Abstract
AGL6-clade genes are a subfamily of MADS-box genes and preferentially expressed in floral organs. OsMADS6 and OsMADS17 are two AGL6-like genes in rice. OsMADS17 has been shown to play a minor role in floral development and appears to result from a duplication of OsMADS6. OsMADS6 was initially named as MFO1 for mosaic floral organs based on its moderate mutant phenotypes. So far, four moderate or weak mutant alleles of OsMADS6 have been described, providing valuable insights into its role in flower development. Here, we report a null allele of OsMADS6 (Osmads6-5), which exhibited a strong mutant phenotype in spikelet without affecting vegetative traits, causing all floral organs except lemma homeotically transformed into lemma-like organs (LLOs) as well as an indeterminate floral meristem, thus resulting in a mutant floret consisting of reiterating whorls of lemma and LLOs. In consistently, over-expression of OsMADS6 led to additional lodicule-, stamen- and carpel-like organs. Expression analysis showed that OsMADS6 controls the formation of the incipient primordia of lodicule, stamen and carpel via regulating the expression of class B, C and SEP-like MADS-box genes. Taken together, our results revealed that OsMADS6 acts as a critical regulator for early flower development in rice and provide novel insights into the molecular mechanism of OsMADS6.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent studies have shown that SEP genes have divergent functions and play fundamental roles in determining floral fate. In Arabidopsis, four SEP genes (SEP1/2/3/4) are necessary for the identity specification of all four floral whorls and for the determinacy of floral meristem (Pelaz et al. 2000; Ditta et al. 2004). In rice, five SEP-like genes have been identified. Among them, OsMADS1 is mainly involved in the specification of lemma/palea and the regulation of floral meristem determinacy (Jeon et al. 2000a; Prasad et al. 2001; Malcomber and Kellogg 2004; Agrawal et al. 2005; Prasad et al. 2005; Chen et al. 2006) and the other four SEP-like genes might function redundantly in specifying floral organ identity or spikelet development (Agrawal et al. 2005; Cui et al. 2010; Kobayashi et al. 2010; Gao et al. 2010).
AGL6-like genes are a subfamily of MADS-box genes. Phylogenetic analysis suggests that AGL6-clade is the closest relative of AGL2-clade (SEP-like) and has been hypothesized to be sister to AGL2-clade (Zahn et al. 2005). In grasses, the expression of AGL6-clade genes was detected in ovules, carpels, lodicules (equivalent to second whorl floral organs), paleas (putative first whorl floral organs), and floral meristems, suggesting that they may act in both perianth and gynoecium development (Reinheimer and Kellogg 2009). In addition, some studies suggested that over-expression of AGL6-clade genes can promote the expression of flowering time genes (FT and SOC1), flower meristem identity genes (LFY and AP1) and floral organ identity genes (AG and SEP1) and consequently result in early flowering and dwarf transgenic plants and homeotic transformation of floral organs (Jeon et al. 2000b; Hsu et al. 2003; Carlsbecker et al. 2004; Tian et al. 2005; Fan et al. 2007). Together, these results show that AGL6-like genes might play roles in the initiation and formation of flowers, though they are not included in the typical ABCDE model of flower development. However, actual functions of AGL6-clade genes largely remain enigmatic due to lack of their loss-of-function mutants. Recently, identification of loss-of-function mutants in Petunia and maize shed lights on the functions of AGL6-clade genes (Rijpkema et al. 2009; Thompson et al. 2009). The Petunia hybrida AGL6-clade gene, PhAGL6, has been found to function redundantly with the SEP-like genes FBP2 (FLORAL BINDING PROTEIN2) and FBP5 in petal, anther and carpel development. In addition, PhAGL6 and FBP2 proteins may share similar biochemical characteristics because they interacted with the same partners (Rijpkema et al. 2009). In maize, an AGL6-clade gene bearded-ear (bde) was considered to resemble E-function genes because it affects floral organ development in all four whorls and floral meristem identity, and likely functions in multiple complexes including BDE proteins and the C-class protein ZAG1 (Thompson et al. 2009).
OsMADS6 and OsMADS17 are two rice AGL6-clade genes. OsMADS17 has been shown to play a minor role in rice floral development and appears to result from a duplication of OsMADS6 (Ohmori et al. 2009; our unpublished results). Recently, two mutant alleles of OsMADS6 named mfo1-1 and mfo1-2, showing a phenotype of mosaic organs in flowers, have been isolated (Ohmori et al. 2009). Also, Li et al. (2010) described another mutant allele Osmads6-1, similar to mfo1-1. Both studies suggested that OsMADS6 appears to regulate floral development redundantly with OsMADS1 (Ohmori et al. 2009; Li et al. 2010). Whereas Zhang et al. (2010) reported that OsMADS6 shows an essential role in endosperm nutrient accumulation and is subject to epigenetic regulation from two additional mutant alleles of OsMADS6 (here we renamed one as Osmads6-2 and the other is identical to mfo1-2). Moreover, Li et al. (2011) suggested that OsMADS6 genetically interacts with rice floral homeotic genes OsMADS16/3/58/13 and DROOPING LEAF (DL) in specifying floral organ identities and meristem fate and, in particular, OsMADS6 protein can directly bind to OsMADS58. However, these four OsMADS6 mutant alleles showed moderate (mfo1-1, Osmads6-1 and Osmads6-2) or weak (mfo1-2) floral phenotypes. Hence, the loss of function phenotype of OsMADS6 remains to be further defined.
In this work, we identified the fifth mutant allele, a null allele, of OsMADS6 (Osmads6-5), which clearly revealed the function of OsMADS6 in flower development, showing that it is critical for determining the floral developmental fate and meristem determinacy by regulating the expression of floral organ identity genes at early stages of flower development and providing a new insight into the molecular mechanism of flower development in rice.
Materials and methods
Plant materials
The Osmads6-5 was discovered in a doubled haploid (DH) line derived from the F1 of a cross between an indica variety Gui-630 and a japonica variety Taiwanjing by anther culture and maintained through heterozygotes.
