Abstract
Hybrid necrosis is a well-known reproductive isolation mechanism in plant species, and an autoimmune response is generally considered to trigger hybrid necrosis through epistatic interaction between disease resistance-related genes in hybrids. In common wheat, the complementary Ne1 and Ne2 genes control hybrid necrosis, defined as type I necrosis. Two other types of hybrid necrosis (type II and type III) have been observed in interspecific hybrids between tetraploid wheat and Aegilops tauschii. Another type of hybrid necrosis, defined here as type IV necrosis, has been reported in F1 hybrids between Triticum urartu and some accessions of Triticum monococcum ssp. aegilopoides. In types I, III and IV, cell death occurs gradually starting in older tissues, whereas type II necrosis symptoms occur only under low temperature. To compare comprehensive gene expression patterns of hybrids showing growth abnormalities, transcriptome analysis of type I and type IV necrosis was performed using a wheat 38k oligo-DNA microarray. Defense-related genes including many WRKY transcription factor genes were dramatically up-regulated in plants showing type I and type IV necrosis, similarly to other known hybrid abnormalities, suggesting an association with an autoimmune response. Reactive oxygen species generation and necrotic cell death were effectively inhibited by ZnCl2 treatment in types I, III and IV necrosis, suggesting a significant association of Ca2+ influx in upstream signaling of necrotic cell death in wheat hybrid necrosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hybrid incompatibility between two diverging lineages acts as a postzygotic reproductive barrier, and plays significant roles in intraspecific differentiation and plant speciation (Bomblies and Weigel 2007; Rieseberg and Willis 2007). Most types of hybrid incompatibility are caused by epistatic interaction of at least two loci or alleles, a basis of the Dobzhansky–Muller model (Bomblies and Weigel 2007; Presgraves 2010). Hybrid necrosis is a major incompatibility observed in higher plants, and is generally considered to be triggered by an autoimmune response. The causative genes of hybrid necrosis have been isolated in Arabidopsis, lettuce and rice, and commonly encode disease resistance-related proteins including NB-LRR-type resistance (R) proteins and R-interacting proteins (Alcázar et al. 2009, 2010; Bomblies et al. 2007; Chae et al. 2014; Jeuken et al. 2009; Yamamoto et al. 2010). In addition, a number of defense-related genes are up-regulated in hybrid plants exhibiting necrotic symptoms in Arabidopsis and Nicotiana (Bomblies et al. 2007; Masuda et al. 2007).
In intraspecific crosses of common wheat (Triticum aestivum L.), hybrid necrosis caused by interaction between dominant alleles of the Ne1 and Ne2 loci is frequently observed (Hermsen 1963; Tsunewaki 1960, 1970). The complementary Ne1 and Ne2 loci are respectively located on chromosome arms 5BL and 2BS, and the induction of necrotic cell death by Ne1–Ne2 is called type I hybrid necrosis (Chu et al. 2006; Takumi et al. 2013; Tsunewaki 1960, 1992). Type I necrosis functions as a reproductive barrier contributing to intraspecific differentiation mainly between European and American varieties and middle Asian and African varieties in common wheat (Pukhalskiy et al. 2000). Distribution of the Ne1 and Ne2 dominant alleles is postulated to be due to selection advantages such as rust fungus resistance (Bomblies and Weigel 2007), although no direct evidence proving a relationship between type I necrosis and disease resistance has been reported. In leaves of wheat showing type I necrosis, superoxide is highly accumulated and antioxidant enzymes show high levels of activity (Dalal and Khanna-Chopra 2001; Khanna-Chopra et al. 1998; Sugie et al. 2007).
Hybrid necrosis has also been reported in interspecific crosses of wheat relatives (Nishikawa 1960; Roy 1955; Sears 1944). In interspecific hybrids showing necrotic symptoms, interplay between the distinct nuclear genomes triggers the incompatible responses, and causal genes are believed to be located in the genome of both parental species. Common wheat is an allohexaploid species (AABBDD genome) that originated through allohexaploidization between cultivated tetraploid wheat Triticum turgidum L. (AABB genome) and the wild diploid relative Aegilops tauschii Coss. (DD genome) (Matsuoka 2011). In many interspecific crosses reflecting the allopolyploid speciation process of common wheat, abnormal growth phenotypes have been found (Matsuoka et al. 2007; Nishikawa 1960, 1962a, b), and the abnormalities in the triploid hybrids could act as postzygotic hybridization barriers inhibiting the formation of common wheat. Two types of hybrid necrosis, types II and III, are observed in many F1 triploid plants (ABD genome) of tetraploid wheat and Ae. tauschii (Mizuno et al. 2010, 2011; Nishikawa 1960, 1962b). Cell death occurs gradually beginning with older tissues in hybrid lines showing type III necrosis, similarly to type I necrosis, whereas type II necrosis lines show a necrotic phenotype under low temperature conditions (Mizuno et al. 2011; Nishikawa 1960). In addition to necrotic symptoms, low temperature represses mitotic cell division in the shoot apices of hybrid plants displaying type II necrosis (Mizuno et al. 2011). Causal D-genome genes, Net2 for type II necrosis and Nec1 for type III necrosis, are respectively assigned to chromosomes 2D and 7D, but chromosome localization of the complementary genes, Net1 and Nec2, on the AB genome is unknown (Mizuno et al. 2010, 2011). A number of defense-related genes are up-regulated in both type II and III necrosis, and some are commonly up-regulated (Mizuno et al. 2010, 2011). Thus, molecular mechanisms involved in expression of the hybrid necrosis phenotypes share signal pathways related to plant disease responses. Two types of hybrid chlorosis, one with severe and one with mild symptoms, are observed to a limited extent in triploid hybrids and synthetic hexaploid wheat lines (Mizuno et al. 2010; Nakano et al. 2015). Unlike type II and III necrosis and the two types of hybrid chlorosis, severe growth abortion (SGA) is always lethal, because SGA-exhibiting ABD triploids cease development and growth after expansion of the second or third leaves (Mizuno et al. 2010; Nishikawa 1960). In spite of their phenotypic difference from type II and III hybrid necrosis, a number of defense-related genes are highly up-regulated in seedlings of SGA-exhibiting hybrids and leaves of synthetic wheat lines with hybrid chlorosis symptoms (Hatano et al. 2012; Nakano et al. 2015).
