Abstract
Paradoxically low nitrogen resorption efficiency in the drought-deciduous desert shrub Fouquieria splendens Engelm (ocotillo) triggered tests of the hypotheses that resorption is often low in this species and that resorption is influenced by zinc. Resorption efficiency and proficiency were measured in 1989 and 1994 at two sites in the Chihuahuan Desert in plants to which zinc, or zinc and nitrogen were added. Resorption of nitrogen, phosphorus, and zinc in unfertilized plants varied temporally and spatially, but was both efficient (66%, 49%, and 40%, respectively) and proficient (0.55%, 0.09%, and 9.4 μg g−1, respectively) as determined by comparison to worldwide resorption patterns in a wide variety of other species. Applications of zinc had no significant effect on the resorption of nitrogen and phosphorus, but did influence the resorption of zinc. Resorption of zinc was significantly less efficient in zinc-treated plants than controls at only one of the two sites in one of the 2 years, yet resorption of zinc was significantly less proficient in zinc-treated plants than controls in both years and at both sites. This pattern of zinc resorption adds insight into the continuing debate regarding the relationship between fertility and resorption because the data used to fuel the debate have almost exclusively described macronutrients, not trace metals. The high variability in resorption among individuals, sites, and years observed for F. splendens may well be an attribute of many desert-dwelling, drought-deciduous plants. When senescence is controlled primarily by water availability rather than photoperiod, especially in a landscape characterized by unpredictable amounts and timing of precipitation, high variability in associated processes such as resorption may be inevitable.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Resorption of nutrients from senescing plant tissues occurs throughout the plant kingdom (Chapin and Kedrowski 1983; Aerts 1996; Killingbeck et al. 2002), varies markedly in both efficiency (Aerts 1996; the content of an element in senesced leaves expressed as a percentage of the content in mature green leaves) and proficiency (Killingbeck 1996; the content of an element in senesced leaves), and increases plant fitness (May and Killingbeck 1992). Its action as a conservation strategy impacts the nutrient budgets of macronutrients such as nitrogen (N) and phosphorus (P), but also acts to conserve trace metals (Killingbeck 1985; Killingbeck 2004). The possibility that macronutrients and trace metals interact to significantly influence the resorption of one or the other has yet to be experimentally addressed.
In 1986, the C3 drought-deciduous desert shrub Fouquieria splendens Engelm. (ocotillo) was the focus of a study in the Chihuahuan Desert initiated to determine the maximum levels to which N could be resorbed (Killingbeck 1992). This combination of species and site was chosen because F. splendens grows on extremely N-poor soils, and can utilize large amounts of N as multiple cohorts of leaves can be formed and lost in a single growing season (Cannon 1905; Scott 1932). The hypothesis explored was that potentially high use and low availability would coincide to select for a high degree of internal N conservation.
Mean resorption efficiency of N was not significantly different from zero in 1986, but what was equally paradoxical was that N resorption was highly related to the amount of zinc (Zn) in leaves of F. splendens (R 2 = 0.60–0.65; Killingbeck 1992). The tentative explanation for this latter link was that Zn was an essential co-factor for enzymes that are necessary to break down proteins and peptide chains into the amino acids that are the carriers of N out of leaves during resorption. In addition to being critical to a wide variety of metabolic functions in plants (Broadley et al. 2007), Zn is known to be a key component of metallo-endopeptidases that appear to be linked to the breakdown of chloroplast proteins (Feller 2004), a major source of foliar protein (Peoples and Dalling 1988; Jones 2004). For example, approximately 90% of N resorbed from Oryza sativa leaves was derived from chloroplasts (Morita 1980). Perhaps, then, low N resorption efficiency was the result of insufficient amounts of foliar Zn, a possibility that has widespread implications.
These counterintuitive results gave rise to manipulative fertilization experiments in two populations of F. splendens growing in disjunct sites in the Chihuahuan Desert. The primary hypotheses addressed were that (1) resorption of N, P, and Zn in F. splendens is significantly lower than averages worldwide, for a range of years and sites, and (2) resorption efficiency and proficiency of N, P, and Zn are enhanced by intensive short-term (1 year) or extensive long-term (5 years) fertilization with Zn. These experiments also provided the opportunity to explore the widely debated issue of whether fertility influences resorption, but with a trace metal rather than one of the customary macronutrients.
