Abstract
In arid and semiarid shrubland ecosystems of the Mediterranean basin, soil moisture is a key factor controlling biogeochemical cycles and the release of CO2 via soil respiration. This is influenced by increasing temperatures. We manipulated the microclimate in a Mediterranean shrubland to increase the soil and air night-time temperatures and to reduce water input from precipitation. The objective was to analyze the extent to which higher temperatures and a drier climate influence soil CO2 emissions in the short term and on an annual basis. The microclimate was manipulated in field plots (about 25 m2) by covering the vegetation during the night (Warming treatment) and during rain events (Drought treatment). Soil CO2 effluxes were monitored in the treatments and compared to a control over a 3-year period. Along with soil respiration measurements, we recorded soil temperature at 5 cm depth by a soil temperature probe. The seasonal pattern of soil CO2 efflux was characterized by higher rates during the wet vegetative season and lower rates during the dry non-vegetative season (summer). The Warming treatment did not change SR fluxes at any sampling date. The Drought treatment decreased soil CO2 emissions on only three of 10 occasions during 2004. The variation of soil respiration with temperature and soil water content did not differ significantly among the treatments, but was affected by the season. The annual CO2 emissions were not significantly affected by the treatments. In the semi-arid Mediterranean shrubland, an increase of soil CO2 efflux in response to a moderate increase of daily minimum temperature is unlikely, whereas less precipitation can strongly affect the soil processes mainly limited by water availability.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Respiration of terrestrial ecosystems is a major flux in the global carbon cycle and a potentially important positive feedback mechanism for climate change (Schimel 1995; Prentice et al. 2001). Where water is a limiting factor—as in arid and semiarid systems around the Mediterranean basin—soil moisture is a key factor controlling microbial respiration and mineralization processes (Cortez 1997; Conant et al. 1998; Leiròs et al. 1999), which constitute part of the CO2 efflux from soil to the atmosphere. Although the rate at which C can be sequestered in dry regions is low, C residence times in dryland soil are long, sometimes even longer than in forest soils. Therefore, arid regions may lose less C than wet regions because lack of water limits soil mineralization and consequently the return of C to the atmosphere (Wohlfart et al. 2008). Several studies demonstrate higher CO2 emissions from soil when the climate becomes warmer, especially when occuring in temperate and boreal ecosystems (Peterjohn et al. 1994; Rustad et al. 2001; Melillo et al. 2002; Bekku et al. 2003). In environments where soil C stocks can reach high values, any temperature increase can lead to high CO2 release to the atmosphere, with a positive feedback on climate warming. Some studies report an acclimation effect when easily decomposable C is depleted due to increasing temperatures. This leads to a transient increase in CO2 emissions, followed by a return to pre-heating emission rates (Oechel 2000; Melillo et al. 2002; Kirschbaum 2004; Eliasson et al. 2005). These studies were frequently conducted increasing soil and/or air temperatures during the whole day. But, according to some authors, global warming has been causing a decrease in daily temperature amplitude due to an increase of minimum (night-time) temperatures (Easterling et al. 1997; Easterling et al. 2000; Vose et al. 2005), therefore specific studies to test the impact of this trend are needed. Only little research has focused on soil CO2 emissions in the Mediterranean area under a moderate change in temperature regime, perhaps based on the perception that sparse vegetation cover and bare soil can only produce low emissions (Wohlfart et al. 2008). Most studies in the Mediterranean environment investigated woodland or maquis formations, which represent later vegetational succession stages. Gariga ecosystems are made up of low shrubs and sparse vegetation, representing the first stages of soil colonization after disturbance. Those ecosystems are widespread in the Mediterranean basin, reflecting thousands of years of human modifications (agriculture, pastures, timber and wood, and, recently, tourism, etc.). According to the FAO Forest Resource Assessment (FAO 2005), 30 million ha are covered by “other wooded lands”, including shrublands in the Mediterranean basin. The Italian 2005 National Inventory of Forest and Carbon INFC (http://www.sian.it/inventarioforestale/jsp/home.jsp) reported that Mediterranean shrublands and maquis cover about 690,100 ha. La Mantia et al. (2007) showed a linear increase of organic C from 13 to 85 t C ha−1 in a Mediterranean soil along a chronosequence of 30 years after land abandonment. Warmer and drier climate conditions are forecasted for Southern Europe during the summer season (Cubasch et al. 1996; Kundzewicz et al. 2001; Christensen et al. 2007). The extensive shrubland around the Mediterranean basin warrants focusing on the potential consequences of higher night-time temperatures and longer summer drought periods on soil CO2 emissions in these ecosystems.
To evaluate the responses of different ecosystem processes to climate changes, we induced a night-time soil and air warming and longer summer drought periods in a semi-arid Mediterranean shrub ecosystem (gariga) for 5 years. The approach involved a field climate manipulation system (Warming and Drought treatments). We analyze the responses of soil CO2 emission to the treatments over a three-year period in order to answer the following questions:
-
(1)
will a moderate increase of night-time temperatures change daytime CO2 emission rates?
-
(2)
will a longer summer drought period change soil CO2 emission rates? And will it alter the following soil CO2 emission rates?
-
(3)
will the natural summer drought alter the response of soil respiration to increasing temperature, in semi-deciduous gariga ecosystems?
-
(4)
will the induced day-by-day increase of night-time temperatures and the induced drought change the annual soil carbon emission? Can the changes be related to the season in which the drought occurs?
In order to answer these questions:
-
(1)
we measured soil CO2 emission at day-time and in drought-treated and non-drought-treated periods over a three-year study period (Q1 and Q2);
-
(2)
we compared the parameters of a set of equations describing the SR variation in response to temperature and soil water content, in wet and dry seasons (Q3);
-
(3)
we compared the estimated annual and seasonal CO2 emissions using a semi-empirical model for the different treatments (Q4 and Q5).
