Abstract
At most sites the magnitude of soil-atmosphere exchange of nitrous dioxide (N2O), carbon dioxide (CO2) and methane (CH4) was estimated based on a few chambers located in a limited area. Topography has been demonstrated to influence the production and consumption of these gases in temperate ecosystems, but this aspect has often been ignored in tropical areas. In this study, we investigated spatial variability of the net fluxes of these gases along a 100 m long slope of a evergreen broadleaved forest in southern China over a whole year. We expected that the lower part of slope would release more N2O and CO2, but take up less atmospheric CH4 than the upper part due to different availability of water and nutrients. Our results showed that the soil moisture (Water Filled Pore Space, WFPS) decreased along the slope from bottom to top as we expected, but among the three gases only N2O emissions followed this pattern. Annual means of WFPS ranged from 27.7% to 52.7% within the slope, and annual emissions of N2O ranged from 2.0 to 4.4 kg N ha−1 year−1, respectively. These two variables were highly and positively correlated across the slope. Neither potential rates of net N mineralization and nitrification, nor N2O emissions in the laboratory incubated soils varied with slope positions. Soil CO2 release and CH4 uptake appeared to be independent on slope position in this study. Our results suggested that soil water content and associated N2O emissions are likely to be influenced by topography even in a short slope, which may need to be taken into account in field measurements and modelling.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Carbon dioxide (CO2), nitrous oxide (N2O) and methane (CH4) are the three main greenhouse gases (GHG) contributing to global warming (IPCC 2001). The increases in their atmospheric concentrations are attributed mainly to anthropogenic activities, such as deforestation, agricultural practices, and fossil fuels combustion. Besides, a considerable amount of atmospheric GHG is produced and consumed through soil processes (IPCC 2001). However, the large temporal and spatial variability of soil processes makes the accurate estimation and prediction of landscape soil–atmosphere exchange of these gases challenging, especially in tropical forests where relatively few sites have been monitored (Breuer et al. 2000; Werner et al. 2007). Furthermore, at most sites the magnitude was estimated based on a few chambers located in a limited area (Breuer et al. 2000; Tang et al. 2006), which very likely causes potential error when estimating regional gas fluxes between land and atmosphere by scaling up from small sampling units over heterogeneous areas (Reiners et al. 1998).
A range of environmental factors, such temperature and moisture, and soil properties have been identified to be controls of soil C and N cycling processes (Davidson et al. 2000; Corre et al. 2002; Saiz et al. 2006; Tang et al. 2006; Mo et al. 2008). These controls are, in turn, influenced by topography, through the movements of surface and subsurface water, nutrients and dissolved soil organic matter (Hairston and Grigal 1994; Hirobe et al. 1998; Hishi et al. 2004). Nitrogen concentrations in living leaves, fresh litter, litter-layer and soil upper layers were shown to be lower in the valley plots than in both slope and plateau plots in a central Amazonian forest (Luizao et al. 2004). At the walker Branch forest watershed (Tennessee, USA), it was shown that valley floors had greater potential net nitrification, and greater microbial activities (Garten et al. 1994). Within a slope of a plantation in Shiga prefecture of Japan, net nitrification and percent nitrification were high in the lower part and very low in the upper part of the slope, although net N mineralization showed no clear gradient (Hirobe et al. 1998). Generally speaking, compared to upper slope well-drained soils, lower slope poorly-drained soils have higher microbial respiration, N mineralization, net nitrification, microbial biomass N, denitrification and lower N immobilization (see Corre et al. 2002). In addition, soil texture and vegetation, that influence soil C and N cycles, can also be affected by topography (Luizao et al. 2004). It can thus be predicted that the patterns of soil C and N processes along a slope will inevitably affect those of soil–atmosphere exchange of GHG. This has been demonstrated by a number of studies in temperate ecosystems (Corre et al. 1996, 2002; Ambus 1998; Holst et al. 2008; Jungkunst et al. 2008; Yu et al. 2008), but in tropics the spatial variability in GHG efflux appeared to be often ignored (Reiners et al. 1998). In some tropical areas with distinct dry and wet seasons, there may be a different spatial pattern of trace gas exchanges between soil and atmosphere along the slope in different seasons.
