Abstract
High levels of selenium can cause adverse effects in plants as well as animals. In a greenhouse experiment, rapeseed (Brassica napus) was grown in an alkaline sandy loam soil treated with different levels of selenate-Se and selenite-Se ranging from 0 to 4 mg kg−1. Total dry matter yield of selenium-treated rapeseed plants decreased significantly as compared to control plants. Plants were stressed at a very early stage of vegetative growth and produced fewer rosettes and flowers. Plant height and leaf production were negatively affected by selenate-Se. Dry matter of leaves was significantly higher in selenite- than in selenate-treated plants. Selenate-treated plants accumulated 75–160 times more Se in shoots and 2–18 times more in roots as compared to selenite-treated plants at the rosette formation stage, with this difference narrowing at peak flowering stage. Rapeseed leaves were subjected to biochemical analysis at rosette and peak flowering stages. Accumulation of selenium in leaves resulted in a significant increase in lipid peroxidation, chlorophyll, vitamin C and free amino acids, and a decrease in phenols, total soluble sugars and starch concentration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Selenium has long been recognised as an essential micronutrient for animal and human nutrition (Birringer et al. 2002; Hartikainen 2005; Lobanov et al. 2008). Unicellular green algae are the only photosynthetic organisms that require Se for their optimum growth (Fu et al. 2002; Novoselov et al. 2002). Selenium has not yet been found to have any specific role in higher plants (Läuchli 1993; Terry et al. 2000; Kweon et al. 2004), although some resistant plants growing on seleniferous soils, including some species of Astragalus and Stanleya, require Se for their growth and development (Rosenfeld and Beath 1964; Feist and Parker 2001; Pickering et al. 2003). Most cultivated plants are Se non-accumulators and are sensitive to its presence in the growth medium (Terry et al. 2000; White et al. 2004; Freeman et al. 2006; Zhang et al. 2007).
Plants absorb Se from soil primarily as selenate and translocate it to foliage specifically to chloroplasts, where it follows the sulfur assimilation pathway (Terry et al. 2000; Ellis and Salt 2003; Sors et al. 2005). Considerable differences in the uptake and transport of various Se compounds have been reported (Yu and Gu 2007). Both selenate and organic Se compounds are taken up into plant roots from the soil by active process, whereas selenite uptake seems to be through passive diffusion (Asher et al. 1977; Arvy 1993; Terry et al. 2000; Zhang et al. 2003; White et al. 2004). Recently, Li et al. (2008) have reported that selenite uptake in wheat is also an active process mediated by proton-coupled phosphate transporters. Selenium toxicity is thought to be due to the non-specific replacement of sulfur by Se in proteins and other sulfur compounds (Minorsky 2003; White et al. 2004). Thus, in most plants, protein activities are adversely affected because of substitution of cysteine and methionine by their selenoanalogs in proteins, causing symptoms including chlorosis and stunting that mimic sulfate starvation, as well as withering and drying of leaves and premature death (Terry et al. 2000). Excess accumulation of selenium leads to significant deterioration in the quality of grains produced on seleniferous soils (Sharma et al. 2008).
Some pockets of seleniferous soils containing total Se from 0.23 to 4.55 mg kg−1 have been identified in northwestern India. Consumption of Se-rich feed based on forage and grain crops grown on these soils has resulted in serious health hazards to animals and humans (Dhillon and Dhillon 2003). Thus, the role of Se in plant growth and impact of dietary selenium continues to be most active area of research in Se-enriched areas. Rapeseed, an important oilseed crop and a major component of the Indian diet, is commonly cultivated in the seleniferous region of Indian Punjab. However, no specific information is available regarding the impact of Se accumulation on growth and biochemical constituents of crops grown on seleniferous soils. Depending on the type of Se species present in soil, different plant species may exhibit quantitative differences in the amount of Se absorbed. The present investigation was therefore undertaken to study accumulation of selenium by rapeseed (Brassica napus L.) at different growth stages as influenced by two sources of selenium, i.e. sodium selenate and sodium selenite. The impact of Se accumulation in rapeseed leaves on biochemical components such as carbohydrate composition, soluble proteins, vitamin C and lipid peroxidation (LPO) was also investigated.