Materials and methods
Description of the area
The study area is located in the Regional Park Porto Conte Capo Caccia in the Capo Caccia peninsula (northeast Sardinia, Italy; 40° 37′ N, 8° 10′ E). The geologic substrate is Mesozoic limestone and the main soil type is Terra Rossa (Lithic Xerorthent and Typic Rhodoxeralfs, USDA 1993). The soil is rocky and shallow (20–30 cm); the texture is sandy loam, with an ABC profile; pH is 7.7; soil organic matter content is 3.9% in the main rooting zone (0–10 cm); bulk density is 1.1 g cm−3. The climate is semi-arid, characterized by a mean annual rainfall of 640 mm occurring mainly in autumn and spring, with a long dry period from May to August. The mean annual temperature is 16.8°C, ranging from 7°C (mean minimum temperatures in the coldest month) to 28°C (mean maximum temperatures in the hottest month) (meteorological station of Fertilia Airport N 40°38′ E 8°17′; altitude 40 m asl; distance to sea about 4 km; period of observation 1961–1990).
The experimental plots (25 m2 each) were located in a firebreak opened in 1973. The firebreak had been managed by controlled fire until 1990, then mechanically in 1991 and 1992. Since 1993, no further interventions were made and the vegetation started to colonize the soil. Presently, vegetation cover inside the plots is made up by plants with a 1 m maximum height, constituted of Cistus monspeliensis L., Helichrysum italicum G. Don and Dorycnium pentaphyllum Scop. The presence of other shrubs (Pistacia lentiscus L., C. creticus L., Daphne gnidium L., etc.) is more sporadic. Bare soil makes up about 20% of the soil surface.
Microclimate manipulation and monitoring
The experimental plots were set up according to Beier et al. (2004). Three treatments were applied (Warming, Drought, Control). The Warming treatment involved covering the vegetation and soil with aluminium curtains during the night, so that the ecosystem retained a portion of the energy accumulated during daylight. This simulated the effect of global warming on daily minimum temperatures. Extended summer drought (Drought treatment) was induced by covering the plots with waterproof plastic curtains, transparent to infrared radiation, during rain events that occurred during two periods per year, in spring and in autumn (1 May–13 June 2002; 27 August–15 November 2002; 1 April–31 May 2003; 1 October–30 November 2003; 1 April–31 May 2004; 1 October–30 November 2004). To avoid damages of the infrastructure, the curtains were automatically opened if the wind reaches a threshold value (10 m s−1). In particular, during the autumn season very intense rains were frequently coupled with strong wind condition, thus reducing the efficiency of the system in removing rains during this period. Metallic barriers were inserted into the soil around the drought plots, to avoid input of water flow when runoff occurs. Each plot was delimited by a metallic frame, 6 × 4 m wide and 1.5 m tall, supporting the mobile curtains (5 × 4 m). The curtains moved along two rails placed on the shorter sides of the scaffolding. An electronic device automatically controlled the movements of the curtains.
The control treatment was delimited in the same manner as the treated ones, except that they did not have any curtains. Nine plots were assigned to the three treatments according to an experimental design with three treatments randomly replicated in three blocks.
In each plot, air temperature (Ta) and relative humidity at 20 cm height from the soil surface (Igromer HP100A, Rotronic, CH), soil temperature at 10 cm depth (Ts10) and at 20 cm depth (Ts20) (LTN NR3, Tecno.El, IT), and soil water content (WCs) at 10 cm depth (ECH2O probe model EC-20, Decagon Devices, Inc.) were continuously monitored. Moreover, an in-situ weather station measured air temperature and relative humidity (Igromer HP100A, Rotronic, CH), wind direction and speed (03002 Young, USA), global solar radiation (PYR SKS1110, Skye Instruments, UK) and precipitation (ARG199, Environmental Measurements, UK) at 2 m height. The opening and closing times of the curtains were recorded for each plot by sensors (Mac-I) installed along the rails. These sensors allow the functioning of the curtains to be checked and to calculate the amount of precipitation excluded. All the data were acquired and stored in a datalogger (CR10X Campbell Scientific, Inc., USA) as half-hour mean values and downloaded on a laptop nearly every week. Given the high soil heterogeneity, we removed the differences of soil water capacity among the plots by calculating the daily mean relative soil water content (RSWC) as a percentage of the annual maximum, representing the value at saturation. These values were used for the model parameterization (see below). The values at saturation ranged between 17 and 28 % v/v among the nine plots, usually recorded during weeks 6–9 of every year or during the last weeks of each year, depending on the precipitation regime; the minimum values always occurred in summer between July and August, and ranged between 3 and 7 % v/v.
Soil CO2 efflux
Soil CO2 (SR) efflux was measured for 3 years (from 2002 to 2004) by a portable infrared gas analyzer EGM-3 (PP Systems, Hertfordshire, UK) connected to a closed-type cuvette SRC1 (PP Systems, Hertfordshire, UK). The measurements were made in the morning at sunrise (after the period of night-time warming), and at midday (12:00–14:00) at maximum temperatures, approximately every 30 days, on permanent 5 PVC collars (10 cm diameter, 5 cm height) per plot placed into the soil surface, and randomly distributed in bare soil and under vegetation cover. Collars were inserted into the soil for only 2–3 cm, in order to avoid root damages, 1 month before the start of measurements. Along with soil respiration measurements, soil temperature at 5 cm depth was recorded next to the collars by a soil temperature probe (PP Systems, Hertfordshire, UK), connected to the EGM.
Because soil water content varied significantly during the year, the model proposed by Hanson et al. (1993) was applied to the data to estimate the temperature sensitivity of SR without the limiting effect of water and to estimate the annual emission. This model combines two equations to describe the response of SR to temperature and soil water content.
The model is described by the following equation:
where
In these equations, SR is the soil respiration rate [µmol CO2 m−2 s−1], Q 10 is the increase in soil respiration with a 10°C increase of soil temperature, Cf is the soil coarse fraction (>2 mm), k is the sensitivity of SR to soil water content variation, R max is the maximum soil respiration rate when water content is at saturation, and Ts is the soil temperature at 5 cm depth.
We calculated the annual soil CO2 emission (SRcum) for each year and for each plot—as the sum of half-hour emissions calculated using Eq. 1 and the measured microclimatic variables. To make consistent, the soil temperatures measured continuously at 10 cm depth were calibrated, in each plot, with the temperatures measured at 5 cm depth during the SR measurements. Significant linear regressions were found for each plot with values of R2 higher than 0.90. The calculated temperatures at 5 cm depth were used for the annual calculations.