In southern China, forests are mainly distributed in mountains and hills, which exhibit a large landscape variability. The magnitude, temporal, and spatial patterns of soil–atmospheric exchanges of greenhouse gases in forests of this region are in particular highly uncertain (Tang et al. 2006; Werner et al. 2006). In an old-growth broadleaf forest of this region, Tang et al. (2006) found a high soil N2O emission rate of 4.7 kg N ha−1 year−1, which is well above the averages of 1.2–1.4 kg N ha−1 year−1 estimated for tropical forests (Stehfest and Bouwman 2006; Werner et al. 2007) and is far higher than the rate (0.5 kg N ha−1 year−1) in a primary tropical forest in southwestern China (Werner et al. 2006). This high rate may be related to local high atmospheric N deposition (20–50 kg N ha−1 year−1, Fang et al. 2008a). We have also found elevated N leaching in soil water below the main rooting zone (67 kg N ha−1 year−1 including organic N, Fang et al. 2008a) in this forest. However, the N leaching in a small stream draining the catchment is much lower (17 kg N ha−1 year−1 including organic N, Fang et al. 2008b). We suspect that the reason for the reduction in N leaching from upslope soils to the stream would be due in part to denitrification (partially emitted as N2O) in the bottom of the catchment near the stream (Xiong et al. 2007; Ouyang et al. 2008).
In the present study, we investigated the spatial pattern of in situ soil–atmosphere exchange of N2O, CO2 and CH4 along a short and steep slope in an evergreen broadleaved forest in southern China over a whole year. At the end of field measurement, soils were taken to quantify the potential rates of net N mineralization and nitrification and these gas fluxes with laboratory incubation method. We hypothesized that soil water availability and soil N chemistry and thereby soil–atmosphere exchange of N2O, CO2, and CH4 would change with slope position, i.e. the lower part of slope would release more N2O and CO2, but take up less CH4 than the upper part.
Methods and materials
Site description
The study site is located in Dinghushan Biosphere Reserve (DHSBR) in the middle part of Guangdong province in southern China (112°33′ E and 23°10′ N). This forest area is representative of the dominant landscape type of vast areas in the region. The climate is warm and humid. The mean annual rainfall of 1,927 mm has a distinct seasonal pattern, with 75% falling from March to August and only 6% from December to February (Huang and Fan 1982). Mean annual relative humidity is 80% and mean annual temperature is 21.0°C, with average temperatures in the coolest month (January) and the hottest month (July) of 12.6°C and 28.0°C, respectively (Huang and Fan 1982). These forests have been exposed to high atmospheric N deposition of 20–50 kg N ha−1 year−1 in the last 15 years (Fang et al. 2008a).
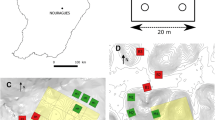
A short and steep slope was selected to conduct this study in a middle size forested catchment in August 2005. The slope is 100 m long from the stream to the ridge, and its slope is 15–35%, with an average of 29% (Fig. 1). This forest has been well protected since the establishment of the reserve in 1956. The major species are Castanopsis chinensis, Machilus chinensis, Schima superba, Cryptocarya chinensis, Syzygium rehderianum in the canopy and sub-canopy layers of this forest. The soil is lateritic red earth formed from sandstone (Oxisols). The pH value (H2O) in upper 10 cm mineral soil is 3.8 and the C/N ratio is 22 (Fang et al. 2008a).
Field measurements
Five sampling plots (5 m × 10 m) were set at 15–25 m long intervals on the slope at the beginning of this study, and were designated as bottom, middle 1, middle 2, middle 3 and top, respectively (Fig. 1). In each plot, three replicate chambers 1–2.5 m apart at similar elevation were anchored 5 cm into the soil permanently. Each chamber was a 25 cm diameter ring made of stainless-steel (Zhang et al. 2008a). In order to minimize the effect of tree distance (Butterbach-Bahl et al. 2002; Saiz et al. 2006), all chambers were at least 1.5 m away from stems. And plants inside ring were cut if any. At the beginning of this study, all living trees higher than 2 m within each plot were tagged, numbered, identified to species, and their height and diameter at breast height (DBH) were recorded.
Gases were collected monthly during the period from September 2005 to August 2006. During each flux measurement, a removable 35 cm high chamber top (made of stainless-steel) was attached to the ring. Gas samples were collected with 100 ml plastic syringes at 0 (time 0) and 30 min (time 1) after the chamber closure and analyzed for gas concentrations within 24 h using gas chromatography (Agilent 4890D, Agilent Co. USA, Tang et al. 2006) to calculate exchange rates (based on the difference in gas concentration between the time 0 and time 1). We did not sample gases in chamber at 10 min intervals during each measurement, as often found in other reports (Tang et al. 2006). This is because the previous study in an adjacent evergreen broadleaved forest showed air concentration in chambers at the same size as those we used, linearly increased within the first hour of field incubation (Tang et al. 2006).
