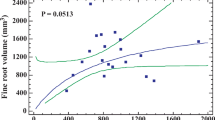
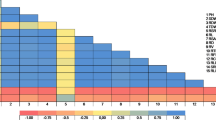
Abstract
Hemp (Cannabis sativa L.) is mainly grown for its fibre and is considered a desirable crop for sustainable production systems. In a field trial carried out over two years in Northern Italy the root system of a hemp crop, cultivated at contrasting plant densities, was sampled and analysed with an image analysis software. Root length density (RLD) was highest in the first 10 cm of soil, almost 5 cm cm−3; it decreased progressively until the depth of 130 cm, a part from a peak at 90–100 cm in response to a perched water table. Roots were found to 130 cm of depth in one year and to 200 cm in the other. Root diameter was finer (190 μm) in the upper soil layer, it increased with depth until 100 cm, and remained constant at 300 μm thereafter. Following the same trend of RLD, root biomass was highest in the first soil layer; 50% of the root biomass was found in the first 20 cm or 50 cm when taproot biomass was considered or not. Total root biomass was 3.21 t ha−1 and 2.41 t ha−1 in the two years of trial, but the ratio between aboveground and below ground biomass was constant at 5.46. None of the root parameters were significantly affected by plant population, which seems to confirm the plastic behaviour that hemp shows for aboveground development. The high root biomass production measured in this study, especially in deeper soil layers, provides additional evidence of the positive role that hemp can play in sustainable cropping systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The study of the root system is needed to understand a crop’s capacity to explore the soil, and take up water and nutrients (Barber 1995). Plant roots constitute a major source of organic matter when decomposed, and while growing are capable of both creating and stabilizing useful soil structural features (Cochrane and Aylmore 1994) depending on soil type, environmental factors and cultivated species (Monroe and Kladivko 1987).
Research on plant root systems under field conditions is difficult (Cheng et al. 1990) and the traditional methods (Newman 1966; Tennant 1975) to estimate the root length were generally laborious and very time consuming. In recent years different methods to estimate root length, root diameter and root biomass have been studied. Fast procedures and accurate analysis of digital images have been developed (Bouma et al. 2000; Costa et al. 2000; Himmelbauer et al. 2004; Vamerali et al. 2003a).
Determination of root parameters along soil profile is useful to understand and simulate root development. Root simulation models have been developed for annual crops embracing a wide range of technical approaches (Gregory 2006a) and root parameters.
Root Length Density (RLD, cm of length per cm−3 of soil) is the most frequently estimated parameter to determine root morphology (Vamerali et al. 2003b), the crop’s potential for nutrient and water uptake (Qin et al. 2006). This parameter is used in the root growth models of Kage et al. (2000) and Robertson et al. (1993). RLD is influenced by soil compaction (Amato and Ritchie 2002), tillage (Chassot et al. 2001), bulk density (De Freitas et al. 1999) and weather (Qin et al. 2004).
Root diameter (RD) is another important trait of the root system that again effects nutrient and water uptake (Qin et al. 2006; Gregory 2006a) and is affected by soil tillage (Holanda et al. 1998; Qin et al. 2004). Root diameter is also linked with root longevity (Eissenstat et al. 2000) and the formation of continuous pores (Passioura 2002). RD is one of the most important input parameters for rhizosphere modelling (Himmelbauer et al. 2004).
Root Biomass (RB) is an important parameter to determine the costs associated with root construction and root maintenance (Bouma et al. 2000; Veen 1980).
Field trials were carried out to evaluate the effect of agronomic treatments on hemp yield and to calibrate and validate a growth model to be used as a basis for a decision support system (Amaducci 2003; Amaducci et al. 2008). During 2004 and 2005, field trials were carried out to study the effect of plant population on various crop parameters. Considering that no information is available on hemp root development, on the same field trials root sampling was carried out. The objective of this paper is to present the characteristics of hemp roots as influenced by growing conditions so to provide data for crop modelling and to evaluate root biomass left in the soil after harvesting. The latter aspect is important to evaluate the potential of hemp as a sustainable crop, in particular regarding its contribution to increase carbon sink into the soil. Hemp is considered a sustainable crop for energy production (Biewinga and Bijl 1996), it is a good precedent crop for wheat productivity (Bocsa and Karus 1998; Gorchs et al. 2000) and it efficiently suppresses weeds without the need for chemical treatments (Lotz et al. 1991; Berger 1969). The sustainability of the whole production chain, from cultivation to the realisation of end products, is a challenging target in agriculture (Kirchmann and Thorvaldsson 2000). Hemp fibre production is close to achieving this target provided that CO2 emissions created during fibre processing are reduced (van der Werf and Turunen 2008) or compensated for by the CO2 assimilated and stored by the plant (i.e. in the fibre and in the roots).