Statistical analysis
The effects of Warming treatments on the air and soil temperatures were tested by a Wilcoxon signed rank test. We tested if the daily minimum temperature differences (Warming minus Control) can be assumed to be higher than zero.
The effects of Drought treatments on the soil relative water content were tested by a Wilcoxon signed rank test. We tested if the daily mean value differences (Drought minus Control) can be assumed to be lower than zero.
Analysis of variance (ANOVA) was performed in order to test the effect of treatments and time on soil respiration, on estimated parameters of Eq. 1 and on cumulated annual CO2 emissions. In the case of significant differences between the treated variables and the control, the post-hoc Bonferroni test was performed. We considered the plot as the smallest independent unit (n = 3).
The statistical software SYSTAT 11 (SPSS Inc., Chicago, Illinois) was used to perform the analyses, using the General Linear Model (GLM) routine.
Results
Climate and treatment effects on microclimate
The climate characteristics of the area during the three-year study period are reported in Table 1. In 2002 and 2004, the mean annual air temperature was about 16°C, similar to the 30-year mean value. In 2002, total rainfall was about 600 mm, with an anomalous frequency in summer; in 2004, total precipitation was about 530 mm, with a prolonged drought from May to early September. The year 2003 was extremely dry, especially in spring and summer, with annual precipitation of 410 mm; during the spring, rainfall amounted to only 51 mm, which is about one-third of the 30-year mean value for this period; furthermore, the mean temperature of August 2003 was 27°C, i.e. 5°C warmer than the same month of the previous year or +2°C if compared with the 30-year mean value.
The Warming treatment significantly (p < 0.001) increased the soil and air temperatures (Table 2). The mean effects, during the three study years, on daily minimum air and soil temperatures were about 1°C, 0.5°C and 0.3°C, respectively at +20 cm (Ta), −10 cm (Ts10) and −20 cm (Ts20). Concerning air temperature, the greatest differences between warming and control were recorded in 2004, with the 2002 and 2003 effects being significantly weaker. The strongest effect on soil temperature at −10 cm occurred in 2002, when it was significantly higher than in 2003 and 2004. The mean effects on soil temperature at −20 cm did not different among the years.
The Drought treatment was deployed during two periods per year, as reported in the Materials & Methods section. The amounts of excluded precipitation (sum of the two periods) expressed as percentage of the annual value were 16%, 36% and 36% in the years 2002, 2003 and 2004, respectively (Table 3). The Drought treatment significantly reduced the relative soil water content when running, except during spring (first period) 2003, when only 2 mm of rain were excluded (Table 3). As a general pattern, the second period was characterized by greater precipitation compared to the first. The largest RSWC reductions occurred in autumn 2002 (−33%) and spring 2004 (−27%).
Soil CO2 efflux
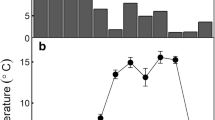
The seasonal variations of soil CO2 efflux, soil temperature and relative soil water content for the three treatments and for the three study years are presented in Fig. 1. During the entire observation period, soil CO2 effluxes (SR) ranged between 1.2 and 5.2 µmol CO2 m−2 s−1 in the control plots (Fig. 1). The highest values were recorded in spring, with soil temperatures (Ts) ranging from 20 to 30°C and relative soil water content (RSWC) between 20 and 50%. Generally, SR followed the same seasonal pattern of Ts when RSWC was above 20%. During late spring and summer, the SR decreased following the drop of RSWC (Fig. 1). The lowest fluxes were recorded in summer, when the RSWC reached the annual minimum (<6%), or during winter at temperatures around 12°C and RSWC close to saturation. CO2 efflux in summer (on average 1.3 µmol CO2 m−2 s−1), when RSWC was lower than 6%, was about 32% of the higher values recorded during the other seasons, at RSWC higher than 6%. After the summer minimum, the SR increased according to the variation of RSWC (autumn 2003 and 2004), even if limited by the contemporary reduction of soil temperature (Fig. 1).
Midday soil CO2 efflux, relative soil water content and soil temperature for the Control and Warming treatments during the study period. Control: full circles with continuous line. Warming: open circles with dashed line. a SR is the mean value per treatment; b RSWC is the soil water content relative to the annual maximum (saturation) at the time of SR measurements; c Ts is the soil temperature recorded at 5 cm depth by the temperature probe connected to the EGM. Error bars indicate standard errors of the mean (n = 3) for all three variables
The Warming treatment did not significantly affect the SR measured at any dates (Fig. 1) or the annual mean values measured at morning and at midday (Table 4).
The Drought treatment reduced the midday values of SR only in spring-summer 2004 (p < 0.01; Fig. 2), with no measurable effect in earlier sampling dates (data not shown). Even though the Drought treatment reduced RSWC only in April and May 2004 (p < 0.01), i.e. during the spring period of rain exclusion, a significant SR reduction was observed in May, June and July 2004 (Fig. 2). The decrease in May was about 30% (p < 0.05), in June about 37% (p < 0.001) and in July about 30% (p < 0.01). During summer, the Drought treatment was terminated and RSWC reached a common minimum value below 6% in both treatments. In contrast, no significant effects were recorded for the morning SR in the same period (2004 May-July; data not shown), even though no interaction between Drought treatment and daily RWC pattern was observed. Because the Drought treatment’s effect on SR occurred only in spring/summer and only on three occasions, the mean annual SRs measured at both morning and midday were not statistically different from the control (Table 4).