The static chamber technique is known to underestimate gases production, like CO2 by about 10–15%, because the rising concentration within the chamber headspace, reduces the diffusion gradient within the soil (Pumpanen et al. 2004). Since we here focused on the comparison between slope positions this underestimation is of minor importance. Soil temperature and moisture at 5 cm below soil surface were recorded at each chamber at the beginning of each gas measurement. Soil temperature was measured using a digital thermometer. Volumetric soil moisture was measured simultaneously using a MPKit ((ICT, Australia). In this paper, these recorded soil moisture values were converted to WFPS (Water Filled Pore Space) by the following formula:
Where SBD is soil bulk density, Vol is volumetric water moisture and 2.65 is the density of quartz.
Laboratory incubation
All organic layer (above the mineral soil) within each chamber were collected immediately by hand after the last field measurement (August 2006), and then the mineral soils (0–10 cm depth) were sampled for soil bulk density determination and laboratory incubations, using a stainless steel corer (3 cm diameter). In laboratory, organic layer was dried and weighed; the mineral soils from each chamber were mixed thoroughly by hand removing fine roots and stones, and then were passed through a 2 mm mesh sieve.
Of the sieved mineral soil, four sub-samples of about 10 g from each chamber were taken to measure soil water content, water holding capacity (WHC), pH value and extractable inorganic N (NH4 + and NO3 −) concentration, respectively, and two replicate sub-samples of 80 g were adjusted soil water content to 60% of WHC and were then put into 30 PVC-containers of 1.2 L for further laboratory incubation. These containers were kept for 30 days in an air-conditioned room at 20°C. The air in each container was sampled five times over the incubation period (0, 7, 14, 21, and 30 days). Prior to each air sampling, the containers were opened for an hour and then sealed with screw-caps fitted with plastic tubes for 12 h. Air from the headspace of the containers was drawn out with 100 ml nylon syringes to analyze concentrations of GHG, using the same method described above. Air samples from five blanks without soils were considered as the initial condition. After each sampling the containers were covered with gas-permeable polyethylene until the next sampling. The incubated soils were kept at constant gravimetric moisture content throughout the incubation period by regular additions of distilled water. At the end of the month-long incubation, extractable soil inorganic N concentration was measured for each incubated container.
For measurement of soil extractable inorganic N, one 10 g mineral soil from each chamber/container was shaken for 1 h in 50 ml 1 mol L−1 KCl, and filtered through pre-leached Whatman no.1 filters. The NH4 + concentration in soil extracts was determined by the indophenol blue method followed by colorimetry, and the NO3 − concentration was determined after cadmium reduction to NO2 −, followed by sulfanilamide–NAD reaction (Liu et al. 1996). Soil pH was measured in deionized water suspension after shaking for 1 h at a ratio of 25 ml water to 10 g mineral soil, using a glass electrode (Liu et al. 1996).
Statistical analysis
Repeated measures ANOVA (RMANOVA) with Turkey’s HSD test was performed to examine the difference in soil temperature, soil moisture and gas fluxes among five slope positions for the study period from September 2005 to August 2006. One-way ANOVA was also performed for each sampling occasion. In order to examine the difference in spatial pattern of these variables along the slope between in dry season and wet season, we designated the period from October 2005 to February 2006 as dry-cool season and the rest of year as humid-warm season (Fig. 2) and performed two-way ANOVA (sampling date and slope position as main factors) for these two seasons separately. For soil variables (for in situ, annual means only) both one-way ANOVA and ANOVA with increasing distance from the bottom of the slope (broken down into orthogonal polynomial components) was used to identify the spatial pattern on the slope. Single correlation analysis was used to examine the relations between soil variables. For the relationship between in situ gas fluxes, soil temperature and soil moisture, both linear and nonlinear regression models (Tang et al. 2006; Mo et al. 2008) were further examined and the best-fitted regressions were chosen in terms of correlation coefficients. All analyses were conducted using SPSS 10.0 for Windows. Statistical significant differences were set at P values <0.05 unless otherwise stated.
Results
Precipitation and air temperature
The data from the weather station in the reserve showed that there was a prolonged drought from October to December 2005 during the measuring campaign (Fig. 2), but annual precipitation (1,880 mm) was close to the long-term average of 1,927 mm (Huang and Fan 1982). Only 118 mm or 6% of annual precipitation fell in the dry-cool season (October 2005 to February 2006). Also the annual mean temperature 21.7°C, as well as the monthly temperature range from 12.1°C (December 2005) to 28.9°C (July 2006) (Fig. 2), were close to the long-term averages (Huang and Fan 1982).
