Abstract
Polyamines are known to play important roles in plant stress tolerance but it has been difficult to determine precise functions for each type of polyamine and their interrelationships. To dissect the roles of putrescine from the higher polyamines spermidine and spermine, we generated transgenic rice plants constitutively expressing a heterologous S-adenosylmethionine decarboxylase (SAMDC) gene from Datura stramonium so that spermidine and spermine levels could be investigated while maintaining a constant putrescine pool. Whereas transgenic plants expressing arginine decarboxylase (ADC) produced higher levels of putrescine, spermidine and spermine, and were protected from drought stress, transgenic plants expressing SAMDC produced normal levels of putrescine and showed drought symptoms typical of wild type plants under stress, but the transgenic plants showed a much more robust recovery on return to normal conditions (90% full recovery compared to 25% partial recovery for wild type plants). At the molecular level, both wild type and transgenic plants showed transient reductions in the levels of endogenous ADC1 and SAMDC mRNA, but only wild type plants showed a spike in putrescine levels under stress. In transgenic plants, there was no spike in putrescine but a smooth increase in spermine levels at the expense of spermidine. These results confirm and extend the threshold model for polyamine activity in drought stress, and attribute individual roles to putrescine, spermidine and spermine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plant cells are abundant with receptors that respond to drought, and these activate signal transduction pathways that culminate in the induction of batteries of stress-response genes, leading to the synthesis of proteins that mediate diverse physiological and biochemical protective strategies against dehydration (Hu et al. 2006). Well-characterized drought-response proteins include chaperons, ion channels, transporters, antioxidant/detoxification enzymes, and enzymes that adjust the levels of osmoprotectant compounds, known as compatible solutes (reviewed by Vinocur and Altman 2005; Bhatnagar-Mathur et al. 2008). One useful strategy to improve drought tolerance is therefore to overexpress enzymes that synthesize compatible solutes such as amino acids, sugars, alcohols and polyamines (Umezawa et al. 2006).
Polyamines are ubiquitous, nitrogen-rich compounds implicated in many stress responses. Putrescine, spermidine and spermine are the most common linear polyamines and all are derived from arginine. In plants, putrescine is synthesized via two pathways, one in which arginine is converted into putrescine via the intermediate agmatine through the activity of arginine decarboxylase (ADC; EC 4.1.1.19) (Hanfrey et al. 2001), and another in which it is converted first to ornithine and then into putrescine through the activity of ornithine decarboxylase (ODC; EC 4.1.1.17). Putrescine is further converted into spermidine and then spermine by the enzymes spermidine synthase (SPDS; EC 2.5.1.16) and spermine synthase (SPMS; EC 2.5.1.22), respectively. These reactions require aminopropyl groups which are derived from S-adenosylmethionine (SAM) by SAM decarboxylase (SAMDC; EC 4.1.1.50) (Bagni and Tassoni 2001; Bassie et al. 2008).
The role of polyamines in stress tolerance was first demonstrated over 50 years ago, when Richards and Coleman (1952) showed that endogenous putrescine levels increase in response to potassium deficiency in barley (Hordeum vulgare L.). More recent studies have shown that many plants accumulate polyamines under biotic and abiotic stresses (reviewed by Alcazar et al. 2006; Groppa and Benavides 2008). Molecular genetic analysis using inhibitors, mutants and transgenic plants over-expressing specific polyamine biosynthetic genes has increased our understanding of the roles of polyamines in stress responses, and we have used rice (Oryza sativa L.; Os) as a model system to study these responses (Capell et al. 1998; Noury et al. 2000; Lepri et al. 2001).
Previously, we showed that transgenic rice overexpressing a Datura stramonium (Ds) SAMDC cDNA accumulated higher levels of putrescine and/or spermidine in leaves compared to wild type plants, mediated by a concomitant increase in ADC and ODC activities, although no changes in OsSAMDC and OsSPDS mRNA levels were measured (Thu-Hang et al. 2002). In subsequent experiments we used an antisense transgene to reduce OsADC mRNA levels and hence ADC activity, but OsODC, OsSAMDC and OsSPDS mRNA levels remained unchanged. We concluded that altering polyamine levels had no effect on the steady-state mRNA levels of endogenous pathway genes, demonstrating that the endogenous and transgenic pathways were uncoupled (Capell et al. 2000; Trung-Nghia et al. 2003). These data were used to derive a uniform model in which the putrescine pool must exceed a certain threshold to induce the synthesis of spermidine and spermine under stress. Such increases not only regulate putrescine levels, but also exert an anti-senescence effect at the whole plant level, resulting in phenotypically normal plants after 6 days (d) of drought-stress (Capell et al. 2004).
Having elucidated a role for putrescine in drought stress tolerance, we sought to determine the roles of spermidine and spermine by generating transgenic plants with elevated spermidine levels but normal levels of putrescine, through constitutive expression of DsSAMDC. Although such plants were not drought tolerant, the normal phenotype returned when stress was removed, whereas wild type plants were unable to recover. The analysis of polyamine and enzyme levels in transgenic and wild type populations throughout the stress and recovery periods showed that the higher polyamines play a key role in stress tolerance. It appears that spermine is the direct effector of stress tolerance whereas spermidine acts indirectly to maintain high spermine levels at crucial time points during stress. The impact of these data on the previously reported threshold model is discussed.
Materials and methods
Transgenic plants
Transgenic rice plants containing the Datura stramonium SAMDC cDNA (Ubi:DsSAMDC; Y07768; Fig. 1a) were generated as previously described (Thu-Hang et al. 2002).