Midday soil CO2 efflux, relative soil water content and soil temperature for the Control and Drought treatments during the year 2004. Control: full circles with continuous line. Drought: open triangle with dashed line. a SR is the mean value per treatment; b RSWC is soil water content relative to the annual maximum (saturation) at the time of SR measurements; c Ts is the soil temperature recorded by the temperature probe connected to the EGM (n = 3). Error bars indicate standard errors of the mean (n = 3). The shaded areas indicate the period with rain exclusion
The inverse correlation between the seasonal variation of temperature and soil water content, typical for the Mediterranean area, prohibits fitting the relationship between SR and Ts on an annual basis by a simple exponential function. For this reason, we tested (but do not present the results) several two-variable models (Hanson et al. 1993; Xu and Qi 2001; Reichstein et al. 2002; Curiel Yuste et al. 2003; Joffre et al. 2003; Borken et al. 2006). The aim was to fit our measurements (mean plot values) over the entire period or over two seasons: wet vegetative season (RSWC > 6%) and dry non-vegetative season (RSWC < 6%). In these models the SR variation is estimated as a function of soil temperature and soil water content. Only the Hanson model (Hanson et al. 1993), on a two-season basis, fitted the data appropriately, with r2 between 0.5 and 0.9. The Rmax parameter, that is the maximum CO2 emissions without water limitation, varied between 2.9 µmol CO2 m−2 s−1 (control plots) and 3.1 µmol CO2 m−2 s−1 (both warming and drought plots) in the wet vegetative season; the treatments were not statistically different (Tab. 5). In the dry non-vegetative season, the mean Rmax was lower (p < 0.001) than in the wet season, without any significant effect of treatment. The Q10 factor, that is the SR sensitivity to temperature, averaged 1.4–1.5 in the wet season, with no statistical differences among treatments. In the dry season, the Q10 factor averaged 1.9–2.0, again with no statistical differences among treatments. Unexpectedly, this latter value was higher (p < 0.05) than that of the wet season (Table 5).
The effect of the progressive water limitation on soil CO2 efflux during the growing season is represented in Fig. 3a, which plots SR against Ts variation without water limitation (theoretical) or under the actual situation (wet vegetative season WVS). The theoretical curve was calculated using the parameterized Hanson model (overall average of parameters, Table 5) with the actual range of Ts variation and fixed, not limiting RSWC (45%), while the WVS was calculated with the actual range of Ts variation and with RSWC as a function of Ts, as shown in Fig. 3b. The effect of water limitation on SR was particularly strong under high temperatures (late spring). The curve NVS (not vegetative period—Fig. 3a) represents the SR for Ts changes, calculated as described above for WVS but with the equation parameters of the dry NVS (Table 5).
a SR-Ts relations estimated by Eq. 1, as an average of all treatments, reported in Table 5 column 4, for the wet vegetative season (WVS, continuous line), dry non-vegetative season (NVS, dotted line) and the theoretical curve for the vegetative period without water limitation (dashed line). b Relationship of relative soil water content (RSWC) with soil temperature (Ts) obtained with the data recorded on SR sampling occasions; RSWC = 714.6e^(−0.15*Ts); R2 = 0.61
The estimated cumulated annual CO2 emissions (SRcum) for the three study years and for the different treatments varied between 3.4 and 4.2 kg CO2 m−2 year−1 (Fig. 4a). The effects of the treatments on SRcum were never significant in any of the 3 years. The average value in 2004 was lower (p < 0.05) than those estimated in the two previous years. On a seasonal basis (Fig. 4b), mean winter emissions were statistically lower than in summer in the year 2002 and 2003 (p < 0.001), while no significant differences were estimated among the other months. Differently, in the year 2004, the summer emissions were lower than in the autumn (p < 0.001), while no significant differences were estimated among the other months.
Cumulated annual soil CO2 efflux (a) and seasonal soil CO2 efflux (b) for the 3 years of experimentation. Different lower case letters indicate significant differences (p < 0.05) among mean cumulative soil CO2 emissions of the 3 years. Different capital letters indicate significant differences (p < 0.05) among the seasons in the 3 years. Statistical differences were estimated by ANOVA. JFM: Winter; AMJ: Spring; JAS: Summer; OND: Autumn.White bar: control. Slanted bar: warming; gray bar: drought. Error bars are s.e. of the mean (n = 3)
Discussion
Several field and laboratory experiments have shown that soil respiration is stimulated by 25–45% when soil temperatures is increased by 0.6–6.0°C (Luo et al. 2001; Rustad et al. 2001; Melillo et al. 2002; Niinistö et al. 2004) during the first year of warming, and then acclimates to lower rates. This phenomenon is due to the depletion of the labile carbon pool, which readily responds to soil warming (Kirschbaum 2004; Gu et al. 2004; Eliasson et al. 2005; Knorr et al. 2005). The result is an apparent change of temperature sensitivity. On the other hand, Bradford et al. 2008) suggested that thermal adaptation can also be invoked to explain this phenomenon. This adaptation would involve a temperature-induced change of microbial communities. Such results were usually obtained in experiments run in temperate cold, boreal or arctic ecosystems, applying air/soil temperature increases of 3–6°C and where the soil organic layer was well developed. Two explanations can be forwarded for the unchanged CO2 emissions rates we report here for the Mediterranean ecosystem: the moderate increase of soil temperatures triggered by the treatment and limited to the nigh-time period (about +1.1°C for the air and about 0.5°C for soil at 10 cm depth), and/or the low contribution of the soil organic layer, which is patchy and practically absent in some parts. We can also exclude an effect of the treatment on the organic matter in the deeper soil layer, where it is present, because of the loss of the warming effect in the deepest layer (−20 cm depth). Additionally, the high C/N ratio and the arid and warm climate of the Mediterranean environment may result in very slow leaf litter decomposition rates (De Angelis et al. 2000; Moro and Domingo 2000; Fioretto et al. 2001; Zhang et al. 2008). At the same site, de Dato (unplublished) reported a C/N ratio of the Cistus monspeliensis (one of the two dominant species) leaf litter between 80 and 90 and a decomposition rate of 0.11–0.17 g dry biomass d−1. On the other hand, as typical in Mediterranean environments, litter is scattered, located in “island of fertility” (Schlesinger and Pilmanis 1998) under the vegetation cover, and this phenomenon can be related to the runoff typically occurring during the first rain events in autumn (intense rains). Under these conditions the main effect of the temperature increase is mediated by the response of the photosynthetic machinery that are not experiencing the warming treatment during the daytime.