Stand characteristics
The density was on average 1,580 stems ha−1 for the trees with height over 2 m (Table 1). The mean height was 6.9–10.5 m, and mean DBH was 10.6 to 19.2 cm. The basal area at breast height varied greatly from 20 to 62 m2 ha−1 within the slope, with a total mean of 38 m2 ha−1. There were no clear gradients for these measurements along the slope. However, the amount of organic soil layer, ranging from 2.6 to 4.3 Mg ha−1, appeared to increase with the slope from bottom to top (Table 1).
Soil characteristics
Soil temperature and moisture exhibited clear seasonal courses, and generally followed those of air temperature and precipitation (Fig. 2). Mean soil temperature across the slope ranged from 14.5°C to 30.3°C, and mean soil moisture (WFPS) ranged from 18.4% to 70.9%. The soil temperature varied significantly between slope positions across the observation period, although the absolute differences were very small (Fig. 2). The differences were statistically significant at most sampling dates and thus also on the seasonal means and annual means (Fig. 2 and Table 2). Soil moisture was significantly different by the slope positions almost throughout the whole year (Fig. 2). Annual mean moisture over the study period ranged from 27.7% to 52.7% on the slope, which was within that of seasonal variation (18.4% to 70.9%). The highest moisture was not located in the lowest part of the slope, but in the middle part, although they were not statistically significant (Figs. 2 and 3). The driest part was observed in the top of the slope as expected (Fig. 3). Soil moisture differed with slope position stronger in the dry-cool season than the humid-warm season (Fig. 3).
At the end of the field measurement campaign, the upper 10 cm mineral soil from all chambers was collected for property analysis. The results showed that soil bulk density was not significantly different by the slope position (Table 3). The soils were strongly acidic, with pH values being 3.63–3.82. The highest pH value was found in the bottom of the slope. Concentrations of total extractable inorganic N (NH4 ++NO3 −) were similar between the slope positions. However, extractable NH4 + concentration was highest in the top of the slope and lowest in the bottom, and it increased significantly with the distance from the slope bottom to top (Table 3). The reverse was observed for extractable NO3 − concentration (Table 3).
In situ soil–atmosphere exchange of N2O, CO2, and CH4
Emission rates of N2O and CO2 were significantly lower in the dry-cool season where there was a 3-month long drought period, than in the humid-warm season (Figs. 2 and 3). Over the slope, emission rates of N2O and CO2 were 68% and 98% higher in humid-warm season, respectively (Fig. 3). These patterns well agreed with those of soil temperature and moisture, as evidenced by significant correlations between these variables across the slope (all P < 0.001, n = 180, Fig. 4). Higher emission rates occurred in soils with temperature 25–30°C and with WFPS 35–65% (Fig. 4). At most sampling times, CH4 was consumed by the soil, but no obvious seasonal trend and thereby no dependency on soil temperature and moisture were found (Figs. 2 and 3).
Soil N2O emissions (4.4–111.7 μg N m−2 h−1) were significantly affected by slope position on three out of 12 sampling dates (Fig. 2), and RMANOVA over the study period showed that soil N2O emissions were significantly different by slope position (P = 0.005). The effect of slope position on N2O emissions was smaller in the dry-cool months than in the humid-warm months (Figs. 2 and 3). Soil N2O emissions exhibited a substantial spatial variability with a range from 22.6 to 50.6 μg N m−2 h−1 over the study period. This range was slightly narrower than the temporal variation of 12.1 to 69.9 μg N m−2 h−1. The highest emission rate was not located in the bottom of the slope. The spatial pattern of N2O emission rates along the slope followed that of soil moisture from bottom to top (Fig. 3, Table 2).
The topographic influence on soil CO2 emissions was not clear in humid-warm season, but significant lowest values were observed on the top position in dry-cool season (Fig. 3). Over the study period, RMANOVA showed the topographic influence was not significant (Fig. 3 and Table 2). The difference of maximum and minimum soil CO2 emissions on the slope was similar between the two seasons (Fig. 3). No significant difference in CH4 uptake was found among slope position, in any season or over the whole study year (Fig. 3 and Table 2), but soils appeared to take up more atmospheric CH4 in dry-cool season than in humid-warm season (Fig. 3).