Hybrid necrosis is frequently observed in interspecific crosses of two wild diploid Triticum species, Triticum monococcum subspecies aegilopoides (Link) Thell. (AmAm genome) and Triticum urartu Tumanian ex Gandilyan (AA genome) (Yamagishi 1987). This hybrid necrosis, called here type IV necrosis, is hypothetically triggered by epistatic interaction between dominant alleles of Ned1 in the nuclear genome of T. monococcum ssp. aegilopoides and Ned2 in the T. urartu genome (Yamagishi 1987). All examined T. urartu accessions contain the dominant allele of Ned2, whereas Ned1-carrier and non-carrier accessions are mixed in T. monococcum ssp. aegilopoides (Yamagishi 1987). The necrotic phenotype in type IV necrosis appears to resemble the symptoms in type I and III necrosis. However, no additional information on type IV necrosis has been reported.
Gene expression profiles are not necessarily consistent with each other, but some genes are mutually up- and down-regulated in type II and III necrosis and SGA of ABD triploids (Hatano et al. 2012; Mizuno et al. 2011). Here, we performed transcriptome analyses for type I and IV necrosis to elucidate the mechanisms inducing necrotic symptoms at the transcriptional level and compared gene expression profiles with those in plants showing growth abnormalities in ABD triploid and synthetic wheat to clarify the common features underlying the various types of hybrid necrosis in Triticum.
Materials and methods
Plant materials
Three accessions of common wheat were used for analysis of type I necrosis. The common wheat variety S-615 has no dominant alleles for Ne1 and Ne2. The near-isogenic lines (NILs) carrying dominant alleles Ne1 and Ne2 are designated Ne1-S615 and Ne2-S615, respectively (Tsunewaki and Koba 1979). The Ne1 and Ne2 alleles originated from common wheat cultivars Prelude and Kharkov, respectively (Tsunewaki 1960), and these dominant alleles were introduced into the S-615 genetic background through 10 backcrosses.
To generate interspecific hybrids with type IV necrosis, we used three accessions of T. monococcum subspecies aegilopoides (Link) Thell. (syn. T. boeoticum Boiss.), and five accessions of wild diploid T. urartu Tumanian ex Gandilyan. KU-101-3 and KU-8001 were used as Ned1 carriers, and KU-3620 as a non-carrier in T. monococcum ssp. aegilopoides. For T. urartu, KU-199-5, KU-199-8, KU-199-9, KU-199-11 and KU-199-16 were used and crossed with pollen of T. monococcum ssp. aegilopoides accessions. Many of the F1 seeds from T. urartu crossed with pollens of T. monococcum ssp. aegilopoides failed to germinate, as reported previously (Yamagishi 1987; Fricano et al. 2014); some plants that germinated were used for phenotyping and RNA extraction.
For studies of type III necrosis, we used two synthetic hexaploid wheat lines showing an early flowering phenotype. They were produced from two cross combinations between a tetraploid wheat accession, T. turgidum ssp. durum (Desf.) Husn. cv. Langdon (Ldn), and two A. tauschii Coss. accessions, KU-2059 and KU-2828 (Mizuno et al. 2010). The Ldn/KU-2059 line showed normal growth (wild type; WT) and the Ldn/KU-2828 line showed type III necrosis.
Transcriptome analysis
Two F1 hybrids of common wheat, Ne2-S615/S-615 for WT and Ne2-S615/Ne1-S615 for type I necrosis, and two interspecific hybrids of diploid wheat, KU-3620/KU-199-11 for WT and KU-101-3/KU-199-5 for type IV necrosis, were used for microarray analysis. Total RNA was extracted using an RNeasy Plant Mini kit (Qiagen, Hilden, Germany) from leaves of seedlings grown at normal temperature (23 °C) for 7 days. Necrotic symptoms started to appear on the 1st (lowest) leaves in the 7-week-old seedlings of the type I and type IV necrosis hybrids. A KOMUGI 38k oligonucleotide DNA microarray (Agilent Technologies, Santa Clara, CA) was supplied by the National BioResource Project (NBRP)-Wheat, Japan (https://www.nbrp.jp) for analysis. Detailed information on the 38k microarray platform can be found in Kawaura et al. (2008) and the Gene Expression Omnibus (GEO) database of the National Center for Biotechnology Information (NCBI) website under GPL9805. Cyanine-3 (Cy3) labeled cRNA was prepared from 0.5 μg RNA using the Quick Amp Labeling kit (Agilent Technologies) according to the manufacturer’s instructions. Hybridization of Cy3-labeled cRNA to the microarrays and washing were performed using a Gene Expression Hybridization kit and Gene Expression Wash Pack (Agilent Technologies). Signal intensities were detected by Feature Extraction Software 9.5 (Agilent Technologies). Slides were scanned immediately after washing on the Agilent DNA Microarray Scanner (G2505B) using one color scan setting for 1×44k array slides. Scanned images were analyzed with Feature Extraction Software using default parameters to obtain background-subtracted and spatially detrended processed signal intensities. Two independent experiments were conducted for each sample. All microarray data were deposited as series GSE64342 in the NCBI GEO database (http://www.ncbi.nlm.nih.gov/geo/), including supplementary files, GSM1568808 through GSM1568811 for type I necrosis and GSM1568812 through GSM1568815 for type IV necrosis.
Functions of probes and genes were predicted by BLASTx searches (E value <1e−3) against the NCBI non-redundant protein database. Based on the 20 top BLASTx hits, Gene Ontology (GO) annotation was performed using the Blast2GO algorithm (Conesa et al. 2005). Annotations were refined using the ANNEX function (Myhre et al. 2006) and mapped to plant-specific GO-Slim terms, both available in Blast2GO. GO terms of differentially expressed (fold change ≥3 or ≤1/3) probes were extracted and compared.
For comparison of the microarray data for type I and type IV necrosis with previously reported data for growth abnormalities in ABD triploids and synthetic wheat hexaploids, data sets including GSE19613, GSE24566, GSE33357 and GSE59640 in the NCBI GEO were respectively used for type III necrosis, type II necrosis, SGA and hybrid chlorosis. RNA samples were extracted from leaves and crown tissues of synthetic hexaploids after 6 weeks of incubation at 4 °C for plant lines with a type II necrosis phenotype, or with no low-temperature treatment for other plants (Hatano et al. 2012; Mizuno et al. 2010, 2011; Nakano et al. 2015). For the analyses of type II and type III necrosis and hybrid chlorosis, the RNA samples were extracted from leaves starting to appear the abnormal growth symptoms (Mizuno et al. 2010, 2011; Nakano et al. 2015). In total, 22,018 probes were selected from the 38k microarray based on evaluation of their signal intensity (non-saturated uniform signal greater than background signal), and used for the comparison. A heat map was constructed using the function “heatmap.2” of the R statistical package “gplots” (version 2.11.0) (Warnes 2012). Hierarchical clustering of samples and probes was performed using the “hclust” function of the “stats” package in R (version 2.15.2) with the agglomeration method set to average (R Core Team 2012). The distance between samples and probes was calculated using the function “Dist”, available in the package “amap” (version 0.8.7) (Lucas 2011), based on Pearson’s correlation.