Materials and methods
Site and plant descriptions
The two populations of F. splendens studied were located approximately 27 km from one another in the northern reaches of the Chihuahuan Desert in southern New Mexico, USA. The first was in the foothills of the Organ Mountains 11 km east of Las Cruces, NM on a rocky south-facing slope (32°19′N, 106°38′W). The second was in the Jornada Basin Long-Term Ecological Research Site/Chihuahuan Desert Rangeland Research Center 20 km north of Las Cruces on a rocky east-facing slope of Mt. Summerford (32°30′N, 106°48′W). Mean annual temperature and precipitation were approximately 15°C and 200–250 mm, respectively (West and Klemmedson 1978) and soils were Aridisols (Morain 1981). The F. splendens plants studied each had 15–100 stems longer than 1 m. The longest stem on any plant in 1989 was 335 cm.
Organ mountains fertilization and leaf collections
In May 1989, 12 mature, leafless F. splendens plants were chosen for study, eight of which were the subject of a previous study indicating a possible link between foliar Zn and resorption of N (Killingbeck 1992). One plant from each pair of plants having similar numbers of stems was randomly chosen as a control (n = 6), the other became a Zn-fertilized treatment (n = 6).
Because F. splendens has the ability to take up liquids through its stems (Lloyd 1905), Zn was delivered to stems of Zn-treatment plants as an aqueous solution, and to roots as solid zinc sulfate. During the initial application of aqueous fertilizer on 24 May 1989, 37 ml of the ZnSO4 solution were sprayed onto the terminal 60 cm of every stem longer than 1 m. This concentration of Zn was chosen because it matched exactly the maximum recommended application rate of Zn in a commercial liquid fertilizer containing both N and Zn that was used in another phase of this research (see Jornada fertilization and leaf collections later in the Methods). The concentration (6.25 g ZnSO4 in 3,785 ml distilled water) and amount (37 ml) of ZnSO4 solution remained constant for all Zn-fertilized treatments in this study, as did the location of application (terminal 30–60 cm of stems) and the amount and application of solid Zn fertilizer (8.72 g of ZnSO4 applied evenly in a 2-m diameter plot centered on each treatment plant).
Subsequent applications of aqueous Zn were applied once per week from 30 May–27 July 1989 to each of 12 marked stems on each plant. Liquid fertilizer was not applied after stems produced leaves, nor was it applied after 1989 in any part of this study. To control for the water added during the spraying of fertilized treatment plants, 37 ml of distilled water were sprayed onto the terminal 60 cm of every stem >1 m on all controls.
Solid fertilizer was applied once per month from 24 May to 16 August 1989, and then again on 11 July 1990, 12 June 1993, and 26 August 1994. The application rate (1.0 g Zn m−2) was the approximate mid-point of the rate recommended for crop plants (0.2–2.2 g m−2; Murphy and Walsh 1972).
In 1989, unsenesced green leaves were collected on 8–9 August from three previously selected stems on each plant that had received either Zn or water. Because the application of liquids to stems ceased before the emergence of leaves, uptake of Zn or water would have occurred only through stems. Senesced leaves were collected on 29 October from three previously selected stems from which leaves had not already been collected, but that had received either Zn or water. Leaves were collected from 5-mm mesh nylon fruit bags that encased the terminal 45 cm of each stem, as was the case throughout the study.
In 1994, unsenesced green leaves were collected on 25 August from three stems at 120° intervals around the canopy of each of the 12 plants. Senesced leaves were collected on 17 October from the three stems per plant that were closest to the stems from which green leaves were collected in August.
Jornada fertilization and leaf collections
In May 1989, 18 mature, leafless F. splendens plants were chosen for study and divided into six groups. One plant from each group was randomly chosen to become 1) a water-sprayed control (n = 6), 2) a Zn-fertilized treatment (n = 6), or 3) a fertilized treatment that received both Zn and N (Zn + N treatments, n = 6). Nitrogen was part of this last treatment because adding N to Zn fertilizers is known to increase the amount of Zn taken up by commercial pecan trees (Smith and Storey 1979; Esteban Herrera personal communication).
As at the Organ Mountains site, Zn was delivered as liquid and solid ZnSO4 to the Zn-treated plants. During the initial application of fertilizer on 22 May 1989, the ZnSO4 solution was sprayed onto every stem >1 m. Subsequent applications of liquid Zn were applied once per week from 30 May–19 July 1989 to 16 marked stems on each plant.
Controls received distilled water sprayed onto every stem >1 m. Subsequent applications of water were applied once per week from 30 May–19 July 1989 to each of 16 marked stems on each control.