When linking annual fluxes of three gases to annual means of soil temperature and moisture across the entire slope, we found that annual N2O emissions were correlated significantly (P = 0.01) with soil temperature and marginally (P = 0.08) with soil moisture (Fig. 5). But no such correlations were found for either soil CO2 emissions or CH4 uptake.
The pH and concentrations of extractable NH4 +–N and NO3 −–N in the top 10 cm mineral soil, were measured on the last sampling date. We related these variables to in situ soil N2O and CO2 emission and CH4 uptake on that sampling date, and found that soil N2O emissions were marginally correlated with concentrations of extractable NO3 −–N (positively, P = 0.06) and NH4 +–N (negatively, P = 0.08). Soil N2O emissions were not correlated with soil temperature or moisture on that sampling date. Soil CO2 emissions were significantly (P = 0.02) correlated with soil pH values only. Soil CH4 uptake was not influenced by any soil variable.
Potential rates of soil N transformations and fluxes of N2O, CO2, and CH4
Monthly rate of potential net N mineralization ranged from 15.5 to 21.1 mg N kg dry soil−1 and all mineralized N was nitrified (Table 4). Neither potential net N mineralization nor nitrification was found to be different by the slope position (Table 4). Nor were accumulative emissions of N2O or CO2 from incubated soils in laboratory. In contrast, the ability to oxide CH4 appeared to decrease from the bottom to the top (Table 4). There were no significant correlations between mean in situ and potential fluxes for any of the three gases. Potential N2O emission or CH4 uptake showed no significant correlations with soil extractable N or with N mineralization and nitrification in the laboratory incubation (Table 4). However, we found good relationships between potential CO2 fluxes and N mineralization and nitrification (P = 0.029 and 0.012, respectively).
Discussion
Annual soil N2O emissions from this study forest are 2.0–4.4 kg N ha−1 year−1, with a mean of 3.0 kg N ha−1 year−1, and annual CO2 are 5.4–9.0 Mg C ha−1 year−1, with a mean of 6.2 Mg C ha−1 year−1 across the slope (Fig. 5). These values are somewhat smaller than the observations from the nearby broadleaved forests which have been protected for more than 400 years (on average, 4.7 kg N ha−1 year−1 and 9.9 Mg C ha−1 year−1, Tang et al. 2006). Likewise, the soil CH4 uptake is smaller in our study forest (−4.3–0.4 kg C ha−1 year−1, on average −1.9 kg C ha−1 year−1) than in that old forest (−7.8 kg C ha−1 year−1, Tang et al. 2006). This could be because we studied a young forest (50 years old), where a stronger N utilization by vegetation and soil organisms are expected than in that old forest. However, our results are slightly higher than those in the adjacent mixed and the pine forests, where N2O emissions were measured to be 2.1–2.7 kg N ha−1 year−1 (Tang et al. 2006; Zhang et al. 2008a). On the other hand, the seasonal pattern of soil variables were observed in the study (Figs. 2 and 3) similarly as the previous reports (Tang et al. 2006; Mo et al. 2008; Zhang et al. 2008a, b).
Riparian zone or wetland area situated at the interface of terrestrial and aquatic environment, have long been identified to be “hotspots” of N2O production in the landscape (Groffman et al. 1998; Hefting et al. 2006). In the present study, the bottom position which was 1.5 m away from the stream, can also be considered to be such a kind of area. Our results did show an increase in N2O emission rates in this part of the slope (Fig. 3), the soil N2O emission rate at the bottom position (6.7–77.9 μg N m−2 h−1, Fig. 2) is, however, much smaller than those reported for temperate riparian forest soils (up to 9,000 μg N m−2 h−1, Hefting et al. 2006). This could be due in part to our bottom position locating by a periodically stream within a small catchment (<10 ha) and low soil water content (SWFP on average 45%, Fig. 3). Similarly, this absolute increase (top to bottom difference 2.2 kg N ha−1 year−1) is too small to explain the observed difference in the leaching N flux below the main rooting zone (67 kg N ha−1 year−1, Fang et al. 2008a) and in streamwater (17 kg N ha−1 year−1, Fang et al. 2008b). On the other hand, this result suggests that other mechanisms, such as plant uptake, sorption, desorption, and microbial decomposition in deeper soil layers (Fang et al. 2008b), the emissions of NO (Li et al. 2007) and N2, might be of importance in the reduction in N leaching from upslope to the stream. Additional N removal, like N uptake and denitrification in stream water might also occur after the water exported from soils but before was sampled for chemical analysis.
