Abstract
Introduction
Patients undergoing transsphenoidal pituitary surgery (TSS) are at risk for several serious complications, including the syndrome of inappropriate antidiuretic hormone and subsequent hyponatremia.
Objective
In this study, we examined the effect of 1 week of post-discharge fluid restriction to 1.0 L daily on rates of post-operative readmission for hyponatremia.
Methods
We retrospectively analyzed all patients undergoing TSS from 2008 to 2014 and prospectively recorded patient data from 2015 to 2017. Patients were divided into a control cohort (2008–2014), who were discharged with instructions to drink to thirst; and an intervention cohort (2015–2017) who were instructed to drink less than 1.0 L daily for 1 week post-operatively.
Results
This study included 788 patients; 585 (74.2%) in the control cohort and 203 (25.8%) in the intervention cohort. Overall, 436 (55.3%) were women, the median age was 47 (range 15–89), and average BMI was 29.4 kg/m2 (range 17.7–101.7). Patients were relatively well matched. Of patients in the intervention group, none was readmitted for hyponatremia (0/203), compared to 3.41% (20/585) in the control group (p = 0.003). Patients in the intervention group also had significantly higher post-operative week one sodium levels (140.1 vs 137.5 mEq/L; p = 0.002). No fluid balance complications occurred in patients who followed this protocol.
Conclusion
Hyponatremia can be a life-threatening complication of TSS, and prevention of readmission for hyponatremia can help improve patient safety and decrease costs. Mandatory post-discharge fluid restriction is a simple and inexpensive intervention associated with decreased rates of readmission for hyponatremia and normal post-operative sodium levels.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Patients undergoing transsphenoidal pituitary surgery (TSS) are at risk for several serious complications, some of which can be life-threatening and all of which delay patient recovery and increase healthcare costs [1,2,3,4,5]. Among these, one of the most common is the syndrome of inappropriate antidiuretic hormone secretion (SIADH), a disorder caused by overproduction of ADH. Although SIADH can be self-limiting, serious cases can result in severe, life-threatening hyponatremia [6, 7]. The incidence of this problem after transsphenoidal surgery ranges from 3.6 to 19.8% [7,8,9,10,11,12,13]. Clinically significant hyponatremia after surgical intervention typically occurs between post-operative day 4–7. This is often after discharge for those undergoing transsphenoidal surgery, potentially placing patients at risk of serious electrolyte dysfunction without the availability of immediate medical attention [11, 14,15,16,17].
The cause of post-TSS hyponatremia is not fully understood, but it has been hypothesized that iatrogenic manipulation of the pituitary gland results in trauma that mechanically releases stores of ADH [12, 14, 16, 18, 19]. An alternative hypothesis suggests cerebral salt wasting syndrome as the cause of hyponatremia following pituitary surgery [14, 20, 21]. There are currently no reliable clinical, pathological or imaging predictors of hyponatremia, despite many studies examining these possibilities [18, 22, 23].
To reduce the rate of this potentially dangerous complication, some have advocated for prophylactic strategies across the entire population of patients undergoing surgery [24,25,26]. One such strategy is mandatory fluid restriction during the post-operative period, which may have the effect of stabilizing post-operative serum sodium concentrations and limiting readmissions for hyponatremia. In this study, we examined the effect of 1 week of routine post-discharge fluid restriction to 1.0 L daily on rates of post-operative hyponatremia and readmission for SIADH by comparing a 7-year historical control group from our center to a 2-year intervention cohort.
Methods
Study design
This study was designed as a historically controlled cohort study comparing patients undergoing transsphenoidal surgery with the senior author (ERL) at Brigham and Women’s Hospital from April 2008 through December 2014 with patients undergoing surgery at the same site with the same surgeon from January 2015 through June 2017.
Cohort definitions
Across both cohorts, all patients were operated upon by the same surgeon at the same center. Additionally, all patients were followed post-operatively with daily measurements of serum sodium concentration as inpatients. After discharge, patients were reassessed at 1 week post-operatively in clinic by a nurse practitioner. At this visit, all patients were clinically evaluated for signs of SIADH and diabetes insipidus (DI), and had serum testing for sodium and cortisol levels.
