Abstract
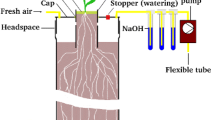
Rhizodeposition, i.e. the release of carbon into the soil by growing roots, is an important part of the terrestrial carbon cycle. However thein situ nature and dynamics of root-derived carbon in the soil are still poorly understood. Here we made an investigation of the latter in laboratory experiments using13CO2 pulse chase labelling of wheat (Triticum aestivum L.). We analyzed the kinetics of13C-labelled carbon and more specially13C carbohydrates in the rhizosphere. Wheat seedlings-soil mesocosms were exposed to13CO2 for 5 hours in controlled chambers and sampled repeatedly during two weeks for13C/C analysis of organic carbon. After a two-step separation of the soil from the roots, the amount of total organic13C was determined by isotope ratio mass spectrometry as well as the amounts of13C in arabinose, fructose, fucose, glucose, galactose, mannose, rhamnose and xylose. The amount and isotopic ratio of monosaccharides were obtained by capillary gas chromatography coupled with isotope ratio mass spectrometry (GC/C/IRMS) after trimethyl-silyl derivatization. Two fractions were analyzed : total (hydrolysable) and soluble monomeric (water extractable) soil sugars. The amount of organic13C found in the soil, expressed as a percentage of the total photosynthetically fixed13C at the end of the labelling period, reached 16% in the day following labelling and stabilised at 9% after one week. We concluded that glucose under the form of polymers was the dominant moietie of rhizodeposits. Soluble glucose and fructose were also present. But after 2 days, these soluble sugars had disappeared. Forty percent of the root-derived carbon was in the form of neutral sugars, and exhibited a time-increasing signature of microbial sugars. The composition of rhizospheric sugars rapidly tended towards that of bulk soil organic matter.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Amelung W, Cheshire M V, and Guggenberg G 1996 Determination of neutral and acidic sugars in soil by capillary gas-liquid chromatography after trifluoroacetic acid hydrolysis. Soil Biol. Biochem. 28, 1631–1639.
Chantigny M, Angers D and Beauchamp C 2000 Decomposition of de-inking paper sludge in agricultural soils as characterized by carbohydrate analysis. Soil Biol. Biochem. 32, 1561–1570.
Cheng W, Coleman D C, Carroll C R and Hoffman C A 1994 Investigating short-term carbon flows in the rhizospheres of different plant species, using isotope trapping. Agron. J. 86, 782–788.
Derrien D, Marol C, Balesdent J and Santaella C 2003 Measurements of the13C/12C ratio of soil-plant individual sugar by chromatography/combustion/isotope-ratio mass spectrometry of silylated derivatives. Rapid Commun. Mass Spectrom. 17, 2626–2631.
Fan T W M, Lane A N, Shenker M, Bartley J P, Crowley D and Higashi R M 2001 Comprehensive chemical profiling of gramineous plant root exudates using high-resolution NMR and MS. Phytochemistry 57, 209–221.
Grayston S J, Vaughan D and Jones D 1996 Rhizosphere carbon flow in trees, in comparison with annual plants: the importance of root exudation and its impact onmicrobial activity and nutrient availability. Appl. Soil Ecol. 5, 29–56.
Jones D L and Darrah P R 1996 Re-sorption of organic compounds by roots ofZea mays L and its consequences in the rhizosphere. 3. Characteristics of sugar influx and efflux. Plant Soil 178, 153–160.
Kuzyakov Y 2002 Review: Factors affecting rhizosphere priming effects. J. Plant Nutr. Soil Sci. 165, 382–396.
Larre-Larrouy M-C and Feller C 1997 Determination of carbohydrates in two ferralitic soils: analysis by capillary gas chromatography after derivatization by silylation. Soil Biol. Biochem. 29, 1585–1589.
Macko S A, Ryan M and Engel M H 1998 Stable isotopic analysis of individual carbohydrates by gas chromatographic/combustion/isotope ratio mass spectrometry. Chemical Geology 152, 205–210.
Meharg A A and Killham K 1995 Loss of exudates from roots of perennial ryegrass inoculated with a range of micro-organisms. Plant Soil 170, 345–349.