Materials and methods
Greenhouse experiment
A greenhouse experiment was carried out with soil collected from the University Research Farm and rapeseed (Brassica napus, var GSC-5) as a test crop. The soil was air-dried in shade and sieved through a 2 mm sieve. The experimental soil was sandy loam in texture, alkaline (pH 8.25), electrical conductivity 0.35 ds m−1, calcium carbonate 2.13%, organic carbon 0.35%, total Se 0.35 mg kg−1 and water soluble Se 0.013 mg kg−1. A sample (4 kg) of experimental soil was weighed, treated with different levels and sources of selenium and placed in a series of pots lined with polythene sheeting. The treatments consisted of two sources of selenium, viz. sodium selenate (as Na2SeO4, MW 188.9) and sodium selenite (as Na2SeO3⋅5H2O, MW 263.0) procured from Sigma (http://www.sigmaaldrich.com); four levels of selenium, viz. 0, 1, 2 and 4 mg Se kg−1 soil, and four replications. A basal dose of 60 mg N kg−1 and 30 mg P2O5 kg−1 soil, supplied through urea and potassium dihydrogen orthophosphate, respectively, was also added in each pot. All the nutrients were applied in solution form and thoroughly mixed with soil. Thereafter the soil was brought to field capacity with distilled water and allowed to equilibrate for 24 h before rapeseed crop was sown. Seeds (15–20 per pot) were placed in the upper layer of soil (about 0.5 cm deep) and the soil was covered with polythene sheet to keep the soil moist until germination. After the seedlings were well established, five seedlings per pot were retained. Irrigation was supplied as distilled water as and when required. Plant tissue samples were collected at different growth stages of rapeseed and subjected to analysis of selenium and biochemical components.
Growth parameters
Various growth parameters, viz. dry matter yield, number of rosettes and flowers, were recorded for each treatment. Leaves (collected from the 2nd, 3rd and 4th positions of inflorescence) and root samples were collected at different growth stages, i.e. at rosette stage (40 days after sowing) and peak flowering stage (80 days after sowing). To remove surface contaminants, the samples were washed gently with tap water followed by rinsing with single-distilled and double-distilled water. The dry weight of different plant tissues was recorded after drying the samples to constant weight in a hot air oven at 55 ± 5°C.
Biochemical analysis
For the estimation of carbohydrates and total soluble proteins, a weighed quantity of plant tissue (0.5–1.0 g) was stored in 80% ethanol and 0.1 N NaOH, respectively, at low temperature until further analysis. For determination of chlorophyll, homogenised fresh leaf material was extracted with prechilled 80% acetone solution and concentrations were measured spectrophotometrically (Witham et al. 1971). LPO in fresh leaves was assayed by measuring thiobarbituric acid reactive substances (TBARS) according to the method of Heath and Parker (1968). Total soluble sugar levels were determined with phenol-sulfuric acid reagent (Dubois et al. 1956) using glucose as standard. Starch was extracted by following the method of Clegg (1956) and starch content was calculated by multiplying the amount of glucose (determined by the Dubois method) by a factor of 0.9. Reducing sugars content was determined by following the method of Nelson (1944). Total phenols were determined according to Swain and Hills (1959) using gallic acid as standard. Quantitative estimation of free amino acids was made according to Lee and Takahashi (1966), and protein contents in leaf tissue were determined according to Lowry et al. (1951) using bovine serum albumin as standard.
Chemical analysis
Dry plant material was digested in a di-acid mixture of nitric acid and perchloric acid at a ratio of 5:2, and the acid digest was subjected to Se analysis following the method described by Levesque and Vandette (1971).
Statistical analysis
There were four replications with duplicate observations in each replication. Data on Se content and biochemical components were analysed by analysis of variance (ANOVA) test for determining the critical differences between different treatments.