The two cohorts, the historical control cohort and the intervention cohort, were defined by the dates above. From April 2008 to December 2014, all patients undergoing transsphenoidal surgery with the senior author were discharged with instructions to drink to thirst and avoid forcing fluids. These patients were included in the historical control group, and data regarding their pre-operative and post-operative course were collected retrospectively.
From January 2015 through June 2017, all patients undergoing transsphenoidal surgery with the senior author were discharged with a 1.0 L water container (standard hospital water pitcher) and were instructed to limit fluid intake to one container of fluids per day. These patients were included in the intervention group, and data regarding their pre-operative and post-operative course were collected prospectively for this study.
Outcome measures
The primary outcome of interest was the rate of readmission for SIADH in both cohorts. Readmission in both patient cohorts was determined by patient clinical status using a standard protocol. At the week one post-operative visit, all patients underwent testing for serum sodium concentration, and readmission was considered if sodium levels were found to be below 130 mEq/L or if the patient demonstrated clinical signs and symptoms characteristic of hyponatremia. Patients were also readmitted if they presented to the emergency department with signs or symptoms of hyponatremia. Because of the subjective nature of readmission, the secondary outcomes of interest were post-operative serum sodium concentrations for all patients at one, two, three, four, five, six, and seven days post-operatively.
Statistical analysis
Statistical comparisons were performed between the control cohort and the intervention cohort to evaluate differences in baseline characteristics, as well as rates of readmission and post-operative sodium levels. Independent t-tests and Chi square tests were conducted using IBM SPSS Version 23 (IBM Corp., Armonk, NY, USA 2015). For all tests, p < 0.05 was considered statistically significant.
Results
Patient demographics and cohort matching
A total of 788 patients were included in this study; 585 (74.2%) were included in the control cohort and 203 (25.8%) were included in the intervention cohort (Table 1). Across all patients, 436 (55.3%) were women, the median age was 47 (range 15–89), and average BMI was 29.4 kg/m2 (range 17.7–101.7). Patients were well matched by pre-operative demographics and tumor characteristics, except there were more women (62.1 vs. 53%; p = 0.027) in the intervention cohort compared to the control cohort. The intervention cohort was also less likely to have a medical history of hypertension (20.2 vs. 29.1%; p = 0.017). Regarding tumor characteristics, the groups were well matched in tumor size and type except for LH staining tumors, which were less common in the intervention cohort (8.4 vs. 22%; p < 0.001) (Table 2). Patients were well matched in clinical diagnoses as well, with similar distributions of tumor types between the control and intervention cohorts. Both groups were equally likely to experience post-operative complications not related to hyponatremia or SIADH, however more patients from the control cohort were readmitted for any reason when including these complications (11.3 vs. 5.4%; p = 0.013).
Outcomes
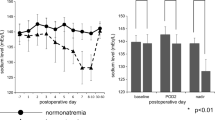
Of patients in the intervention group, none was readmitted for hyponatremia (0/203), compared to 3.41% (20/585) readmitted in the control group (p = 0.003) (Table 3). Patients in the intervention group also had significantly higher post-operative day one serum sodium levels (138.3 ± 3.5 vs. 136.7 ± 4.0 mEq/L; p < 0.001), post-operative day two (140.3 ± 3.1 vs. 138.9 ± 3.2 mEq/L; p < 0.001), post-operative day three (140.2 ± 3.1 vs. 139.2 ± 2.9 mEq/L; p = 0.003), post-operative day six (140.4 ± 4.6 vs. 138 ± 4.0 mEq/L; p < 0.001), and post-operative day seven (137.5 ± 5.3 vs. 140.1 ± 3.8 mEq/L; p < 0.001) than patients in the control group (Fig. 1). No fluid balance complications occurred in patients who followed this protocol.
Discussion
TSS is the most effective treatment for many lesions of the pituitary gland and surrounding sellar space, including pituitary adenomas, craniopharyngiomas, and Rathke cleft cysts [27,28,29,30,31]. With relatively low complication rates, transsphenoidal surgery often offers favorable outcomes, particularly among patients with debilitating symptoms caused by tumor expansion, such as hypopituitarism, visual field deficits, headaches, or overproduction of pituitary hormones [31, 32]. Despite its relative safety, one of the most common complications of TSS is SIADH with subsequent hyponatremia, which in rare cases can be life-threatening [6, 7]. In a recent study, hyponatremia was reported to be the most common reason for 30-day unplanned readmission among patients undergoing TSS [1, 22].