Molecular and biochemical characterization of transgenic plants carrying the DsSAMDC transgene. a Map of Ubi:DsSAMDC showing transcription unit, relevant restriction sites, and primers used for analyses. The Datura SAMDC cDNA is 1,839 bp (AJ251898) in size. KpnI has one restriction site in the plasmid. Nos, nopaline synthase. Arrows represent primers and length of amplified fragment. b Gel blot analysis of KpnI digested genomic DNA (13 μg) from primary transformant (T0) and progeny plants (T1 and T2). Blot was probed with the 900 bp-DIG-labelled PCR product from Ubi:DsSAMDC; wt, wild type; 93 T0 primary transformant; 93-4 T1 representative progeny plant; numbers indicated different homozygous progeny plants (T2) from 93-4; Molecular mass of marker (1-kb DNA ladder, Invitrogen, UK) indicated in kilobases (kb). c Gel blot analysis of total RNA from leaf tissue (wt, wild-type, 93 T0 primary transformant; 93-4 T1 and T2 representative progeny; a 900 bp-DIG-labeled PCR probe from Ubi:DsSAMDC was used. Exposure time 45 min). UV fluorescence of ethidium bromide-stained gel showing equal loading of total RNA from plants used in the hybridizations shown above, lower panel. d SAMDC enzyme activity in different transgenic lines compared to appropriated controls (upper panel). Values are mean ± SE for control lines (n = 6) and mean ± SE in transgenic lines (n = 4). ADC activity in plants 1 and 4 was significantly different from control at P < 0.001 and for plants 2 and 3 at P < 0.01. Cellular polyamine levels (lower panel) in controls and four representative transgenic plants. Values are mean ± SE in control lines (n = 36) and mean ± SE in transgenic lines (n = 12). Significance of data is as follows: ***P < 0.001; **0.01 > P > 0.001; remaining values were not significantly different from wild type at P > 0.05
Drought stress treatment
Two-month-old wild type rice plants (Oryza sativa L. subsp. Japonica cv. EYI105) were used to determine a drought stress regime that gave a clear phenotype, which was then applied to experimental wild type and transgenic plants. We selected 40 homozygous R 2 plants derived from transgenic line 93-4 and divided them into groups of five with five wild type controls. The plants were grown at 26 ± 2°C, with a 12-h photoperiod (900 μmohn m−2 s−1 photosynthetically active radiation) and 80% relative humidity. After 60 d, the water was replaced with 20% polyethyleneglycol (Mr 8000) (Perez-Molphe-Balch et al. 1996) for 6 d. Leaf samples were collected at 0, 3 and 6 d after PEG treatment. The PEG solution was then replaced with water. The plants were allowed to recover for 20 d and final samples were collected. Recovery was defined as the return to a near normal phenotype including new growth. The drought stress experiment was carried out four times, over a 2-year period.
DNA and RNA gel blot analysis
Genomic DNA was extracted from leaves according to Edwards et al. (1991), and 13 μg was digested with KpnI (which cuts once in the transforming plasmid backbone), separated by 0.8% agarose gel electrophoresis and transferred to a positively charged nylon membrane (Roche UK). DIG-labeled Ds and Os SAMDC probes were prepared by PCR as described (Thu-Hang et al. 2002; Trung-Nghia et al. 2003), denatured at 68°C for 10 min and hybridized to the membrane overnight at 42°C. After high-stringency washing, the signal was detected using a DIG Luminescence Detection Kit (Roche, UK) according to the manufacturer’s instructions. Stripping and reprobing of the membranes was performed as described (Lahaye et al. 1996).
Total RNA was extracted from leaves from 2- to 3-month-old plants using Trizol® (Invitrogen), and 30 μg of denatured RNA was separated by 1.2% agarose-formaldehyde gel electrophoresis in 1× MOPS buffer (Sambrook et al. 1989). Primers used to generate the OsADC1 specific probe were: For5 (5′-TCT GTA CGC CGA CCA GCT GAA-3′; in the position +1,350 bp, defining position +1 the ATG from OsADC1 ORF) and Rev6 (5′-TGC AAG AAC TCC TCG CGT GAA CAC CAA GAA-3′) amplifying a fragment of 810 bp of the AY604047 sequence (Akiyama and Jin 2007). Blotting, hybridization and signal detection were carried out as described above. All experiments were carried out four times using independent RNA preparations, and the steady state levels of OsADC1, OsSAMDC1 and DsSAMDC mRNA were quantified using Quantity One (Quantification Software; Bio-Rad) normalized against RNA loading levels. OsSAMDC1 (Y07766) and DsSAMDC (Y07768) cDNAs share 44% nucleotide sequence identity and do not cross-hybridize under high stringency conditions.
Synthesis of polyclonal antibodies against rice ADC1
In order to synthesize polyclonal antibodies against ADC1 we selected a specific epitope based on antigenicity prediction using the Jameson and Wolf algorithm (Jameson and Wolf 1988). The 14-residue peptide (ASGESSGMSSDSEG) located 668 residues from the ADC1 C-terminus had the highest predicted antigenic index (Fig. 2a). This peptide was used to raise polyclonal antibodies in New Zealand white rabbits (Genscript Corp. Piscataway, NJ, USA).
a Deduced amino acid sequence of OsADC1 (GenBank accession no. AY644047). Amino acids are designated in single letter code. Antigenic sequences are shown in bold. Arrowhead in position 668 indicates the antigenic sequence we used to generate anti ADC1 antibodies. b Western blot analysis of total rice protein extracts from leaves and roots (30 μg per lane) from 2-month old plants. After SDS-PAGE analysis and transfer, blots were probed overnight at 4°C with 1:5000 dilution of (I) pre-immune serum and (II) anti-ADC1. L, New England BioLabs Pre-stained protein marker, broad range (#P7701)
Protein analysis
Leaf proteins were extracted in two volumes of buffer (0.2 M Tris–HCl pH 7.5, 5 mM EDTA, 0.1% Tween 20), vortexed for 1 h at 4°C and centrifuged for 10 min at 13,800×g The supernatant protein concentration was determined using the Bradford method (Bradford 1976) with BSA as the standard. For immunoblotting, 50 μg aliquots of protein were separated by 10% SDS-PAGE (Laemmli 1970) and transferred to Immun-Blot PVDF membranes (BioRad, Hercules CA, USA). The primary antibody (see above) was diluted 1:5000 and the secondary alkaline phosphatase-conjugated goat anti-rabbit IgG (Sigma) was diluted 1:30000. Rabbit pre-immune serum was diluted 1:5000.
SAMDC assay
Leaf tissue (300 mg) was extracted in 1 ml buffer (1 M Tris pH 7.5, 2 mM DTT, 1 mM EDTA) and ground with 100 mg polyvinylpyrrolidone. Following centrifugation at 12,000×g for 20 min, the supernatant was used directly in enzyme activity assays. Tissue was always processed immediately after harvest and all assays were performed using fresh extracts. Enzyme assays were carried out as described (Thu-Hang et al. 2002). One nKat of SAMDC activity was defined as 1 μmol 14CO2 released min−1 g−1 fresh weight (fw).