Measurement of photosynthetic activity
A portable JUNIOR-PAM fluorometer (Heinz Walz GmbH, Effeltrich, Germany) was used for measurement of the maximum photochemical quantum yield of photosystem II (Fv/Fm). The first leaves of 1-, 2- and 3-week-old WT and hybrid necrosis plants were incubated in the dark for 1 h. Fv/Fm was measured at ten independent locations on each leaf blade of three F1 plants according to the instructions in the operating manual for the fluorometer. For type IV necrosis, three F1 plants for each cross were analyzed, and the first leaves of 4-, 6- and 8-day-old WT and hybrid necrosis plants were used for measurement of photosynthetic activity.
Quantitative reverse transcription (qRT-PCR) analysis
qRT-PCR was performed using RNA isolated from leaves of WT and hybrid necrosis plants. Each plant was grown at 23 °C, and total RNA was extracted using Sepasol-RNA I (Nacalai Tesque, Kyoto, Japan). First-strand cDNA was synthesized from DNase I-treated RNA samples using ReverTra Ace Reverse Transcriptase (Toyobo, Osaka, Japan) and an oligo(dT)20 primer. Accumulation of transcripts of each gene was detected using a LightCycler 480 Real-Time PCR System (Roche Diagnostics, Mannheim, Germany) with THUNDERBIRD SYBR qPCR Mix (Toyobo) and gene-specific primer sets. The gene-specific primer sets for qRT-PCR are listed in Supplemental Table 1. The Actin gene was used as an internal control, and relative expression was calculated as 2−ΔΔCt (Livak and Schmittgen 2001), representing the value relative to the transcript levels in leaves of WT (Ne2-S615/S-615 or KU-3620/KU-199-16). Mean values were calculated based on three technical replicates.
Zinc chloride treatment and trypan blue and diaminobenzidine (DAB) staining
Each hybrid plant was grown at 23 °C, watering every day with 0.2 % Hyponex (Hyponex Japan, Osaka, Japan) solution with or without 0.02 mM ZnCl2, which functions as a calcium antagonist. The ZnCl2-treated plants were used for staining with trypan blue and DAB, measurement of photosynthetic activity and extraction of RNA.
Trypan blue was used for detection of dead cells. Leaf samples were submerged in 0.5 % trypan blue staining solution (Nacalai Tesque). After boiling for 1 min, the samples were incubated at room temperature for 25 min and washed with water containing 2.5 mg/mL chloral hydrate. DAB (Wako, Osaka, Japan) was used for H2O2 detection. Leaf samples were infiltrated with 2 mg/mL DAB solution (pH 3.8) for 1 h, and then treated with 99 % ethanol for chlorophyll removal. The trypan blue- and DAB-stained leaves were observed under bright field illumination under an Olympus BX60 microscope (Olympus, Tokyo, Japan).
Results
Phenotypes of type I and type IV necrosis
Hybrid plants showing type I necrosis were generated by crossing Ne1-S615 and Ne2-S615 NILs. All of the F1 plants clearly showed a hybrid necrosis phenotype of gradual premature death of leaves or plants (Fig. 1a). At the four-leaf stage, F1 plants with type I necrosis exhibited complete necrotic cell death of the first leaf. Type IV necrosis plants were produced by interspecific crossing between KU-101-3 and KU-199-16, and all F1 plants exhibited necrotic symptoms at the seedling stage (Fig. 1b). The first leaves showed complete cell death at the two-leaf stage. F1 plants showing type I or IV necrosis were killed during the vegetative growth phase at normal ambient temperature (23 °C).
Transcriptome analysis of type I and type IV necrosis. a Leaves of hexaploid wheat hybrids with (lower) and without (upper) type I necrosis symptoms. b Leaves of interspecific hybrids of two wild diploid wheat species with (lower) and without (upper) type IV necrosis symptoms. c, d Summary of microarray analyses. Up- and down-regulated genes in type I (c) and type IV (d) necrosis were categorized into 17 functional categories
According to a previous report (Yamagishi 1987), some wild einkorn wheat accessions show hybrid necrosis in interspecific crosses with T. urartu accessions, and others exhibit a WT phenotype without any necrotic symptoms. We confirmed appearance of the necrotic phenotype in some cross combinations between wild einkorn wheat and T. urartu (Table 1). KU-3620 showed a WT phenotype without any necrotic symptoms in all examined crosses with four T. urartu accessions, whereas necrotic symptoms were clearly observed in leaves of hybrid plants between two other ssp. aegilopoides accessions and T. urartu accessions.
Microarray analysis of type I and type IV necrosis
To comprehensively compare gene expression profiles between WT (Ne2-S615/S-615) and type I necrosis plants (Ne2-S615/Ne1-S615), we analyzed the transcriptomes using a wheat-specific 38k oligo DNA microarray (Kawaura et al. 2008). After hybridization with the RNA samples, probes showing significant (P < 0.05) difference in signal intensity compared to the WT were defined as either up- or down-regulated genes. Of the 37,826 probes on the wheat microarray, 405 (1.1 %) and 1254 (3.3 %) probes were respectively regarded as genes up- and down-regulated in leaves of the type I necrosis plants compared with WT plants (Supplemental Table 2). Based on a BlastX search against the NCBI nr protein database, 1219 differentially expressed probe sequences were assigned to one or more GO annotations. GO analysis showed that the up- and down-regulated genes could be categorized into 17 functional categories (Fig. 1c). Expression of genes related to carbohydrate metabolism, defense and transport were dramatically altered, and many photosynthesis-related genes were down-regulated in leaves of type I necrosis plants.
Similarly, gene expression profiles were compared between WT (KU-3620/KU-199-11) and type IV necrosis plants (KU-101-3/KU-199-5). Of the 37,826 microarray probes, 1323 (3.5 %) and 2805 (7.4 %) probes were respectively regarded as genes up- and down-regulated in leaves of type IV necrosis plants compared with WT plants (Supplemental Table 3). Up- and down-regulated probes were encountered more in type IV necrosis than in type I necrosis. GO annotations of the 2661 differentially expressed probe sequences showed that the up- and down-regulated genes in type IV necrosis could be categorized into 16 functional categories that appeared in type I necrosis (Fig. 1d). The accumulation of transcripts related to carbohydrate metabolic processes, stress and transport was dramatically up-regulated in leaves of type IV necrosis plants. As found in type I necrosis, a number of photosynthesis-related genes were down-regulated in type IV necrosis.