Materials and methods
Field setting
Field trials were carried out in 2004 and 2005 at the experimental farm of the University of Bologna, in Cadriano (32 m a.s.l., 44° 33′ N, 11° 21′ E). The experimental lay out was a completely randomized block design with three target densities (120, 240, 360 plants m−2) replicated three times. In both years nitrogen fertilisation was applied before sowing at a rate of 60 kg ha−1, which proved to be the optimal dose in the area where the experiments were carried out (Amaducci et al. 2002). Sowing was carried out with an experimental machine (Vignoli) on 8th April in 2004 and 6th April in 2005. Inter-row distance was 13 cm and the genotype was the monoecious Futura 75. In both years the preceding crops was wheat and the soil had been ploughed at 35 cm during the summer after wheat harvest and then harrowed before sowing for seed bed preparation.
Soil samplings were carried out in both years at the end of flowering (cod. 2305, Mediavilla et al. 1998) on 28 July in 2004 and 14 July in 2005. Two soil samples per plot 130 cm total depth were taken placing the auger (a self constructed “Shelby” tube sampler) in the inter-row and pressing it with the hydraulic arm of a digger. One sample was used to determine soil texture and the other to study the root system. In both cases the soil core (7.4 cm diameter) was divided in 10 cm long sub samples. In both years and for each plants density, but only in one replicate, soil samples were carried out until 200 cm to investigate the maximum depth of the hemp root system.
Considering that soil within plant rows contained only fine roots, after harvesting a cubic soil sample (3375 cm3) was collected along the row in each plot. These samples were washed in water so as to separate the taproots. These were counted, dried at 105°C for 24 h, and weighed.
Meteorological data
The meteorological data were collected at the agrometeorological station of the Agricultural Faculty of the University of Bologna in Cadriano. Water table depth was monitored with phreatimeters to 2.5 m (Fig. 1).
Analysis of soil texture
The texture obtained by particle-size analysis using Esenwein levigators and according to the USDA classification was loam (42% sand, 37% silt, 21% clay) in 2004 and silt-clay-loam (8% sand, 61% silt, 31% clay) in 2005.
Root analysis
Prior to root extraction soil samples were kept in a solution of oxalic acid (2%) for 2 hours. Thereafter soil samples were washed in a hydraulic sieving-centrifugation device (Vamerali et al. 2003a). Roots were recovered from the water using a 0.5 mm mesh sieve. Every root sample, after being cleaned from organic debris by hand, was put into tins filled with a 10% ethyl alcohol solution to prevent mould growth.
Root development was studied measuring root length density (RLD, cm cm−3) i.e. the total root length per unit of soil volume, using images of roots samples. To facilitate image acquisition roots were coloured in a methyleneblue solution (4%) for 10 minutes (Himmelbauer et al. 2004), washed from the colouring excess and then spread over a thin layer of water inside a Plexiglas box. Images were acquired using a normal scanner device (Canon, CanoScan 3000 ex) with a resolution of 300 DPI (Vamerali et al. 2003a).
To prevent overestimation of RLD values due to shadow effect the scanner cover was modified with the addition of neon lights (Bauhus and Messier 1999).
Photo scans were analysed with Axio Vision 4.5 (Zeiss), which uses the high – precision algorithm Fibrelength. This software can use different file extensions. The images were analysed as bitmap. The resolution adopted for Axio Vision was 300 dpi (Vamerali et al. 2003a).
Root biomass was measured after drying root samples at 105°C for 24 h.