Soil moisture varied greatly with the slope position, and it decreased in the upper part of the slope as we expected. But the wettest area was not located in the bottom of the slope (the lowest part), and soil water content showed quadratic relationship with slope position over the study year with being more pronounced in dry-cool season (Fig. 3). Among the three gases we investigated, however, only N2O emissions followed the pattern of soil moisture along the slope (Fig. 3). Soil N2O emissions exhibited a substantial spatial variability, with a range being slightly narrower than the temporal variation. This result indicates that the effect of slope position and associated soil moisture on soil N2O emissions is comparable to that of season in the study forest. However, we observed decreased soil N2O emissions on the middle 2 position where the soils were the wettest. One of explanations is that high denitrification activity occurred on this position, by which a large fraction of N2O produced was reduced to N2 or NO. This is consistent with the fact that higher N2O emission rate take place in soils with WFPS of 35–65%, not over 70% (Fig. 4). Low root and/or microbial activity indicated by low soil respiration can also in part account for the decreased N2O emission therein (Fig. 3). Both nitrification and denitrification can produce N2O, but the present study does not allow us to distinguish their respective contribution. Thus further research need to address which process is the main source of N2O in the study soil and if it varies with slope position.
The measurement of extractable NO3 − and NH4 + in the upper 10 cm mineral soil at the end of sampling period can provide a snap-shot index of the relative sized of N availability and N processes as shaped by topographic conditions within the slope. Extractable NO3 − concentration was significantly higher in the lower part of the slope than in the upper, and the reverse was true for extractable NH4 + concentration (Table 3). The percentage of NO3 − of the total extractable inorganic N decreased from 61–82% in the bottom and middle 1 of the slope to 24–46% in the top (Table 3). The soil N2O emissions on the same sampling date were found to positively relate to the concurrent extractable NO3 − concentration (P = 0.06). These results indicated that the soils might have a stronger nitrification process in the lower part than in the upper part, during which N2O was produced. More NO3 − concentration in the lower part also meant that there was more NO3 − source therein to fuel the denitrifier and thereby promote denitrification in combination with higher soil water content (lower oxygen concentration).
There were no differences in the potential rates of net N mineralization and nitrification, and cumulative N2O emissions as well, across the slope position in the laboratory incubation where moisture was kept constant. The vegetation was not shown to differ significantly along this short and steep slope either (Table 1). Soil temperature decreased from the top to the bottom of the slope, but the difference was very small (Fig. 3). The observed negative correlation between soil temperature and N2O emissions (Fig. 5), opposite to the normal effect of temperature on microbial processes, was due to the covariant soil temperature and moisture over the gradient. We can therefore conclude that the difference in in situ soil N2O emissions along the slope may mainly result from the environmental control, soil water availability in field.
In this study, no clear gradient along the slope was found for soil CO2 emission over the whole study year, although slope position has a significant influence in dry-cool season (Fig. 3). It indicated that soil CO2 emissions might be influenced by other factors in addition to soil water, such as amount of organic materials on the floor (Table 1), root biomass and soil depth (the latter two were not measured in this study). In situ soil CH4 uptake was not significantly influenced by slope position either (Figs. 2 and 3), despite that the ability to oxide atmosphere CH4 in the laboratory incubation appeared to be stronger in the soil from the lower part compared to that from upper part (Table 4).
The wettest part was not observed in the bottom of the slope in our study. This might be related to soil texture, bedrock depth, water flow pattern and organic matter content. At the bottom of the slope close to the stream, the soil was well drained, because the stream channel was about 0.5 m deep. Some organic materials, a key component holding moisture and providing fuels for microbes including nitrifying and denitrifying bacteria, in this part of the slope is likely to be taken away by seasonal floodings, which may also explain partially why the amount of organic layer was the lowest in this part of the slope (Table 1). Similar trend of soil moisture was found in a 108 m long slope in a Japanese forest (Hirobe et al. 1998).
In conclusion, this study demonstrated that soil water content and N2O emission rates were significantly different by slope position even within a short and steep slope. Annual soil N2O emission was highly related to mean soil moisture across the slope. No clear trends were observed for soil CO2 release and CH4 uptake in this study. Our results nonetheless indicated that soil water content and associated soil N2O emissions are likely to be influenced by topography, with the spatial variation by slope positions being comparable to the temporal variation by seasons. This may need to be taken into account in field measurements and modelling.