Several transcription factor genes, encoding MADS-box, WRKY, NAC and bHLH family proteins, were highly up-regulated in leaves of F1 plants showing type I or IV necrosis (Tables 2, 3). Many WRKY-type transcription factor genes, which play important roles in immune responses, senescence, and various other processes in plants (Rushton et al. 2010; Ishihama and Yoshioka 2012), were found among the up-regulated transcription factor genes in both types of hybrid necrosis. In addition, transcripts of a number of defense-related genes, including chitinase, hypersensitive response protein and sulfur-rich/thionin-like protein genes, accumulated abundantly in F1 plants showing type I or IV necrosis (Supplemental Tables 4 and 5). Up-regulation of reactive oxygen species (ROS)-related genes, such as curcuminoid synthase-like, oxalate oxidase and germin-like genes, was also observed in type I necrosis. In both types of hybrid necrosis plants, photosynthesis-related genes showed a high rate of down-regulation, including the genes for chlorophyll a/b binding protein and ribulose-1,5-bisphosphate carboxylase (Supplemental Tables 6 and 7).
Photosynthetic activity in type I and type IV necrosis
To examine the effects of down-regulation of photosynthesis-related genes, photosynthetic activities were compared between the WT and hybrid necrotic plants. Photosystem II activity of F1 plants showing type I necrosis was significantly reduced in the first leaves of 1-, 2- and 3-week-old seedlings compared to WT F1 plants without any necrotic symptoms (Fig. 2a). A reduction of photosystem II activity was also observed in three cross combinations showing type IV necrosis (Fig. 2b). The Fv/Fm values in F1 plants showing type IV necrosis and grown for 6 and 8 days were significantly different from corresponding values for WT F1 plants. These observations were consistent with the down-regulation of photosynthesis-related genes in plants showing hybrid necrosis.
Comparison of chlorophyll fluorescence between wild-type (WT) and necrosis-showing hybrid plants. Means with standard deviations were calculated from data of nine experiments. Student’s t test was used to test for statistical significance (*P < 0.05, **P < 0.01, ***P < 0.001) between WT and necrosis-showing hybrid plants. a Type I necrosis. The first leaves of 1-, 2- and 3-week-old seedlings were incubated in the dark and then used to calculate the ratio of variable to maximum fluorescence (Fv/Fm). b Type IV necrosis. The first leaves of 4-, 6- and 8-day-old seedlings were incubated in the dark and then used to calculate Fv/Fm
Expression analysis of defense-related genes in type I and type IV necrosis
To validate the microarray data, qRT-PCR was conducted for 10 genes, nine up- and one down-regulated (Supplemental Table 8). For convenience, the progression of symptoms was classified into three levels in leaves of type I necrosis-showing seedlings, depending on the observed extent of necrotic symptoms (Fig. 3a). In WT F1 plants, total RNA was extracted from leaves of seedlings at the same age as in type I necrosis. Transcripts of four defense-related genes were more markedly accumulated at level 2 in F1 plants showing type I necrosis than in WT F1 plants (Fig. 3b), indicating that the up-regulation of defense-related genes occurred before the necrotic symptoms appeared in type I necrosis plants. A similarly dramatic up-regulation was also observed at level 2 for three of four WRKY transcription factor genes in type I necrosis plants (Fig. 3c). The catalase gene, which functions in ROS alleviation, was highly induced at level 3 in type I necrosis plants (Fig. 3d). Rapid down-regulation of the photosynthesis-related gene PsbB was observed at level 2 before necrotic symptoms appeared in type I necrosis plants (Fig. 3e).
Comparison of accumulation of transcripts of the indicated genes between WT and necrosis-showing plants in common wheat. a Three levels of type I necrosis symptoms on leaves. Total RNA was extracted from leaves showing each level of necrosis. b Transcript accumulation patterns of four up-regulated genes related to the defense response from qRT-PCR. The transcript levels are shown as values relative to those in level 1 leaves of the WT line. Means ± standard deviations were calculated from data in three experiments. The Actin gene was used as an internal control. c Transcript accumulation patterns of four up-regulated WRKY transcription factor genes. d Transcript accumulation patterns of an up-regulated gene related to ROS alleviation. e Transcript accumulation patterns of a down-regulated gene related to photosynthesis
qRT-PCR analysis was also performed to compare the expression patterns of the 10 up-regulated genes and one down-regulated gene between two WT lines of wild diploid wheat and three F1 lines showing type IV necrosis. Total RNA was extracted from the first leaves of 4-, 6- and 8-day-old seedlings. The four defense-related gene transcripts were more abundantly accumulated in type IV necrosis plants than in WT (Fig. 4a). Transcript levels of the four WRKY transcription factor genes and the ROS-alleviation gene were markedly higher 4 days after sowing in type IV necrosis plants than in WT, and then gradually decreased in the type IV necrosis plants (Fig. 4b, c). Transcript accumulation of PsbB was lower in three F1 lines with type IV necrosis than in WT (Fig. 4d).
Comparison of accumulation of transcripts of the indicated genes between WT and necrosis-showing plants in wild diploid wheat. Total RNA was extracted from the first leaves of 4-, 6-, 8-day-old seedlings and used for qRT-PCR. The transcript levels are shown as values relative to those in 4-day-old seedlings of WT (T. urartu KU199-11). Means ± standard deviations were calculated from data in three experiments. The Actin gene was used as an internal control. a Transcript accumulation patterns of four up-regulated genes related to the defense response. b Transcript accumulation patterns of four up-regulated WRKY transcription factor genes. c Transcript accumulation patterns of an up-regulated gene related to ROS alleviation. d Transcript accumulation patterns of a down-regulated gene related to photosynthesis
Comparison of gene expression profiles among wheat hybrids with growth abnormalities
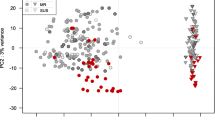
We compared gene expression profiles among the four types (type I to IV) of wheat hybrid necrosis, mild and severe hybrid chlorosis, and SGA in wheat ABD triploid plants based on differences in signal intensity of 22,018 microarray probes relative to WT. These probes selected for the comparison revealed non-saturated uniform signals that were greater than the background signal. A heat map showed the relationships between the comprehensive gene expression profiles of the eight growth abnormalities in various wheat hybrids (Fig. 5). Types I and IV necrosis had gene expression profiles that were closely related to SGA. Hybrid chlorosis and type III necrosis in wheat synthetic hexaploids were categorized into the same clade. Type II necrosis was distinct from other hybrid growth abnormalities.