The fertilizer applied to the Zn + N treatment plants was a solution of commercially available Nitro-Zinc (Agricultural Products Co., Inc; 15% N, 5% Zn) mixed at the maximum recommended concentration (38 ml Nitro-Zinc in 3450 ml distilled water). Procedures for applying this solution to the Zn + N-treatments were identical to that for the Jornada Zn-treatments, and the Zn concentration in this solution was identical to that in the Zn-treatment aqueous spray.
Solid fertilizers were applied once per month from 20 May–25 August 1989, and then again on 11 July 1990, 10 June 1993, and 30 August 1994. The Zn + N-treated plants received an additional 92.35 g of ammonium nitrate (NH4NO3), an application rate (9.8 g N m−2) similar to that used in other resorption studies (e.g., 8.3 g N m−2; Chapin and Moilanen 1991).
In 1989, unsenesced green leaves were collected on 25 July from three previously selected stems on each of the control, Zn-treated, and Zn + N-treated plants. Senesced leaves were collected from these same plants on 6–10 November from three previously selected stems from which leaves had not already been collected, but that had received water, Zn, or Zn + N.
In 1994, unsenesced green leaves were collected on 29 August from each of the controls, Zn-treated, and Zn + N-treated plants. Senesced leaves were collected on 3–15 October from the three stems per plant that were closest to the stems from which green leaves were collected in August.
Jornada leaf-treated plants
To see the effects of adding ZnSO4 directly to leaves as opposed to adding Zn to unfoliated stems, green leaves were collected from three fully foliated stems on each of five plants at the Jornada site on 23 July 1989. Later that day, nine additional stems per plant were sprayed with either water (three stems), ZnSO4 solution (three stems), or Nitro-Zinc solution (three stems; n = 5 per treatment). The same spray regimen was repeated on 1 and 8 August. Senesced leaves were collected on 7–10 November. Solid fertilizers were not applied to these plants.
Laboratory analyses
For all foliage, leaf surface area was measured with a LI-COR Model 3100 area meter. Leaves were oven-dried at 60 °C to constant mass, weighed, and ground in a Wiley mill. Nutrient analyses were performed at the University of Rhode Island or at the New Mexico State University Soil, Plant, and Water Testing Laboratory. Total N was extracted from leaves with a Kjeldahl N digestion or with a sulfuric acid-hydrogen peroxide wet ashing technique (Thomas et al. 1967) and measured with a Technicon AutoAnalyzer. Phosphorus was extracted with the same wet ashing technique or with perchloric acid (Williams 1984) and measured with a Technicon AutoAnalyzer or a Perkin Elmer inductively coupled plasma spectrophotometer. Zinc was extracted with perchloric acid, as above, or with a microwave digestion technique (Jones et al. 1991) and measured with a Varian atomic absorption spectrophotometer or a Perkin Elmer inductively coupled plasma spectrophotometer.
Calculations of resorption efficiency and proficiency
Resorption efficiency, the percentage of a nutrient withdrawn from green leaves as the leaves senesce, was calculated as the difference in the content (nutrient mass per unit leaf surface area) of a nutrient between green and senesced leaves, divided by the content in green leaves, the quantity multiplied by 100. Resorption proficiency, the level to which a nutrient is reduced in senesced leaves (Killingbeck 1996), was calculated as the concentration of a nutrient in fully senesced leaves expressed on a mass basis [% (=g 100 g−1) for N and P; μg g−1 for Zn].
Because leaching by rainfall does not account for a significant amount of the reductions of N, P, and Zn in senescing leaves of F. splendens (Killingbeck 1992; Killingbeck unpublished data), or for significant reductions in the senescing leaves of other species (e.g., Ryan and Bormann 1982; Killingbeck et al. 1990), the measured differences between green and senesced leaves were reductions due to resorption.