Results
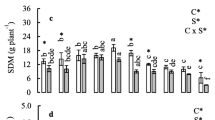
At the rosette stage, sodium-selenate-treated rapeseed plants displayed slower growth (as indicated by height) as compared to control or selenite-treated plants (Fig. 1). Plant growth was reduced significantly with higher concentration of Se (4 mg selenate-Se kg−1 soil) and these plants produced fewer leaves than those grown in soil with no selenium addition. The dry matter content of leaves at stage Ι (rosette stage) and stage II (peak flowering) decreased when the crop was grown in soil with increasing concentrations of both forms of Se (Table 1). However, the dry matter accumulation in leaves at different stages of growth was higher in selenite as compared to selenate treatments. Dry matter yield of selenate-treated plants decreased significantly at stage I and II as compared to control and selenite-treated plants (Fig. 2). The higher decrease in total dry matter yield of selenate-treated plants as compared to selenite treatments might be due to the slow rate of growth of these plants (Fig. 1), especially when 4 mg selenate-Se kg−1 soil was applied and the decrease was 30–35% as compared to plants grown without Se. Selenate and selenite-treated plants displayed significantly slower flowering rates than those grown without any added Se (Table 1). The number of rosettes formed at stage I and flowers produced at stage II were significantly lower due to the slower flowering rates in Se-treated plants as compared to control plants, and this effect was dose dependent (Table 1). Although there was a delay in flowering, those plants that ultimately did produce flowers were able to produce fruits.
Data recorded in Table 2 show that Se concentrations in shoot and root tissues from control plants were 0.75 and 0.58 mg kg−1, respectively, at stage I, and 0.14 and 0.42 mg kg−1, respectively, at stage II. Selenium application at the rate of 1 mg kg−1 soil increased Se shoot concentration up to 200- and 2.5-fold in selenate- and selenite-treated plants, respectively, at stage I. The corresponding increases were 800- and 5-fold at 4 mg Se kg−1 soil. In selenite-treated plants, Se concentrations were higher in roots than in shoots, whereas selenate-treated plants showed the reverse trend. Selenium accumulation in shoot tissue decreased while it increased in roots with the growth of the plant. Shoots showed greatest selenium accumulation at stage Ι whereas roots accumulated maximum Se at stage II. The dissimilar response to the application of selenate and selenite was evident at all the Se dosages applied to the soil, i.e. lower Se accumulation in selenite-treated plants and higher accumulation in selenate-treated plants in all tissues studied.
Addition of both forms of Se up to 2 mg kg−1 soil increased the total chlorophyll content and LPO in leaves at stages I and II (Fig. 3). However, the chlorophyll content of leaves decreased with 4 mg Se kg−1 as compared to 1 and 2 mg Se kg−1 soil, but the values were still higher compared to the control. At peak flowering stage, selenate-Se (4 mg Se kg−1 soil) increased LPO but the extent of LPO decreased with increasing concentrations of selenite, and LPO values were still higher with selenite treatments as compared to control leaves. A similar trend was observed when the data was expressed on a milligram protein basis. In rapeseed leaves collected from Se-treated soil, content of vitamin C increased and that of total phenols decreased significantly as compared to control leaves at both stages. Vitamin C content in leaves of control as well as Se-treated plants was higher at stage II as compared to stage I. Total soluble sugars, reducing sugars and starch content decreased significantly in leaves at stages I and II when different concentrations of selenate/selenite were supplied to the soil (Table 3), and this decrease was not dose dependent. The starch, total soluble sugars and reducing sugar content was higher in Se-treated leaves at stage II as compared to stage I.
Total soluble protein at stages I and II in rapeseed leaves from control plants was determined as 15.4% and 6.2%, respectively (Table 3). Rapeseed leaves from plants grown on soil supplied with various concentrations of Se exhibited a non-significant decrease in total soluble protein content at stage I. A significant increase in protein content at stage II was observed with selenate treatments only. The protein content of the leaves decreased in all treatments with the growth of the plant, i.e. higher protein content was observed at stage I as compared to stage II. The total free amino acid content of leaves increased significantly with selenate/selenite treatments, with higher values of total amino acid content in leaves with selenite as compared to selenate (Table 3).