Polyamine analysis
Crude leaf extracts were dansylated, separated by thin layer chromatography and quantified as previously described (Capell et al. 1998; Bassie et al. 2000). Results were expressed as nmol g−1 fw.
Statistical analysis
We used hpt-transformed as well as wild-type plants (average of three samples each from six independent lines; n = 36) to determine control values for biochemical analyses (enzyme activity and polyamine content). Hygromycin-resistant transformants and wild-type values were not significantly different in terms of enzyme activity and polyamine levels (P > 0.05). The polyamine content of wild type and transgenic plants was determined using the average value of three samples from each sibling (n = 3). Drought stress experiments were carried out four times, each experiment involving five transgenic and five wild type plants. Three samples were collected from each plant for polyamine measurements at each time point (0, 3 and 6 d and recovery). The polyamine levels presented are the average of four experiments (n = 12). The data were analyzed by two-way analysis of variance followed by a t-test using the Residual Mean Square in the ANOVA as the estimate of variability.
Results
Molecular and biochemical characterization of DsSAMDC transgenic rice plants
Transformed plants containing an intact Ubi:DsSAMDC transgene (Fig. 1a) were identified by amplifying a 900-bp diagnostic fragment. Testing for polyamine levels revealed six lines with increased levels of spermidine but normal levels of putrescine (Thu-Hang et al. 2002). We selected line 93 for further experiments. This line was representative of the DsSAMDC lines and was characterized fully molecularly and biochemically through the T2 homozygous generation. DNA blots using the 900-bp PCR product described above as a probe revealed that the transgene segregated in a 3:1 ratio, confirming single locus integration (Fig. 1b). Digestion was carried out with KpnI, which cuts once within the backbone sequence of the transforming plasmid (Fig. 1a) revealing that multiples copies of the transgene had integrated at the same locus. Progeny plants (T1 and T2) produced the same transgene banding patterns as the primary transformant (a representative sample from 40 T2 progeny is shown in Fig. 1b). This homozygous population was used in all the stress experiments.
RNA gel blot analysis showed that the steady-state DsSAMDC mRNA level was similar in T0, T1 and homozygous T2 plants from line 93 (Fig. 1c). SAMDC activity was analyzed in leaves simultaneously with mRNA and polyamine measurements. Background SAMDC activity in wild type plants was 0.40–0.50 nKat, whereas homozygous transgenic plants showed significantly higher levels (up to 1.69 nKat; P < 0.001). Representative examples are shown in Fig. 1d, upper panel. Putrescine, spermidine and spermine levels were determined in the same samples allowing polyamine levels to be compared to SAMDC activities (Fig. 1d, lower panel). A two-fold increase in spermidine was observed in the primary transformant (676.23 ± 39 nmol g−1 fw; P < 0.001) compared to wild-type (315.2 ± 29 nmol g−1 fw). T1 plants that inherited the transgene also showed a 1.5 to 2-fold increase in spermidine levels over wild type. Similarly, spermidine levels in homozygous T2 progeny varied from 1.5-fold (485.43 ± 60 nmol g−1 fw; P < 0.01) to 2-fold (639.7 ± 45 nmol g−1 fw; P < 0.001) compared to wild-type (320.7 ± 26 nmol g−1 fw). No significant variation (P > 0.05) was detected in putrescine or spermine levels in the leaves of any of these plants, all of which were phenotypically normal and fertile.
Anti-ADC1 antibodies detect a 63-kDa protein in rice leaves
Polyclonal antibodies specific for OsADC1 (Fig. 2a) were used to detect the enzyme ADC1 in immunoblots of total rice protein from different tissues. A 63-kDa species was detected in leaves, but not in roots (Fig. 2b). The size of this protein is consistent with a previously purified rice ADC (Reggiani 1994). Some cross-reactivity with other proteins was observed, but these were not specific interactions since the same bands were also seen following incubation with the pre-immune serum. The antiserum raised against OsADC1 was used to characterize the behavior of the enzyme under drought stress in subsequent experiments in wild type and transgenic plants.
Drought stress response in wild type rice plants
Leaf samples from 2 month old plants were collected on days 0 (Fig. 3a-I), 3 (Fig. 3a-II) and 6 (Fig. 3a-III). In each panel, the two plants on the left were maintained under normal submerged conditions whereas the two plants on the right were subjected to PEG treatment to induce drought stress. The symptoms of dehydratation (wilting and curling of the leaves) were clear after 3 d and became more severe at 6 d. The PEG solution was replaced with water on day seven and recovery was assessed after 20 d. Twenty-five percent of wild type plants recovered to a near normal phenotype and initiated new growth, producing one new tiller per plant.
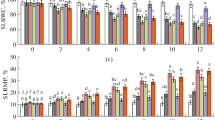
Molecular and biochemical characterization of wild type plants under drought stress. a I—Phenotype of 2 month old wild type plants under submerged conditions; II—Phenotype of plants: normal submerged conditions (2 plants on the left) and after 3 d treatment (2 plants on the right); III—Phenotype of plants: normal submerged conditions (2 plants on the left) and after 6 d treatment (two plants on the right). b Gel blot analysis of total RNA from leaf tissue (numbers represent individual plants). A 800-bp rice OsADC1 (AY644047) DIG labeled PCR probe was used. Exposure time 1 h 30 min. Membrane was re-probed with a 800 bp-DIG-labeled PCR probe from OsSAMDC (Trung-Nghia et al. 2003). Exposure time 1 h. UV fluorescence of ethidium bromide-stained gel showing equal loading of total RNA from plants used in the hybridizations shown above, lower panel. c Putrescine, spermidine and spermine levels in wild type plants after different periods of drought stress. Values are mean ± SE (n = 12). Period 6–20 d on the X axes not to scale
Steady-state OsADC1 mRNA levels were also monitored at 0, 3 and 6 d after stress induction (Fig. 3b). The OsADC1 probe hybridized to two transcripts, the most prominent 3 kb in length and a weaker moiety of 2.4 kb. Although differing in abundance, the accumulation profile of each transcript was the same, reducing to 25% of the normal level after 3 d and increasing after 6 d to approximately 50% of normal levels. Twenty days after release from stress, OsADC1 mRNA returned to near its initial level (Fig. 3b). All mRNA levels reported in this experiment were relative rather than absolute, but the same profiles were seen when the experiment was carried out four times. There were no changes in the levels of ADC1 protein as detected by immunoblot over the same period (data not shown).
