–108
de Gouw J, Warneke C (2007) Measurements of volatile organic compounds in the Earth’s atmosphere using proton-transfer-reaction mass spectrometry. Mass Spectrom Rev 26:223–257
Di Carlo P, Brune WH, Martinez M, Harder H, Lesher R, Ren X, Thornberry T, Carroll MA, Young V, Shepson PB, Riemer D, Apel E, Campbell C (2004) Missing OH reactivity in a forest: evidence for unknown reactive biogenic VOCs. Science 304:722–725
Drebs A, Nordlund A, Karlsson P, Helminen J, Rissanen P (2002) Climatological statistics of Finland 1971–2000. Finnish Meteorological Institute, Helsinki
Fall R, Karl T, Jordan A, Lindinger W (2001) Biogenic C5 VOCs: release from leaves after freeze–thaw wounding and occurrence in air at a high mountain observatory. Atmos Environ 35:3905–3916
Gray CM, Monson RK, Fierer N (2010) Emissions of volatile organic compounds during the decomposition of plant litter. J Geophys Res 115. doi:10.1029/2010JG001291
Greenberg JP, Asensio D, Turnipseed A, Guenther AB, Karl T, Gochis D (2012) Contribution of leaf and needle litter to whole ecosystem BVOC fluxes. Atmos Environ 59:302–311
Guenther A (1997) Seasonal and spatial variations in natural volatile organic compound emissions. Ecol Appl 7:3445
Guenther AB, Zimmerman PR, Harley PC (1993) Isoprene and monoterpene emission rate variability: model evaluations and sensitivity analyses. J Geophys Res 98:12609–12617
Guenther A, Karl T, Harley P, Wiedinmyer C, Palmer PI, Geron C (2006) Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atmos Chem Phys 6:3181–3210
Hakola H, Laurila T, Rinne J, Puhto K (2000) The ambient concentrations of biogenic hydrocarbons at a northern European, boreal site. Atmos Environ 34:4971–4982
Hakola H, Hellén H, Tarvainen V, Bäck J, Patokoski J, Rinne J (2009) Annual variations of atmospheric VOC concentrations in a boreal forest. Boreal Environ Res 14:722–730
Hari P, Kulmala M (2005) Station for measuring ecosystem-atmosphere relations (SMEAR II). Boreal Environ Res 10:315–322
Hari P, Keronen P, Bäck J, Altimir N, Linkosalo T, Pohja T, Kulmala M, Vesala T (1999) An improvement of the method for calibrating measurements of photosynthetic CO2 flux. Plant Cell Environ 22:1297–1301
Hayward S, Muncey RJ, James AE, Halsall CJ, Hewitt CN (2001) Monoterpene emissions from soil in a Sitka spruce forest. Atmos Environ 35:4081–4087
Hellén H, Hakola H, Pystynen K-H, Rinne J, Haapanala S (2006) C2–C10 hydrocarbon emissions from a boreal wetland and forest floor. Biogeosciences 3:167–174
Ilvesniemi H, Levula J, Ojansuu R, Kolari P, Kulmala L, Pumpanen J, Launiainen S, Vesala T, Nikinmaa E (2009) Long-term measurements of the carbon balance of a boreal Scots pine dominated forest ecosystem. Boreal Environ Res 14:731–753
Insam H, Seewald MSA (2010) Volatile organic compounds (VOCs) in soils. Biol Fert Soils 46:199–213
Isidorov V, Jdanova M (2002) Volatile organic compounds from leaves litter. Chemosphere 48:975–979
Isidorov VA, Vinogorova VT, Rafałowski K (2003) HS-SPME analysis of volatile organic compounds of coniferous needle litter. Atmos Environ 37:4645–4650
Janson R, De Serves C, Romero R (1999) Emission of isoprene and carbonyl compounds from a boreal forest and wetland in Sweden. Agr Forest Meteorol 98–99:671–681
Kesselmeier J, Staudt M (1999) Biogenic volatile organic compounds (VOC): An overview on emission, physiology and ecology. J Atmos Chem 33:23–88
Kolari P, Bäck J, Taipale R, Ruuskanen TM, Kajos MK, Rinne J, Kulmala M, Hari P (2012) Evaluation of accuracy in measurements of VOC emissions with dynamic chamber system. Atmos Environ 62:344–351
Kulmala M, Toivonen A, Mäkelä JM, Laaksonen A (1998) Analysis of the growth of nucleation mode particles observed in Boreal forest. Tellus 50B:449–462
