Abstract
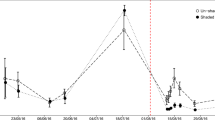
It remains a challenge to quantify and assess the importance of the direct plant below-ground flux of photosynthate carbon (C) to soil microorganisms, especially in forests because of the size of the plants and the great spatial heterogeneity of soils. We studied the importance of labile C inputs from trees on the respiratory activity of soil microorganisms by comparing the response of plots with and without girdled pine trees (Pinus sylvestris L.) to additions of C4-sucrose, thus enabling us to differentiate between utilization of endogenous C3-soil C sources and exogenous C4-sucrose. In both girdled and non-girdled plots the respiration rate after sucrose application, i.e. substrate induced respiration measured in the field, was on average ca. double that of basal respiration rate measured in the field. However, the C4-sucrose-induced increase in respiration of endogenous C3-C was significantly higher in non-girdled plots. Expression of C3-respiration as a percentage of induced respiration in the field showed that in girdled plots, C3-respiration decreased after sucrose addition and, consequently, the induced respiration in the field was totally C4-C based. A previous laboratory experiment found no increase in total respiration rate when C4-sucrose was added to the soil substrate of non-mycorrhizal and ectomycorrhizal pine plants. Hence, we see no reason to attribute the increased respiration to (mycorrhizal) roots. Thus, our results indicate that despite the alleged C limitation of the soil microorganisms there is a fraction of SOM, or C within the microbial biomass that is available to microbial metabolism if their C limitation is relieved by the supply of labile C. This fraction corresponds to roughly 10–20% of biomass C of the heterotrophic organisms and seems to become exhausted in the long-term absence of supply of photosynthate to roots.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- BRfield :
-
The basal respiration of the untreated undisturbed soil measured directly in the field
- IRfield :
-
Induced respiration in the field is SIRfield−BRfield
- SIRfield :
-
Substrate induced respiration in the field is the total respiration rate after addition of sucrose measured in the field
References
Alexander M (1977) Introduction to soil microbiology. John Wiley, New York, pp 134–233
Anderson JPE, Domsch KH (1978) A physiological method for the quantitative measurement of microbial biomass in soils. Soil Biol Biochem 10:215–221
Bhupinderpal-Singh, Nordgren A, Löfvenius MO, Högberg MN, Mellander PE, Högberg P (2003) Tree root and soil heterotrophic respiration as revealed by girdling of boreal Scots pine forest: extending observations beyond the first year. Plant Cell Environ 26:1287–1296
Chapin FS III, Matson PA, Mooney HA (2002) Principles of terrestrial ecosystem ecology. Springer, New York
Cheng WX, Zhang QL, Coleman DC, Carroll CR, Hoffman CA 1996 Is available carbon limiting microbial respiration in the rhizosphere? Soil Biol Biochem 28:1283–1288
Dalenberg JW, Jager G (1981) Priming effect of small glucose additions to 14C-labelled soil. Soil Biol Biochem 13:219–223
Dalenberg JW, Jager G (1989) Priming effect of some organic additions to 14C-labelled soil. Soil Biol Biochem 21:443–448
De Nobili M, Contin M, Mondini C, Brookes PC (2001) Soil microbial biomass is triggered into activity by trace amounts of substrate. Soil Biol Biochem 33:1163–1170
Ekblad A, Högberg P (2000) Analysis of δ13C of CO2 distinguishes between microbial respiration of added C4-sucrose and other soil respiration in a C3-ecosystem. Plant Soil 219:197–209
Ekblad A, Nordgren A (2002) Is growth of soil microorganisms in boreal forests limited by carbon or nitrogen availability? Plant Soil 242:115–122
Ekblad A, Nyberg G, Högberg P (2002) 13C-discrimination during microbial respiration of added C3-, C4- and 13C-labelled sugars to a C3-forest soil. Oecologia 131:245–249
Högberg P, Ekblad A (1996) Substrate-induced respiration measured in situ in a C3-plant ecosystem using additions of C4-sucrose. Soil Biol Biochem 28:1131–1138
Högberg MN, Högberg P (2002) Extramatrical ectomycorrhizal mycelium contributes one-third of microbial biomass and produces, together with associated roots, half the dissolved organic carbon in a forest soil. New Phytol 154:791–795
Högberg P, Nordgren A, Buchmann N, Taylor AFS, Ekblad A, Högberg MN, Nyberg G, Ottosson-Löfvenius M, Read DJ (2001) Large-scale forest girdling shows that current photosynthesis drives soil respiration. Nature 411:789–792
Jones DL, Hodge A, Kuzyakov Y (2004) Plant and mycorrhizal regulation of rhizodeposition. New Phytol 163:459–480
Keeling CD (1958) The concentration and isotopic abundances of atmospheric carbon dioxide in rural areas. Geochim Cosmochim Acta 13:322–334
Kelliher FM, Barbour MM, Hunt JE (2005) Sucrose application, soil microbial respiration and evolved carbon dioxide isotope enrichment under contrasting land uses. Plant Soil 268:233–242
Kuzyakov Y, Friedel JK, Stahr K (2000) Review of mechanisms and quantification of priming effects. Soil Biol Biochem 32:1485–1498
Mead R, Curnow RN, Hasted AM (1993) Statistical methods in agriculture and experimental biology. Chapman and Hall, London, p 415
O’Leary MH (1988) Carbon isotopes in photosynthesis. Bioscience 38:328–336
Pataki DE, Ehleringer JR, Flanagan LB, Yakir D, Bowling DR, Still CJ, Buchmann N, Kaplan JO, Berry JA (2003) The application and interpretation of Keeling plots in terrestrial carbon cycle research. Global Biogeochem Cy 17, 1022, doi:10.1029/2001GB001850
Paul EA, Clark FE (1996) Soil microbiology and biochemistry. Academic Press, San Diego, p 182
Phillips DL, Gregg JW (2001) Uncertainty in source partitioning using stable isotopes. Oecologia 127:171–179
Schulze WX, Gleixner G, Kaiser K, Guggenberger G, Mann M, Schulze ED (2005) A proteomic fingerprint of dissolved organic carbon and of soil particles. Oecologia 142:335–343
Subke JA, Hahn V, Battipaglia G, Linder S, Buchmann N, Cotrufo MF (2004) Feedback interactions between needle litter decomposition and rhizosphere activity. Oecologia 139:551–559
Tate RL (1995) Soil microbiology. John Wiley, New York, pp 103–256
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: D. E. Crowley
Rights and permissions
About this article
Cite this article
Göttlicher, S.G., Steinmann, K., Betson, N.R. et al. The dependence of soil microbial activity on recent photosynthate from trees. Plant Soil 287, 85–94 (2006). https://doi.org/10.1007/s11104-006-0062-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-006-0062-8