The tissue C and N content in fine root (from non-coloured subsamples) and taproots were determined by Elemental Combustion System CHNO-S Costech ECS 4010 (Costech Instruments).
According to Gale and Grigal (1987) the distribution of root biomass along soil depth was analysed fitting the following function to experimental data:
where Y is the fraction of root biomass cumulated from the soil surface to the depth d. β is a dimensionless coefficient that describes the shape of the cumulative distribution of root biomass with depth.
Statistics
The experimental design was one factor (planting density) randomized complete block design with split plot (soil depth) combined over years. Analysis of Variance was carried out on all data and Bartlett’s test was performed to assess the homogeneity of variance between years. Means resulted significant after ANOVA were separated according to Fisher’s LSD test for P ≤ 0.05.
Results
Soil and meteorological data
The soils in the two years had different textures throughout the entire sampling depth (Fig. 2). In 2004 the soil was sandy loam. Sand content was on average 45–50% in the top 100 cm and 20–30% from 100 to 130 cm. Clay content was approximately 20% along the whole soil profile, while silt was 30% in the top 100 cm increasing to over 50% at 130 cm depth. In 2005 the soil was mainly silty clay loam, with an average silt content of 60% with values ranging from 50 to 75%, it had a low sand content (10–15%) and clay ranging from 30 to 45%. Soil organic matter in the top 40 cm was 1.5% and 1.7% in 2004 and 2005, respectively; the pH (in H2O) was 7.1 in both years. Total rainfall, from sowing until harvesting, was 282 mm in 2004 (close to the long term mean of the region), and 178 mm in 2005 (Fig. 1). From sowing date until the end of June water table was 20 cm higher in 2004 compared to 2005, due to the higher winter precipitation, 578 mm from October 2003 until April 2004 and 440 mm from October 2004 and April 2005.
Root analysis
RLD values were significantly affected by soil depth but not by plant population or year of cultivation. RLD values were highest in the first 10 cm of soil, they decreased sharply from 10 to 30 cm, kept stable from 30 to 70 cm and then showed a progressive and significant increase until 80–110 cm. Under 110 cm RLD showed a linear decrease until 130 cm. In both years root sampling was carried out on all the plots of one block until 200 cm, in 2004 roots were found until the deepest layer, while in 2005 no roots were found below 130 cm (Fig. 3). Despite this difference it can be noted that in both years almost 50% of the root system (represented by RLD) was concentrated in the first 50 cm of soil.
Root Diameter was another parameter influenced only by soil depth. In particular root diameter was lowest in the top layer of soil, increased linearly until 60 cm, was constant from 60 to 110 and then increased until 130 cm (Fig. 4).
Root dry matter (RDM) was significantly affected by soil depth and year of cultivation. Within the explored soil layer RDM was significantly higher in 2004, when it summed to 1.83 t ha−1, than in 2005 when it was 1.40 t ha−1.
Below ground biomass of hemp was 74% larger when tap roots are taken into account. Dry matter of the taproots was 1.38 t ha−1 in 2004 and 1.01 t ha−1 in 2005. As a consequence total below ground dry biomass for hemp was on average 3.21 t ha−1 in 2004 and 2.41 t ha−1 in 2005. Taking into account the dry matter accumulated in the tap root it was calculated that 50% of total RDM was concentrated in the first 20 cm of soil in both years. The above ground biomass measured at the moment of root sampling was 17.6 t ha−1 and 13.0 t ha−1 in 2004 and 2005 respectively, consequently the ratio between above ground dry biomass and root dry biomass was similar in both years and on average equal to 5.46.
Average RDM decreased down the soil profile following the same trend already shown for RLD: it was highest (244 kg ha−1) in the first 10 cm, then it decreased until 30 cm to then remain fairly constant from 30 to 80 cm (approximately 120 kg ha−1). It showed a slight increase at 90–100 cm and then decreased sharply to reach its minimum value at 130 cm depth (42 kg ha−1) (Fig. 5). Similar to RLD, 50% of RDM (excluding taproot biomass) was found in the first 50 cm of soil in both years. Root biomass distribution down the soil profile was studied fitting the simple asymptotic function firstly proposed by Gerwitz and Page (1974) and thereafter widely adopted by other authors (from Gregory 2006a). The value of the dimensionless coefficient β, obtained fitting this function to the hemp root biomass data in the first 130 cm, was 0.984 (R 2 = 0,94).