Microscopic observation
Young panicles (1–10 mm) were observed by scanning electron microscopy (SEM, Duan et al. 2010). Panicles were fixed in 2.5 % glutaric dialdehyde and washed with a sodium phosphate buffer (0.1 M, pH 7.0); further fixed in 1 % osmic acid for 1–2 h and again washed with the sodium phosphate buffer (0.1 M, pH 7.0); dehydrated with an ethanol series, incubated in ethanol-isoamyl acetate and then in isoamyl acetate; dried, mounted and coated with gold; and finally observed with a XL30 ESEM scanning electronic microscope (PHLIPS Company, Amsterdam, The Netherlands).
Histochemical analysis
Rice flowers fixed in 2.5 % glutaraldehyde solution were dehydrated using a graded ethanol series and embedded in Leica 7022 historesin (Leica, Nussloch, Germany). Samples were sectioned to 4 μm, stained with 0.1 % Toluidine Blue-O (Sigma, St. Louis, MO, USA) and observed under an Olympus AX-80 light microscope.
Isolation of Osmads6-5
Heterozygotes of Osmads6-5 were crossed with an indica cultivar Minghui-77, and 1,128 F2 plants with the Osmads6-5 phenotype were selected for gene mapping. Publicly available RM-series simple sequence repeat (SSR) markers as well as some new SSR markers (data not shown) developed by us were used for fine mapping. Reverse transcription PCR (RT-PCR) analysis of the expression of Osmads6-5 allele was performed using a pair of primers (5′-ATGACTCCTACCCACCATTG-3′ and 5′-TCAAAGAACCCATCCCAGCATGAAG-3′). For complementation test, a 13,488-bp OsMADS6 genomic DNA fragment covering 3,702-bp promoter and 2,198-bp 3′ region was sub-cloned into a binary vector pCAMBIA1300 and introduced into Osmads6-5 mutant embryonic calli by Agrobacterium tumefaciens-mediated transformation (Hiei et al. 1994).
Function analysis of a truncated polypeptides of Osmads6-5
The 477 bp truncated OsMADS6 coding sequence was cloned into a binary vector pTCK303 driven by a rice ubiquitin promoter (Wang et al. 2004). The binary construct was introduced into EHA105 and further into rice. Seven independent transgenic plants were acquired.
Over-expression of OsMADS6
Full-length OsMADS6 cDNA was introduced into a modified binary vector pCAMBIA1301 with restriction sites of BamHI and SalI (Chen et al. 2006). The binary construct 35S:OsMADS6 was introduced into EHA105 and further into rice. Eight independent 35S:OsMADS6 plants were obtained.
Real-time quantitative PCR (qRT-PCR) analysis
Reverse transcription of total RNA was performed using SuperScript III First-Strand Synthesis kit (Invitrogen, USA). As previously described (Lan et al. 2004), the cDNA samples were diluted into 8 ng/μl and 2 ng/μl. Triplicate quantitative assays were performed using the SYBR Green Master Mix (Applied Biosystems, CA, USA) with an ABI 7900 sequence detection system. The relative quantification method (∆ΔCT) was used to evaluate quantitative variation between replicates examined. Amplification of 18S rRNA was used as an internal control to normalize all data. Primers used for qRT-PCR analysis are listed in Table S1.
mRNA in situ hybridization
mRNA in situ hybridization was essentially performed as previously described by Lai et al. (2002). Wild-type and Osmads6-5 young panicles (2–10 mm) were fixed with formalin-acetic acid-alcohol solution and embedded in paraffin. Probe of OsMADS1 was designed as previously described by Li et al. (2009); OsMADS3/58 by Yamaguchi et al. (2006); OsMADS7/8 by Cui et al. (2010) and OsMADS17 by Ohmori et al. (2009). The OsMADS4/16 probe primers were 5′-CGGCTACCACCACGACGACA-3′/5′-CTGAGTGCTAATGCTGGGAG-3′ and 5′-ACCGTATCATCGCTCGATCT-3′/5′-GCACACCACGCATACATAAT-3′, respectively.
Protoplast transient expression assay
The full-length and truncated OsMADS6 coding sequences (deletion the MADS-domain) were cloned into a rebuilt vector pBI221 driven by CaMV 35S promoter to generate p221-35S:OsMADS6-GFP, respectively. The GFP fusion construct was transformed into Arabidopsis protoplasts using the protocol described previously (Yoo et al., 2007). Localization of OsMADS6-GFP fusion protein was observed with a Zeiss LSM 510 META confocal microscope (Zeiss, Jena, GER).
Results
The Osmads6-5 floret comprises many whorls of lemma-like organs
A mature wild-type floret of rice comprises of a lemma and a palea, a pair of lodicules, six stamens and a carpel (Fig. 1a). We identified a floral organ mutant of rice initially named lemmata because its flower only consisted of reiterating lemma or lemma-like organs (LLOs). We showed that the mutant was caused by a mutation of OsMADS6 (see below). Since four different OsMADS6 alleles have been described previously (Ohmori et al. 2009; Li et al. 2010; Zhang et al. 2010), to follow the previous work, we hereafter renamed lemmata as Osmads6-5. Osmads6-5 appeared normal for heading time and all vegetative morphological characters except for floret phenotype (Fig. 1b). The Osmads6-5 floret appeared noticeably bulgy in comparison with the wild-type floret (Fig. 1c, d). The lemma of the mutant floret was normal in size, but the palea was significantly enlarged (Fig. 1e). It is known that lemma and palea normally have five and three vascular bundles, respectively (Fig. 1g). The number of vascular bundles remained normal in the Osmads6-5 lemma, but greatly increased (mostly up to 8–10) in its palea (Fig. 1h). In addition, the Osmads6-5 palea became curled inward on the margin like a lemma, rather than formed the membranous margin and the barbs that exist in the normal palea (Fig. 1g, h). These results suggest that the identity of lemma remains unchanged, whereas the identity of palea is evidently altered, losing the feature of palea but gaining the feature of lemma in the Osmads6-5 floret.