References
Ambus P (1998) Nitrous oxide production by denitrification and nitrification in temperate forest, grassland and agriculture soils. Eur J Soil Sci 49:495–502 doi:10.1046/j.1365-2389.1998.4930495.x
Breuer L, Panen H, Butterbach-Bahl K (2000) N2O emission from tropical forest soils of Australia. J Geophys Res 105(D21):26353–26367 doi:10.1029/2000JD900424
Butterbach-Bahl K, Rothe A, Papen H (2002) Effect of tree distance on N2O and CH4-fluxes from soils in temperate forest ecosystems. Plant Soil 240:91–103 doi:10.1023/A:1015828701885
Corre MD, van Kessel C, Pennock DJ (1996) Landscape and seasonal patterns of nitrous oxide emissions in a semiarid region. Soil Sci Soc Am J 60:1806–1815
Corre MD, Schnabel RR, Stout WL (2002) Spatial and seasonal variation of gross nitrogen transformations and microbial biomass in a Northeastern US grassland. Soil Biol Biochem 34:445–457 doi:10.1016/S0038-0717(01)00198-5
Davidson EA, Verchot LV, Cattânio JH, Ackerman IL, Carvalho JEM (2000) Effects of soil water content on soil respiration in forests and cattle pastures of eastern Amazonia. Biogeochemistry 48:53–69 doi:10.1023/A:1006204113917
Fang YT, Gundersen P, Mo JM, Zhu WX (2008a) Input and output of dissolved organic and inorganic nitrogen in subtropical forests of South China under high air pollution. Biogeosciences 5:339–352
Fang Y, Zhu W, Gundersen P, Mo J, Zhou G, Yoh M (2008b) Large loss of dissolved organic nitrogen in nitrogen-saturated forests in subtropical China. Ecosystems (N Y, Print). doi:10.1007/s10021-008-9203-7
Garten CT Jr, Huston MA, Thoms CA (1994) Topographic variation of soil nitrogen dynamics at Walker Branch watershed, Tennessee. For Sci 40:497–512
Groffman PM, Gold AJ, Jacinthe PA (1998) Nitrous oxide production in riparian zones and groundwater. Nutr Cycl Agroecosyst 52:179–186 doi:10.1023/A:1009719923861
Hairston AB, Grigal DF (1994) Topographic variation in soil water and nitrogen for two forested landforms in Minnesota, USA. Geoderma 64:125–138 doi:10.1016/0016-7061(94)90093-0
Hefting MM, Bobbink R, Janssen M (2006) Spatial variation in denitrification and N2O emission in relation to nitrate removal efficiency in a N-stressed riparian buffer zone. Ecosystems (N Y, Print) 9:55–563 doi:10.1007/s10021-006-0160-8
Hirobe M, Tokuchi N, Iwatsubo G (1998) Spatial variability of soil nitrogen transformation patterns along a forest slope in a Cryptomeria japonica D. Don plantation. Eur J Soil Biol 34:123–131 doi:10.1016/S1164-5563(00)88649-5
Hishi T, Hirobe M, Tateno R, Takeda H (2004) Spatial and temporal patterns of water-extractable organic carbon (WEOC) of surface mineral soil in a cool temperate forest ecosystem. Soil Biol Biochem 36:1731–1737 doi:10.1016/j.soilbio.2004.04.030
Holst J, Liu C, Yao Z, Brüggemann N, Zheng X, Han X, Butterbach-Bahl K (2008) Importance of point sources on regional nitrous oxide fluxes in semi-arid steppe of Inner Mongolia, China. Plant Soil 296:209–226 doi:10.1007/s11104-007-9311-8
Huang ZF, Fan ZG (1982) The climate of Ding Hu Shan. Trop Subtrop For Ecosyst 1:11–23 in Chinese with English abstract
IPCC (2001) Third assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Jungkunst HF, Flessa H, Scherber C, Fiedler S (2008) Groundwater level controls CO2, N2O and CH4 fluxes of three different hydromorphic soil types of a temperate forest ecosystem. Soil Biol Biochem 40:2047–2054 doi:10.1016/j.soilbio.2008.04.015
Li DJ, Wang XM, Mo JM, Sheng GY, Fu JM (2007) Soil nitric oxide emissions from two subtropical humid forests in south China. J Geophys Res 112:D23302 doi:10.1029/2007JD008680
Liu GS, Jiang NH, Zhang LD (1996) Soil physical and chemical analysis and description of soil profiles. Standards Press of China, Beijing (in Chinese)
Luizao RCC, Luizao FJ, Paiva RQ, Monteiro TF, Sousa LS, Kruijt B (2004) Variation of carbon and nitrogen cycling processes along a topographic gradient in a central Amazonian forest. Glob Change Biol 10:592–600 doi:10.1111/j.1529-8817.2003.00757.x