Steady-state OsSAMDC1 mRNA levels were also monitored at 0, 3 and 6 d after stress induction (Fig. 3b). Like OsADC1 mRNA, the levels of OsSAMDC1 mRNA were reduced after 3 d of stress, but after 6 d of stress and throughout the recovery period, steady-state OsSAMDC1 mRNA levels returned to normal (Fig. 3b).
The amount of putrescine doubled in stressed plants after 6 d (711.2 ± 32 nmol g−1 fw, P < 0.001) compared to non-stressed controls (369.7 ± 52 nmol g−1 fw; Fig. 3c). After 3 d of drought stress, the plants accumulated up to 2-fold spermidine (440.3 ± 48 nmol g−1 fw; P < 0.01) and 1.5-fold spermine (336.70 ± 59 nmol g−1 fw; P < 0.05) compared to non-stressed controls (spermidine 230.8 ± 30 nmol g−1 fw; spermine 218.6 ± 63 nmol g−1 fw; Fig. 3c). No significant differences in spermidine or spermine accumulation were detected after 6 d of drought stress (Fig. 3c). After 20 d of recovery leaf polyamine content was not significantly different to levels measured at day 0 (Fig. 3c).
Drought stress response in transgenic rice plants expressing DsSAMDC
Drought stress responses were evaluated in 2-month-old transgenic plants expressing DsSAMDC (Fig. 4). Leaf samples from 2-month-old plants were collected on days 0 (Fig. 4a-I), 3 (Fig. 4a-II) and 6 (Fig. 4a-III). Each panel shows two wild type plants as controls (left) and two transgenic plants (right). The wild type and transgenic plants showed similar drought stress phenotypes (wilting and leaf curling) after 3 d. Although the T2 plants were siblings there was variation in the level of DsSAMDC mRNA, but the relative changes in expression over the treatment and recovery course were similar in each plant (two representative examples with medium and high mRNA expression levels are shown in Fig. 4b, top panel). There also appeared to be no difference in the expression profile of OsSAMDC1 in wild type and transgenic plants, showing first a dip after 3 d under stress, a slight rise after 6 d and then a return to basal levels during recovery (Fig. 4b, second panel).
Molecular characterization of wild type (2 plants on the left) and transformed plants (2 plants on the right) under drought stress. a I—Phenotype of two month old 93-4 progeny (T2) plants; II—after 3 d and III—after 6 d of drought stress. b Gel blot analysis of total RNA from leaf tissue (wt, wild-type, numbers represent individual progeny T2 plants from 93-4). A 900-bp DIG-labelled probe from DsSAMDC (exposure time 45 min) and a 800-bp-DIG-labeled PCR probe from OsSAMDC1 (exposure time 1 h) were used (Thu-Hang et al. 2002; Trung-Nghia et al. 2003). Membrane was re-probed with a 800-bp OsADC1 (AY644047) DIG labeled PCR probe. Exposure time 1 h 30 min. UV fluorescence of ethidium bromide-stained gel showing equal loading of total RNA from plants used in the hybridizations shown above, lower panel. c Western blot analysis of protein extracts from 2 month old wild type and individual progeny plants from 93-4, using anti-ADC1 antibodies (wt, wild type); L, New England BioLabs Prestained protein marker, broad range (#P7701). Each lane was loaded with 30 μg of protein
The OsADC1 probe hybridized to two transcripts in transgenic and wild type plants, each showing the same relative changes in steady state mRNA levels during stress and recovery. In all plants, there was a dip to approximately 20% normal levels after 3 d under stress conditions, a partial recovery to approximately 50% normal levels after 6 d and then full recovery after 20 d in both transgenic plants and surviving wild type plants (Fig. 4b, third panel). The level of ADC protein was monitored by immunoblot over the same timescale and a 63-kDa polypeptide detected by the antibody in all wild type and transgenic plants was present at constant levels throughout (Fig. 4c).
No substantial changes in putrescine levels were observed in transgenic plants after 3 and 6 d under stress (360.2 ± 36 nmol g−1 fw to 483.9 ± 62 nmol g−1 fw; P > 0.05; Fig. 5a) which contrasts sharply with the dramatic accumulation of putrescine in wild type plants under the same conditions (Figs. 3d and 5a). The spermidine content of transgenic plants remained the same after 3 d under stress (561.25 ± 67 nmol g−1 fw, P < 0.001), double the level observed in wild-type plants at day 0 (292.8 ± 36 nmol g−1 fw). However, spermidine levels in transgenic plants reverted to basal levels after 6 d (241.3 ± 31 nmol g−1 fw; Fig. 5a). There was a concomitant increase in spermine, commencing 3 d after stress induction (313.5 ± 38 nmol g−1 fw P < 0.01; Fig. 5a) reaching a maximum 2.5-fold increase after 6 d under stress (449.8 ± 28 nmol g−1 fw P < 0.001; Fig. 5a) compared to spermine levels in wild type and transgenic plants at day 0 (186.25 ± 41 nmol g−1 fw) and compared to wild type plants after 6 d (255.6 ± 47 nmol g−1 fw; Fig. 5a).
Biochemical characterization of wild type and transformed plants under drought stress. a Putrescine, spermidine and spermine levels in wild type and the line 93-4-4 plants after different periods of drought stress. Period 6–20 d on the X axes not to scale. b Phenotype of 2 month old wild type (wt) and plants expressing Ubi:DsSAMDC (93-4-4) after 20 d of recovery
Development of plants under stress and during recovery
In four experiments each involving five wild type plants, a total of five plants recovered when stress was removed (two in the first two experiments, one in the third, none in the fourth; Fig. 5b). This 25% recovery rate was poor compared to the transgenic plants, where a total of 18 plants recovered (four in each of the first two experiments and all five in the third and fourth). This is a 90% recovery rate (Fig. 5b). All the recovered plants were able to set seeds, but the wild type plants could only manage to produce one new tiller apiece, whereas the transgenic plants formed two or three new tillers.