Next, we compared gene expression profiles among three types (type I, III and IV) of wheat hybrid necrosis in which necrotic symptoms appear on leaves of seedlings under normal temperature (23 °C) (Hermsen 1963; Mizuno et al. 2010; Tsunewaki 1960; Yamagishi 1987). Differences in signal intensity of all 37,826 probes relative to WT were compared among leaves showing hybrid necrosis. Significant positive correlations were detected between the hybrid necrosis plants (Table 4). Moreover, expression profiles were compared for each growth abnormality relative to WT using probes related to defense, carbohydrate metabolism, and photosynthesis. For up-regulated genes, significantly strong positive correlations were observed between the profiles of defense-related genes in type I and type III necrosis and between the profiles of carbohydrate metabolism-related genes in type III and type IV necrosis. For down-regulated genes, significantly strong positive correlations were found in the expression profiles of photosynthesis-related genes between type I and type IV necrosis. However, significant positive correlations were not necessarily detected in the other combinations, and negative correlations were found in some combinations.
Alleviation of necrotic symptoms via zinc chloride treatment
ZnCl2 functions as a Ca2+ antagonist preventing calcium metabolism, and inhibits victorin-induced apoptotic cell death in oat (Hoat et al. 2006). To examine the effect of ZnCl2 on necrotic cell death in wheat hybrids, we treated seedlings showing the three types of hybrid necrosis with ZnCl2. ZnCl2 treatment dramatically suppressed necrotic symptoms on the leaf surface in these types of hybrid necrosis (Fig. 6a). In addition, the numbers of trypan blue-stained cells, which indicate dead cells, in leaves of F1 seedlings showing type I necrosis and synthetic hexaploid seedlings with type III necrosis, were greatly reduced by the ZnCl2 treatment (Fig. 6b). This result indicated that ZnCl2 treatment alleviated necrotic cell death in wheat hybrid necrosis. ROS generation was detected by DAB staining in leaves of type I and type III hybrid necrosis wheat seedlings, and the numbers of brown-colored cells were decreased by ZnCl2 treatment in both types (Fig. 6b). Thus, ZnCl2 treatment repressed ROS generation in wheat hybrid necrosis. Effects of ZnCl2 treatment on photosynthetic activity were also examined for the three types of hybrid necrosis. Time-course studies clearly showed that ZnCl2 treatment led to significant recovery from the decrease in photosynthetic activity of leaves of hybrid necrosis wheat seedlings (Fig. 6c).
Effects of ZnCl2 treatment on necrotic cell death in three types of hybrid necrosis. a Leaf phenotype of WT and necrosis-showing plants after 10 days of ZnCl2 treatment. b Trypan blue- and DAB-stained leaves in type I and type III necrosis. c Comparison of chlorophyll fluorescence among WT, necrosis-showing plants without ZnCl2 treatment and necrosis-showing plants after ZnCl2 treatment. The first leaves were incubated in the dark and then used to calculate the ratio of variable to maximum fluorescence (Fv/Fm). Means ± standard deviations were calculated from data in nine experiments. Student’s t test was used to test for statistical significance (*P < 0.05; **P < 0.01; ***P < 0.001) between the WT and necrosis-showing plants
To examine the effects of ZnCl2 treatment on defense-related gene expression, qRT-PCR was conducted for genes encoding seven defense-related proteins. We chose these defense-related genes which were commonly up-regulated in the hybrid necrosis lines. According to the data from the microarray analyses, transcript accumulation levels of the pathogenesis-related protein 4 (PR4) gene were 4.5-, 30.7- and 8.8-fold increased in type I, type III and type IV necrosis, respectively. For the PR4 gene, two primer sets derived from different cDNAs were used (Supplemental Table 1). From the microarray data, class I and class III chitinases, RPP13-like protein1-like, and phenylalanine ammonia lyase (PAL) genes were 3.2- to 112.7-times up-regulated in type I and type III necrosis. The WRKY3 transcription factor gene was 25.0- and 14.9-fold activated in type I and type IV necrosis based on the microarray data, respectively. The qRT-PCR analyses showed that the transcripts of most examined genes accumulated more abundantly in leaves of the hybrid necrosis wheat seedlings than in leaves of WT plants (Fig. 7). Significant differences in the transcript levels were also observed in leaves of ZnCl2-treated seedlings. Transcript accumulation of some examined genes including PR4 and class I chitinase was markedly reduced by ZnCl2 treatment in type I and type III necrosis plants, whereas no remarkable reduction was observed for PAL, WRKY3 and RPP13-like protein1-like. Thus, ZnCl2 treatment did not necessarily affect transcript accumulation of defense-related genes in theses three types of hybrid necrosis plants.
Effects of ZnCl2 treatment on transcript accumulation of two defense-related genes in three types of hybrid necrosis. Total RNA was extracted from the first leaves of plants after 10 days of ZnCl2 treatment and used for qRT-PCR. The transcript levels are shown as values relative to those in leaves of WT without ZnCl2 treatment. Means ± standard deviations were calculated from data in three experiments. The Actin gene was used as an internal control. Student’s t test was used to test for statistical significance (*P < 0.05; **P < 0.01; ***P < 0.001) between WT and necrosis-showing lines
Discussion
Recent studies have suggested that autoimmune responses trigger hybrid necrosis in higher plants, and are accompanied by up-regulation of a number of defense-related genes (Alcázar et al. 2009, 2010; Bomblies et al. 2007; Chae et al. 2014; Jeuken et al. 2009; Yamamoto et al. 2010). Several types of hybrid necrosis have been reported in inter- and intraspecific crosses of wheat and its relatives, and previous studies suggest that all known types of wheat hybrid necrosis are genetically programmed through interaction of two complementary genes (Hermsen 1963; Mizuno et al. 2010; Nishikawa 1962b; Tsunewaki 1960; Yamagishi 1987). The up-regulation of defense-related genes is observed before necrotic symptoms appear in leaves of hybrid plants exhibiting type II and type III necrosis, and necrotic cell death is accompanied by ROS generation and reduction in photosynthetic activity (Mizuno et al. 2010, 2011). Similar alterations in gene expression patterns, photosynthetic activity and ROS generation were found prior to appearance of necrotic symptoms in leaves of hybrid plant seedlings exhibiting type I and type IV necrosis (Figs. 1, 2, 3, 4, 6b). These characteristics indicate that type I and type IV necrosis share at least some similar mechanisms inducing necrotic cell death with type II and type III necrosis. In fact, significant positive correlations were observed in the gene expression profiles among plants showing the three types of hybrid necrosis (Table 4). Hypersensitive response (HR)-induced cell death typically occurs as a plant defense response to various plant pathogens (Mur et al. 2008). The programmed cell death in type III necrosis could be regarded as HR-like cell death, and the autoimmune response triggered by interaction of the causal genes (Nec1 and Nec2) appears to result in this type of necrosis, as reported for hybrid necrosis in other plant species (Mizuno et al. 2010). Thus, our observations strongly suggest that an autoimmune response might be triggered by epistatic interactions between Ne1 and Ne2 in type I necrosis of common wheat and between Ned1 and Ned2 in type IV necrosis of wild diploid wheat.