Morphological and anatomical observation of wild-type and Osmads6-5 spikelets. a An opened wild-type spikelet. b, c Intact spikelets and panicles of wild-type and Osmads6-5. d, e Opened mature Osmads6-5 spikelets. f Opened young Osmads6-5 spikelets. Lemma, palea and two LLOs transformed from two lodicules were removed. g, h Transverse section of a wild-type spikelet (g) and an Osmads6-5 mutant spikelet (h). Arrowheads indicate vascular bundles in palea. Arrow indicates vascular bundle in LLO. Red circle in (e) and (f) indicates a developing centre of a young mutant spikelet, where many whorls of LLO would be continuously formed. Numbers 2 and 3 in (d–f) and (h) indicate LLOs transformed from lodicules and stamens in whorl 2 and 3, respectively; numbers 4–6 indicate whorls of LLOs transformed from pistil. eg empty glume, le lemma, pa palea, lo lodicule, st stamen, ca carpel. Scale bars 1 mm in (a–f), and 100 μm in (g, h)
Inside the Osmads6-5 floret, the lodicules, stamens and carpel all disappeared and the space was filled with many green organs (Fig. 1d). Anatomical observation clearly revealed that these green organs all possessed the inner structure similar to that of lemma/palea, namely, consisting of four layers of different tissues (cell types), including upper epidermis (silicified cells), fibrous sclerenchyma cell layer, spongy parenchymatous cell layer, and lower epidermis non-silicified cells (Prasad et al. 2005); in addition, they all had the morphological feature of curled margin, like a lemma (Fig. 1h). Hence, it is reasonable to identify these green organs as LLOs. The LLOs were arranged in many (usually 6–10, occasionally 12–15) whorls (Fig. 1f, Table 1). The numbers of LLOs in whorls 2 and 3 were different. In the most Osmads6-5 florets, there were two and six LLOs in whorls 2 and 3, respectively (Fig. 1e, Table 1), suggesting that the LLOs in whorls 2 and 3 might be homeotically transformed from the two lodicules and six stamens, respectively. Nevertheless, a few Osmads6-5 florets with fewer and one or two larger LLOs in whorls 2 and 3 were observed, suggesting that two or more LLOs might have merged into a larger one. Occasionally, we also observed that 3–4 LLOs developed in whorl 2 (Fig. 1e, h). Interestingly, the carpel in the Osmads6-5 floret was replaced by many (often 3–7) whorls of LLOs (Fig. 1f, h, Table 1), suggesting that the determinacy of the floral meristem was lost.
To further clarify the morphogenesis of Osmads6-5 flowers, we examined young spikelets of both the mutant and wild type at different developmental stages by SEM (Fig. 2). The results clearly demonstrated that the floral primordium in the wild-type spikelet developed in an order of that empty glumes developed first (Fig. 2a), followed by a lemma and a palea (Fig. 2a), two lodicules, six stamens (Fig. 2b) and a carpel (Fig. 2c, d). In the mutant, the development of floral primordium also occurred from the periphery to the center. The primordia of empty glumes and lemma could normally emerge and develop into the correct organs in the Osmads6-5 spikelet (Fig. 2e). The palea primordium could also emerge normally in the Osmads6-5 spikelet, but an obvious notch and multiple bumps formed as the development progressed (Fig. 2e–g), while in the wild-type spickelet only a single bump developed in the central region of palea (Fig. 2b, c). This could explain why the Osmads6-5 palea was larger in size and had more vascular bundles. The primordia at whorls 2 and 3 could be formed as expected in the Osmads6-5 spikelet, but they were all homeotically transformed and developed into LLOs, instead of lodicules and stamens as in the wild type (Fig. 2h). Occasionally, some anther-shaped primordia that would develop into LLOs were observed in whorl 3 in the Osmads6-5 spikelet (Fig. 2i). This provides a piece of direct evidence for a homeotic transformation of stamens taking place in the mutant. The central region (whorl 4) of floral primordium did not develop into a single LLO corresponding to carpel in the Osmads6-5 spikelet. Instead, a process similar to that occurred in whorl 3 was reiterated in the central region, resulting in multiple (ranging 3–7) whorls of LLOs (Figs. 1h, 2j, k, Table 1).
SEM observations of wild-type and Osmads6-5 spikelets during early developmental stages. a–d Wild-type spikelets. Lemma and palea were removed in (d). e–k Osmads6-5 spikelets. White arrowheads in (e) and (f) indicate notch formed in the mutant palea. White arrows in (f) and (g) indicate new bump emerged at the mutant palea. Red arrowheads in (i) indicate anther-shaped LLO organs in the whorl 3. Lemma and palea were removed in (h–k). Numbers 2 and 3 indicate LLOs transformed from lodicules and stamens in whorls 2 and 3, respectively; numbers 4–6 incidate whorls of LLOs transformed from pistil. rg rudimentary glume, eg empty glume, le lemma, pa palea, lo lodicule, st stamen, ca capel, fm floral meristem. Scale bars 50 μm
In summary, all the floral organs of Osmads6-5 that develop after lemma are transformed into LLOs, and potentially endless whorls of LLOs are generated due to the loss of floral meristem determinacy, indicating that the OsMADS6 gene is critical for the identity specification of palea, lodicule, stamen and carpel, and for the determinacy of floral meristem.