Discussion
Polyamines have long been associated with stress responses in plants, but it has been difficult to pin down their precise role and determine how the levels of different polyamines influence different stress response pathways. Our studies of drought tolerance in transgenic plants expressing different components of the polyamine metabolic pathway have shown that the accumulation of putrescine is a key factor, and that exceeding a threshold level of putrescine may be an important trigger for downstream events mediated by the higher polyamines spermidine and spermine (Capell et al. 2004).
Our strategy to elucidate the individual roles of different polyamines in drought tolerance has been to generate populations of transgenic plants expressing individual enzymes and to investigate the responses of these plants under normal and stress conditions (Capell et al. 1998, 2000; Noury et al. 2000; Lepri et al. 2001; Thu-Hang et al. 2002; Trung-Nghia et al. 2003). These studies have helped to generate the ‘threshold model’ (Capell et al. 2004) in which excess putrescine is converted into the higher polyamines spermidine and spermine, which mediate the direct effects of stress tolerance by inhibiting senescence. Spermidine and spermine also feedback to regulate the putrescine pool, with the result that normal metabolic and morphological phenotypes are restored under stress. In wild type plants, the putrescine pool never exceeds the threshold level, and drought symptoms become evident in a few days. In transgenic plants expressing ADC, putrescine synthesis is enhanced even under normal conditions, stress results in the trigger level being exceeded and the accumulation of spermidine and spermine leads to the restoration of normal growth and development, even under stress.
To refine this model, we generated transgenic plants constitutively expressing a heterologous SAMDC gene (DsSAMDC), which should increase the levels of higher polyamines without affecting putrescine levels, therefore allowing us to determine the contribution of the higher polyamines without the confounding effects of putrescine. The conversion of putrescine to spermidine and spermine is mediated by the enzymes SPDS and SPMS, respectively, which transfer the aminopropyl groups provided by SAMDC to their respective substrates (Bagni and Tassoni 2001; Bassie et al. 2008). However, since neither SPDS nor SPMS is limiting for higher polyamine synthesis, increasing the level of SAMDC activity is sufficient to increase the spermidine and spermine pools in transgenic plants. Noh and Minocha (1994) over-expressed the human SAMDC cDNA in transgenic tobacco plants resulting in a 2–3 fold increase in spermidine although spermine levels were not reported. Similar results were observed when the homologous SAMDC cDNA was re-introduced into potato driven by the tuber-specific patatin promoter (Pedros et al. 1999). The spermine level also doubled in transgenic mice hearts expressing human SAMDC with a concomitant decrease in spermidine and putrescine levels (Nisenberg et al. 2006). It has been suggested that the toxicity of spermidine places a cap on the maximum levels that can be achieved in transformed cells (Noh and Minocha 1994; Schipper et al. 2000) which may explain why we never see higher than double the normal amount of spermidine in transgenic rice plants. The expression of SAMDC should channel the flux from spermidine into spermine, and although spermine is ubiquitous in eukaryotic cells at high levels (Hamasaki-Katagiri et al. 1998), we postulated that tighter regulation of cellular spermine metabolism, compared to putrescine (Noury et al. 2000) or spermidine (Bassie et al. 2000), occurs because spermine may be the most active polyamine in terms of stress tolerance (Jiménez-Bremont et al. 2007 and references therein).
Transgenic rice overexpressing DsSAMDC accumulated higher levels of putrescine and/or spermidine in leaves compared to wild type plants, mediated by a concomitant increase in ADC and ODC activities, although no changes in OsSAMDC and OsSPDS mRNA levels were observed (Thu-Hang et al. 2002). Under stress, both wild type and DsSAMDC transgenic rice plants showed a significant reduction in the levels of endogenous ADC1 and SAMDC mRNAs, although in the transgenic plants, the loss of endogenous SAMDC was compensated by the transgene. Both the wild type and transgenic plants showed equivalent phenotypic signs of drought stress after 3 and 6 d, but only the transgenic plants mounted a strong recovery, with 90% of the plants returning to a normal phenotype after 20 d compared to only 25% of wild type plants, which returned to a partially-restored phenotype. The wild type plants underwent a spike of putrescine accumulation under stress but this was not converted into an increase in the higher polyamines. In contrast, the transgenic plants showed no evidence of putrescine accumulation but smoothly accumulated spermine, at the expense of spermidine, over the same stress period.
All DsSAMDC transgenic plants were able to develop multiple tillers in the course of their growth in sharp contrast to the few surviving wild type plants which at best could only produce one tiller. It appears, therefore, that DsSAMDC-expressing plants are able to compensate for long term effects of the drought stress in contrast to wild type plants.
No changes in AtADC1 expression were detected in dehydrated Arabidopsis plants (Urano et al. 2003) or detached leaf disks subjected to osmotic stress (Soyka and Heyer 1999) but AtADC2 underwent significant stress-dependent changes in expression. In rice, the apparent ADC1 ortholog OsADC1 was down-regulated 3 d after the onset of stress and remained suppressed for up to 6 d (Figs. 3b and 4b) in wild type plants and transformants. However, transcriptional regulation of the ADC1 gene was reported 6 h after wounding in Arabidopsis (AtADC1; Perez-Amador et al. 2002) and during pea ovary growth and early fruit development (Perez-Amador et al. 1995). Several ADC cDNA clones have been isolated and characterised from various species. Whereas in some plants a single gene encodes ADC (Kakar and Sawhney 2002; Delis et al. 2005; Eller et al. 2006), Arabidopsis has two copies of the ADC gene and duplication has also been found in 12 species of the Brassicaceae family (Galloway et al. 1998; Mo and Pua 2002). Akiyama and Jin (2007) cloned the first OsADC1 cDNA (AY604047) and indicated that a putative OsADC2 sequence was present in the rice genome. This suggests that rice plants activate a second ADC gene under drought stress that might regulate flux through the polyamine pathway. Furthermore, using the OsADC1 gene-specific probe we detected two different OsADC mRNA species (Figs. 3b and 4b). The expression patterns of the two mRNA species remain stable throughout the stress period. Surveying the rice genome database we identified a putative transcriptional start site 530 bp upstream of the site mapped by Akiyama and Jin (2007). Whether this overlapping transcription unit is responsible for the two mRNA species we observed has yet to be determined.