Discussion
Selenium exists in the soil mainly as selenate, selenite and organic forms, although elemental Se and selenide-Se also exist (Mazzafera 1998) depending upon the oxidation–reduction potential of soil. Alkaline soil pH favours the formation of selenate under well-oxidising, and selenite in mildly-oxidising, environments (Geering et al. 1968). Transformation between selenite and selenate is a slow process; thus, both forms may co-exist in alkaline soils. Our results revealed that higher concentrations of Se supplied to the soil had negative effects on the vegetative and reproductive growth of rapeseed plants during the initial stages of development. Visual symptoms of sodium selenate (4 mg Se kg−1) toxicity such as growth reduction observed in the present study are similar to those reported earlier in wheat, mustard and pea plants (Tripathi and Misra 1974), maize (Dhillon et al. 1977) and wheat (Lyons et al. 2005). Selenium stunted plant growth and caused them to produce fewer leaves and flowers. Similar effects on vegetative and reproductive growth were reported previously by Bañuelos et al. (1997) and Euliss (2004), who reported delayed emergence and stunted growth of canola when grown on seleniferous soil. Although non-accumulators are sensitive to high Se concentration, they can tolerate as well as accumulate even high concentrations of Se without growth reduction when grown in Se-enriched soils. The critical level of Se in plants above which significant decrease in yield would occur was found to be 104.8 µg g−1 in raya (Brassica juncea Czern L.), 76.9 µg g−1 in maize (Zea mays L.), 41.5 µg g−1 in rice (Oryza sativa L.) and 18.9 µg g−1 in wheat (Triticum aestivum L.) shoots (Rani et al. 2005).
Our data revealed that Se-treated plants had lower leaf dry matter content than control plants. A significant reduction in dry matter accumulation by maize (Dhillon et al. 1977), pearl millet, oat and berseem (Bawa et al. 1992) was reported when more than 0.5 mg Se kg−1 soil was applied. Dry matter yield decreased in wheat and sunflower (Singh and Singh 1978), various Brassica land races (Bañuelos et al. 1997) and rice (Prasad and Arora 1980) when grown on seleniferous soil. Coffee plants exhibited lower dry matter accumulation in roots and leaves when irrigated with 1 mM sodium selenite (Mazzafera 1998).
In the present study, selenium uptake was found to be higher in shoots and roots from selenate as compared to a selenite source. The highest Se shoot concentration was obtained in selenate-treated plants but, for both sources of selenium, there was a significant positive correlation between shoot Se concentration and soil Se concentration. Shoot Se concentration decreased between rosette formation and peak flowering stage, whereas root Se concentration showed the reverse trend. This trend was also observed in Arabidopsis thaliana, Astragalus bisculactus and other plants (Rosenfeld and Beath 1964; Xue et al. 2001; Turakainen et al. 2004) whose shoot Se concentrations increase to a maximum during seedling growth, then decline prior to, or upon, flowering. White et al. (2007b) reported that in Arabidopsis thaliana accessions Columbia and Landsberg erecta grown on agar containing 0.3 μM selenate, there was an initial increase in shoot and root Se concentrations up to 20 days after sowing, followed by a gradual decline in Se concentration in these tissues from 20 to 40 days after sowing. The decrease in Se content in shoots during crop development might be due to its translocation towards other plant parts, including reproductive organs. A number of authors have investigated the absorption and transport of selenate and selenite in plants like Phaseolus vulgaris (Arvy 1989, 1993), Lycopersicon esculentum (Asher et al. 1977) and several other vegetables (Bañuelos and Meek 1989), and considerable differences in uptake and transport of various selenium compounds have been reported (Yu and Gu 2007). Most Se was reported to be translocated to the aerial organs in plants receiving selenate (Asher et al. 1977; Arvy 1989, 1993), whereas the majority of Se supplied as selenite accumulated in the roots. The absorption of selenate by roots and its distribution into plants is much faster than that of selenite (White et al. 2004), and total Se accumulation in a plant has been reported to be about 10-fold higher from selenate as compared to selenite. A dissimilar response in shoot selenium concentration to the application of selenite and selenate was also reported earlier (Cartes et al. 2005). Selenate salts are very soluble (Elrashidi et al. 1987) and are readily taken up by plants (Gissel-Nielsen and Bisbjerg 1970; Eisler 1985) whereas availability of selenite to the plants may be influenced by soil composition, as selenite is absorbed by clay minerals and iron oxides (Hamdy and Gissel-Nielsen 1977) present in the soil. Barrow et al. (2005) reported that in the same type of soil selenite behaves like phosphate and is more strongly sorbed than selenate to soil surfaces, thus becoming less bioavailable than selenate at equal rate of soil application.