Statistical analyses
Statistical analyses were performed with SAS JMP Version 5 software (SAS Institute Inc. 2002). Resorption efficiencies, which are proportional percentages, were arcsine transformed before analysis. Normality of distribution was determined with the Shapiro-Wilk W Test. For comparisons of pairs of means, Student’s t test was used for data that were normally distributed, and the Wilcoxon Test was used for data that were not normally distributed. For simultaneous comparisons of more than two means, ANOVA was used throughout, even when multiple means were not normally distributed because ANOVA is “quite robust to non-normality” (Underwood 1997). ANOVA analyses were followed by the Tukey-Kramer HSD multiple means comparison. Entire plants, not individual stems, were used as replicates in all statistical analyses. Green-leaf N and P concentrations for one sample (Organ Mountains plant number 6 in 1994) were extreme outliers and were winsorized (Sokal and Rohlf 1981) to prevent spurious results. Linear regressions comparing resorption of Zn to green-leaf Zn were run with log10-transformed data for both resorption efficiency and proficiency. Because the assumption of independence of variables cannot be met for regressions examining resorption efficiency (green-leaf Zn itself is used in the calculation of efficiency), statistical significance was determined by comparing the slopes of the regression lines to a slope of 1.0 (see Kobe et al. 2005).
Results
Resorption patterns in the absence of fertilization
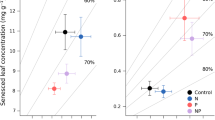
Resorption in unfertilized controls was typically both efficient and proficient compared to plants worldwide, and varied among sites and years (Fig. 1). Mean N, P, and Zn resorption efficiency was 66, 49, and 40%, respectively, across years and sites. Mean resorption proficiency of these same elements was 0.55%, 0.09%, and 9.4 μg g−1, respectively. Nitrogen resorption proficiency differed between sites in 1994, and between years at both sites, but mean N resorption efficiency remained almost unchanged throughout the study (Fig. 1). Phosphorus resorption efficiency and proficiency differed between sites in 1989 and differed markedly among individuals at the Jornada site in 1989. Zinc resorption was more than twice as proficient in 1989 as in 1994 at the Jornada site, but high variance precluded the statistical significance of an apparent trend in efficiency between sites.
Mean nitrogen, phosphorus, and zinc resorption efficiency and proficiency in unfertilized Fouquieria splendens plants growing at the Organ Mountains and Jornada sites during the leafless-stem experiment in 1989 and 1994. The horizontal dashed line represents the grand mean for each of the six panels. Error bars represent the standard error of the mean. Within each of the six panels, bars with different letters are significantly different (P < 0.05; ANOVA and Tukey-Kramer HSD; n = 6 for each year at each site)
Effects of leafless-stem fertilization
Fertilization of leafless stems with Zn or a combination of Zn and N had no significant effect on N and P resorption efficiency and proficiency. Fertilization did, however, have a significant impact on Zn resorption in 1989 at both the Organ Mountains (Fig. 2) and Jornada (Fig. 3) sites. Zinc resorption was less efficient in Zn-treated plants than in controls at the Organs site, and less proficient in Zn-treated plants than in controls at both sites. Five years later, these significant differences no longer existed. Although Zn resorption in the Zn + N-treatments was significantly less proficient than in controls in 1989 at the Jornada site, such was not the case in 1994 (Fig. 3).
Mean zinc resorption efficiency and proficiency in controls (C) and zinc-treatments (ZN) at the Organ Mountains site during the leafless-stem experiment in 1989 and 1994. Error bars represent the standard error of the mean. The probability that the two means in each panel are significantly different is given in the nested box (Student’s t test or Wilcoxon test; n = 6 for each treatment in each year)
Mean zinc resorption efficiency and proficiency in controls (C), zinc-treatments (ZN), and zinc + nitrogen treatments (ZN + N) at the Jornada site during the leafless-stem experiment in 1989 and 1994. Error bars represent the standard error of the mean. Within each of the four panels, bars with different letters are significantly different (P < 0.05; ANOVA and Tukey-Kramer HSD; n = 6 for each treatment in each year)
Effects of foliated-stem fertilization
As was the case when the application of solid fertilizers was combined with the application of liquid fertilizers to leafless stems, the application of liquid fertilizers to fully foliated stems had no significant impact on N and P resorption efficiency or proficiency (Fig. 4). However, Zn resorption was less efficient and proficient in both Zn-bearing treatments than in controls, and in Zn + N-treatments than in treatments containing Zn alone.