An oxidative burst occurs in the early stage of HR to pathogen infection (Wojtaszek 1997). ROS generation appears prior to cell death in type III necrosis, and the generated ROS may function as initiators of cell death in type III necrosis, as they do in HR (Mizuno et al. 2010). The HR-like reaction is assumed to occur in type I and type IV necrosis, as observed in type III necrosis, and thus ROS would play important roles in initiation and/or progression of necrotic cell death in type I and type IV necrosis. In addition, Ca2+-dependent processes mediate various types of programmed cell death including both apoptotic and non-apoptotic cell death (Orrenius et al. 2003), and a Ca2+ signal plays important roles in the signal transduction pathways leading to innate immune-associated cell death in plants (Ma and Berkowitz 2007). Ca2+ influx into plant cells is associated with NADPH oxidase-dependent ROS generation (Bindschedler et al. 2001; Davies et al. 2006; Song et al. 2006). Inhibition of the cytosolic Ca2+ elevation leads to ROS generation and delays plant HR to an avirulent pathogen (Ma and Berkowitz 2007). In fact, ZnCl2, as a Ca2+ antagonist, strongly suppresses victorin-induced cell death, an example of plant defense-associated host cell apoptosis, in oat (Hoat et al. 2006). Our present study showed that ZnCl2 treatment effectively alleviated ROS generation, reduction in photosynthetic activity, and necrotic cell death in three types of wheat hybrid necrosis (Fig. 6). These results indicated that inhibition of Ca2+ signaling repressed necrotic symptoms through reduction in the generated ROS in plants showing wheat hybrid necrosis. Taking these observations together suggests that epistatic interactions between causative genes of wheat hybrid necrosis might result in Ca2+ influx and that the Ca2+ influx might play a role in upstream signaling of necrotic cell death.
On the other hand, no clear reduction in gene expression was observed in two plant defense-related genes following ZnCl2 treatment in three types of wheat hybrid necrosis (Fig. 7). These results indicated that defense-related gene activation might be independent of ROS generation in wheat hybrid necrosis. In general, Ca2+ influx is considered to act as a mediator for induction of a set of defense-related genes (Ma and Berkowitz 2007). At least in wheat hybrid necrosis, however, epistatic interaction of the causative genes appears to activate defense-related genes without a Ca2+ influx. Thus, defense-related gene activation might proceed in parallel with ROS generation-mediated cell death in wheat hybrid necrosis. To clarify the regulation of defense-related genes via causative gene interaction in hybrid necrosis, much earlier responses in necrotic cells of wheat hybrids should be analyzed in further studies.
Comprehensive gene expression profiles in type I and IV necrosis were positively correlated with that observed in type III necrosis (Table 4). In addition, phenotypic and transcriptional responses to ZnCl2 treatment resembled each other among the three types of hybrid necrosis (Figs. 6, 7). Comparison of the comprehensive expression profiles with those associated with other growth incompatibilities showed that profiles of type III necrosis and type IV necrosis were similar to those of hybrid chlorosis in synthetic hexaploid wheat and SGA in ABD triploid wheat hybrids, but the profile of type II necrosis was distinct from those observed for other growth incompatibilities (Fig. 5). Necrotic cell death requires low-temperature conditions, and normal growth temperatures such as 23 °C cannot induce any necrotic symptoms in plants showing type II necrosis (Mizuno et al. 2011; Takumi and Mizuno 2011). Low-temperature treatment activates a number of wheat cold-responsive genes, resulting in differences in gene expression profiles for plants showing type II necrosis from profiles seen for other growth incompatibilities. Weakly positive correlations of gene expression profiles were observed even between leaves of plants showing type III necrosis and low temperature-treated leaves and crown tissues in type II necrosis (Mizuno et al. 2011). Thus, hybrid growth incompatibilities, in spite of their phenotypic variation, share at least in part similar alterations in gene expression profiles such as activation of defense- and carbohydrate metabolism-related genes and down-regulation of photosynthesis-related genes in inter- and intra-specific crosses of wheat and its relatives.
In wild diploid wheat, hybrid necrosis was observed in many interspecific cross combinations (Table 1). Epistatic interaction between Ned1 in T. monococcum ssp. aegilopoides and Ned2 in T. urartu was previously assumed to trigger necrotic cell death (Yamagishi 1987). All five of the T. urartu accessions examined in the present study were considered to carry the dominant allele of Ned2, whereas one ssp. aegilopoides accession KU-3620 contained no dominant Ned1 allele. This result agreed with a previous report (Yamagishi 1987). The dominant Ned2 allele would be fixed in the T. urartu population, while distribution of the dominant Ned1 allele appears to be limited in the ssp. aegilopoides population. Thus, post-zygotic reproductive isolation via the Ned1–Ned2 interaction is incomplete between the two wild diploid wheat species. However, other types of reproductive isolation additively underlie the two wild diploid wheat species (Fricano et al. 2014; Johnson and Dhaliwal 1976; Yamagishi 1987). For example, when ssp. aegilopoides accessions are used as pollen parents, F1 hybrid seeds with T. urartu accessions can be obtained, but most of them fail to germinate (Johnson and Dhaliwal 1976; Yamagishi 1987). The failure of germination has been attributed to nuclear-cytoplasm incompatibility or abnormal endosperm development in the hybrid seeds (Dhaliwal 1977; Gill and Waines 1978; Johnson and Dhaliwal 1976). The nuclear genomes of the two wild diploid wheat species are clearly differentiated, as reported by several research groups (Brandolini et al. 2006; Castagna et al. 1994, 1997; Mizumoto et al. 2002). Nuclear genome differentiation might be at least partly caused by reproductive isolation between the two wild diploid wheat species. To clarify the contribution of type IV necrosis to the post-zygotic reproductive isolation between the two wild diploid wheat species, intraspecific distribution of the dominant Ned1 allele in the ssp. aegilopoides population should be elucidated in further study. Moreover, the cytoplasmic effects of T. urartu on viable seed development should also be studied to understand more comprehensively the reproductive isolation of wild diploid wheat.