Accumulation of Se in leaves resulted in significant increases in chlorophyll, vitamin C and LPO, and decreased phenolic content (Fig. 3). Variations in chlorophyll content of plants grown on soil supplemented with different forms of Se have been reported by earlier researchers (Mazzafera 1998; Xue et al. 2001; Huang et al. 2005). Mazzafera (1998) reported decreased chlorophyll content in coffee seedlings receiving sodium selenite treatment. In contrast, higher chlorophyll concentration in edible spinach and ground tomato was observed in plants treated with 5 μM selenium (Hawrylak and Szymanska 2004). Selenium treatment resulted in increased vitamin C content in lettuce cultivars (Shang et al. 1998), celery (Lee et al. 1999), mustard and spinach leaves (Saggoo et al. 2004), tea leaves (Hu et al. 2003; Huang et al. 2005) and Brassica chinensis (Kweon et al. 2004). Increasing concentration of Se in broccoli from <1.0 to >800 μg g−1 inhibited production of most phenolic acids (Finley et al. 2005), although the relative distribution of specific phenolic acids was not altered.
Our studies showed that extent of LPO was higher with selenate (4 mg Se kg−1 soil) whereas a similar concentration of selenite has a lowering effect on LPO extent. Both antioxidant and prooxidant roles for Se have been reported depending upon its concentration in the growth medium as well as in plant tissues. At lower concentrations, Se acts as antioxidant and results in decreased LPO, whereas higher concentrations resulted in increased LPO in lettuce (Xue et al. 2001), wheat (Nowak et al. 2004) and ryegrass (Cartes et al. 2005). The level of LPO was dependent on the shoot Se concentration rather than the chemical form of Se supplied to plants. Evidence of Se-induced lipid peroxidation, and consequently oxidative stress, was obtained with the increase level of TBARS in coffee cells exposed to 0.05 and 0.5 mM selenite (Gomes-Junior et al. 2007).
Our results on the carbohydrate composition of rapeseed leaves are contradictory to earlier studies in other plants, which reported that selenium induced starch accumulation (Pennanen et al. 2002), increased soluble sugars in coffee (Mazzafera 1998), mustard and spinach leaves (Saggoo et al. 2004) and glucose in bean plants (Arvy 1989). The data presented in Table 3 showed a nonsignificant change in protein and a significant increase in free amino acid content in rapeseed leaves due to selenate/selenite treatment at stage I, and by selenite at stage II. The increased amino acid content at various Se concentrations might be due to the synthesis of non-protein selenoamino acids. Higher values of total amino acid content in leaves with selenite treatments as compared to selenate treatments might be attributed to the fact that selenate reduction by ATP sulfurylase is a rate-limiting step (Terry et al. 2000; Raspor et al. 2003). Peterson and Butler (1962) suggested that Se accumulator species have evolved a mechanism whereby Se may be excluded from protein incorporation. Selenium exclusion may be due to formation of non-protein selenoamino acids such as selenomethylselenocysteine, and synthesis of these compounds may divert selenium from formation of SeCys and SeMet (Brown and Shrift 1981) in Se-tolerant species. In our study, Brassica leaves showed higher concentrations of Se, which is consistent with their unique sulfur metabolism (Willey and Wilkins 2006) and useful for phytoremediation of Se-laden soils (Bañuelos et al. 1997; Terry et al. 2000). The high Se concentration in Brassica leaves can result in the accumulation of several unusual metabolites, such as Semethylselenocysteine, γ-glutamylSemethylselenocysteine and Semethylselenomethionine, in these tissues (White et al. 2007b). Although such changes in protein content has not been proved, the higher accumulation of Se might explain the chemical alterations observed in the present study.
Consumption of Brassica leaves, used for example in a popular Punjabi dish (sarson ka saag) throughout the world, may vary from 20 to 25 g day−1 person−1 on a dry weight basis (200 to 250 g day−1 person−1 on fresh weight basis). Thus, we can conclude that consumption of 25–50 g (dry weight) of Brassica leaves grown in soil treated with Selenite-Se up to 4 mg kg−1 may be sufficient to meet the daily requirement for Se. However, consumption of the same amount of Brassica leaves grown on soils supplied with 1–4 mg kg−1 selenate-Se will result in a much higher intake of Se than the recommended daily allowance of 50–400 µg day−1.