Osmotic shock in darkness induces ADC activity and results in putrescine accumulation in oat leaf segments (Capell et al. 1998). Borrell et al. (1995, 1996) showed that there was a significant increase in the levels of a 24-kD polypeptide in the osmotically-stressed oat leaves when compared with the 0 h control. In oat, ADC is synthesized as a pre-protein (66 kD) which is self-processed into an active 24-kD enzyme (Bell and Malmberg 1990; Malmberg and Cellino 1994) that can be inhibited by spermine (Borrell et al. 1996). We found that ADC protein accumulation in wild type and transgenic plants did not change during stress and recovery (Fig. 4c) and was not affected by either spermidine (Fig. 5a; 0 d) or spermine (Fig. 5a; 6 d), which contradicts the mRNA data and previous results with ADC protein. Since our antiserum detected a peptide near the C-terminus of ADC1 (Fig. 2a), it should also detect the smaller, processed form of the protein. However, we cannot rule out that conformation changes during processing make the processed enzyme undetectable with this antiserum, or that ADC is not cleaved post-translationally in rice and instead undergoes some other form of modification to control its activity. While post-translational cleavage has been demonstrated in oat and Arabidopsis (Watson and Malmberg 1996) it appears no such processing occurs in Brassica campestris (Primikirios and Roubelakis-Angelakis 2001).
The key to understanding the role of polyamines in drought stress is to determine the individual roles of each polyamine in isolation. Since our threshold model suggested that the role of putrescine was to maintain a pool to facilitate the synthesis of spermidine and spermine under stress (Capell et al. 2004) the creation of plants with immediate access to the higher polyamines was crucial to advance our understanding. A remarkable difference between wild type and DsSAMDC transgenic plants under stress was that the significant increase in putrescine levels detected 3 d after the onset of stress in wild type plants (Fig. 3c) was not detected in the transgenic plants (Fig. 5a). Instead, these plants accumulated spermine directly, suggesting that SAMDC is a rate limiting enzyme that prevents putrescine converting to higher polyamines under stress. In the same transgenic plants we observed an induction of the OsSAMDC gene after 3 d of stress (Figs. 3b and 4b) and those with the highest levels of spermine recovered fully when stress was removed, compared to wild type plants which were unable to recover fully, if at all.
There was no perturbation in putrescine homeostasis in transgenic plants growing normally or under stress, i.e. spermine synthesis was uncoupled from putrescine metabolism in this transgenic system. This allowed us to establish an unequivocal physiological role for the higher polyamines, with spermidine acting as an intermediate and spermine as a key protective molecule with a direct impact on stress recovery. A double knock-out of Arabidopsis ACL5 and SPMS generated plants that were unable to produce spermine and were defective in stem elongation (Hanzawa et al. 2000). This mutant was also hypersensitive to drought but could be rescued by pre-exposure to spermine, but not to putrescine or spermidine (Yamaguchi et al. 2007). Our experiments thus provide a basis to further refine and extend our ‘threshold model’. Plants expressing ADC can accumulate putrescine, and once this exceeds a certain threshold the excess putrescine is converted to spermidine and spermine, providing stress tolerance after 6 d under stress conditions (Capell et al. 2004). In contrast, plants expressing SAMDC accumulate the higher polyamines while putrescine levels remain normal. These plants do not show stress tolerance but can recover more readily when stress is removed. A direct protective effect has been reported for spermine in stabilization of the membrane structure in mesophyll protoplasts isolated from osmotically stressed oat leaves (Tiburcio et al. 1994); spermine may also act as a free radical scavenger in the nucleus (Ha et al. 1998) and a spermine-dependent signaling pathway involved in the hypersensitive response has been demonstrated in tobacco leaves (Takahashi et al. 2003). We thus conclude that drought tolerance depends on the balance between putrescine and spermine, with spermidine acting as a key intermediate. Plants with elevated putrescine are able to tolerate stress because putrescine has a direct protective role in preventing the symptoms of dehydration, but it also feeds the spermine pool (via spermidine) which exerts additional effects that aid recovery when the stress conditions are removed. When spermine synthesis is uncoupled from putrescine levels, we see an interesting effect in which the immediate protective effects of putrescine are absent but the lasting protective effects of spermine on plant recovery are maintained. Such effects not only manifest at the molecular and phenotypic levels but crucially extend to long term development. The later effects are important in our continuing quest to develop plants that are better able to tolerate environmental stresses thus improving agricultural productivity.