The phenotype of Osmads6-5 is caused by a large insertion-deletion mutation in OsMADS6
Genetic analysis showed that Osmads6-5 is controlled by a single recessive gene and the mutant phenotype was stable across different cropping seasons (Table 2). To reveal the molecular cause of the mutant phenotype, we employed the positional cloning strategy to isolate Osmads6-5 gene. Using publicly available RM-series SSR markers as well as some new SSR markers developed in this study, we fine mapped Osmads6-5 to a 66.1-kilobase (kb) region on the long arm of chromosome 2. According to the annotation provided by the NCBI database, this region contains 12 genes including OsMADS6 (data not shown). Using a pair of primers designed according to the DNA sequence of OsMADS6, we found that the OsMADS6 locus was completely co-segregated with the mutant trait (data not shown). Sequence analysis of the two bands amplified from the wild-type and the mutant genomic DNA revealed a 2,915-bp deletion (including 2,783-bp of the promoter and 132-bp of the first exon) replaced by an 852-bp insertion (Dataset S1) in the OsMADS6 locus (Fig. 3a). We compared the sequences of other 11 genes in this region between the wild type and the mutant, but no variation was found. Therefore, OsMADS6 should be the only candidate gene. To validate the candidate gene, a complementation vector containing a genomic fragment covering a 3,702-bp promoter region and a 2,198-bp 3′ end region of OsMADS6 was introduced into Osmads6-5, and the mutant phenotype was rescued (Fig. 3b), confirming that the mutant phenotype was indeed caused by the mutation of OsMADS6 gene.
Map-based cloning of OsMADS6 and analysis of the schematic transcript of Osmads6-5. a Genomic organization of OsMADS6 gene and Osmads6-5 allele. OsMADS6 consists of eight exons indicated by vertical bars (the 5′- and 3′-UTR are not shown). Osmads6-5 has a large insertion-and-deletion mutation in OsMADS6. The deleted segment is indicated in gray. The intergenic region upstream of the deletion is indicated in green. b Complementation of Osmads6-5 by pCAMBIA1300-OsMADS6. Left an opened mutant spikelet. Right a spikelet of complemented line (rescued line-1) with the palea/lemma removed from the flower. Scale bars 1 mm. c Schematic transcript structures of OsMADS6 in wild-type and Osmads6-5. The chimeric RNA transcript in Osmads6-5 encompasses a 41 bp inserted sequence (indicated in white zone) at 5′ end of the OsMADS6 transcript. DNA sequence analysis revealed that all the three possible reading frames of the chimeric transcript had premature stop codons. d Possible ORF of schematic transcript of Osmads6-5. Four possible reading frames encoding truncated OsMADS6 polypeptides were found in the downstream sequences of the chimeric transcript of Osmads6-5. The MADS-box domain, I-region, K-region and C-terminal region are shown in green, gray, red and blue in c and d, respectively
Osmads6-5 is a null allele of OsMADS6
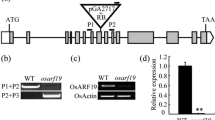
In order to confirm that Osmads6-5 is a null allele of OsMADS6, we examined the expression of Osmads6-5. The results of RT-PCR analysis indicated that a chimeric RNA transcript consisting of a 41 bp of the inserted sequence and its downstream sequence of OsMADS6 was detected (Fig. 3c). Sequence analysis revealed that the predicted three reading frames all have premature stop codons, indicating that there is no in-frame start codon ATG in the insertion sequence (Fig. 3c). Next, we checked the downstream sequences of the chimeric transcript and found four possible ORFs encoding truncated OsMADS6 polypeptides (Fig. 3d). The largest predicted ORF, with the deletion of the MADS domain, I-region and partial K-region, was predicted to be capable of encoding of 159 amino acids identical to the region of OsMADS6 from 93 aa to 251 aa (Fig. 3d). In order to examine whether the truncated OsMADS6 could possibly cause a weak dominant negative effect resulting in the observed phenotype in the double recessive plant but not in the heterozygous one, we expressed the 477 bases of transcription sequence under the control of an ubiquitin promoter. Compared with the wild type, all the transgenic lines displayed a normal floral phenotype except for a slight enclosure of panicles (Fig. S1). This result showed that the predicted truncated polypeptide of Osmads6-5, if any, could not cause a weak dominate negative effect on rice floral organ development. Therefore, the recessive mutant Osmads6-5 is a null allele of OsMADS6.
Over-expression of OsMADS6 results in over production of palea, lodicule, stamen and carpel but has little influence on vegetative traits
To further characterize the function of OsMADS6, we produced transgenic rice plants with over-expressed OsMADS6 driven by the constitutive CaMV 35S promoter. Eight transgenic plants were obtained, in which the expression of OsMADS6 was significantly increased (Fig. S2). No obvious phenotypic changes were observed in the transgenic plants except for spikelets. A few transgenic spikelets were found to possess an additional incomplete floret (Fig. 4b–d, Table 2). In most of the transgenic spikelets, the lodicule, stamen and carpel were all overdeveloped with some morphological abnormalities (Fig. 4e; Table 3). Many additional lodicules developed in the second whorl (Fig. 4f, g). Those lodicules looked normal except a little flatter in shape (Fig. 4e–g). Interestingly, two palea-like organs (PLOs) also developed in this whorl (Fig. 4e–g). Judging from their position, we speculated that the two PLOs might be homeotically transformed from the two original lodicules. In the third and fourth whorls, the most of the transgenic florets had 7–10 stamens and 2–4 carpels (Fig. 4f). Occasionally, two or more carpels merged at the base and developed into a larger one with some unclear tissues on the top (Fig. 4g). The lemma was normal in size, but the palea became obviously similar to lemma in both morph and size in the transgenic florets (Fig. 4e). In sum, these results indicated that OsMADS6 plays a specific role in the regulation of floral organ identity except for the lemma and of flower meristem determinacy in rice.