Mean nitrogen, phosphorus, and zinc resorption efficiency and proficiency in controls (C), zinc-treatments (ZN), and zinc + nitrogen treatments (ZN + N) at the Jornada site during the foliated-stem experiment in 1989. Error bars represent the standard error of the mean. Within each of the six panels, bars with different letters are significantly different (P < 0.05; ANOVA and Tukey-Kramer HSD; n = 6 for each treatment)
Specific leaf mass
Specific mass of F. splendens leaves (SLM; g cm−2) was significantly higher in senesced than in green leaves in all combinations of site, year, and treatment in the leafless stem experiments, with only one exception (controls, 1994, Organs site; unpublished data). The same was true for the foliated-stem experiment, with no exceptions. This is likely due to a reduction in surface area as the nonrigid, nonxeromorphic leaves of F. splendens (Lersten and Carvey 1974) senesce and dry. Even though resorption efficiencies were appropriately calculated as percentage changes in nutrient mass per unit area (e.g., Woodwell 1974), reductions in surface area could result in underestimates of resorption efficiency (van Heerwaarden et al. 2003). However, any such reductions (a) would have affected almost all treatments equally because SLM differed among treatments in only one combination of site and year (1994, Jornada site), and (b) would have had no impact on the measurement of resorption proficiency.
Discussion
Unlike F. splendens at the Organs site in 1986 whose N resorption efficiencies were not statistically different from 0% (Killingbeck 1992), the same plants, and other unfertilized controls at the Organ Mountains and Jornada sites, consistently resorbed 60% or more of the N in their senescing leaves. Phosphorus resorption, although much more variable than that of N, was also much more efficient (49%) than in 1986 (31%). This clearly indicates that resorption of N and P in F. splendens is not consistently inefficient.
But what about resorption proficiency? Resorption of N in unfertilized controls was highly proficient in three of the four combinations of years and sites. In those three combinations, N was reduced to 0.32–0.55% in senesced leaves, levels well below the 0.7% threshold that demarcates the upper limit of what is considered to be highly proficient resorption of N (Killingbeck 1996). The most proficient individual at resorbing N during the study reduced N to 0.24% in its senesced leaves, further indicating that F. splendens has the physiological capacity for highly proficient resorption of this element.
Resorption proficiency was again more variable for P than for N, but controls at the Organs site reduced P to 0.02–0.03%, levels well below the 0.05% threshold that demarcates the upper limit of what is considered to be highly proficient resorption of P (Killingbeck 1996). The most proficient individual at resorbing P during the study reduced this element to the extremely low level of 0.01%. Therefore, resorption of N and P is neither consistently inefficient nor consistently unproficient. In fact, mean resorption of N in F. splendens was considerably more efficient (66%) and proficient (0.55%) than that in a wide variety of species worldwide (50%, Aerts 1996 and 0.87%, Killingbeck 1996, respectively).
Effects of fertilization on N and P resorption
Neither short-term nor long-term applications of fertilizers containing Zn, or Zn and N had any positive effect on N and P resorption efficiency or proficiency. Even when Zn was applied directly to leaves, N and P resorption was not affected. Therefore, the hypothesis that N and P resorption is influenced by fertilization with Zn alone, or Zn and N, was rejected.
Effects of fertilization on Zn resorption
In contrast to resorption of N and P, the hypothesis that Zn resorption is influenced by short-term additions of Zn-bearing fertilizers was accepted. It has long been known that Zn is mobile enough to be transported out of leaves during resorption (Mukhjerjee 1969; Killingbeck 1985), but little is known about the factors that affect this process. At the Organs site in 1989, and in the Jornada foliated-stem experiment, resorption of Zn became less efficient and less proficient as Zn was added separately, or in combination with N. Zinc resorption was also significantly less proficient in Zn- and Zn + N-treatments than in controls at the Jornada site in 1989.
Long-term additions of Zn did not impact Zn resorption. Five years after the applications of Zn fertilizer began, the initial significant differences between Zn-bearing treatments and controls had disappeared. For the plants at the Organs site, it was initially tempting to suggest that the differences in Zn resorption between 1989 and 1994 could be attributed to a shift in the relative amounts of green-leaf Zn in controls and Zn-treatments. Green-leaf Zn was significantly higher in Zn-treatments than controls in 1989, but not in 1994 (unpublished data). However, the actual differences were rather small and no such shift in green-leaf Zn between 1989 and 1994 was evident in F. splendens at the Jornada site. Reducing the intensive applications of both solid and liquid fertilizer after 1989 to periodic applications of solid fertilizer resulted in the loss of Zn-treatment effects on Zn resorption.
The pattern of Zn resorption seen in 1989 bears on the continuing debate regarding the relationship between fertility and resorption. Studies supporting (Richardson et al. 1999; Kobe et al. 2005; Rejmánková 2005; Richardson et al. 2005) and refuting (Birk and Vitousek 1986; Minoletti and Boerner 1994; Aerts 1996; Milla et al. 2005) the hypothesis that fertility or foliar nutrient status significantly influence resorption have focused almost exclusively on N and P. Adding Zn fertilization and resorption to the debate increases the breadth of our understanding of this notoriously complex relationship.