Emmett et al. (2004), using a mobile cover as in this study, reported the impact of a prolonged drought on SR in different European shrublands. Similarly to our findings, they showed significant drought-related effects in both summer and winter, outside the extent of the periodic experimental drought, in their dry Spanish site. In our study, the SR reduction after prolonged drought was limited to springtime, when photosynthesis sustains soil respiration via highly labile fresh C. Therefore, the SR reduction can be an indirect effect of reduced photosynthesis, as shown for temperate and Mediterranean forests (Janssens et al. 2001; Högberg et al. 2001; Xu and Qi 2001; Rey et al. 2002; Curiel Yuste et al. 2003; Curiel Yuste et al. 2004), where the vegetative season spans from spring through summer. Reduced plant photosynthesis and more limited growth were also experimented in this and in similar experiments (Peñuelas et al. 2004; Peñuelas et al. 2007; Sardans et al. 2008; de Dato et al. 2008) in response to lower soil water availability induced by the drought treatment. Considering the leafless habit of the dominant species at our site during the dry non-vegetative season, the SR reduction might be mostly related to the absence of the rhizo-microbial component. In fact, even in late spring and/or early summer, when RSWC is still above the 6%, SR remained relatively high. Under these same climate conditions, plants still have green leaves and N mineralization rates are high (de Dato, unpublished). Part of this tolerance might also be related to the water lift by deep roots from deeper soil layers. Under these conditions, plants can also maintain the superficial roots in an active state (enabling rhizo-microbial respiration), even at very low soil water content (Caldwell et al. 1998; Lee et al. 2005).
The amplitude of SR reduction between the wet vegetative (RSWC > 6%) and dry non-vegetative season (RSWC < 6%) in our experiment was about 41%. Our reduction was lower than reported by Reichstein et al. (2003), that estimated for a wide set of ecosystems a SR reduction by 24%, 62%, and 76%, at RSWC contents of 50%, 10%, and 5%, respectively. Taking into account the deciduous habit of our community during summer, this lower reduction suggest a relative high resilience of the microbial community to water limitation. Additionally, the possible input of death fine root biomass during summer could have increased the substrates for the microbial activity, partially compensating the water limitation. Del Grosso et al. (2005) reported that the specific soil respiration rate (the M factor) fell significantly during the non-vegetative period in different natural ecosystems, independent of soil temperature and water content. Moreover, they found that, compared across different sites, the M factor tended to vary with NPP during the growing season and with SOC during the non-growing season. Accordingly, during the vegetative season respiration depends on fresh carbon supplied by plants, while during the non-growing season it is sustained by old carbon. Comparison with published data on partitioning of soil CO2 efflux in Mediterranean shrublands is hard, given the lack of soil CO2 efflux studies on these ecosystems (Subke et al. 2006). A quantitative estimation of the contribution of the two fractions was realised by Tang and Baldocchi (2005), in a natural Mediterranean oak-grass ecosystem. The authors estimated an heterotrophic/total respiration ratio in the range 39–41%, depending on the season.
On an annual basis, Emmett et al. (2004) found a good relationship between SR and Ts only when soil temperatures were in the range 0–20°C; over this threshold, water limitations masked the SR relationship with temperature, especially at the Spanish site. Similarly, other studies have reported no temperature dependence of SR at low water content (Bryla et al. 1997; Rey et al. 2002). On the other hand, other studies showed a reduced sensitivity at low water availability and high temperatures (Xu and Qi 2001; Janssens and Pilegaard 2003; Conant et al. 2004; Reichstein et al. 2005; Tedeschi et al. 2006; Sowerby et al. 2008).
Our results showed a significant change on the parameters of the Hanson model, in wet and dry seasons. The Rmax (the potential CO2 efflux without water limitation) reduction in the dry versus wet season (−78%) was more pronounced than the mean SR reduction (−60%). This is explained by the contemporary increase of the parameter Q10 (the SR sensitivity to temperature) of about 32% in the dry season. Knorr et al. (2005) showed that an altered temperature sensitivity of soil C efflux might reflect a shift in the carbon source, with a higher sensitivity when the SOC pool is the dominating source. Davidson and Janssens (2006), using an Arrhenius type model, also showed that a higher temperature sensitivity of soil respiration can be explained by the higher activation energy needed for the reactions of complex carbon molecules. Conversely, Fang et al. (2006) asserted that no experimental evidence sustains the underlying assumption of the Davidson and Janssens (2006) model, concluding no definitive demonstration of different sensitivities of different pools. Furthermore, Waldrop and Firestone (2006) reported a strong seasonal change in the soil microbial community composition of oak and open grassland soils, and Allen et al. (1995) and Zhang et al. (2005) documented changes in these communities in response to climatic changes and/or disturbances. In our experiment, both the carbon source and soil microbial community composition may well have changed: the deciduous habit of the plants during summer stopped the input of recently fixed carbon to the roots, which together with an increased fine root mortality (López et al. 1998) could have triggered a change in the active fraction of the microbial community.
The comparison of seasonal emissions, demonstrated compensation between the reduction of the instantaneous fluxes in summer with the increase of the hot hours in that season: the degree-sum of a typical day in summer was about twofold than in winter, but no in the year 2004.
Conclusion
In the semi-arid, water-limited Mediterranean shrubland, an increase of soil CO2 efflux in response to moderately increased daily minimum temperature is unlikely. On the other hand, less precipitation can affect the soil processes, especially if it occurred in spring, when plants are growing. The significant seasonal changes of the model parameters describing soil CO2 emission as a function of climatic factors, should be tested in different environment to take into account that the fraction of the different source of carbon can change on a seasonal basis, reflecting on temperature sensitivity. In particular, the SR response to temperature during the wet (vegetative) and dry (non-vegetative) seasons in semi-deciduous communities needs more deep investigation, under natural conditions.
A prolonged drought season may reduce soil CO2 emissions over the short term, but not on an annual basis because the compensation with the daily length and temperatures. On the other hand, the decrease of biomass accumulation in response to drought reduces the capacity of this ecosystem to remove CO2 from the atmosphere.