The analysis of C/N ratio of root biomass was not affected by plant density, year of cultivation or soil depth and it was 88 for the tap root and 31 for the fine roots. In both roots fraction the carbon content was approximately 41%, while the nitrogen content was 1.31% in the fine roots and 0.48% in the tap roots.
Discussion
Multiple experiments proved that above ground biomass of hemp is not affected by plant populations, within a large range, due to the plasticity of this crop (Amaducci et al. 2002; Venturi and Amaducci 1997; van der Werf et al. 1995). None of the root parameters evaluated in this manuscript were significantly affected by plant population, this seems to confirm the plastic behaviour of hemp also for below ground development at least in the range of plant populations herein evaluated and for the given inter-row distance.
The heterogeneous development of hemp root system, as expressed by the change of RLD and RDM values down the soil profile, is a consequence of the mathematical branching structure underpinning root architecture and especially of the heterogeneous resource availability in the soil in terms of nutrients and water (Lynch 1995; Doussan et al. 2003). In particular, RLD and RDM values in both years (Figs. 3 and 5) were higher in the first soil layers which is common to most plants (Amato and Ritchie 2002; Vamerali et al. 2003a; Qin et al. 2004; Wilhelm et al. 1982) and is a consequence of the more favourable conditions in terms of nutrient and oxygen availability of the top soil (Anderson 1987; Cheng et al. 1990; Gregory 2006b; Pietola 2005). The increase of RLD registered in both years around 100 cm soil depth could be explained by the presence of a perched water table at approximately 120 cm from the surface (data not shown). The RLD increase observed in 2004 at 170–180 cm could also be explained by the presence of a deep water table that was never lower than 190 cm (Fig. 1).
Considering that root growth and rooting depth of crop plants is restricted by physical and chemical impediments (Gregory 2006a) the absence of roots under 130 cm in 2005 (Fig. 3) can be explained by the presence of a silt layer at approximately the same depth (Fig. 2). Moreover, the depth containing 95% of all roots is generally deeper in coarser soil, as in 2004, than in clay or loam soils (Gregory 2006b).
The comparison of RLD distribution of hemp with that of maize, winter wheat, oat, barley and sugar beet reported by other authors (Vamerali et al. 2003a; Pietola 2005; Qin et al. 2006), seems to indicate that in deep soil layer hemp has a higher development of roots (Fig. 6). This finding is in agreement with the high value of the coefficient β that describes the shape of the cumulative distribution of root biomass with depth according to the function proposed by Gerwitz and Page (1974). Comparing the β value found for hemp (0.984) to that of other agricultural crops and natural biomes (King et al. 2003; Jackson et al. 1996) it is apparent that hemp has a deep root profile distribution. Since deep rooting is favourable and is usually found in water limiting environments (Schenk and Jackson 2002), this feature of hemp likely contributes to its suitability in Mediterranean environments, where in fact it is traditionally cultivated without irrigation (Venturi and Amaducci 1999). Another positive implication of a deep root system is linked to the accumulation of biomass in soil zones where mineralization is limited and consequently carbon is stored for longer times (Liebig et al. 2005; Lemus and Lal 2005). Overall comparing the results of fine root biomass of hemp with that of other agricultural annual crops indicates that hemp has a larger production of fine roots compared to wheat, cotton and sorghum (Wechsung et al. 1995; Upendra et al. 2005) and similar to that of maize (Piper and Weiss 1993).