To more effectively treat the population of patients undergoing TSS, it is important to predict and control the occurrence of hyponatremia, which does not currently have any reliable or consistent clinical, pathologic, or imaging predictors. Previously, at least two smaller studies have analyzed the effect of post-operative water restriction on the occurrence of hyponatremia [24, 25]. The first, a study by Takeuchi et al., limited patients to 2.5 L post-operatively and included 185 patients, of which 93 received the intervention. This study reported a lower rate of hyponatremia in the water restricted group compared to the control group (5.4 vs. 16.3%, p = 0.017), but did not evaluate readmission or serum sodium data directly. In a separate study, Matsuyama et al. studied 207 patients who were subject to a regimen of prophylactic water restriction if certain clinical criteria suggestive but not diagnostic of SIADH were present [24]. This algorithm was associated with lower rates of SIADH in the intervention group compared to the control group (14 vs. 38%, p < 0.001).
After retrospective review of the control cohort and prospective treatment in the intervention cohort, there is a clear difference in rates of readmission for hyponatremia between groups. The rate of readmission decreased from 3.41% to none, with water restriction being the only significant change in the patients’ postoperative treatment paradigm between cohorts. There is some evidence in the literature regarding the effectiveness of water restriction on hyponatremia readmission, but no prior studies were found that directly examined serum sodium levels through post-operative week one [25]. Our data suggest that not only is water restriction associated with a decrease in rates of hyponatremia that is clinically severe enough for readmission, it is also associated with an overall increase in sodium levels through the first post-operative week. Although post-operative day four and five sodium levels were not significantly different between the two cohorts, this could be due in part to these days being the least frequently collected. At post-operative day six and seven, when a greater proportion of patients returned for evaluation, the difference in sodium levels remained statistically significant.
It is also important to note that the two cohorts were similar in makeup with regard to tumor type and tumor staining, with the exception of LH staining. Previous studies have suggested that post-operative hyponatremia may be more common in patients with certain sellar lesions, such as ACTH-producing adenomas causing Cushing’s disease and craniopharyngiomas [16, 33,34,35,36]. Our data suggest that post-operative water restriction may reduce the occurrence of hyponatremia across various types of sellar lesions.
Although this intervention was associated with lower rates of readmission and higher post-operative sodium values, it is necessary to situate it in the context of clinical care for patients undergoing TSS. Typically, patients who undergo uncomplicated TSS are discharged on post-operative day two or three. As such, there is limited immediate post-operative follow-up unless 1 week appointments are mandated, as in our practice. The occurrence of hyponatremia can therefore go unnoticed for several days, at which point patients may present to the emergency department or by phone with severe hyponatremia resulting in headache, nausea, and vomiting. Mandated water restriction can reduce the occurrence of this condition, thereby optimizing patient safety during the most dangerous period post-operatively, the first post-operative week.
Additionally, in 2016, the estimated cost for a readmission for hyponatremia after TSS was $6011 at an academic tertiary care center, based on estimated costs provided directly by that hospital’s financial department [37]. Costs can reach even higher levels when patients require admission to an intensive care unit with frequent invasive monitoring of their electrolyte disturbance. Because this intervention is associated with minimal cost, the savings in readmission costs may be impressive, particularly in patient populations where the baseline rate of post-operative hyponatremia is higher than in this study.
Limitations to this study include the inherent bias related to retrospective patient review. Because this was not a randomized trial, the two cohorts were not perfectly matched, with some differences in baseline characteristics such as the percentage of women and the prevalence of hypertension in each cohort. Additionally, it was not possible for this study to be blinded, to either patients or the care team. Perhaps most significantly, the rate of SIADH even in the control cohort (3.41%) was extremely low compared to the existing literature, where it ranges as high as 30% [25]. Because of this, the absolute magnitude of reduction in rates of hyponatremia with the intervention may be underestimated by this study, and should be examined in a patient population with a higher, more generalizable baseline rate of post-operative hyponatremia.
Conclusion
Hyponatremia can be a devastating and life-threatening complication of TSS, and prevention of readmission for hyponatremia can help improve patient safety and decrease costs. Mandatory post-discharge fluid restriction is a simple and inexpensive intervention associated with decreased rates of readmission for hyponatremia and increased one-week post-operative sodium levels.