Abbreviations
- DW:
-
Dry weight
- LPO:
-
Lipid peroxidation
- Se:
-
Selenium
- TBARS:
-
Thiobarbituric acid reactive substances
References
Arvy MP (1989) Some factors influencing the uptake and distribution of selenite in the bean plant (Phaseolus vulgaris). Plant Soil 117:129–133
Arvy MP (1993) Selenate and selenite uptake and translocation in bean plants. J Exp Bot 44:1083–1087
Asher CJ, Butler GW, Peterson PJ (1977) Selenium transport in root systems of tomato. J Exp Bot 28:279–291
Bañuelos GS, Meek DW (1989) Selenium accumulation in selected vegetables. J Plant Nutr 12:1255–1272
Bañuelos GS, Ajwa HA, Terry N, Zayed A (1997) Phytoremediation of selenium laden soils: a new technology. J Environ Qual 52(6):426–430
Barrow NJ, Cartes P, Mora ML (2005) Modifications to the Freundlich equation to describe anion sorption over a large range and to describe competition between pairs of ions. Eur J Soil Sci 56:601–606
Bawa SS, Dhillon KS, Dhillon SK (1992) Screening of different fodders for selenium absorption capacity. Indian J Dairy Sci 45:457–460
Birringer M, Pilawa S, Flohe L (2002) Trends in selenium biochemistry. Nat Prod Rep 19:693–718
Brown TA, Shrift A (1981) Exclusion of selenium from proteins of selenium tolerant Astragalus species. Plant Physiol 67:1051–1053
Cartes P, Gianfreda L, Mora ML (2005) Uptake of selenium and its antioxidant activity in ryegrass when applied as selenate and selenite forms. Plant Soil 276:359–367
Clegg KM (1956) The application of the anthrone reagent to the estimation of starch in cereals. J Sci Food Agric 7:40–44
Dhillon KS, Dhillon SK (2003) Distribution and management of seleniferous soils. Adv Agron 79:119–184
Dhillon KS, Randhawa NS, Sinha MK (1977) Selenium status of some common fodders and natural grasses of Punjab. Indian J Dairy Sci 30:218–224
Dubois M, Gills KA, Hamilton JK, Rober PA (1956) Calorimetric method for the determination of sugars and related substances. Anal Chem 28:350–356
Eisler R (1985) Selenium hazards to fish, wildlife and invertebrates: a synoptic review. Biological Report 85 (1.5), US Fish and Wildlife Service, Washington, DC
Ellis DR, Salt DE (2003) Plants, selenium and human health. Curr Opin Plant Biol 6:273–279
Elrashidi MA, Adriano DC, Workman SM, Lindsay WL (1987) Chemical equilibria of selenium in soils: a theoretical development. Soil Sci 144:141–152
Euliss KW (2004) The effects of selenium accumulation in hydroponically grown canola (Brassica napus). J Young Invest 1:1–12
Feist LJ, Parker DR (2001) Ecotypic variation in selenium accumulation among populations of Stanleya pinnata. New Phytol 149:61–69
Finley JW, Sigrid-Keck A, Robbins RJ, Hintze KJ (2005) Selenium enrichment of broccoli: interactions between selenium and secondary plant compounds. J Nutr 135:1236–1238
Freeman JL, Zhang LH, Marcus MA, Fakra S, McGrath SP, Pilon-Smits EAH (2006) Spatial imaging, speciation, and quantification of selenium in the hyperaccumulator plants Astragalus bisulcatus and Stanleya pinnata. Plant Physiol 142:124–134
Fu LH, Wang XF, Eyal Y, She YM, Donald LJ, Standing KG, Ben Hayyim G (2002) A selenoprotein in the plant kingdom. Mass spectrometry confirms that an opal codon (UGA) encodes selenocysteine in Chlamydomonas reinhardtii glutathione peroxidase. J Biol Chem 277:25983–25991
Geering HR, Cary EE, Jones LHP, Allaway WH (1968) Solubility and redox criteria for the possible forms of selenium in soils. Soil Sci Soc Am J 32:35–40
Gissel-Nielsen G, Bisbjerg B (1970) The uptake of applied selenium from soils by plants. 2. The utilization of various selenium compounds. Plant Soil 32:382–396