Perhaps the most salient point regarding this fertility issue is this; there was not always a significant relationship between foliar Zn concentration and Zn resorption in F. splendens. The consistent trend in the ANOVA analyses was that Zn treatments resulted in less efficient and less proficient resorption of Zn. However, in three of four combinations of site and year, regression analyses indicated no significant trend between green-leaf Zn and (a) efficiency of Zn resorption (P > 0.05; slope of log10 green-leaf Zn/log10 Zn resorption efficiency was not significantly different from a slope of 1.0; see Kobe et al. 2005) and b) proficiency of Zn resorption (P > 0.05; log10 green-leaf Zn/log10 Zn resorption proficiency). Interestingly, as green-leaf Zn increased, resorption of Zn became significantly less proficient at the Organs site in 1989, the year liquid Zn fertilizer was applied (P ≤ 0.01; R 2 = 0.450). Therefore, the results are mixed for the existence of a strong, consistent link between the resorption and availability of Zn, just as they are for N and P in the literature at large.
Influence of cofactors and variability in resorption
Rejecting the hypothesis that foliar Zn plays a consistent role in influencing N resorption in F. splendens still does not address two important questions: (1) is there merit in the hypothesis that metallic enzyme cofactors such as Zn may influence N resorption in general? and (2) what caused the large temporal variation in ocotillo resorption?
There is no reason, from a theoretical standpoint, to abandon the hypothesis that deficiencies in Zn, or other metallic cofactors, could reduce resorption of N. Zinc is known to be an integral component of metallo-endopeptidases (Barrett 1986), which in turn have been implicated in the breakdown of cellular proteins (Feller 2004). This catabolism is necessary for the effective resorption of N from senescing tissues because N is moved out of senescing leaves as amino acids (Chapin et al. 1986; Hörtensteiner and Feller 2002; Mae 2004). Feller (2004) has already suggested that observed reductions of proteolysis in the presence of the metal chelator EDTA were likely the result of the inhibition of a metallo-endopeptidase.
Therefore, when Zn reaches some critical low level, some fraction of the enzymatic machinery responsible for protein catabolism will be rendered non-functional. What those critical levels are for any combination of plant species and site is presently unknown. Zinc is considered to be deficient for plant growth at concentrations below 20 μg g−1 (Jones 1972). Nonetheless, Zn concentrations were below that level in 1989 when resorption of N was both highly efficient and proficient. This, along with the fact that the plants grew vigorously, is clear evidence that the 20 μg g−1 deficiency threshold is not universal. Because factors other than Zn availability may have dominated the control of resorption in this study, laboratory experiments in which factors such as water availability are optimized, while Zn availability is varied widely, would improve our ability to determine whether cofactors can influence macronutrient resorption.
As for the second question, some degree of temporal variability in resorption is always expected, but extreme variability has rarely been reported. The unpredictable timing of leaf senescence and abscission in drought-deciduous plants in general, and F. splendens in particular, may be responsible for highly variable resorption. Senescence controlled primarily by water availability alone, rather than by a combination of photoperiod and additional factors as it is in many deciduous plants, may lead to abnormally high spatial and temporal variability in the resorption of drought-deciduous plants such as F. splendens.
Because drought and water availability are known to impact resorption (Hocking 1982; del Arco et al. 1991; Heckathorn and DeLucia 1994; Minoletti and Boerner 1994) and because water availability is known to be highly variable in deserts worldwide (Whitford 2002), it seems logical to hypothesize that resorption in desert-dwelling, drought-deciduous plants may in fact be highly variable. The relatively small number of multi-year, multi-site studies of resorption in drought-deciduous species prevents a definitive test of this hypothesis at present, but for F. splendens, the hypothesis has been borne out.
