Abstract
Secondary plant metabolites, and in particular monoterpenes, have been recognised as potential medicinal agents for centuries. As such, terpenes have been the focus of a plethora of scientific studies examining various aspects of their bioactivity. In particular, antimicrobial activity and anticancer potential have been studied extensively. Whilst the antimicrobial and anticancer activity of terpenes has been demonstrated in vitro, fewer studies have been conducted examining specific aspects of the mechanisms of antimicrobial action and anticancer efficacy in vivo. The purpose of this review is therefore to examine recent advances in the areas of antimicrobial and anticancer activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Our understanding of the bioactivity of terpenes is constantly evolving with the frequent publication of both new data and comprehensive reviews (Bakkali et al. 2008; Burt 2004; Hammer and Carson 2011). Given this, the aim of this paper is to review a selection of recent, illuminating studies in the arena of terpene bioactivity, including those presented by the two authors at the 2010 Phytochemical Society of Europe International Symposium entitled ‘‘Terpenes—Application, Activity and Analysis’’, and to discuss these in the context of our existing knowledge.
Anticancer efficacy and mechanisms of action
There have been abundant reports over the recent years highlighting the anticancer efficacy of essential oils and their terpene components, demonstrating in vitro anticancer activity against a variety of mammalian cancer cell lines and animal models of cancer. This short review will focus on more recent findings regarding the anticancer activity of terpenes that have progressed to animal research in addition to human clinical trials.
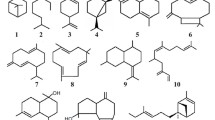
At the Terpene symposium, Dr Greay summarised published research demonstrating the in vitro and in vivo anticancer activity of tea tree oil (Greay et al. 2010a, b). The presentation highlighted that both tea tree oil and its major monoterpene component terpinen-4-ol (Fig. 1) inhibited murine tumour cell proliferation at concentrations non-cytotoxic to non-tumour cell lines. The anticancer modes of action of both the whole oil and its major component involve low level apoptosis, primary necrosis through cellular and mitochondrial membrane disruption and G1 cell cycle arrest of murine tumour cells (Greay et al. 2010b). Our more recent findings that topically applied tea tree oil combined with DMSO inhibited growth and temporarily regressed subcutaneous tumours in immunocompetent mice were also presented (Greay et al. 2010a). The antitumour effect observed was accompanied by skin irritation which completely resolved post treatment, in addition to a marked neutrophil infiltration into the tumour site (Greay et al. 2010b). Further, it was reported that topically applied tea tree oil induces significant dendritic cell activation in mice, but the relationship between this immune activation and antitumour mechanism(s) of action remain to be elucidated (Greay, unpublished data). Calcabrini and colleagues (2004) were one of the first groups to report that tea tree oil and terpinen-4-ol can induce cancer cell cytotoxicity via caspase dependent apoptosis mediated by membrane lipid reorganisation of human melanoma cells. Moreover, both TTO and terpinen-4-ol were more cytotoxic to adriamycin resistant melanoma cells, suggested that neither are substrates for P-glycoprotein (Pgp) (Calcabrini et al. 2004).
The diterpene ester ingenol-3-angelate (Fig. 1) isolated from the sap of Euphorbia peplus (also known as milk or radium weed) was first reported by Ogbourne and colleagues (2004) to have anticancer activity against both murine cancer cells in addition to murine tumours (Ogbourne et al. 2004). The basis of this research followed reports that topical application of milkweed sap was reported to be effective against skin cancers. Ingenol-3-angelate was cytotoxic to a range of murine and human tumour cell lines, and when applied to both subcutaneous tumours in immunocompetent mice and human xenograft tumors in immunodeficient mice, induced tumour regression by mitochondrial and plasma membrane disruption ultimately resulting in primary necrotic tumour cell death (Ogbourne et al. 2004). Further research has shown that ingenol-3-angelate induces activation of PKC and a neutrophil mediated antibody dependent cellular cytotoxicity-dependent prevention of tumour relapse (Challacombe et al. 2006). Most recently it was reported that topically applied ingenol-3-angelate crosses the epidermis by absorptive transport as a Pgp substrate where it can enter the tumour site and injure tumour vasculature. This damage in addition to CsA dependent subcutaneous haemorrhage at the tumour periphery appears to be necessary for anticancer efficacy (Li et al. 2010). Its topical formulation PEP005 has shown significant efficacy against AKs in phase II clinical trials (Anderson et al. 2009; Siller et al. 2009) and is currently in phase III clinical trials.