Gomes-Junior RA, Gratao PL, Gaziola SA, Mazzafera P, Lea PJ, Azevedo RA (2007) Selenium-induced oxidative stress in coffee cell suspension cultures. Funct Plant Biol 34:449–456
Hamdy AA, Gissel-Nielsen G (1977) Fixation of selenium by clay mineral and iron oxides. Z Pflanzenernaehr Bodenkd 140:63–70
Hartikainen H (2005) Biogeochemistry of selenium and its impact on food chain quality and human health. J Trace Elem Med Biol 18:309–318
Hawrylak B, Szymanska M (2004) Selenium as a sulphydrylic group inductor in plants. Cell Mol Biol Lett 9:329–336
Heath RL, Parker L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hu Q, Xu J, Pang G (2003) Effect of selenium on the yield and quality of green tea leaves harvested in early spring. J Agric Food Chem 51(11):3379–3381
Huang Y, Xu J, Hu Q (2005) Effect of selenium on preservative quality of green tea during autumn tea-processing season. J Agric Food Chem 53:7444–7447
Kweon Y, Cheol S, Kum P, Young C, Ah J (2004) Effect of selenium source and concentrations on growth and quality of endive and pak-choi in deep flow culture. Korean J Hortic Sci Technol 22(2):151–155
Läuchli A (1993) Selenium in plants: uptake, functions and environmental toxicity. Bot Acta 106:455–468
Lee GP, Park K, Lee JM, Gross KC, Watada AE, Lee SK (1999) Quality improvement of Seoul celery by selenium in nutrient solution culture. Acta Hortic 483:185–192
Lee YP, Takahashi T (1966) An improved colorimetric determination of amino acids with use of ninhydrin. Anal Biochem 14:71–77
Levesque M, Vandette ED (1971) Selenium determination in soil and plant materials. Can J Soil Sci 51:85–93
Li HF, McGrath SP, Zhao FJ (2008) Selenium uptake, translocation and speciation in wheat supplied with selenate or selenite. New Phytol 178:92–102
Lobanov AV, Hatfield DL, Gladyshev VN (2008) Reduced reliance on the trace element selenium during evolution of mammals. Genome Biol 9:R62
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Lyons GH, Stangoulis JCR, Graham RD (2005) Tolerance of wheat (Triticum aestivum L.) to high soil and solution selenium levels. Plant Soil 270:179–188
Mazzafera P (1998) Growth and biochemical alterations in coffee due to selenite toxicity. Plant Soil 201:189–196
Minorsky PV (2003) The hot and the classic selenium in plants. Plant Physiol 133:14–15
Nelson N (1944) A photometric adaptation of the Somogyi method for determination of glucose. J Biol Chem 153:375–380
Novoselov S, Rao M, Onoshko N, Zhi H, Kryukov G, Xiang Y, Weeks D, Hatfield D, Gladyshev V (2002) Selenoproteins and selenocysteine insertion system in the model plant cell system, Chlamydomonas reinhardtii. EMBO J 21:3681–3693
Nowak J, Kaklewski K, Ligocli M (2004) Influence of selenium on oxidoreductive enzymes activity in soil and in plants. Soil Biol Biochem 36:1553–1558
Pennanen A, Xue T, Hartikainen H (2002) Protective role of selenium in plant subjected to severe UV irradiation stress. J Appl Bot 76:66–76
Peterson PJ, Butler GW (1962) The uptake and assimilation of selenite by higher plants. Aust J Biol Sci 15:126–146
Pickering IJ, Wright C, Bubner B, Ellis D, Persans MW, Yu EY, George GN, Prince RC, Salt DE (2003) Chemical form and distribution of selenium and sulfur in the selenium hyperaccumulator Astragalus bisulcatus. Plant Physiol 131:1460–1467
Prasad T, Arora SP (1980) Studies on 75Se accumulation in rice plants and its effect on yield. J Nuclear Agric Biol 9:77–78