Phenotypes of wild-type and the 35S:OsMADS6 spikelets. a Wild-type spikelet. White arrow indicates the palea marginal tissue. b–d 35S:OsMADS6 spikelets with an additional incomplete floret indicated by white arrowheads. e–g Opened 35S:OsMADS6 spikelets. Lemma and palea were removed in (f) and (g), one and two PLOs were removed in (f) and (g), respectively. White arrow in (e) indicates the loss of palea marginal tissue in palea (likely LLO). Red arrows in (e) and (f) indicate PLOs transformed from lodicules. Red arrowheads in (g) indicate unclear tissues. eg empty glume, le lemma, pa palea, lo lodicule, st stamen, ca capel. Scale bars 1 mm
Several class B, C and E genes are down-regulated but OsMADS1 and OsMADS17 are up-regulated in Osmads6-5
To examine the relationships between OsMADS6 and other MADS-box genes in rice, we investigated the transcription levels of three AP1-like (OsMADS14/15/18), three class B (OsMADS2/4/16), two class C (OsMADS3/58), five class SEP-like (OsMADS1/5/7/8/19) and one AGL6-clade (OsMADS17) genes in the young panicles of Osmads6-5 and wild type at the early stages of flower development using qRT-PCR. The results showed that in Osmads6-5 mutant, one SEP-like gene (OsMADS1) and the AGL6-like member (OsMADS17) were found to be significantly up-regulated, but several class B (OsMADS4/16), class C/D (OsMADS3/13/58) and class E (OsMADS7/8) genes were found to be significantly down-regulated; whereas the mutation of OsMADS6 appeared not to affect the expression of AP1-like genes (Fig. 5). These results suggested that OsMADS6 regulates the morphogenesis and development of floral organs probably by regulating the expression of other MADS-box genes, especially the floral organ identity genes.
The expression patterns of floral organ identity genes are significantly altered in Osmads6-5
To further discern the relationships between OsMADS6 and other floral organ identity genes in rice, we examined the expression of six down-regulated genes (OsMADS3/4/7/8/16/58) and two up-regulated genes (OsMADS1/17) by RNA in situ hybridization in the wild type and Osmads6-5.
The two class B genes (OsMADS4/16) had similar expression patterns. In wild-type spikelets, the two genes first expressed in the incipient primordia of lodicule and stamen, and their expression persisted during the development of the two floral organs (Fig. 6A[a–c], B[a–c]; Nagasawa et al. 2003). In Osmads6-5 spikelets, however, the expression patterns of OsMADS4 and 16 were obviously altered. The two genes showed a delayed expression, and almost no expression signal was detected in the presumptive initiating regions of whorls 2 and 3 where two and six LLOs would develop, respectively (Fig. 6A[d], B[d]). Weak expression signal was observed in the LLOs primordia of inner whorls until later developmental stages (Fig. 6A[e, f], 6B[e, f]).
Expression of Class B, C, SEP-like genes and OsMADS17 in wild type and Osmads6-5 spikelets. A–H Expression of OsMADS4, OsMADS16, OsMADS3, OsMADS58, OsMADS7, OsMADS8, OsMADS1 and OsMADS17 in wild-type (a–c) and Osmads6-5 spikelets (d–f), respectively. Black arrowheads indicate LLOs. le lemma, pa palea, lo lodicule, st stamen, ca carpel, fm floral meristem, LLO lemma-like organ. Bars 100 μm
The two class C genes (OsMADS3/58) started to be expressed when the lemma and palea primordia just appeared in wild-type flowers (Fig. 6C[a], D[a]). OsMADS3 RNA accumulated in the stamen primordia at early stages and in the carpel primordia later (Fig. 6C[b, c]; Yamaguchi et al. 2006). OsMADS58 signal was detected in the primordia of stamen and carpel throughout the developing stages (Fig. 6D[b, c]; Yamaguchi et al. 2006). In Osmads6-5 spikelets, the expression of the two class C genes was all delayed, similar to that of the two class B genes. There was little detectable signal until whorl 3 was initiated (Fig. 6C[d], D[d]). Later, only weak expression was detectable, restricted to the inner whorls and the central floral meristem where many whorls of LLOs would develop subsequently (Fig. 6C[e, f], D[e, f]).
The expression domains of two SEP-like genes OsMADS7/8 in the wild type were largely overlapped during spikelet development (Fig. 6E[a–c], F[a–c]); Cui et al. 2010). When the lemma and palea primordia were just initiating, both of the genes showed a strong expression in the floral meristem where the floral organ primordia of three inner whorls would subsequently initiate (Fig. 6E[a], F[a]). After that, strong transcription was restricted to lodicules, stamens and carpels (Fig. 6E[b, c], 6F[b, c]; Cui et al. 2010). In Osmads6-5, after the lemma and palea primordia emerged, the floral meristem did not show any detectable signal (Fig. 6E[d], 6F[d]). During later stages, the expression domain was confined to the LLOs primordia of inner whorls (Fig. 6E[e, f], F[e, f]). In addition, the expression levels of OsMADS7/8 were significantly reduced in Osmads6-5 spikelets.
Another SEP-like gene, OsMADS1, was principally expressed in palea and lemma in the wild type when their incipient primordia emerged, with a weak expression in carpel (Fig. 6G[a–c]; Prasad et al. 2005). In Osmads6-5, the initial expression of OsMADS1 was not altered (Fig. 6G[d]). But the expression domain was clearly extended to the primordia of the inner-whorl LLOs during their development (Fig. 6G[e, f]), resulting in the up-regulated expression of OsMADS1.
The other AGL6-like gene, OsMADS17, was expressed intensely in the floral meristem when lemma and palea were initiated in wild-type spikelets. Subsequently, the signal was detected in the lemma, palea, lodicules, stamens and ovule (Fig. 6H[a–c]; Ohmori et al. 2009). In Osmads6-5, the early expression level and domain of OsMADS17 were not significantly altered (Fig. 6H[d]). As the development proceeded, the signal of OsMADS17 expression appeared in all whorls of LLOs from their initiation. Consistent with the fact that there are more whorls formed in Osmads6-5, OsMADS17 had an increased expression level in the mutant compared with the wild type (Fig. 6H[e, f]).
In summary, the temporal-spatial expression patterns of the floral organ identity genes examined were significantly altered in Osmads6-5, supporting the notion that OsMADS6 is required for their correct spatial and temporal expression.