Mo JM, Zhang W, Zhu WX, Gundersen P, Fang YT, Li DJ, Wang H (2008) Nitrogen addition reduces soil respiration in a mature tropical forest in southern China. Glob Change Biol 14:403–412
Ouyang XJ, Zhou Gy, Huang ZL, Liu JX, Zhang DQ, Li J (2008) Effect of simulated acid rain on potential carbon and nitrogen mineralization in forest soils. Pedosphere 18(4):503–514
Pumpanen J, Kolari P, Ilvesniemi H, Minkkinen K, Vesala T, Niinistö S, Lohila A, Larmola T, Morero M, Pihlatie M, Janssens I, Yuste JC, Grünzweig JM, Reth S, Subke JA, Savage K, Kutsch W, Østreng G, Ziegler W, Anthoni P, Lindroth A, Hari P (2004) Comparison of different chamber techniques for measuring soil CO2 efflux. Agric For Meteorol 123:159–176 doi:10.1016/j.agrformet.2003.12.001
Reiners WA, Keller M, Gerow KG (1998) Estimating rainy season nitrous oxide and methane fluxes across forest and pasture landscapes in Costa Rica. Water Air Soil Pollut 105:117–130 doi:10.1023/A:1005078214585
Saiz G, Green C, Butterbach-Bahl K, Kiese R, Avitabile V, Farrell EP (2006) Seasonal and Spatial variability of soil respiration in four Sitka spruce stands. Plant Soil 287:161–176 doi:10.1007/s11104-006-9052-0
Stehfest E, Bouwman L (2006) N2O and NO emission from agricultural fields and soils under natural vegetation: summarizing available measurement data and modeling of global annual emissions. Nutr Cycl Agroecosyst 74:207–228 doi:10.1007/s10705-006-9000-7
Tang XL, Liu SG, Zhou GY, Zhang DQ, Zhou CY (2006) Soil–atmospheric exchange of CO2, CH4, and N2O in three subtropical forest ecosystems in southern China. Glob Change Biol 12:546–560 doi:10.1111/j.1365-2486.2006.01109.x
Werner C, Zheng X, Tang J, Xie B, Liu C, Kiese R, Butterbach-Bahl K (2006) N2O, CH4 and CO2 emissions from seasonal tropical rainforests and a rubber plantation in Southwest China. Plant Soil 289:335–353 doi:10.1007/s11104-006-9143-y
Werner C, Butterbach-Bahl K, Haas E, Hickler T, Kiese R (2007) A global inventory of N2O emissions from tropical rainforest soils using a detailed biogeochemical model. Global Biogeochem Cycles 21:GB3010 doi:10.1029/2006GB002909
Xiong ZQ, Xing GX, Zhu ZL (2007) Nitrous oxide and methane emissions as affected by water, soil and nitrogen. Pedosphere 17(2):146–155
Yu KW, Faulkner SP, Baldwin MJ (2008) Effect of hydrological conditions on nitrous oxide, methane, and carbon dioxide dynamics in a bottomland hardwood forest and its implication for soil carbon sequestration. Glob Change Biol 14:1–15 doi:10.1111/j.1365-2486.2008.01545.x
Zhang W, Mo JM, Yu GR, Fang YT, Li DJ, Lu XK, Wang H (2008a) Emissions of nitrous oxide from three tropical forests in Southern China in response to simulated nitrogen deposition. Plant Soil 306:221–236 doi:10.1007/s11104-008-9575-7
Zhang W, Mo JM, Zhou GY, Gundersen P, Fang YT, Lu XK, Zhang T, Dong SF (2008b) Methane uptake responses to nitrogen deposition in three tropical forests in southern China. J Geophys Res 113:D11116 doi:10.1029/2007JD009195
Acknowledgements
This study was funded by the National Natural Science Foundation of China (Nos. 30725006, 40703030, 30670392), Postdoctoral Fellowship of South China Botanical Garden, the Chinese Academy of Sciences (200611), and the NitroEurope IP (EU-contract No. 17841). We thank X. M. Fang and others for their kind assistances in the field and laboratory. Comments from two anonymous reviewers have greatly improved the quality of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Per Ambus.
Rights and permissions
About this article
Cite this article
Fang, Y., Gundersen, P., Zhang, W. et al. Soil–atmosphere exchange of N2O, CO2 and CH4 along a slope of an evergreen broad-leaved forest in southern China. Plant Soil 319, 37–48 (2009). https://doi.org/10.1007/s11104-008-9847-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-008-9847-2