Abstract
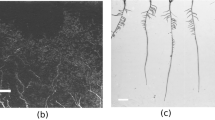
The objective of the present work was to develop a method to distinguish between metabolically inactive and active parts of plant roots. White clover (Trifolium repens L.) roots were stained with 2,3,5-triphenyltetrazolium chloride (TTC) followed by root colour classification with an interactive scanner-based image analysis programme (WinRHIZO). Roots inactivated by boiling were unstained and pale brown, whereas fresh samples with predominantly metabolically active roots turned dark red, red or pale red after staining. A small amount of very young, presumable active roots (0.8% of total active root length) failed to stain red with TTC. The colour analysis of inactive and active roots was based on four colour classes for boiled roots and seven classes for fresh roots, respectively, as defined upon visual examination of images. Pixel colours falling outside the defined classes were allocated to the nearest defined class – an option that increased objectivity and stability and reduced the required number of colour classes. For the fresh white clover roots, 75–86% of the total root length was determined as active, while 3–7% of the boiled roots fell into the same category. The percentage of total root length measured by WinRHIZO that was identified as metabolically active was linearly correlated with the percentage of fresh roots in mixtures of fresh and boiled roots (R2=0.99). Colour classes chosen à priori from one experiment could be used to distinguish fairly satisfactorily between active and inactive roots of another white clover cultivar grown under other conditions, but failed to classify activity in ryegrass (Lolium multiflorum Lam.) root samples. In the latter case, colour classes needed to be re-defined in order to produce reliable data. Our work shows that WinRHIZO’s colour identification sub-module provides a new promising tool to classify root activity as identified after staining with TTC, but colour classes must be carefully evaluated on every new occasion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
T J Bouma K Hengst B P Koutstaal J Soelen Particlevan (2002) ArticleTitleEstimating root lifespan of two grasses at contrasting elevation in a salt marsh by applying vitality staining on roots from in-growth cores Plant Ecol. 165 235–245 Occurrence Handle10.1023/A:1022290530303
T J Bouma K L Nielsen B Koutstaal (2000) ArticleTitleSample preparation and scanning protocol for computerised analysis of root length and diameter Plant Soil 218 185–196 Occurrence Handle10.1023/A:1014905104017 Occurrence Handle1:CAS:528:DC%2BD3cXhvVGrtb8%3D
A Clemensson-Lindell (1994) ArticleTitleTriphenyltetrazolium chloride as an indicator of fine-root vitality and environmental stress in coniferous forest stands: applications and limitations Plant Soil 159 297–300 Occurrence Handle1:CAS:528:DyaK2cXivFehtLk%3D
L H Comas D M Eissenstat A N Lakso (2000) ArticleTitleAssessing root death and root system dynamics in a study of grape canopy pruning New Phytol. 147 171–178 Occurrence Handle1:CAS:528:DC%2BD3cXms1yltLw%3D
C Costa L M Dwyer C Hamel D F Muamba X L Wang L Nantais D L Smith (2001) ArticleTitleRoot contrast enhancement for measurement with optical scanner-based image analysis Can. J. Bot. 79 23–29
K Fiala (1990) ArticleTitleLive and dead underground plant biomass in a natural meadow hydrosere Folia Geobotanica 25 112–135
K Fiala (2000) ArticleTitleProportion of living biomass in the total dry mass of belowground organs of various plant communities Preslia, Praha 72 73–85
WA Jacques RH Schwass (1957) ArticleTitleRoot development in some common New Zealand pasture plants. VII.Seasonal root placement in perennial ryegrass (Lolium perenne), Italian ryegrass (L. multiflorum) and tall fescue (Festuca arundinacea) N. Zeal. J. Sci. Technol. A. 37 569–583
J D Joslin G S Henderson (1984) ArticleTitleThe determination of percentages of living tissue in woody fine root samples using triphenyltetrazolium chloride Forest Sci. 30 965–970
D P Knievel (1973) ArticleTitleProcedure for estimating ratio of live and dead root dry matter in root core samples Crop Sci. 13 124–126
D A Musser A R Oseroff (1994) ArticleTitleThe use of tetrazolium salts to determine sites of damage to the mitochondrial electron-transport chain in intact-cells following in-vitro photodynamic therapy with photofrin-II Photobiol. 59 621–626 Occurrence Handle1:CAS:528:DyaK2cXkslOntLc%3D
Regent Instruments Inc. 2002 Win/MacRHIZO 2002a Basic, Reg & Pro For Washed Root Measurement. Quebec, Canada, 92 pp.
M Ruf I Brunner (2003) ArticleTitleVitality of tree fine roots: reevaluation of the tetrazolium test Tree Physiol. 23 257–263 Occurrence Handle12566261
J J Schuurman M A J Goedewaagen (1971) Methods for the Examination of Root Systems and Roots Centre for Agricultural Publishing and Documentation Wageningen 86
T F Slater B Sawyer U Sträuli (1963) ArticleTitleStudies on a succinate-neotetrazolium reductase system of rat liver III.Points of coupling of four different tetrazolium salts Biochim. Biophys. Acta. 77 383–393 Occurrence Handle1:CAS:528:DyaF2cXlt1Wk Occurrence Handle14089413
A A Titlyanova I P Romanova N P Mironycheva-Tokareva (1999) ArticleTitlePattern and process in above-ground and below-ground components of grassland ecosystems J. Veg. Sci. 10 307–320
Z Wang W H Burch P Mou R J Jones R J Mitchell (1995) ArticleTitleAccuracy of visible and ultraviolet light for estimating live root proportions with minirhizotrons Ecology 76 2330–2334
KJ Ward B Klepper RW Rickman RR Allmaras (1978) ArticleTitleQuantitative estimation of living wheat-root lengths in soil cores Agron. J. 70 675–677
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stūrīte, I., Henriksen, T.M. & Breland, T.A. Distinguishing between metabolically active and inactive roots by combined staining with 2,3,5-triphenyltetrazolium chloride and image colour analysis. Plant Soil 271, 75–82 (2005). https://doi.org/10.1007/s11104-004-2027-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11104-004-2027-0