Finer roots, that provide a high interface of contact with soil favour water and nutrient uptake (Eissenstat 1992), are usually found in favourable soil conditions where gas exchange and nutrients are higher and soil compaction lower; this is in fact the condition of the top 30–40 cm of soil that had been ploughed in our experiment and where roots of lowest diameter were found (Fig. 4). The increase of root diameter along the soil profile that we found for hemp was also reported for sugar beet (Vamerali et al. 2003a) and in the first soil layers for maize grown under conventional tillage (Qin et al. 2006) and for wheat (Mosca et al. 1992). The increase of root diameter is generally associated with the ability of thicker roots to exert higher growth pressure (Rosolem et al. 2002) and consequently to grow in high resistance soil layers. This is the case when compacted soil layers are encountered or in general with the increase of soil bulk density along soil depth (Liebig et al. 2005; Gregory 2006b). The further increases of root diameter measured around 120 cm can be explained by the presence of a compacted soil layer (Chassot and Richner 2002; Qin et al. 2004; Matechera et al. 1992), the presence of which seems confirmed by a suspended water table at 120 cm of depth.
The difference in the C/N ratio between fine roots (88) and tap root (31) is an effect of the higher nitrogen content of the fine roots (1.31%), which being the active part of the root system have a larger concentration of transport protein (Gregory 2006a), compared to that of the tap roots (0.48%). The carbon content of both fine and tap roots was 41%, which is in accordance with analyses carried out in previous experiments with different crops (Hadley and Causton 1984).
Conclusions
None of the root parameters evaluated in this manuscript were significantly affected by plant population, which seems to confirm the plastic behaviour of hemp not only for above ground but also for below ground development. The plasticity of hemp root development was also confirmed by the uniformity of root parameters despite the notable differences in the soil properties in the two years. When roots are not blocked by compacted layers they can reach up to 200 cm depth to reach the water table (if present).
Hemp is considered an ideal crop for organic agriculture (Stickland 1995); high root biomass production measured in this study and particularly its distribution in deep soil layers provides additional evidence of the positive role that hemp can play in sustainable cropping systems.
References
Amaducci S (2003) HEMP-SYS: design, development and up-scaling of a sustainable production system for HEMP textiles – an integrated quality SYStem approach. J Ind Hemp 8:79–83
Amaducci S, Errani M, Venturi G (2002) Response of hemp to plant population and nitrogen fertilization. Ital J Agron 6(2):103–111
Amaducci S, Colauzzi M, Bellocchi G, Venturi G (2008) Modelling post-emergent hemp phenology (Cannabis sativa L.): theory and evaluation. Eur J Agron 28:90–102
Amato M, Ritchie JT (2002) Spatial distribution of roots and water uptake of maize (Zea mays L.) as affected by soil structure. Crop Sci 42:773–780
Anderson EL (1987) Corn root growth and distribution as influenced by tillage and nitrogen fertilization. Agron J 79:544–549
Barber SA (1995) Soil nutrient bioavailability. A mechanistic approach. Wiley, New York, NY, p 414
Bauhus J, Messier C (1999) Evaluation of fine root length and diameter measurements obtained using RHIZO image analysis. Agron J 91:142–147
Berger J (1969) The world’s major fibre crops: their cultivation and manuring. Centre D’Etude de l’Azote, Zurich, p 219
Biewinga EE, Van der Bijl G (1996) Sustainability of energy crops in Europe. Centre for Agriculture & Environment, CLM 234, Utrecht, p 209
Bòcsa I, Karus M (1998) The cultivation of hemp: botany, varieties, cultivation and harvesting. Hemptech, Sebastopol, CA (USA), p 184
Bouma TJ, Nielsen KL, Koutstaal B (2000) Sample preparation and scanning protocol for computerised analysis of root length and diameter. Plant Soil 218:185–196