To overcome reproductive barriers among the related plant species, the causal genes for the genetic incompatibilities should be identified and molecular cloning of them should be required. However, large efforts should be needed for the identification and isolation of the reproductive barrier-causative genes. Therefore, the alleviation effects of the ZnCl2 application on the expression of necrotic symptoms suggest an alternative strategy to overcome at least some of the reproductive barriers. If the ZnCl2-applied suppression of necrotic symptoms effectively continued until the grain maturation stage of the hybrid plants, gene flow over hybrid necrosis could be realized. The effects of long-term ZnCl2 application on development of the hybrid necrosis plants should be tried in the future studies.
References
Alcázar R, García AV, Parker JE, Reymond M (2009) Incremental steps toward incompatibility revealed by Arabidopsis epistatic interactions modulating salicylic acid pathway activation. Proc Natl Acad Sci USA 106:334–339
Alcázar R, García AV, Kronholm I, de Meaux J, Koornneef M, Parker JE, Reymond M (2010) Natural variation at Strubbelig Receptor Kinase 3 drives immune-triggered incompatibilities between Arabidopsis thaliana accessions. Nat Genet 42:1135–1139
Bindschedler LV, Minibayeva F, Gardner SL, Gerrish C, Davies DR, Bolwell GP (2001) Early signalling events in the apoplastic oxidative burst in suspension cultured French bean cells involve camp and Ca2+. New Phytol 151:185–194
Bomblies K, Weigel D (2007) Hybrid necrosis: autoimmunity as a potential gene-flow barrier in plant species. Nat Rev Genet 8:382–393
Bomblies K, Lempe J, Epple P, Warthmann N, Lanz C, Dangl JL, Weigel D (2007) Autoimmune response as a mechanism for Dobzhansky–Muller-type incompatibility syndrome in plants. PLoS Biol 5:e236
Brandolini A, Vaccino P, Boggini G, Özkan H, Kilian B, Salamini F (2006) Quantification of genetic relationships among A genomes of wheats. Genome 49:297–305
Castagna R, Maga G, Perenzin M, Heun M, Salamini F (1994) RFLP-based genetic relationships of einkorn wheats. Theor Appl Genet 88:818–823
Castagna R, Gnocchi S, Perenzin M, Heun M (1997) Genetic variability of the wild diploid wheat Triticum urartu revealed by RFLP and RAPD markers. Theor Appl Genet 94:424–430
Chae E, Bomblies K, Kim ST, Karelina D, Zaidem M, Ossowski S, Martín-Pizarro C, Laitinen RAE, Rowan BA, Tenenboim H, Lechner S, Demar M, Habring-Müller A, Lanz C, Rätsch G, Weigel D (2014) Species-wide genetic incompatibility analysis identifies immune genes as hot spots of deleterious epistasis. Cell 159:1341–1351
Chu CG, Faris JD, Friesen TL, Xu SS (2006) Molecular mapping of hybrid necrosis genes Ne1 and Ne2 in hexaploid wheat using microsatellite markers. Theor Appl Genet 112:1374–1381
Conesa A, Götz S, García-Gómez JM, Terol J, Talón M, Robles M (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676
Dalal M, Khanna-Chopra R (2001) Differential response of antioxidant enzymes in leaves of necrotic wheat hybrids and their parents. Physiol Plant 111:297–304
Davies DR, Bindschedler LV, Strickland TS, Bolwell GP (2006) Production of reactive oxygen species in Arabidopsis thaliana cell suspension cultures in response to an elicitor from Fusarium oxysporum: implications for basal resistance. J Exp Bot 57:1817–1827
Dhaliwal HS (1977) Basis of difference between reciprocal crosses involving Triticum boeoticum and T. urartu. Theor Appl Genet 49:283–286
Fricano A, Brandolini A, Rossini L, Sourdille P, Wunder J, Effgene S, Hidalgo A, Erba D, Piffanelli P, Salamini F (2014) Crossability of Triticum urartu and Triticum monococcum wheats, homoeologous recombination and description of a panel of interspecific introgression lines. G3 4:1931–1941
Gill BS, Waines JG (1978) Paternal regulation of seed development in wheat hybrids. Theor Appl Genet 51:265–270
Hatano H, Mizuno N, Matsuda R, Shitsukawa N, Park P, Takumi S (2012) Dysfunction of mitotic cell division at shoot apices triggered severe growth abortion in interspecific hybrids between tetraploid wheat and Aegilops tauschii. New Phytol 194:1143–1154
Hermsen JGT (1963) The genetic basis of hybrid necrosis in wheat. Genetica 33:245–287
Hoat TX, Nakayashiki H, Tosa Y, Mayama S (2006) Specific cleavage of ribosomal RNA and mRNA during victorin-induced apoptotic cell death in oat. Plant J 46:922–933
Ishihama N, Yoshioka H (2012) Post-translational regulation of WRKY transcription factors in plant immunity. Curr Opin Plant Biol 15:431–437
Jeuken MJW, Zhang NW, McHale LK, Pelgrom K, den Boer E, Lindhout P, Michelmore RW, Visser RGF, Niks RE (2009) Rin4 causes hybrid necrosis and race-specific resistance in an interspecific lettuce hybrid. Plant Cell 21:3368–3378
Johnson BL, Dhaliwal HS (1976) Reproductive isolation of Triticum boeoticum and Triticum urartu and the origin of the tetraploid wheat. Am J Bot 63:1088–1094
Kawaura K, Mochida K, Ogihara Y (2008) Genome-wide analysis for identification of salt-responsive genes in common wheat. Funct Integr Genomics 8:277–286
Khanna-Chopra R, Dalal M, Kumar PG, Laloraya M (1998) A genetic system involving superoxide causes F1 necrosis in wheat (Triticum aestivum L.). Biochem Biophys Res Commun 248:712–715
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Lucas A (2011) amap: another multidimentional analysis package. R package version 0.8-7. http://CRNA.R-project.org/package=amap
Ma W, Berkowitz GA (2007) The grateful dead: calcium and cell death in plant innate immunity. Cell Microbiol 9:2571–2585
Masuda Y, Yamada T, Kuboyama T, Marubashi W (2007) Identification and characterization of genes involved in hybrid lethality in hybrid tobacco cells (Nicotiana suaveolens x N. tabacum) using suppression subtractive hybridization. Plant Cell Rep 26:1595–1604