References
Bohl MA, Ahmad S, White WL, Little AS (2017) Implementation of a postoperative outpatient care pathway for delayed hyponatremia following transsphenoidal surgery. Neurosurgery. doi:10.1093/neuros/nyx151
Corona G, Giuliani C, Parenti G et al (2016) The economic burden of hyponatremia: systematic review and meta-analysis. Am J Med 129(8):823–835.e4
Asemota AO, Ishii M, Brem H, Gallia GL (2017) Comparison of complications, trends, and costs in endoscopic vs microscopic pituitary surgery: analysis from a US Health Claims Database. Neurosurgery 81(3):458–472
Halvorsen H, Ramm-Pettersen J, Josefsen R et al (2014) Surgical complications after transsphenoidal microscopic and endoscopic surgery for pituitary adenoma: a consecutive series of 506 procedures. Acta Neurochir 156(3):441–449
Ciric I, Ragin A, Baumgartner C, Pierce D (1997) Complications of transsphenoidal surgery: results of a national survey, review of the literature, and personal experience. Neurosurgery 40(2):225–236 (discussion 236–227)
Ausiello JC, Bruce JN, Freda PU (2008) Postoperative assessment of the patient after transsphenoidal pituitary surgery. Pituitary 11(4):391–401
Sata A, Hizuka N, Kawamata T, Hori T, Takano K (2006) Hyponatremia after transsphenoidal surgery for hypothalamo-pituitary tumors. Neuroendocrinology 83(2):117–122
Hussain NS, Piper M, Ludlam WG, Ludlam WH, Fuller CJ, Mayberg MR (2013) Delayed postoperative hyponatremia after transsphenoidal surgery: prevalence and associated factors. J Neurosurg 119(6):1453–1460
Jahangiri A, Wagner J, Han SW et al (2014) Morbidity of repeat transsphenoidal surgery assessed in more than 1000 operations. J Neurosurg 121(1):67–74
Barber SM, Liebelt BD, Baskin DS (2014) Incidence, etiology and outcomes of hyponatremia after transsphenoidal surgery: experience with 344 consecutive patients at a single tertiary center. J Clin Med 3(4):1199–1219
Kelly DF, Laws ER Jr, Fossett D (1995) Delayed hyponatremia after transsphenoidal surgery for pituitary adenoma: report of nine cases. J Neurosurg 83(2):363–367
Lee JI, Cho WH, Choi BK, Cha SH, Song GS, Choi CH (2008) Delayed hyponatremia following transsphenoidal surgery for pituitary adenoma. Neurol Med Chir 48(11):489–492 (discussion 492–484).
Cusick JF, Hagen TC, Findling JW (1984) Inappropriate secretion of antidiuretic hormone after transsphenoidal surgery for pituitary tumors. N Engl J Med 311(1):36–38
Cote DJ, Alzarea A, Acosta MA et al (2016) Predictors and rates of delayed symptomatic hyponatremia after transsphenoidal surgery: a systemastic review. World Neurosurg 88:1–6
Zada G, Liu CY, Fishback D, Singer PA, Weiss MH (2007) Recognition and management of delayed hyponatremia following transsphenoidal pituitary surgery. J Neurosurg 106(1):66–71
Hensen J, Henig A, Fahlbusch R, Meyer M, Boehnert M, Buchfelder M (1999) Prevalence, predictors and patterns of postoperative polyuria and hyponatraemia in the immediate course after transsphenoidal surgery for pituitary adenomas. Clin Endocrinol 50(4):431–439
Belzer JS, Williams CN, Riva-Cambrin J, Presson AP, Bratton SL (2014) Timing, duration, and severity of hyponatremia following pediatric brain tumor surgery*. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and. Crit Care Soc 15(5):456–463
Blair ET, Clemmer JS, Harkey HL, Hester RL, Pruett WA (2017) Physiological mechanisms of water and electrolyte disturbances following transsphenoidal pituitary surgery. World Neurosurg. doi:10.1016/j.wneu.2017.07.175
Hannon MJ, Thompson CJ (2014) Neurosurgical hyponatremia. J Clin Med 3(4):1084–1104
Andrews BT, Fitzgerald PA, Tyrell JB, Wilson CB (1986) Cerebral salt wasting after pituitary exploration and biopsy: case report. Neurosurgery 18(4):469–471