Chassot A, Richner W (2002) Root characteristics and phosphorus uptake of maize seedlings in a bilayered soil. Agron J 94:118–127
Chassot A, Stamp P, Richner W (2001) Root distribution and morphology of maize seedling as affected by tillage and fertilizer placement. Plant Soil 231:123–135
Cheng W, Coleman DC, Box JE Jr (1990) Root dynamics, production and distribution in agroecosystems on the Georgia Piedmont using minirhizotrons. J Appl Ecol 27:592–604
Cochrane HR, Aylmore LAG (1994) The effects of plant roots on soil structure, Proceedings of 3rd Triennial Conference “Soils 94”, p 207–212
Costa C, Dwyer LM, Hamilton RI, Hamel C, Nantais L, Smith DL (2000) A sampling method for measurement of large root systems with scanner-based image analysis. Agron J 92:621–627
De Freitas PL, Zobel RW, Snyder VA (1999) Corn root growth in soil columns with artificially constructed aggregates. Crop Sci 39:725–730
Doussan C, Pagès L, Pierret A (2003) Soil exploration and resource acquisition by plant roots: an architectural and modelling point of view. Agronomie 23:419–431
Eissenstat DM (1992) Costs and benefits of constructing roots of small diameter. J Plant Nutr 15:763–782
Eissenstat DM, Well CE, Yanai RD, Whitbeck JL (2000) Building roots in a changing environment: implications for root longevity. New Phytol 147:33–42
Gale MR, Grigal DF (1987) Vertical root distributions of northern tree species in relation to successional status. Can J For Res 17:829–834
Gerwitz A, Page ER (1974) An empirical mathematical model to describe plant root systems. J Appl Ecol 11:773–781
Gorchs G, Llovers J, Comas J (2000) Effect of hemp (Cannabis sativa L.) in a crop rotation hemp–wheat in the humid cool areas of North-eastern of Spain. In proceeding of the Conference: Crop development for the cool and wet regions of Europe (COST Action 814), Pordenone, Italy, pp 581–589
Gregory PJ (2006a) Plant roots: growth, activity and interaction with soils. Blackwell, UK
Gregory PJ (2006b) Roots, rhizosphere and soil: the route to a better understanding of soil science? Eur J Soil Sci 57:2–12
Hadley P, Causton DR (1984) Changes in percentage organic carbon content during ontogeny. Planta 160:97–101
Himmelbauer ML, Loiskandl W, Kastanek F (2004) Estimating length, average diameter and surface area of roots using two different image analyses systems. Plant Soil 260:111–120
Holanda FSR, Mengel DB, Paula MB, Carvaho JG, Bertoni JC (1998) Influence of crop rotations and tillage systems on phosphorus and potassium stratification and root distribution in the soil profile. Commun Soil Sci Plant Anal 29:2383–2394
Jackson RB, Canadell J, Ehleringer JR, Mooney HA, Sala OE, Schulze ED (1996) A global analysis of root distributions for terrestrial biomes. Oecologia 108:389–411
Kage H, Kochler M, Stutzel H (2000) Root growth of cauliflower (Brassica oleracea L. botrytis) under unstressed condition: measurement and modeling. Plant Soil 223:131–145
King J, Gay A, Sylvester-Bradley R, Bingham I, Foulkes J, Gregory PJ, Robinson D (2003) Modelling cereal root system for water and nitrogen capture: towards an economic optimum. Ann Bot 91:383–390
Kirchmann H, Thorvaldsson G (2000) Challenging targets for future agriculture. Eur J Agron 12:145–161
Lemus R, Lal R (2005) Bioenergy crop and carbon sequestration. Crit Rev Plant Sci 24:1–21
Liebig MA, Johnson HA, Hanson JD, Frank AB (2005) Soil carbon under switchgrass stands and cultivated cropland. Biomass Bioenergy 28:347–354
Lotz LAP, Groeneveld RMW, Habekotté B, van Oene H (1991) Reduction of growth and reproduction of Cyperus esculentus by specific crops. Weed Res 31:153–160
Lynch JP (1995) Root architecture and plant productivity. Plant Physiol 109:7–13
Matechera SA, Alston AM, Kirby JM, Dexter AR (1992) Influence of root diameter on the penetration of seminal roots into a compacted subsoil. Plant Soil 144:297–303
Mediavilla V, Jonquera M, Schmid-Slembrouck I, Soldati A (1998) A decimal code for growth stages of hemp (Cannabis sativa L.). J Int Hemp Assoc 5(2):65, 68–74