References
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–608
Barrett AJ (1986) The classes of proteolytic enzymes. In: Dalling MJ (ed) Plant proteolytic enzymes, vol. I. CRC Press, Boca Raton, FL, USA, pp 1–16
Birk EM, Vitousek PM (1986) Nitrogen availability and nitrogen use efficiency in loblolly pine stands. Ecology 67:69–79
Broadley MR, White PJ, Hammond JP, Zelko I, Lux A (2007) Zinc in plants. New Phytol. 173:677–702
Cannon WA (1905) On the transpiration of Fouquieria splendens. Bull Torrey Bot Club 32:397–414
Chapin FS III, Kedrowski RA (1983) Seasonal changes in nitrogen and phosphorus fractions and autumnal retranslocation in evergreen and deciduous taiga trees. Ecology 64:376–391
Chapin FS III, Moilanen L (1991) Nutritional controls over nitrogen and phosphorus resorption from Alaskan birch leaves. Ecology 72:709–715
Chapin FS III, Shaver GR, Kedrowski RA (1986) Environmental controls over carbon, nitrogen and phosphorus fractions in Eriophorum vaginatum in Alaskan tussock tundra. J Ecol 74:167–195
del Arco JM, Escudero A, Vega Garrido M (1991) Effects of site characteristics on nitrogen retranslocation from senescing leaves. Ecology 72:701–708
Feller U (2004) Proteolysis. In: Nooden LD (ed) Plant cell death processes. Elsevier Academic Press, San Diego, CA, pp 107–123
Heckathorn SA, DeLucia EH (1994) Drought-induced nitrogen retranslocation in perennial C4 grasses of tallgrass prairie. Ecology 75:1877–1886
Hocking PJ (1982) Effect of water stress on redistribution of nutrients from leaflets of narrow-leaved lupin (Lupinus angustifolius L.). Ann Bot 49:541–543
Hörtensteiner S, Feller U (2002) Nitrogen metabolism and remobilization during senescence. J Exp Bot 53:927–937
Jones JB Jr (1972) Plant tissue analysis for micronutrients. In: Mortvedt JJ, Giordana PM, Lindsay WL (eds) Micronutrients in agriculture. Soil Science Society of America, Madison, WI, pp 319–346
Jones ML (2004) Changes in gene expression during senescence. In: Nooden LD (ed) Plant cell death processes. Elsevier Academic Press, San Diego, CA, USA, pp 51–71
Jones JBJ, Wolf B, Mills HA (1991) Plant analysis handbook 1: methods of plant analysis and interpretation. Macro-Micro Publishing, Inc., Athens, GA, USA
Killingbeck KT (1985) Autumnal resorption and accretion of trace metals in gallery forest trees. Ecology 66:283–286
Killingbeck KT (1992) Inefficient nitrogen resorption in a population of ocotillo (Fouquieria splendens), a drought-deciduous desert shrub. Southwest Nat 37:35–42
Killingbeck KT (1996) Nutrients in senescing leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727
Killingbeck KT (2004) Nutrient resorption. In: Nooden LD (ed) Plant cell death processes. Elsevier Academic Press, San Diego, CA, USA, pp 215–226
Killingbeck KT, May JD, Nyman S (1990) Foliar senescence in an aspen (Populus tremuloides) clone: the response of element resorption to interramet variation and timing of abscission. Can J For Res 20:1156–1164
Killingbeck KT, Hammen-Winn SL, Vecchio PG, Goguen ME (2002) Nutrient resorption efficiency and proficiency in fronds and trophopods of a winter-deciduous fern, Dennstaedtia punctilobula. Int J Plant Sci 163:99–105
Kobe RK, Lepczyk CA, Iyer M (2005) Resorption efficiency decreases with increasing green leaf nutrients in a global data set. Ecology 86:2780–2792
Lersten NR, Carvey KA (1974) Leaf anatomy of ocotillo (Fouquieria splendens; Fouquieriaceae), especially vein endings and associated veinlet elements. Can J Bot 52:2017–2021
Lloyd FE (1905) The artificial induction of leaf formation in the ocotillo. Plant World 8:56–59
Mae (2004) Leaf senescence and nitrogen metabolism. In: Nooden LD (ed) Plant cell death Processes. Elsevier Academic Press, San Diego, CA, USA, pp 157–168
May JD, Killingbeck KT (1992) Effects of preventing nutrient resorption on plant fitness and foliar nutrient dynamics. Ecology 73:1868–1878
Milla R, Castro-Diez P, Maestro-Martinez M, Montserrat-Marti G (2005) Does the gradualness of leaf shedding govern nutrient resorption from senescing leaves in Mediterranean woody plants? Plant Soil 278:303–313
Minoletti ML, Boerner REJ (1994) Drought and site fertility effects on foliar nitrogen and phosphorus dynamics and nutrient resorption by the forest understory shrub Viburnum acerifolium L. Am Midl Nat 131:109–119