Rani N, Dhillon KS, Dhillon SK (2005) Critical levels of selenium in different crops grown in an alkaline silty loam soil treated with selenite-Se. Plant Soil 277:367–374
Raspor P, Fujs S, Banszky L, Maraz A, Batic M (2003) The involvement of ATP sulfurylase in Se (VI) and Cr (VI) reduction processes in the fission yeast Schizosaccharomyces pombe. Appl Microbiol Biotechnol 63:89–95
Rosenfeld I, Beath OA (1964) Selenium. Geobotany, biochemistry, toxicity and nutrition. Academic, New York
Saggoo MIS, Dhillon KS, Dhillon SK, Kaur J (2004) Evaluation of consumability potentials of leafy vegetables harvested from selenium rich soil. Environ Inf Arch 2:479–489
Shang QM, Guo LH, Li SJ (1998) Effect of selenium on quality of hydroponics lettuce. J China Agric Univ 3:67–71
Sharma S, Dhillon KS, Dhillon SK, Munshi SK (2008) Changes in biochemical components of wheat and rapeseed grown on selenium-contaminated soil. Arch Agron Soil Sci 54(1):33–40
Singh M, Singh N (1978) Selenium toxicity and its detoxication by phosphorus. Soil Sci 126:255–262
Sors TG, Ellis DR, Salt DE (2005) Selenium uptake, translocation, assimilation and metabolic fate in plants. Photosynth Res 86:373–389
Swain T, Hills WE (1959) The phenolic constituents of Prunus domestica: the quantitative analysis of phenolic constituents. J Sci Food Agric 10:63–68
Terry N, Zayed AM, deSouza MP, Tarun AS (2000) Selenium in higher plants. Annu Rev Plant Physiol Plant Mol Biol 51:401–432
Tripathi N, Misra SG (1974) Uptake of applied selenium by plants. Indian J Agr Sci 44:804–807
Turakainen M, Hartikainen H, Seppänen MM (2004) Effects of selenium treatments on potato (Solanum tuberosum L.) growth and concentrations of soluble sugars and starch. J Agric Food Chem 52:5378–5382
White PJ, Bowen HC, Parmaguru P, Fritz M, Spracklen WP, Spiby RE, Meacham MC, Mead A, Harriman M, Trueman LJ, Smith BM, Thomas B, Broadley MR (2004) Interactions between selenium and sulphur nutrition in Arabidopsis thaliana. J Exp Bot 55:1927–1937
White PJ, Bowen HC, Marshall B, Broadley MR (2007a) Extraordinarily high leaf selenium to sulfur ratios define Se accumulator plants. Ann Bot 100:111–118
White PJ, Broadley MR, Bowen HC, Johnson SE (2007b) Selenium and its relationship with sulfur. In: Hawkesford MJ, de Kok LJ (eds) Sulfur in plants-an Ecological prospective. Springer, Dordrecht, pp 225–252
Willey N, Wilkins J (2006) An analysis of intertaxa differences in sulfur concentration in angiosperms. J Plant Nutr Soil Sci 169:717–727
Witham FH, Blaydes DF, Devlin RM (1971) Experiments in plant physiology. Van Nostrand, New York, p 245
Xue T, Hartikainen H, Piironen V (2001) Antioxidative and growth promoting effect of selenium on senescing lettuce. Plant Soil 237:55–61
Yu XZ, Gu JD (2007) Metabolic responses of weeping willows to selenate and selenite. Environ Sci Pollut Res Int 14(7):510–517
Zhang L, Ackley AR, Pilon-Smits EAH (2007) Variation in selenium tolerance and accumulation among 19 Arabidopsis thaliana accessions. J Plant Physiol 164:327–336
Zhang Y, Pan G, Chen J, Hu Q (2003) Uptake and transport of selenite and selenate by soybean seedlings of two genotypes. Plant Soil 253:437–443
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Peter Christie.
Rights and permissions
About this article
Cite this article
Sharma, S., Bansal, A., Dhillon, S.K. et al. Comparative effects of selenate and selenite on growth and biochemical composition of rapeseed (Brassica napus L.). Plant Soil 329, 339–348 (2010). https://doi.org/10.1007/s11104-009-0162-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-009-0162-3