Discussions
The possible cause of the phenotypic variations of OsMADS6 alleles
Recent studies have shown that AGL6-clade genes are a subfamily of floral MADS-box genes (Becker and Theissen 2003), and significant effects of AGL6-clade genes on floral development have been found in monocots but not in dicots (Schauer et al. 2007, 2008; Rijpkema et al. 2009; Thompson et al. 2009; Ohmori et al. 2009; Li et al. 2010; Zhang et al. 2010). In rice, the AGL6-clade gene OsMADS6 has been shown to function in floral development based on the phenotypes of its four previously described mutants. The first and second mutants, mfo1-1 and mfo1-2, were caused by an amino acid substitution in the MADS domain and a retrotransposon insertion at the position of 119 bp downstream of the stop codon, respectively (Ohmori et al. 2009); the third mutant Osmads6-1 resulted from a 4-bp deletion in the seventh exon (Li et al. 2010); and the fourth mutant Osmads6-2 (initially named as Osmads6-1) was generated by a T-DNA insertion in the first intron (Zhang et al. 2010). However, the altered floral phenotypes displayed in these mutants appeared to be inexplicit and incomplete, mainly consisting of mosaic floral organs. This suggests that they are all probably partial loss-of-function mutants. Hence, the complete loss-of-function of OsMADS6 remained to be further defined.
In this study, we found the fifth mutant allele Osmads6-5, caused by a large insertion-deletion mutation with the deletion of the MADS-domain. All Osmads6-5 spikelets showed that the floral organs except lemma all lose their identities and are transformed into LLOs, and the floral meristem is indeterminate, leading to potentially endless whorls of LLOs developing in the central area. Notably, unlike the four previously reported mutants, no mosaic floral organs were observed in Osmads6-5 (Fig. 1). As a DNA-binding domain, mutations in the MADS domain should directly affect the function of the MADS genes. In fact, over-expression of the MADS domain of OsMADS1 produced abnormal floral phenotype, acting as a dominant negative allele, whereas over-expression of OsMADS1 mutant without the MADS domain showed no phenotypic alteration (Jeon et al. 2000a). We obtained the similar result for OsMADS6 showing that the over-expression of the C-terminal 477-bp ORF of Osmads6-5 produced normal flower phenotype. In addition, the truncated OsMADS6:GFP fusion protein redistribution in cell localization supports a key role of the MADS domain (Fig. S3). These results indicated that a mutant lacking the MADS domain would lose the function of MADS box genes because of its dimerization and DNA binding capacity (Mizukami et al. 1996; Krizek et al. 1999). Therefore, Osmads6-5 is a bona fide null allele of OsMADS6, confirming that it plays a key role in rice flower development. This conclusion is also supported by the phenotype of OsMADS6 over-expression in rice where the number of lodicules, stamens and carpels are increased (Fig. 4), and by the results of in situ RNA hybridization analysis as well (Fig. 6).
It is necessary to point out that Osmads6-2 was suggested to be a null allele because no expression signal was detected by RT-PCR in its 3′-terminal region (Zhang et al. 2010). However, in light of its incomplete mutant phenotype (Zhang et al. 2010), Osmads6-2 appears to still maintain a partial function. Since only the 3′-terminal region was examined while the T-DNA was inserted in the first intron in Osmads6-2, the evidence provided by the RT-PCR was not sufficient to exclude the possibility whether the whole coding sequence of the gene was not transcribed. It is possible that at least the first exon, which is located upstream to the inserted T-DNA, could be transcribed to some extent. If this is true, the protein sequence translated from the putative Osmads6-2 transcript would probably still possess a partial function because the MADS domain of OsMADS6 is just located in the first exon (Fig. 3).
OsMADS6 is a critical regulator for early flower development in rice
The phenotypes of Osmads6-5 resemble that of the B + C double mutants. The Arabidopsis ap3/ag and pi/ag consist of indeterminate number of whorls of sepals replacing all other floral organs (Bowma et al. 1989, 1991), and the maize si1-R/zag1-mum1 shows normal glumes that enclose reiterated lemma/palea-like organs (Ambrose et al. 2000). AGL6-clade is sister to AGL2-clade (SEP-like) and expected to have a similar function to that of AGL2-clade. Interestingly, the main features of the Arabidopsis sep1/2/3 triple mutant, rice Osmads1 mutant and OsMADS7/8 double knockdown plants all look similar to Osmads6-5 except for two distinguishable floral phenotypes between them. Firstly, the central region of Osmads6-5 floret is filled with whorls of LLOs instead of an additional abnormal floret with an elongated pedicel as in sep1/2/3 and Osmads1. Secondly, several allelic mutants of OsMADS1 and OsMADS1/5/7/8-RNAi knockdown lines showed the primary character of “leafy hull”, but this phenotype has never been observed in the five allelic mutants of OsMADS6 (Pelaz et al. 2000; Agrawal et al. 2005; Cui et al. 2010). The morphological similarity and dissimilarity indicates that OsMADS6 and SEP-like genes might have similar but not the identical functions in regulating floral organ specification and the floral meristem determinacy. Indeed, several previous studies have suggested that AGL6-clade might play redundant roles with or even resemble AGL2-clade in controlling floral development (Rijpkema et al. 2009; Thompson et al. 2009; Ohmori et al. 2009; Liu et al. 2009; Li et al. 2010; Koo et al. 2010).
The phenotype resemblance among Osmads6-5, the B + C double mutants and the mutants/knockdown plants of AGL2-clade implies that OsMADS6 might directly regulate the expression of B-/C-class and AGL2-clade genes. Indeed, in this study, we have found that the temporal and spatial expression patterns of the class B (OsMADS4/16) and C (OsMADS3/58) and SEP-like (OsMADS1/7/8) genes showed significant alterations in Osmads6-5 (Fig. 5). Similar results were obtained by Li et al. (2011). Together, these results indicate that OsMADS6 probably acts as an early regulator of floral meristem identity by controlling the expression of floral organ identity genes. Such a regulatory relationship appears to be consistent with the mutant phenotype of Osmads6-5. It is possible that the delayed and reduced expression of most of the class B and C and SEP-like genes and the increased expression of OsMADS1 (SEP-like) make inner floral organs transformed into LLOs in Osmads6-5. Hence, OsMADS6 acts as a critical regulator controlling the early development of inner floral organs via activating the expression of class B and C and SEP-like genes.