References
Akiyama T, Jin S (2007) Molecular cloning and characterization of an arginine decarboxylase gene up-regulated by chilling stress in rice seedlings. J Plant Physiol 164:645–654. doi:10.1016/j.jplph.2006.04.006
Alcazar R, Marco F, Cuevas JC, Patron M, Ferrando A, Carrasco P, Tiburcio AF, Altabella T (2006) Involvement of polyamines in plant response to abiotic stress. Biotechnol Lett 28:1867–1876. doi:10.1007/s10529-006-9179-3
Bagni N, Tassoni A (2001) Biosynthesis, oxidation and conjugation of aliphatic polyamines in higher plants. Amino Acids 20:301–307. doi:10.1007/s007260170046
Bassie L, Noury M, Lepri O, Lahaye T, Christou P, Capell T (2000) Promoter strength influences polyamine metabolism and morphogenic capacity in transgenic rice tissues expressing the oat arginine decarboxylase cDNA constitutively. Transgenic Res 9:33–42. doi:10.1023/A:1008997822463
Bassie L, Zhu C, Romagosa I, Christou P, Capell T (2008) Transgenic wheat plants expressing an oat arginine decarboxylase cDNA exhibit increases in polyamine content in vegetative tissue and seeds. Mol Breed 22:39–50. doi:10.1007/s11032-007-9154-2
Bell E, Malmberg RL (1990) Analysis of cDNA encoding arginine decarboxylase from oat reveals similarity to the Escherichia coli arginine decarboxylase and evidence of protein processing. Mol Gen Genet 224:431–436. doi:10.1007/BF00262438
Bhatnagar-Mathur P, Vadez V, Sharma KK (2008) Transgenic approaches for abiotic stress tolerance in plants: retrospect and prospects. Plant Cell Rep 27:411–424. doi:10.1007/s00299-007-0474-9
Borrell A, Culianez-Macia FA, Altabella T, Besford RT, Flores D, Tiburcio AF (1995) Arginine decarboxylase is localized in chloroplasts. Plant Physiol 109:771–776
Borrell A, Besford RT, Altabella T, Masgrau C, Tiburcio AF (1996) Regulation of arginine decarboxylase by spermine in osmotically-stressed oat leaves. Physiol Plant 98:105–110
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein using the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Capell T, Escobar C, Lui H, Burtin D, Lepri O, Christou P (1998) Over-expression of the oat arginine decarboxylase cDNA in transgenic rice (Oryza sativa L.) affects normal development patterns in vitro and results in putrescine accumulation in transgenic plants. Theor Appl Genet 97:246–254. doi:10.1007/s001220050892
Capell T, Bassie L, Topsom L, Hitchin E, Christou P (2000) Simultaneous reduction of the activity of two related enzymes, involved in early steps of the polyamine biosynthetic pathway, by a single antisense cDNA in transgenic rice. Mol Gen Genet 264:470–476. doi:10.1007/s004380000317
Capell T, Bassie L, Christou P (2004) Modulation of the polyamine biosynthetic pathway in transgenic rice confers tolerance to drought stress. Proc Natl Acad Sci USA 101:9909–9914. doi:10.1073/pnas.0306974101
Delis C, Dimou M, Efrose RC, Flemetakis E, Aivalakis G, Katinakis P (2005) Ornithine decarboxylase and arginine decarboxylase gene transcripts are co-localized in developing tissues of glycine max etiolated seedlings. Plant Physiol Biochem 43:19–25. doi:10.1016/j.plaphy.2004.11.006
Edwards K, Johnstone C, Thompson C (1991) A simple and rapid method for the preparation of plant genomic DNA for PCR. Nucleic Acids Res 19:1349. doi:10.1093/nar/19.6.1349
Eller MH, Warner AL, Knap HT (2006) Genomic organization and expression analyses of putrescine pathway genes in soybean. Plant Physiol Biochem 44:49–57. doi:10.1016/j.plaphy.2006.01.006
Galloway GL, Malmberg RL, Price RA (1998) Phylogenetic utility of the nuclear gene arginine decarboxylase: an example from Brassicaceae. Mol Biol Evol 15:1312–1320
Groppa MD, Benavides MP (2008) Polyamines and abiotic stress: recent advances. Amino Acids 34:35–45. doi:10.1007/s00726-007-0501-8
Ha HC, Sirisoma NS, Kuppusamy P, Zweier JL, Woster PM, Casero RA (1998) The natural polyamine spermine functions directly as a free radical scavenger. Proc Natl Acad Sci USA 95:11140–11145. doi:10.1073/pnas.95.19.11140
Hamasaki-Katagiri N, Katagiri Y, Tabor CW, Tabor H (1998) Spermine is not essential for growth of Saccharomyces cerevisiae: identification of the SPE4 gene (spermine synthase) and characterization of a spe4 deletion mutant. Gene 210:195–201. doi:10.1016/S0378-1119(98)00027-4
Hanfrey C, Sommer S, Mayer MJ, Burtin D, Michael AJ (2001) Arabidopsis polyamine biosynthesis: absence of ornithine decarboxylase and the mechanism of arginine decarboxylase. Plant J 27:551–560. doi:10.1046/j.1365-313X.2001.01100.x
Hanzawa Y, Takahashi T, Michael AJ, Burtin D, Long D, Pineiro M, Coupland G, Komeda Y (2000) ACAULIS5, an Arabidopsis gene required for stem elongation, encodes a spermine synthase. EMBO J 19:4248–4256. doi:10.1093/emboj/19.16.4248
Hu H, Dai M, Yao J, Xiao B, Li X, Zhang Q, Xiong L (2006) Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proc Natl Acad Sci USA 35:12987–12992. doi:10.1073/pnas.0604882103
Jameson BA, Wolf H (1988) The antigenic index: a novel algorithm for predicting antigenic determinants. Comput Appl Biosci 4:181–186
Jiménez-Bremont JF, Ruiz OA, Rodríguez-Kessler M (2007) Modulation of spermidine and spermine levels in maize seedlings subjected to long-term salt stress. Plant Physiol Biochem 45:812–821. doi:10.1016/j.plaphy.2007.08.001
Kakar RK, Sawhney VK (2002) polyamine research in plants—a changing perspective. Physiol Plant 116:281–292. doi:10.1034/j.1399-3054.2002.1160302.x
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. doi:10.1038/227680a0
Lahaye T, Rueger B, Toepsch S, Thalhammer J, Schulze-Lefert P (1996) Detection of single-copy sequences with digoxigenin-labeled probes in a complex plant genome after separation on pulsed-field gels. Biotechniques 21:1067–1072. doi:10.1002/9783527620883
Lepri O, Bassie L, Safwat G, Thu-Hang P, Trung-Nghia P, Hölttä E, Christou P, Capell T (2001) Over-expression of the human ornithine decarboxylase cDNA in transgenic rice plants alters the polyamine pool in a tissue-specific manner. Mol Gen Genet 266:303–312
Malmberg RL, Cellino ML (1994) Arginine decarboxylase of oats is activated by enzymatic cleavage into two polypeptides. J Biol Chem 269:2703–2706