Kulmala M, Suni T, Lehtinen KEJ, Dal Maso M, Boy M, Reissell A, Rannik Ü, Aalto P, Keronen P, Hakola H, Bäck J, Hoffmann T, Vesala T, Hari P (2004) A new feedback mechanism linking forests, aerosols, and climate. Atmos Chem Phys 4:557–562
Kulmala L, Launiainen S, Pumpanen J, Lankreijer H, Lindroth A, Hari P, Vesala T (2008) H2O and CO2 fluxes at the floor of a boreal pine forest. Tellus 60B:167–178
Kulmala L, Pumpanen J, Hari P, Vesala T (2011) Photosynthesis of ground vegetation in different aged pine forests: Effect of environmental factors predicted with a process-based model. J Veg Sci 22:96–110
Leff JW, Fierer N (2008) Volatile organic compound (VOC) emissions from soil and litter samples. Soil Biol Biochem 40:1629–1636
Lindinger W, Hansel A, Jordan A (1998) On-line monitoring of volatile organic compounds by means of Proton-Transfer-Reaction Mass Spectrometry (PTRMS) Medical applications, food control and environmental research. Int J Mass Spectrom 173:191–241
Mogensen D, Smolander S, Sogachev A, Zhou L, Sinha V, Guenther A, Williams J, Nieminen T, Kajos MK, Rinne J, Kulmala M, Boy M (2011) Modelling atmospheric OH-reactivity in a boreal forest ecosystem. Atmos Chem Phys 11:9709–9719
Niinemets Ü, Kuhn U, Harley PC, Staudt M, Arneth A, Cescatti A, Ciccioli P, Copolovici L, Geron C, Guenther A, Kesselmeier J, Lerdau MT, Monson RK, Peñuelas J (2011) Estimations of isoprenoid emission capacity from enclosure studies: measurements, data processing, quality and standardized measurement protocols. Biogeosciences 8:2209–2246
Noe SM, Hüve K, Niinemets Ü, Copolovici L (2012) Seasonal variation in vertical volatile compounds air concentrations within a remote hemiboreal mixed forest. Atmos Chem Phys 12:3909–3926
Nölscher AC, Williams J, Sinha V, Custer T, Song W, Johnson AM, Axinte R, Bozem H, Fischer H, Pouvesle N, Phillips G, Crowley JN, Rantala P, Rinne J, Kulmala M, Gonzales D, Valverde-Canossa J, Vogel A, Hoffmann T, Ouwersloot HG, Vilà-Guerau de Arellano J, Lelieveld J (2012) Summertime total OH reactivity measurements from boreal forest during HUMPPA-COPEC 2010. Atmos Chem Phys Discuss 12:7419–7452
Ortega J, Helmig D (2008) Approaches for quantifying reactive and low-volatility biogenic organic compound emissions by vegetation enclosure techniques – Part A. Chemosphere 72:343–364
Ortega J, Helmig D, Daly RW, Tanner DM, Guenther AB, Herrick JD (2008) Approaches for quantifying reactive and low-volatility biogenic organic compound emissions by vegetation enclosure techniques – Part B: Applications. Chemosphere 72:365–380
Owen SM, Clark S, Pompe M, Semple KT (2007) Biogenic volatile organic compounds as potential carbon sources for microbial communities in soil from the rhizosphere of Populus tremula. FEMS Microbiol Lett 268:34–39
Peñuelas J, Staudt M (2010) BVOCs and global change. Trends Plant Sci 15:133–144
Persson T, Bååth E, Clarholm M, Lundkvist H, Söderström BE, Sohlenius B (1980) Trophic structure, biomass dynamics and carbon metabolism of soil organisms in a Scots pine forest. In: Persson T (ed) Structure and function of northern coniferous forests: an ecosystem study. Ecol Bull 32:419–459
Pihlatie M, Pumpanen J, Rinne J, Ilvesniemi H, Simojoki A, Hari P, Vesala T (2007) Gas concentration driven fluxes of nitrous oxide and carbon dioxide in boreal forest soil. Tellus 59B:458–469
Räisänen T, Ryyppö A, Kellomäki S (2009) Monoterpene emission of a boreal Scots pine (Pinus sylvestris L.) forest. Agr Forest Meteorol 149:808–819
Rinne J, Hakola H, Laurila T, Rannik Ü (2000) Canopy scale monoterpene emissions of Pinus sylvestris dominated forest. Atmos Environ 34:1099–1107