Abbreviations
- Os:
-
Oryza sativa
- AGL:
-
Agamous-like
- MFO:
-
Mosaic floral organ
- SEP:
-
Sepallata
- FT:
-
Flowering time
- SOC1:
-
Suppressor of overexpression of CO 1
- LFY:
-
Leafy
- AP1:
-
Apetala 1
- AG:
-
Agamous
- BDE:
-
Bearded-ear
- FBP:
-
Floral binding protein2
- DL:
-
Drooping leaf
- SEM:
-
Scanning electron microscopy
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- qRT-PCR:
-
Real-time quantitative RT-PCR
- SSR:
-
Simple sequence repeat
- GFP:
-
Green fluorescent protein
- LLO:
-
Lemma-like organ
- PLO:
-
Palea-like organ
References
Agrawal GK, Abe K, Yamazaki M, Miyao A, Hirochika H (2005) Conservation of the E-function for floral organ identity in rice revealed by the analysis of tissue culture-induced loss-of-function mutants of the OsMADS1 gene. Plant Mol Biol 59:125–135
Ambrose BA, Lerner DR, Ciceri P, Padilla CM, Yanofsky MF, Schmidt RJ (2000) Molecular and genetic analyses of the silky1 gene reveal conservation in floral organ specification between eudicots and monocots. Mol Cell 5:569–579
Becker A, Theissen G (2003) The major clades of MADS-box genes and their role in the development and evolution of flowering plants. Mol Phyl Evo 29:464–489
Bowma JL, Smyth DR, Meyerowitz EM (1989) Genes directing flower development in Arabidopsis. Plant Cell 1:37–52
Bowman JL, Smyth DR, Meyerowitz EM (1991) Genetic interactions among floral homeotic genes of Arabidopsis. Development 112:1–20
Carlsbecker A, Tandre K, Johanson U, Englund M, Engstro P (2004) The MADS-box gene DAL1 is a potential mediator of the juvenile-to-adult transition in Norway spruce (Picea abies). Plant J 40:546–557
Chen ZX, Wu JG, Ding WN, Chen HM, Wu P, Shi CH (2006) Morphogenesis and molecular basis on naked seed rice, a novel homeotic mutation of OsMADS1 regulating transcript level of AP3 homologue in rice. Planta 223:882–890
Cui R, Han J, Zhao S, Su K, Wu F, Du X, Xu Q, Chong K, Theißen G, Meng Z (2010) Functional conservation and diversification of class E floral homeotic genes in rice (Oryza sativa). Plant J 6:767–781
Ditta G, Pinyopich A, Robles P, Pelaz S, Yanofsky MF (2004) The SEP4 gene of Arabidopsis thaliana functions in floral organ and meristem identity. Curr Biol 14:1935–1940
Duan YL, Diao ZJ, Liu HQ, Cai MS, Wang F, Wu WR (2010) Molecular cloning and functional characterization of OsJAG gene based on a complete-deletion mutant in rice (Oryza sativa L.). Plant Mol Biol 74:605–615
Fan J, Li W, Dong X, Guo W, Shu H (2007) Ectopic expression of a hyacinth AGL6 homolog caused earlier flowering and homeotic conversion in Arabidopsis. Sci China C Life Sci 50:676–689
Gao X, Liang W, Yin C, Ji S, Wang H, Su X, Guo C, Kong H, Xue H, Zhang D (2010) The SEPALLATA-Like gene OsMADS34 is required for rice inflorescence and spikelet development. Plant Physiol 153:728–740
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J 6:271–282
Hsu HF, Huang CH, Chou LT, Yang CH (2003) Ectopic expression of an Orchid (Oncidium Gower Ramsey) AGL6-like gene promotes flowering by activating flowering time genes in Arabidopsis thaliana. Plant Cell Physiol 44:783–794
Jeon JS, Jang S, Lee S, Nam J, Kim C, Lee SH, Chung YY, Kim SR, Lee YH, Cho YG, An G (2000a) leafy hull sterile1 is a homeotic mutation in a rice MADS box gene affecting rice flower development. Plant Cell 12:871–884
Jeon JS, Lee S, Jung KH, Yang WS, Yi GH, Oh BG, An G (2000b) Production of transgenic rice plants showing reduced heading date and plant height by ectopic expression of rice MADS-box genes. Mol Breed 6:581–592
Kobayashi K, Maekawa M, Miyao A, Hirochika H, Kyozuka J (2010) PANICLE PHYTOMER2 (PAP2), encoding a SEPALLATA subfamily MADS-box protein, positively controls spikelet meristem identity in rice. Plant Cell Physiol 51:47–57
Koo SC et al (2010) Control of lateral organ development and flowering time by the Arabidopsis thaliana MADS-box Gene AGAMOUS-LIKE6. Plant J 62:807–816
Krizek BA, Riechmann JL, Meyerowitz EM (1999) Use of the APETALA1 promoter to assay the in vivo function of chimeric MADS box genes. Sex Plant Reprod 12:14–26
Lai Z, Ma W, Han B, Liang L, Zhang Y, Hong G, Xue Y (2002) An F-box gene linked to the self-incompatibility (S) locus of Antirrhinum is expressed specifically in pollen and tapetum. Plant Mol Biol 50:29–41
Lan L, Chen W, Lai Y, Suo J, Kong Z, Li C, Lu Y, Zhang Y, Zhao X, Zhang X, Han B, Cheng J, Xue Y (2004) Monitoring of gene expression profiles and isolation of candidate genes involved in pollination and fertilization in rice (Oryza sativa L.) using a 10 K cDNA microarray. Plant Mol Biol 54:471–487
Li H, Xue D, Gao Z, Yan M, Xu W, Xing Z, Huang D, Qian Q, Xue Y (2009) A putative lipase gene EXTRA GLUME1 regulates both empty-glume fate and spikelet development in rice. Plant J 57:593–605
Li H, Liang W, Jia R, Yin C, Zong J, Kong H, Zhang D (2010) The AGL6-like gene OsMADS6 regulates floral organ and meristem identities in rice. Cell Res 20:299–313