Matsuoka Y (2011) Evolution of polyploid Triticum wheats under cultivation: the role of domestication, natural hybridization and allopolyploid speciation in their diversification. Plant Cell Physiol 52:750–764
Matsuoka Y, Takumi S, Kawahara T (2007) Natural variation for fertile triploid F1 formation in allohexaploid wheat speciation. Theor Appl Genet 115:509–518
Mizumoto K, Hirosawa S, Nakamura C, Takumi S (2002) Nuclear and chloroplast genome genetic diversity in the wild einkorn wheat, Triticum urartu, revealed by AFLP and SSLP analyses. Hereditas 137:208–214
Mizuno N, Hosogi N, Park P, Takumi S (2010) Hypersensitive response-like reaction is associated with hybrid necrosis in interspecific crosses between tetraploid wheat and Aegilops tauschii Coss. PLoS One 5:e11326
Mizuno N, Shitsukawa N, Hosogi N, Park P, Takumi S (2011) Autoimmune response and repression of mitotic cell division occur in inter-specific crosses between tetraploid wheat and Aegilops tauschii Coss. that show low temperature-induced hybrid necrosis. Plant J 68:114–128
Mur LAJ, Kenton P, Lioyd AJ, Ougham H, Prats E (2008) The hypersensitive response; the centenary is upon us but how much do we know? J Exp Bot 59:501–520
Myhre S, Tveit H, Mollestad T, Laegreid A (2006) Additional gene ontology structure for improved biological reasoning. Bioinformatics 22:2020–2027
Nakano H, Mizuno N, Tosa Y, Yoshida K, Park P, Takumi S (2015) Accelerated senescence and enhanced disease resistance in hybrid chlorosis lines derived from interspecific crosses between tetraploid wheat and Aegilops tauschii. PLoS One. doi:10.1371/journal.pone.0121583
Nishikawa K (1960) Hybrid lethality in crosses between Emmer wheats and Aegilops squarrosa, I. Vitality of F1 hybrids between emmer wheats and Ae. squarrosa var. typica. Seiken Ziho 11:21–28
Nishikawa K (1962a) Hybrid lethality in crosses between Emmer wheats and Aegilops squarrosa, II. Synthesized 6x wheats employed as test varieties. Jpn J Genet 37:227–236
Nishikawa K (1962b) Hybrid lethality in crosses between Emmer wheats and Aegilops squarrosa, III. Gene analysis of type-2 necrosis. Seiken Ziho 14:45–50
Orrenius S, Zhivotovsky B, Nicotera P (2003) Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol 4:552–565
Presgraves DC (2010) The molecular evolutionary basis of species formation. Nat Rev Genet 11:175–180
Pukhalskiy VA, Martynov SP, Dobrotvorskaya TV (2000) Analysis of geographical and breeding-related distribution of hybrid necrosis genes in bread wheat (Triticum aestivum L.). Euphytica 114:233–240
R Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Rieseberg LH, Willis JH (2007) Plant speciation. Science 317:910–914
Roy RP (1955) Semi-lethal hybrids in crosses of species and synthetic amphidiploids of Tritium and Aegilops. Indian J Genet Plant Breed 14:88–98
Rushton PJ, Somssich IE, Ringler P, Shen QJ (2010) WRKY transcription factors. Trend Plant Sci 15:247–258
Sears ER (1944) Inviability of intergeneric hybrids involving Triticum monococcum and T. aegilopoides. Genetics 29:113–127
Song CJ, Steinebrunner I, Wang X, Stout SC, Roux SJ (2006) Extracellular ATP induces the accumulation of superoxide via NADPH oxidases in Arabidopsis. Plant Physiol 140:1222–1232
Sugie A, Murai K, Takumi S (2007) Alteration of respiration capacity and transcript accumulation levels of alternative oxidase genes in necrosis lines of common wheat. Genes Genet Syst 82:231–239
Takumi S, Mizuno N (2011) Low temperature-induced necrosis shows phenotypic plasticity in wheat triploid hybrids. Plant Signal Behav 6:1431–1433
Takumi S, Motomura Y, Iehisa JCM, Kobayashi F (2013) Segregation distortion caused by weak hybrid necrosis in recombinant inbred lines of common wheat. Genetica 141:463–470
Tsunewaki K (1960) Monosomic and conventional analyses in common wheat. III. Lethality. Jpn J Genet 35:71–75
Tsunewaki K (1970) Necrosis and chlorosis genes in common wheat and its ancestral species. Seiken Ziho 22:67–75
Tsunewaki K (1992) Aneuploid analyses of hybrid necrosis and hybrid chlorosis in tetraploid wheats using the D genome chromosome substitution lines of durum wheat. Genome 35:594–601
Tsunewaki K, Koba T (1979) Production and genetic characterization of the co-isogenic lines of a common wheat Triticum aestivum cv. S-615 for ten major genes. Euphytica 28:579–592
Warnes GR (2012) gplots: various R programming tools for plotting data. R package version 2.11.0. http://CRAN.R-project.org/package=gplots
Wojtaszek P (1997) Oxidative burst: an early plant response to pathogen infection. Biochem J 322:681–692
Yamagishi Y (1987) Phylogenetic differentiation between two species of the wild diploid wheats. Genbunsha, Kyoto, Japan. ISBN4-87609-144-7
Yamamoto E, Takashi T, Morinaka Y, Lin S, Wu J, Matsumoto T, Kitano H, Matsuoka M, Ashikari M (2010) Gain of deleterious function caused an autoimmune response and Bateson–Dobzhansky–Muller incompatibility in rice. Mol Genet Genomics 283:305–315
Acknowledgments
We thank emeritus professor Dr. Koichiro Tsunewaki for supplying seeds of NILs, Ne1-S615 and Ne2-S615. We are grateful to Professor Dr. Hitoshi Nakayashiki for his useful suggestions. The diploid wheat seeds used in this study were supplied by the National BioResource Project-Wheat, Japan (www.nbrp.jp). This work was supported by Grants-in-Aid for Scientific Research (B) Nos. 21380005 and 25292008 from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Takamatsu, K., Iehisa, J.C.M., Nishijima, R. et al. Comparison of gene expression profiles and responses to zinc chloride among inter- and intraspecific hybrids with growth abnormalities in wheat and its relatives. Plant Mol Biol 88, 487–502 (2015). https://doi.org/10.1007/s11103-015-0338-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-015-0338-6