References
Allen MF, Morris SJ, Edwards F, Allen EB (1995) Microbe-plant interactions in Mediterranean-type habitats: shifts in fungal symbiontic and saprophytic functioning in response to Global Change. In: Moreno JM, Oechel WC (eds) Global changes and Mediterranean-type ecosystems. Springer-Verlag, New York, pp 287–303
Beier C, Emmett B, Gundersen P, Tietema A, Peñuelas J, Estiarte M, Gordon C, Gorissen A, Llorens L, Roda F, Williams D (2004) Novel approaches to Study climate change effects on terrestrial ecosystems in the field—drought and passive night time warming. Ecosystems (N Y, Print) 7:583–597. doi:10.1007/s10021-004-0178-8
Bekku YS, Nakatsubo T, Kume A, Adachi M, Koizumi H (2003) Effect of warming on the temperature dependence of soil respiration rate in arctic, temperate and tropical soils. Appl Soil Ecol 22:205–210. doi:10.1016/S0929-1393(02)00158-0
Borken W, Savage K, Davidson EA, Trumbore SE (2006) Effects of experimental drought on soil respiration and radiocarbon efflux from a temperate forest soil. Glob Change Biol 12:177–193. doi:10.1111/j.1365-2486.2005.001058.x
Bradford MA, Davies CA, Frey SD, Maddox TR, Melillo JM, Mohan JE, Reynolds JF, Treseder KK, WM D (2008) Thermal adaptation of soil microbial respiration to elevated temperature. Ecol Lett 11:1316–1327. doi:10.1111/j.1461-0248.2008.01251.x
Bryla DR, Bouma TJ, Eissenstat DM (1997) Root respiration in Citrus acclimates to temperature and slows during drought. Plant Cell Environ 20:1411–1420. doi:10.1046/j.1365-3040.1997.d01-36.x
Caldwell MM, Dawson TE, Richards JH (1998) Hydraulic lift: consequences of water efflux from the roots of plants. Oecologia 113:151–161. doi:10.1007/s004420050363
Christensen JH, Hewitson B, Busuioc A, Chen A, Gao XIH, Jones R, Kolli RK K, Kwon W-T, Laprise R, Magaña Rueda V, Mearns L, Menéndez CG, Räisänen J, Rinke A, Sarr A, Whetton P (2007) Regional climate projections. Climate Change 2007: The physical science basis contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, United Kingdom, and New York, NY, USA, pp 847–879
Conant RT, Klopatek JM, Malin RC, Klopatek CC (1998) Carbon pools and fluxes along an environmental gradient in northern Arizona. Biogeochem 43:43–61. doi:10.1023/A:1006004110637
Conant RT, Dalla-Betta P, Klopatek CC, Klopatek JM (2004) Controls on soil respiration in semiarid soils. Soil Biol Biochem 6:945–951. doi:10.1016/j.soilbio.2004.02.013
Cortez J (1997) Field decomposition of leaf litters: relationships between decomposition rates and soil moisture, soil temperature and earthworm activity. Soil Biol Biochem 30:783–793. doi:10.1016/S0038-0717(97)00163-6
Cubasch U, von Storch H, Waszkewitz J, Zorita E (1996) Estimates of climate change in Southern Europe derived from dynamical climate model output. Clim Res 7:129–149. doi:10.3354/cr007129
Curiel Yuste J, Janssens IA, Carrara A, Meiresonne L, Ceulemans R (2003) Interactive effects of temperature and precipitation on soil respiration in a temperate maritime forest pine. Tree Physiol 23:1263–1270
Curiel Yuste J, Janssens IA, Carrara A, Ceulemans R (2004) Annual Q10 of soil respiration reflects plant phenological patterns as well as temperature sensitivity. Glob Change Biol 10:161–169. doi:10.1111/j.1529-8817.2003.00727.x
Davidson EA, Janssens IA (2006) Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440:165–173. doi:10.1038/nature04514
De Angelis P, Chigwerewe KS, Scarascia-Mugnozza GE (2000) Litter quality and decomposition in a CO2—enriched Mediterranean forest ecosystem. Plant Soil 224:31–41. doi:10.1023/A:1004790328560
de Dato GD, Pellizzaro G, Cesaraccio C, Sirca C, De Angelis P, Duce P, Spano D, Scarascia Mugnozza G (2008) Effects of warmer and drier climate conditions on plant composition and biomass production in a Mediterranean shrubland community. iForest 39–48 doi: 10.3832/ifor0418-0010039
Del Grosso SJ, Parton WJ, Mosier AR, Holland EA, Pendall E, Schimel DS, Ojima DS (2005) Modeling soil CO2 emissions from ecosystems. Biogeochem 73:71–91. doi:10.1007/s10533-004-0898-z
Easterling DR, Horton B, Jones PD, Peterson TC, Karl TR, Parker DE, Salinger MJ, Razuvayev V, Plummer N, Jamason P, Folland CK (1997) Maximum and minimum temperature trends for the globe. Science 277:364–367. doi:10.1126/science.277.5324.364
Easterling DR, Karl TR, Gallo KP, Robinson DA, Trenberth KE, Dai A (2000) Observed climate variability and change of relevance to the biosphere. Geophys Res Lett 105
Eliasson PE, McMurtrie RE, Pepper DA, Strömgren M, Linder S, Ågren GI (2005) The response of heterotrophic CO2 flux to soil warming. Glob Change Biol 11:167–181. doi:10.1111/j.1365-2486.2004.00878.x
Emmett BA, Beier C, Estiarte M, Tietema A, Kristensen HL, Williams D, Peñuelas J, Schmidt I, Sowerby A (2004) The response of soil processes to climate change: results from manipulation studies across an environmental gradient. Ecosystems (N Y, Print) 7:625–637. doi:10.1007/s10021-004-0220-x
Fang C, Smith P, Smith JU (2006) Is resistant soil organic matter more sensitive to temperature than the labile organic matter? Biogeosci 3:65–68
FAO (2005) Global forest resources assessment 2005. Progress towards sustainable forest management. FAO, Rome
Fioretto A, Papa S, Sorrentino G, Fuggi A (2001) Decomposition of Cistus incanus leaf litter in a Mediterranean maquis ecosystem: mass loss, microbial activities and nutrient changes. Soil Biol Biochem 33:311–321. doi:10.1016/S0038-0717(00)00142-5