Guerrero R, Pumar A, Soto A et al (2007) Early hyponatraemia after pituitary surgery: cerebral salt-wasting syndrome. Eur J Endocrinol 156(6):611–616
Cote DJ, Dasenbrock HH, Muskens IS et al (2017) Readmission and other adverse events after transsphenoidal surgery: prevalence, timing, and predictive factors. J Am Coll Surg 224(5):971–979
Jahangiri A, Wagner J, Tran MT et al (2013) Factors predicting postoperative hyponatremia and efficacy of hyponatremia management strategies after more than 1000 pituitary operations. J Neurosurg 119(6):1478–1483
Matsuyama J, Ikeda H, Sato S, Yamamoto K, Ohashi G, Watanabe K (2014) Early water intake restriction to prevent inappropriate antidiuretic hormone secretion following transsphenoidal surgery: low BMI predicts postoperative SIADH. Eur J Endocrinol 171(6):711–716
Takeuchi K, Nagatani T, Okumura E, Wakabayashi T (2014) A novel method for managing water and electrolyte balance after transsphenoidal surgery: preliminary study of moderate water intake restriction. Nagoya J Med Sci 76(1–2):73–82
Choong K, Arora S, Cheng J et al (2011) Hypotonic versus isotonic maintenance fluids after surgery for children: a randomized controlled trial. Pediatrics 128(5):857–866
Jane JA Jr, Kiehna E, Payne SC, Early SV, Laws ER (2010) Jr. Early outcomes of endoscopic transsphenoidal surgery for adult craniopharyngiomas. Neurosurg Focus 28(4):E9
Barkhoudarian G, Zada G, Laws ER (2014) Endoscopic endonasal surgery for nonadenomatous sellar/parasellar lesions. World Neurosurg 82(6 Suppl):S138–S146
Jagannathan J, Kanter AS, Sheehan JP, Jane JA Jr, Laws ER Jr (2007) Benign brain tumors: sellar/parasellar tumors. Neurol Clin 25(4):1231–1249
Cote DJ, Wiemann R, Smith TR, Dunn IF, Al-Mefty O, Laws ER (2015) The expanding spectrum of disease treated by the transnasal, transsphenoidal microscopic and endoscopic anterior skull base approach: a single-center experience 2008–2015. World Neurosurg 84(4):899–905
Cote DJ, Besasie BD, Hulou MM, Yan SC, Smith TR, Laws ER (2016) Transsphenoidal surgery for Rathke’s cleft cyst can reduce headache severity and frequency. Pituitary 19(1):57–64
Smith TR, Hulou MM, Huang KT et al (2015) Complications after transsphenoidal surgery for patients with Cushing’s disease and silent corticotroph adenomas. Neurosurg Focus 38(2):E12
Breshears JD, Jiang B, Rowland NC, Kunwar S, Blevins LS (2013) Use of conivaptan for management of hyponatremia following surgery for Cushing’s disease. Clin Neurol Neurosurg 115(11):2358–2361
Adams JR, Blevins LS Jr, Allen GS, Verity DK, Devin JK (2006) Disorders of water metabolism following transsphenoidal pituitary surgery: a single institution’s experience. Pituitary 9(2):93–99
Pratheesh R, Swallow DM, Rajaratnam S et al (2013) Incidence, predictors and early post-operative course of diabetes insipidus in paediatric craniopharygioma: a comparison with adults. Child’s Nerv Syst 29(6):941–949
Mukherjee KK, Dutta P, Singh A et al (2014) Choice of fluid therapy in patients of craniopharyngioma in the perioperative period: a hospital-based preliminary study. Surg Neurol Int 5:105
Hendricks BL, Shikary TA, Zimmer LA (2016) Causes for 30-day readmission following transsphenoidal surgery. Otolaryngol–Head Neck Surg 154(2):359–365
Funding
National Institutes of Health Training Grant T32 CA 009001 (DJC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
We have nothing to disclose.
Rights and permissions
About this article
Cite this article
Burke, W.T., Cote, D.J., Iuliano, S.I. et al. A practical method for prevention of readmission for symptomatic hyponatremia following transsphenoidal surgery. Pituitary 21, 25–31 (2018). https://doi.org/10.1007/s11102-017-0843-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-017-0843-5