Mo H, Pua EC (2002) Up-regulation of arginine decarboxylase gene expression and accumulation of polyamines in mustard (Brassica juncea) in response to stress. Physiol Plant 114:439–449. doi:10.1034/j.1399-3054.2002.1140314.x
Nisenberg O, Pegg AE, Welsh PA, Keefer K, Shantz LM (2006) Overproduction of cardiac S-adenosylmethionine decarboxylase in transgenic mice. Biochem J 393:295–302. doi:10.1042/BJ20051196
Noh EW, Minocha SC (1994) Expression of a human S-adenosylmethionine decarboxylase cDNA in transgenic tobacco and its effects on polyamine biosynthesis. Transgenic Res 3:25–53. doi:10.1007/BF01976024
Noury M, Bassie L, Lepri O, Kurek I, Christou P, Capell T (2000) A transgenic rice cell lineage expressing the oat arginine decarboxylase (adc) cDNA constitutively accumulates putrescine in callus and seeds but not in vegetative tissues. Plant Mol Biol 43:357–544. doi:10.1023/A:1006480304879
Pedros AR, MacLeod MR, Ross HA, McRae D, Tiburcio AF, Davies HV, Taylor MA (1999) Manipulation of S-adenosylmethionine decarboxylase activity in potato tubers. An increase in activity leads to an increase in tuber number and a change in tuber size distribution. Planta 209:153–160. doi:10.1007/s004250050617
Perez-Amador MA, Carbonell J, Granell A (1995) Expression of arginine decarboxylase is induced during early fruit development and in young tissues of Pisum sativum (L.). Plant Mol Biol 28:997–1009. doi:10.1007/BF00032662
Perez-Amador MA, Leon J, Green PJ, Carbonell J (2002) Induction of the arginine decarboxylase ADC2 gene provides evidence for the involvement of polyamines in the wound response in Arabidopsis. Plant Physiol 130:1454–1463. doi:10.1104/pp.009951
Perez-Molphe-Balch E, Gidekel M, Segura-Nieto M, Herrera-Estrella L, Ochoa-Alejo N (1996) Effects of water stress on plant growth and root proteins in three cultivars of rice (Oryza sativa) with different levels of drought tolerance. Physiol Plant 96:284–290
Primikirios NI, Roubelakis-Angelakis KA (2001) Indications for post-translational regulation of Vitis vinifera L. arginine decarboxylase. Plant Mol Biol 45:669–678. doi:10.1023/A:1010665520326
Reggiani R (1994) Purification and synthesis under anaerobic conditions of rice arginine decarboxylase. Plant Cell Physiol 35:1245–1249
Richards FJ, Coleman RG (1952) Occurrence of putrescine in potassium-deficient barley. Nature 170:460. doi:10.1038/170460a0
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schipper RG, Penning LC, Verhofstad AA (2000) Involvement of polyamines in apoptosis. Facts and controversies: effectors or protectors? Semin Cancer Biol 10:55–68. doi:10.1006/scbi.2000.0308
Soyka S, Heyer AG (1999) Arabidopsis knockout mutation of ADC gene reveals inducibility by osmotic stress. FEBS Lett 458:219–223. doi:10.1016/S0014-5793(99)01125-4
Takahashi Y, Berberich T, Miyazaki A, Seo S, Ohashi Y, Kusano T (2003) Spermine signaling in tobacco: activation of mitogen-activated protein kinases by spermine is mediated through mitochondrial dysfunction. Plant J 36:820–829. doi:10.1046/j.1365-313X.2003.01923.x
Thu-Hang P, Bassie L, Safwat G, Trung-Nghia P, Christou P, Capell T (2002) Expression of a heterologous S-adenosylmethionine decarboxylase cDNA in plants demonstrates that changes in SAMDC activity determine level of the higher polyamines spermidine and spermine. Plant Physiol 129:1744–1754. doi:10.1104/pp.010966
Tiburcio AF, Besford RT, Capell T, Borrell A, Testillano PS, Risueño MC (1994) Mechanism of polyamine action during senescence responses induced by osmotic stress. J Exp Bot 45:1789–1800. doi:10.1093/jxb/45.12.1789
Trung-Nghia P, Bassie L, Safwat G, Thu-Hang P, Lepri O, Rocha P, Christou P, Capell T (2003) Reduction in the endogenous arginine decarboxylase transcript levels in rice leads to depletion of the putrescine and spermidine pools with no concomitant changes in the expression of downstream genes in the polyamine biosynthetic pathway. Planta 218:125–134. doi:10.1007/s00425-003-1079-3
Umezawa T, Fujita M, Fujita Y, Yamaguchi-Shinozaki K, Shinozaki K (2006) Engineering drought tolerance in plants: discovering and tailoring genes to unlock the future. Curr Opin Biotechnol 17:113–122
Urano K, Yoshiba Y, Nanjo T, Igarashi Y, Seki M, Sekiguchi F, Yamaguchi-Shinozaki K, Shinozaki K (2003) Characterization of Arabidopsis genes involved in biosynthesis of polyamines in abiotic stress responses and developmental stages. Plant Cell Environ 26:1917–1926. doi:10.1046/j.1365-3040.2003.01108.x
Vinocur B, Altman A (2005) Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Curr Opin Biotechnol 16:1–10. doi:10.1016/j.copbio.2005.02.001
Watson MB, Malmberg RL (1996) Regulation of Arabidopsis thaliana (L.) Heynh arginine decarboxylase by potassium deficiency stress. Plant Physiol 111:1077–1083. doi:10.1104/pp.111.4.1077
Yamaguchi K, Takahashi Y, Berberich T, Imai A, Takahashi T, Michael AJ, Kusano T (2007) A protective role for the polyamine spermine against drought stress in Arabidopsis. Biochem Biophys Res Commun 352:486–490. doi:10.1016/j.bbrc.2006.11.041
Acknowledgments
We would like to thank C. Zhu for helpful discussions. This work was supported by grant AGL2004-00444 from the Ministerio de Ciencia y Tecnologia (MEC, Spain) to T. Capell. A. Peremarti is funded by a MEC FPI fellowship (BES-2005-8900); L. Bassie is funded by a Juan de la Cierva fellowship; T. Capell is a tenure-track scientist of the Ramón y Cajal Programme; P. Christou is an ICREA researcher, all at the UdL. Sequence data from this article can be found in the GenBank/EMBL data libraries under accession numbers Y07768; X56802; AY604047.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peremarti, A., Bassie, L., Christou, P. et al. Spermine facilitates recovery from drought but does not confer drought tolerance in transgenic rice plants expressing Datura stramonium S-adenosylmethionine decarboxylase. Plant Mol Biol 70, 253–264 (2009). https://doi.org/10.1007/s11103-009-9470-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-009-9470-5