Morain S (1981) Soils. In: Williams JL, McAllister PE (eds) New Mexico in Maps, Univ. New Mexico Press, Albuquerque, NM, USA, pp 20–21
Morita K (1980) Release of nitrogen from chloroplasts during leaf senescence in rice (Oryza sativa L.). Ann Bot 26:297–302
Mukherjee KL (1969) Microelement composition of sugarcane leaves during their growth and senescence. J Indian Bot Sci 48:180–184
Murphy LS, Walsh LM (1972) Correction of micronutrient deficiencies with fertilizers. In: Mortvedt JJ, Girodano PM, Lindsay WL (eds) Micronutrients in agriculture, 1st edn. Soil Science Society of America, Madison, WI, USA, pp 347–387
Peoples MB, Dalling MJ (1988) The interplay between proteolysis and amino acid metabolism during senescence and nitrogen reallocation. In: Nooden LD, Leopold AC (eds) Senescence and aging in plants. Academic Press, San Diego, CA, pp 181–217
Rejmankova E (2005) Nutrient resorption in wetland macrophytes: comparison across several regions of different nutrient status. New Phytol 167:471–482
Richardson CJ, Ferrell GM, Vaithiyanathan P (1999) Nutrient effects on stand structure, resorption efficiency, and secondary compounds in Everglades sawgrass. Ecology 80:2182–2192
Richardson SJ, Peltzer DA, Allen RB, McGlone MS (2005) Resorption proficiency along a chronosequence: responses among communities and within species. Ecology 86:20–25
Ryan DF, Bormann FH (1982) Nutrient resorption in northern hardwood forests. BioSci 32:29–32
SAS Institute Inc. (2002) JMP Version 5. SAS Institute Inc., Cary, NC, USA
Scott FM (1932) Some features of the anatomy of Fouquieria splendens. Amer J Bot 19:673–678
Smith MW, Storey JB (1979) Zinc concentration of pecan leaflets and yield as influenced by zinc source and adjuvants. J Amer Soc Hort Sci 104:474–477
Sokal RR, Rohlf FJ (1981) Biometry, 2nd edn. W.H. Freeman and Company, San Francisco, CA, USA
Thomas RL, Sheard RW Jr, Moyer JR (1967) Comparison of conventional and automated procedures for nitrogen, phosphorus, and potassium analysis of plant material using a single digestion. J Agron 59:240–243
Underwood AJ (1997) Experiments in ecology: their logical design and interpretation using analysis of variance. Cambridge Univ. Press, Cambridge, UK
van Heerwaarden LM, Toet S, Aerts R (2003) Current measures of nutrient resorption efficiency lead to a substantial underestimation of real resorption efficiency: facts and solutions. Oikos 101:664–669
West NE, Klemmedson JO (1978) Structural distribution of nitrogen in desert ecosystems. In: West NE, Skujins J (eds) Nitrogen in desert ecosystems. Dowden, Hutchinson & Ross, Inc., Stroudsburg, PA, USA, pp 1–16
Whitford WG (2002) Ecology of desert systems. Academic Press, San Diego, CA, USA
Williams S (1984) Official methods of analysis of the asociation of official analytical chemists, 14th edn. Association of Official Analytical Chemists, Arlington, VA, USA
Woodwell GM (1974) Variations in the nutrient content of leaves of Quercus alba, Quercus coccinea, and Pinus rigida in the Brookhaven Forest from bud-break to abscission. Amer J Bot 61:749–753
Acknowledgements
I thank Michaela Lane-Sanchez for her exceptional work in the field, Walt Whitford for his sage counsel and logistical support, Rich Spellenberg and the Department of Biology at New Mexico State University for office space and staff support during the field and lab portions of this study, Kris Havstad for space, staff, and financial support at the USDA-ARS Jornada Experimental Range during the writing of this manuscript, Vince Gutschick for use of lab space and a Li-COR leaf area meter, and Alejandro Castellanos, Alfonso Escudero, Root Gorelick, and two anonymous reviewers for their insightful comments on a draft of this manuscript. I also thank the University of Rhode Island Foundation and the National Science Foundation (BSR-8604421 to KK) for their financial support of this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Alfonso Escudero.
Rights and permissions
About this article
Cite this article
Killingbeck, K.T. Can zinc influence nutrient resorption? A test with the drought-deciduous desert shrub Fouquieria splendens (ocotillo). Plant Soil 304, 145–155 (2008). https://doi.org/10.1007/s11104-007-9530-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-007-9530-z