Gu L, Post WM, King AW (2004) Fast labile carbon turnover obscures sensitivity of heterotrophic respiration from soil to temperature: a model analysis. Global Biogeochem Cycles 18:GB1022. doi:10.1029/2003GB002119
Hanson PJ, Wullschleger SD, Bohlman SA, Todd DE (1993) Seasonal and topographic patterns of forest floor CO2 efflux from an upland oak forest. Tree Physiol 13:1–15
Högberg P, Nordgren A, Buchmann N, Taylor AFS, Ekblad A, Högberg MN, Nyberg G, Ottosson-Löfvenius M, Read DJ (2001) Large-scale forest girdling shows that current photosynthesis drives soil respiration. Nature 411:789–792. doi:10.1038/35081058
Janssens IA, Pilegaard K (2003) Large seasonal changes in Q10 of soil respiration in a beech forest. Glob Change Biol 9:911–918. doi:10.1046/j.1365-2486.2003.00636.x
Janssens IA, Lankreijer H, Matteucci G, Kowalski AS, Buchmann N, Epron D, Pilegaard K, Kutsch W, Longdoz B, Grünwald T, Montagnani L, Dore S, Rebmann C, Moors EJ, Grelle A, Rannik Ü, Morgenstern K, Oltchev S, Clement R, Gumundsson J, Minerbi S, Berbigier P, Ibrom A, Moncrieff J, Aubinet M, Bernhofer C, Jensen NO, Vesala T, Granier A, Schulze ED, Lindroth A, Dolman AJ, Jarvis PG, Ceulemans R, Valentini R (2001) Productivity overshadows temperature in determining soil and ecosystem respiration across European forests. Glob Change Biol 73:269–278. doi:10.1046/j.1365-2486.2001.00412.x
Joffre R, Ourcival J-M, Rambal S, Rocheteau A (2003) The key-role of topsoil moisture on CO2 efflux from a Mediterranean Quercus ilex forest. Ann Sci 60:519–526. doi:10.1051/forest:2003045
Kirschbaum MUF (2004) Soil respiration under prolonged soil warming: are rate reductions caused by acclimation or substrate loss? Glob Change Biol 10:1870–1877. doi:10.1111/j.1365-2486.2004.00852.x
Knorr W, Prentice IC, House JI, Holland EA (2005) Long-term sensitivity of soil carbon turnover to warming. Nature 433:298–301. doi:10.1038/nature03226
Kundzewicz ZW, Parry ML, Cramer W, Holten JI, Kaczmarek Z, Martens P, Nicholls RJ, Öquist M, Rounsevell MDA, Szolgay J (2001) Europe. In: Bullock PLHH, Hoffman MT, Rounsvell M, Seghal J, Várallyay G (eds) Climate change 2001: impacts, adaptation, and mitigation. Contribution of working group II to the third assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp 642–692
La Mantia T, Oddo G, Rühl J, Furnari G, Scalenghe R (2007) Variazione degli stock di carbonio del suolo in seguito ai processi di abbandono dei coltivi: il caso studio dell’isola di Pantelleria (TP). Forest 4:102–109. doi:10.3832/efor0433-0040102
Lee JE, Oliveira RS, Dawson TE, Fung I (2005) Root functioning modifies seasonal climate. Proc Natl Acad Sci USA 102:17576–17581. doi:10.1073/pnas.0508785102
Leiròs MC, Trasar-Cepeda C, Seoane S, Gil-Sotres F (1999) Dependence of mineralization of soil organic matter on temperature and moisture. Soil Biol Biochem 31:327–335. doi:10.1016/S0038-0717(98)00129-1
López B, Sabaté S, Gracia C (1998) Fine roots dynamics in a Mediterranean forest: effects of drought and stem density. Tree Physiol 8:601–606
Luo Y, Wan S, Hui D, Wallace LL (2001) Acclimatization of soil respiration to warming in a tallgrass prairie. Nature 413:622–625. doi:10.1038/35098065
Melillo JM, Steudler PA, Aber JD, Newkirk K, Lux H, Bowles FP, Catricala C, Magill A, Ahrens T, Morrisseau S (2002) Soil warming and carbon-cycle feedbacks to the climate system. Science 298:2173–2176. doi:10.1126/science.1074153
Moro MJ, Domingo F (2000) Litter decomposition in four woody species in a Mediterranean climate: weight loss, N and P dynamics. Ann Bot (Lond) 86:1065–1071. doi:10.1006/anbo.2000.1269
Niinistö SM, Silvola J, Kellomäki S (2004) Soil CO2 efflux in a boreal pine forest under atmospheric CO2 enrichment and air warming. Glob Change Biol 108:1363–1376. doi:10.1111/j.1365-2486.2004.00799.x
Oechel WC (2000) Acclimation of ecosystem CO2 exchange in the Alaskan Arctic in response to decadal climate warming. Nature 406:978–981. doi:10.1038/35023137
Peñuelas J, Gordon C, Llorens L, Nielsen T, Tietema A, Beier C, Bruna P, Emmett B, Estiarte M, Gorissen A, Williams D (2004) Non-intrusive field experiments show different plant responses to warming and drought among sites, seasons and species in a North-South European gradient. Ecosystems (N Y, Print) 7:598–612. doi:10.1007/s10021-004-0179-7
Peñuelas J, Prieto P, Beier C, Cesaraccio C, De Angelis P, de Dato G, Emmett BA, Estiarte M, Garadnai J, Gorissen A, Kovacs LE, Kroel-Dulay G, Llorens L, Pellizzaro G, Riis-Nielsen T, Inger KS, Sirca C, Sowerby A, Spano D, Tietema A (2007) Response of plant species richness and primary productivity in shrublands along a north-south gradient in Europe to seven years of experimental warming and drought: reductions in primary productivity in the heat and drought year of 2003. Glob Change Biol 13:2563–2581. doi:10.1111/j.1365-2486.2007.01464.x
Peterjohn WT, Melillo JM, Steudler PA, Newkirk KM, Bowles FP, Aber JD (1994) Responses of trace gas fluxes and N availability to experimentally elevated soil temperatures. Ecol Appl 4:617–625. doi:10.2307/1941962
Prentice IC, Farquhar GD, Goulden ML, Heimann M, Jaramillo VJ, Kheshgi HS, Le Quere C, Scholes RJ, Wallace DWR (2001) The carbon cycle and atmospheric carbon dioxide. In: Cubasch U, Meehl GA, Boer GJ, Stouffer RJ, Dix M, Noda A, Senior CA, Raper S, Yap KS (ed) Climate change 2001: The scientific basis. Contribution of working group I to the third assessment report of the intergovernmental panel on climate change. Cambridge University Press, pp.