Merbach W, Mirus E, Knof G, Remus R, Ruppel S, Russow R, Gransee A and Schulze J 1999 Release of carbon and nitrogen compounds by plant roots and their possible ecological importance. J. Plant Nutr. Soil Sci. 162, 373–383.
Miltner A, Richner H H, Kopinke F D and Kästner M 2003 Assimilation of CO2 C in soil and its distribution on different soil organic matter components.In Proceedings of the international conference ‘Mecanisms and regulation of organic matter stabilisation in soils’, October 5–8, 2003, Munich, Germany, 21 pp.
Neumann G and Römheld V 2001 The release of roots exudates as affected by plant’s physiological status.In The rhizosphere, biochemistry and organic substances at the soil-plant interface. Eds. R Pinton, Z Varanini and P Nannipieri. pp. 41–94. Marcel Dekker, Inc New-York, Basel.
Nguyen C 2003 Rhizodeposition of organic C by plants: mechanisms and controls. Agronomie 23, 375–396.
Oades J M, Kirkman M A and Wagner G H 1970 The use of gas-liquid chromatography for the determination of sugar extracted from soils by sulfuric acid. Soil Sci. Soc. Am. Proc. 34, 230–235.
Ogier S, Disnar J R, Alberic P and Bourdier G 2001 Neutral carbohydrate geochemistry of particulate material (trap and core sediments) in an eutrophic lake (Aydat, France). Organic Geochem. 32, 151–162.
Osborn H M I, Lochey F, Mosley L and Read D 1999 Analysis of polysaccharides and monosaccharides in the root mucilage of maize (Zea mays L.) by gas chromatography. J. Chromatogr. A 831, 267–276.
Ostle N, Whiteley A S, Bailey M J, Sleep D, Ineson P and Manefield M 2003 Active microbial RNA turnover in a grassland soil estimated using a (CO2)-C-13 spike. Soil Biol. Biochem. 35, 877–885.
Palta J A and Gregory P J 1997 Drought affects the fluxes of carbon to roots and soil in13C-pulse labelled plants of wheat. Soil Biol. Biochem. 29, 1395–1404.
Pinton R, Varanini Z and Nannipieri P 2001 The rhizosphere, biochemistry and organic substances at the soil-plant interface. Marcel Dekker, Inc New-York, Basel, 424 pp.
Rovira A D, Foster R C and Matin J K 1979 Note on terminology: origin, nature and nomenclature of organic materials in the rhizosphere.In The soil-root interface. Eds. J L Harley and R S Russel. pp. 1–4. Academic Press London.
Swinnen J, Van Veen J A and Merckx R 1994a14C pulselabelling of field-frown spring wheat: an evaluation of its use in rhizosphere carbon budget estimation. Soil Biol. Biochem. 26, 161–170.
Swinnen J, Van Veen J A and Merckx R 1994b Rhizosphere carbon fluxes in field-grown spring wheat: model calculation based on14C partitioning after pulse labelling. Soil Biol. Biochem. 26, 171–182.
van Dongen B E, Schouten S and Sinnoghe Damsté J S 2001 Gas chromatography/combustion/isotope-ratio-monitoring mass spectrometric analysis of methylboronic derivatives of monosccharides: a new method for determinignatural13C abundance of carbohydrates. Rapid Commun. Mass Spectrom. 15, 496–500.
Warembourg F R and Billès G 1979 Estimating carbon transfers in the plant rhizosphereIn The soil-root interface. Eds. J L Harley and R S Russel. pp. 183–196. Academic Press London.
Warembourg F R and Paul E A 1977 Seasonal transfers of assimilated14C in grassland: Plant production and turnover, soil and plant respiration. Soil Biol. Biochem. 9, 295–301.
Warembourg F R, Roumet C and Lafont F 2003 Differences in rhizosphere carbon-partitioning among plant species of different families. Plant Soil 256, 347–357.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Derrien, D., Marol, C. & Balesdent, J. The dynamics of neutral sugars in the rhizosphere of wheat. An approach by13C pulse-labelling and GC/C/IRMS. Plant Soil 267, 243–253 (2004). https://doi.org/10.1007/s11104-005-5348-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11104-005-5348-8