Li H, Liang W, Hu Y, Zhu Lu, Yin C, Xu J, Dreni L, Kater MM, Zhang D (2011) Rice MADS6 interacts with the floral homeotic genes SUPERWOMAN1, MADS3, MADS58, MADS13, and DROOPING LEAF in specifying floral organ identities and meristem Fate. Plant Cell 23:2536–2552
Liu C, Xi WY, Shen LS, Tan CP, Yu H (2009) Regulation of floral patterning by flowering time genes. Dev Cell 16:711–722
Malcomber ST, Kellogg EA (2004) Heterogeneous expression patterns and separate roles of the SEPALLATA gene LEAFY HULL STERILE1 in grasses. Plant Cell 16:1692–1706
Mizukami Y, Huang H, Tudor M, Hu Y, Ma H (1996) Functional domains of the floral regulator AGAMOUS: characterization of the DNA binding domain and analysis of dominant negative mutations. Plant Cell 8:831–845
Nagasawa N, Miyoshi M, Sano Y, Satoh H, Hirano H, Sakai H, Nagato Y (2003) SUPERWOMAN1 and DROOPING LEAF genes control floral organ identity in rice. Development 130:705–718
Ohmori S, Kimizu M, Sugita M, Miyao A, Hirochika H, Uchida E, Nagato Y, Yoshida H (2009) MOSAIC FLORAL ORGANS1, an AGL6-Like MADS box gene, regulates floral organ identity and meristem fate in rice. Plant Cell 21:3008–3025
Pelaz S, Ditta GS, Baumann E, Wisman E, Yanofsky MF (2000) B and C floral organ identity functions require SEPALLATA MADS-box genes. Nature 405:200–203
Prasad K, Sriram P, Kumar CS, Kushalappa K, Vijayraghavan U (2001) Ectopic expression of rice OsMADS1 reveals a role in specifying the lemma and palea, grass floral organs analogous to sepals. Dev Genes Evol 211:281–290
Prasad K, Parameswaran S, Vijayraghavan U (2005) OsMADS1, a rice MADS-box factor, controls differentiation of specific cell types in the lemma and palea and is an early-acting regulator of inner floral organs. Plant J 43:915–928
Reinheimer R, Kellogg EA (2009) Evolution of AGL6-like MADS box genes in grasses (Poaceae): ovule expression is ancient and palea expression is new. Plant Cell 21:2591–2605
Rijpkema AS, Zethof J, Gerats T, Vandenbussche M (2009) The petunia AGL6 gene has a SEPALLATA-like function in floral patterning. Plant J 60:1–9
Schauer SE, Baskar R, Brand L, Bolanos A, Grobei M, Federer MJ, Gagliardini V (2007) Examination of the role of the Arabidopsis MADS-box transcription factors AGL6 and AGL13 in reproduction. Dev Biol 306:312
Schauer SE, Baskara R, Schlüter P, Gheyselincka J, Bolaños A, Curtisa MD, Grossniklaus U (2008) Regulatory elements encoded in the first intron are necessary for proper expression of the MADS-box transcription factors AGL6 and AGL13 in Arabidopsis thaliana. Dev Biol 319:587
Thompson BE, Bartling L, Whipple C, Hall DH, Sakai H, Schmidt R, Hake S (2009) Bearded-ear encodes a MADS box transcription factor critical for maize floral development. Plant Cell 21:2578–2590
Tian B, Chen Y, Yan Y, Li D (2005) Isolation and ectopic expression of a bamboo MADS-box gene. Chin Sci Bull 50:217–224
Wang Z, Chen C, Xu Y, Jiang R, Han Y, Xu Z, Chong K (2004) A practical vector for efficient knockdown of gene expression in rice (Oryza sativa L.). Plant Mol Biol Rep 22:409–417
Yamaguchi T, Lee DY, Miyao A, Hirochika H, An G, Hirano HY (2006) Functional diversification of the two C-class genes OsMADS3 and OsMADS58 in Oryza sativa. Plant Cell 18:15–28
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572
Zahn LM, Kong H, Leebens-Mack JH, Kim S, Soltis PS, Landherr LL, Soltis DE, Depamphilis CW, Ma H (2005) The evolution of the SEPALLATA subfamily of MADS-box genes: a preangiosperm origin with multiple duplications throughout angiosperm history. Genetics 169:2209–2223
Zhang J, Nallamilli BR, Mujahid H, Peng Z (2010) OsMADS6 plays an essential role in endosperm nutrient accumulation and is subject to epigenetic regulation in rice. Plant J 64:604–617
Acknowledgments
We thank Yuanchang Zhou, Zhiwei Chen, Runsen Pan, Lihui Lin, Huazhong Guan, Xuzhang Zhang, Lijun Zhuang and Leilei Zheng of Fujian Agricultural & Forestry University and Yansheng Zhang, Yu’e Zhang, Qun Li and Xingming Hu of the Institute of Genetics and Developmental Biology of Chinese Academy of Sciences for their helps in the experiment, and Kang Chong of the Institute of Botany of Chinese Academy of Sciences for supplying the RNAi vector pTCK303. This work was supported by the National High Technology Research and Development Program of China (2006AA10Z128), the National Basic Research Program (2011CB100202), the Natural Science Foundation of China (30671122) and Natural Science Foundation of Fujian Provincial of China (B0620001).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yuanlin Duan and Zhuo Xing contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duan, Y., Xing, Z., Diao, Z. et al. Characterization of Osmads6-5, a null allele, reveals that OsMADS6 is a critical regulator for early flower development in rice (Oryza sativa L.). Plant Mol Biol 80, 429–442 (2012). https://doi.org/10.1007/s11103-012-9958-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-012-9958-2