Reichstein M, Tenhunen JD, Roupsard O, Ourcival J-M, Rambal S, Dore S, Valentini R (2002) Ecosystem respiration in two Mediterranean evergreen holm Oak forest: drought effects and decomposition dynamics. Funct Ecol 16:27–39. doi:10.1046/j.0269-8463.2001.00597.x
Reichstein M, Rey A, Freibauer A, Tenhunen J, Valentini R, Banza J, Casals P, Cheng Y, Grunzweig JM, Irvine J, Joffre R, Law BE, Loustau D, Miglietta F, Oechel W, Ourcival J-M, Pereira JS, Peressotti A, Ponti F, Qi Y, Rambal S, Rayment M, Romanya J, Rossi F, Tedeschi V, Tirone G, Xu M, Yakir D (2003) Modeling temporal and large-scale spatial variability of soil respiration from soil water availability, temperature and vegetation productivity indices. Global Biogeochem Cycles 17:1104. doi:10.1029/2003GB002035
Reichstein M, Subke J-A, Angeli AC, Tenhunen JD (2005) Does the temperature sensitivity of decomposition of soil organic matter depend upon water content, soil horizon, or incubation time? Glob Change Biol 11:1754–1767. doi:10.1111/j.1365-2486.2005.001010.x
Rey A, Pegoraro E, Tedeschi V, De Parri I, Jarvis PJ, Valentini R (2002) Annual variation in soil respiration and its components in a coppice oak forest in Central Italy. Glob Change Biol 8:851–866. doi:10.1046/j.1365-2486.2002.00521.x
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, Cornelissen JCH, Gurevitch J (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562. doi:10.1007/s004420000544
Sardans J, Peñuelas J, Estiarte M, Prieto P (2008) Warming and drought alter C and N concentration, allocation and accumulation in a Mediterranean shrubland. Glob Change Biol 14:2304–2316. doi:10.1111/j.1365-2486.2008.01656.x
Schimel DS (1995) Terrestrial ecosystem and the carbon cycle. Glob Change Biol 1:77–91. doi:10.1111/j.1365-2486.1995.tb00008.x
Schlesinger WH, Pilmanis AM (1998) Plant-soil Interactions in deserts. Biogeochem 42:169–187. doi:10.1023/A:1005939924434
Sowerby A, Emmett BA, Tietema A, Beier C (2008) Contrasting effects of repeated summer drought on soil carbon efflux in hydric and mesic heathland soils. Glob Change Biol 14:2388–2404. doi:10.1111/j.1365-2486.2008.01643.x
Subke J-A, Inglima I, Cotrufo MF (2006) Trends and methodological impacts in soil CO2 efflux partitioning: a metaanalytical review. Glob Change Biol 12:921–943. doi:10.1111/j.1365-2486.2006.01117.x
Tang J, Baldocchi DD (2005) Spatial–temporal variation in soil respiration in an oak–grass savanna ecosystem in California and its partitioning into autotrophic and heterotrophic components. Biogeochem 73:183–207. doi:10.1007/s10533-004-5889-6
Tedeschi V, Rey A, Manca G, Valentini R, Jarvis PG, Borghetti M (2006) Soil respiration in a Mediterranean oak forest at different developmental stages after coppicing. Glob Change Biol 12:110–121. doi:10.1111/j.1365-2486.2005.01081.x
USDA (1993) Soil survey manual. Washington D.C.
Vose RS, Easterling DR, Gleason B (2005) Maximum and minimum temperature trends for the globe: an update through 2004. Geophys Res Lett 32:L23822. doi:10.1029/2005GL024379
Waldrop M, Firestone M (2006) Seasonal dynamics of microbial community composition and function in oak canopy and open grassland soils. Microb Ecol 52:470–479. doi:10.1007/s00248-006-9100-6
Wohlfart G, Fenstermaker LF, Arnone JA III (2008) Large annual net ecosystem CO2 uptake of a Mojave Desert ecosystem. Glob Change Biol 14:1475–1487. doi:10.1111/j.1365-2486.2008.01593.x
Xu M, Qi Y (2001) Soil-surface CO2 efflux and its spatial and temporal variations in a young ponderosa pine plantation in Northern California. Glob Change Biol 7:667–677. doi:10.1046/j.1354-1013.2001.00435.x
Zhang W, Parker KM, Luo Y, Wan S, Wallace LL, Hu S (2005) Soil microbial responses to experimental warming and clipping in a tallgrass prairie. Glob Change Biol 11:266–277. doi:10.1111/j.1365-2486.2005.00902.x
Zhang D, Hui D, Luo Y, Zhou G (2008) Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. J Plant Ecol 1:85–93. doi:10.1093/jpe/rtn002
Acknowledgments
The research was funded by the EU under the project VULCAN (contract No. EVK2-CT-2000-00094). The authors wish to thank the anonymous reviewers for their useful and careful suggestions and comments. We would like to thank Mr. Oro and several PhD students of the University of Sassari for the fieldwork when the experimental installations were set up. We also would like to acknowledge the Regional Forest Service in the persons of Dr. Nudda, Dr. Caporali and Dr. D’Angelo for supporting this research. Finally, we acknowledge Mr. Bianchi and all the workers of the Regional Forest Service for their practical support and logistic help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Angela Hodge.
Rights and permissions
About this article
Cite this article
de Dato, G.D., De Angelis, P., Sirca, C. et al. Impact of drought and increasing temperatures on soil CO2 emissions in a Mediterranean shrubland (gariga). Plant Soil 327, 153–166 (2010). https://doi.org/10.1007/s11104-009-0041-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-009-0041-y