Rinne J, Taipale R, Markkanen T, Ruuskanen TM, Hellén H, Kajos MK, Vesala T, Kulmala M (2007) Hydrocarbon fluxes above a Scots pine forest canopy: measurements and modeling. Atmos Chem Phys 7:3361–3372
Rinne J, Bäck J, Hakola H (2009) Biogenic volatile organic compound emissions from the Eurasian taiga: current knowledge and future directions. Boreal Environ Res 14:807–826
Ruuskanen TM, Kolari P, Bäck J, Kulmala M, Rinne J, Hakola H, Taipale R, Raivonen M, Altimir N, Hari P (2005) On-line field measurements of monoterpene emissions from Scots pine by proton-transfer-reaction mass spectrometry. Boreal Environ Res 10:553–567
Schaub A, Blande JD, Graus M, Oksanen E, Holopainen JK, Hansel A (2010) Real-time monitoring of herbivore induced volatile emissions in the field. Physiol Plantarum 138:123–133
Sinha V, Williams J, Lelieveld J, Ruuskanen T, Kajos M, Patokoski J, Hellén H, Hakola H, Mogensen D, Boy M, Rinne J, Kulmala M (2010) OH reactivity measurements within a boreal forest: evidence for unknown reactive emissions. Environ Sci Technol 44:6614–6620
Spanke J, Rannik Ü, Forkel R, Nigge W, Hoffmann T (2001) Emission fluxes and atmospheric degradation of monoterpenes above a boreal forest: field measurements and modeling. Tellus B 53:406–422
Sumner AL, Menke EJ, Dubowski Y, Newberg JT, Penner RM, Hemminger JC, Wingen LM, Brauers T, Finlayson-Pitts BJ (2004) The nature of water on surfaces of laboratory systems and implications for heterogeneous chemistry in the troposphere. Phys Chem Chem Phys 6:604–613
Taipale R, Ruuskanen TM, Rinne J, Kajos MK, Hakola H, Pohja T, Kulmala M (2008) Technical Note: Quantitative long-term measurements of VOC concentrations by PTR-MS – measurement, calibration, and volume mixing ratio calculation methods. Atmos Chem Phys 8:6681–6698
Tarvainen V, Hakola H, Rinne J, Hellén H, Haapanala S (2007) Towards a comprehensive emission inventory of terpenoids from boreal ecosystems. Tellus 59B:526–534
Vesala T, Haataja J, Aalto P, Altimir N, Buzorius G, Garam E, Hämeri K, Ilvesniemi H, Jokinen V, Keronen P, Lahti T, Markkanen T, Mäkelä JM, Nikinmaa E, Palmroth S, Palva L, Pohja T, Pumpanen J, Rannik Ü, Siivola E, Ylitalo H, Hari P, Kulmala M (1998) Long-term field measurements of atmosphere-surface interactions in boreal forest combining forest ecology, micrometeorology, aerosol physics and atmospheric chemistry. Trends in Heat, Mass & Momentum Transfer 4:17–35
Wallander H, Nilsson LO, Hagerberg D, Bååth E (2001) Estimation of the biomass and seasonal growth of external mycelium of ectomycorrhizal fungi in the field. New Phytol 151:753–760
Acknowledgments
We thank the staff of SMEAR II and the Hyytiälä Forestry Field station for their help and for the use of the facilities for the study. The financial support from the Vilho, Yrjö and Kalle Väisälä Fund, the Maj and Tor Nessling Foundation, the Academy of Finland Centre of Excellence program (project no 1118615) and Academy of Finland projects 130984 and 1127756 are gratefully acknowledged. The research leading to these results has received funding also from the European Union Seventh Framework Programme (FP7/2007–2013) under grant agreement n° 262254, and the Nordic Center of Excellence CRAICC (Cryosphere-Atmosphere Interactions in a Changing Arctic Climate).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Juha Mikola.
Appendix 1
Appendix 1
Rights and permissions
About this article
Cite this article
Aaltonen, H., Aalto, J., Kolari, P. et al. Continuous VOC flux measurements on boreal forest floor. Plant Soil 369, 241–256 (2013). https://doi.org/10.1007/s11104-012-1553-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1553-4