Monroe CD, Kladivko EJ (1987) Aggregate stability of a silt loam soil as affected by roots of corn, soybeans, and wheat. Commun Soil Sci Plant Anal 18(9):1077–1087
Mosca G, Govi G, Archetti R, Bonciarelli F, Mazzoncini M, Rubino P, Ruggiero C e Venezia G (1992) Effetti della lavorazione del terreno sullo sviluppo degli apparati radicali di frumento (Triticum aestivum L. e Triticum durum Desf.). Riv Agron 26(3):223–231
Newman EI (1966) A method of estimating the total length of root in a sample. J Appl Ecol 3:139–145
Passioura JB (2002) Soil conditions and plant growth. Plant Cell Environ 25:311–318
Pietola LM (2005) Root growth dynamics of spring cereals with discontinuation of mouldboard ploughing. Soil Till Res 80:103–114
Piper EL, Weiss A (1993) Defoliation during vegetative growth of corn: the shoot:root ratio and yield implications. Field Crops Res 31:145–153
Qin RJ, Stamp P, Richner W (2004) Impact of tillage on root systems of winter wheat. Agron J 96:1523–1530
Qin RJ, Stamp P, Richner W (2006) Impact of tillage on maize in a cambisol and luvisol in Switzerland. Soil Till Res 85:50–61
Robertson WS, Fukai S, Hammer GL, Ludlow MM (1993) Modelling root growth of grain sorghum using CERES approach. Field Crop Res 33:113–130
Rosolem CA, Foloni JSS, Tiritan CS (2002) Root growth and nutrient accumulation in cover crops as affected by soil compaction. Soil Till Research 65:109–115
Schenk HJ, Jackson RB (2002) Rooting depths, lateral spreads, and belowground/aboveground allometries of plants in water-limited ecosystems. J Ecol 90:480–494
Stickland D (1995) Suitability of hemp for ecological agriculture. In Proceedings of the Symposium Bioresurce Hemp, pp 255–258
Tennant D (1975) A test of a modified line intersect method of estimating root length. J Ecol 63:995–1001
Upendra MS, Bharat P, Singh B, Wayne Whitehead F (2005) Tillage, cover crops, and nitrogen fertilization effects on cotton and sorghum root biomass, carbon, and nitrogen. Agron J 97:1279–1290
Vamerali T, Guarise M, Ganis A, Bona S, Mosca G (2003a) Analysis of root image from auger sampling with a fast procedure: a case of application to sugar beet. Plant Soil 255:387–397
Vamerali T, Saccomani M, Bona S, Mosca G, Guarise M, Ganis A (2003b) A comparison of root characteristics in relation to nutrient and water stress in two maize hybrids. Plant Soil 255:157–167
Veen BW (1980) Energy cost of ion transport. In: Rains DW, Valentine RC, Holaender C (eds) Genetic engineering of osmoregulation. Impact on plant productivity for food, Chemicals and Energy. Plenum, New York, pp 187–195
Venturi G, Amaducci MT (1997) Effetti di dosi di azoto e densità di semina su produzione e caratteristiche tecnologiche di Cannabis sativa L. Riv Agron 3:616–623
Venturi G, Amaducci MT (1999) Le colture da fibra. Ed agricole ISBN-88-206-4288-3
Wechsung G, Wechsung F, Wall GW, Adamsen FJ, Kimball BA, Garcia RL, Pinter PJ Jr, Kartschall T (1995) Biomass and growth rate of a spring wheat root system grown in free-air CO2 enrichment (FACE) and ample soil moisture. J Biogeogr 22:623–634
van der Werf HMG, Turunen L (2008) The environmental impacts of the production of hemp and flax textile yarn. Ind Crop Prod 27:1–10
van der Werf HMG, Wijlhuizen M, de Schutter JAA (1995) Plant density and self-thinning yield and quality of fibre hemp (Cannabis sativa L.). Field Crop Res 40:153–164
Wilhelm WW, Mielke LN, Fenster CR (1982) Root development of winter wheat as related to tillage practice in western Nebraska. Agron J 74:85–88
Acknowledgments
Research described in this paper was financed by the European Commission in the frame of the project HEMP SYS (Design, Development and Up-Scaling of a Sustainable Production System for HEMP Textiles: An Integrated Quality SYStems Approach) contact number QLK5-CT-2002-01363.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Jan Vos.
Rights and permissions
About this article
Cite this article
Amaducci, S., Zatta, A., Raffanini, M. et al. Characterisation of hemp (Cannabis sativa L.) roots under different growing conditions. Plant Soil 313, 227–235 (2008). https://doi.org/10.1007/s11104-008-9695-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-008-9695-0