Perillyl alcohol (Fig. 1), a monoterpene abundant in mint, caraway, sage, lemongrass and cherries amongst others, has been reported efficacious against a number of cancer cell lines, inducing apoptosis in glioma cells (Fernandes et al. 2005), leukaemia cells (Clark 2006) and breast cancer cells (Yeruva et al. 2010), and in addition to apoptosis, G1 and or G2/M cell cycle arrest in tongue squamous cell carcinoma and lung adenocarcinoma cells (Elegbede et al. 2003) and in non-small cell lung carcinoma cells (Yeruva et al. 2007). Apoptosis induced by perillyl alcohol has been reported to involve caspase 3 activity, increased expression of Bcl-2 (Yeruva et al. 2007) and has also been demonstrated as c-myc dependent (Clark 2006). G1 cell cycle arrest of cancer cells by perillyl alcohol appears to be mediated by the cyclin kinase inhibitors p21 and p27 (Wiseman et al. 2007; Yeruva et al. 2007).
Studies examining topical efficacy of perillyl alcohol in mice showed a protective effective via Ras inhibition, apoptosis and inhibition of oxidative stress against TPA induced inflammation and tumour incidence (Chaudhary et al. 2009). However, phase IIa studies examining efficacy of topical perillyl alcohol cream against patients with AKs showed only a modest effect in reversing sun damage by decreasing the proportion of nuclei deviating from normal and that the lack of significant activity may be due to poor skin penetration (Stratton et al. 2010).
The observation that perillyl alcohol may suppress Ras signalling pathways, rationalised the examination of efficacy against glioma in human patients. A phase I/II study revealed inhalation of perillyl alcohol stabilised disease in 13 out of 29 patients with recurrent malignant gliomas, with case reports on tumour regression in 3 patients (da Fonseca et al. 2008). In addition, it was more recently reported that 89 patients with recurrent malignant glioma treated with perillyl alcohol inhalation survived significantly longer compared with a historical untreated control group, with no reported adverse side effects (da Fonseca et al. 2010).
These terpene agents have demonstrated enormous potential for the treatment of various cancers. The importance of highlighting efficacy not only in cell culture systems but also in animal models which can subsequently lead to promising clinical studies, show that these broadly ignored terpene agents may in fact be a fantastic source of future chemotherapy and/or immunotherapy.
Antimicrobial effects and mechanisms of action
The antimicrobial activity of terpenes is well established. In particular, many studies have been published describing the biocidal activity of terpenes and terpene-rich plant extracts, such as essential oils (Bakkali et al. 2008; Burt 2004). Previous research has also demonstrated that monoterpenes act largely against microorganisms by compromising the integrity and function of the cell membrane. Some of the specific membrane-associated effects that have been demonstrated to date include changes in membrane potential (Fisher and Phillips 2009; Xu et al. 2008), the leakage of intracellular components (Inoue et al. 2004; Lambert et al. 2001; Oussalah et al. 2006) and inhibition of respiration (Cox et al. 2000). In contrast, what is not well established is the potential for terpene resistance to develop, subtle aspects of the mechanism of action and effects elicited by sublethal terpene concentrations, such as adaptation. Advancements in this area come largely from the use of newer technologies and techniques, which illustrate antimicrobial actions that can not be demonstrated using traditional microbiological methods.
At the Terpene symposium Dr Hammer described some recent findings on the effects of the essential oil from Melaleuca alternifolia (tea tree oil), on the development of antibiotic resistance in Escherichia coli. This research built on earlier work which found that the frequencies at which bacteria showed spontaneous resistance to tea tree oil was extremely low and below the detection level (Hammer et al. 2008). The major finding presented was that significantly fewer single-step kanamycin resistant mutants were found when tea tree oil was present compared to when tea tree oil was absent (Hammer, unpublished data). However, no significant differences were evident for ciprofloxacin, ampicillin and rifampicin. Together, these data indicate that the presence of tea tree oil does not increase numbers of antibiotic resistant mutants and may in fact decrease numbers. Similar data have not been published in the scientific literature for plant essential oils or terpene components.
Other recent findings have utilised newer technologies such as flow cytometry and molecular techniques to investigate terpene bioactivity. This has provided researchers with data and information that could not be generated using more traditional laboratory techniques. For example, using flow cytometry to examine Listeria monocytogenes treated with oregano, thyme or cinnamon oils showed three bacterial populations; viable, dead and injured (Paparella et al. 2008). This ‘injured’ subpopulation would be difficult to visualise by conventional viable counting techniques. Furthermore, differences in the effects of the three oils on L. monocytogenes suggest that oregano and thyme act similarly by causing extensive membrane damage whereas cinnamon oil causes death without membrane damage (Paparella et al. 2008). Flow cytometry has also been used to demonstrate that clove (Syzygium aromaticum) essential oil increases the permeability of Propionibactium acnes (Fu et al. 2009). The terpenes thymol and carvacrol (Fig. 1) both reduce membrane potential and permeability in Escherichia coli (Xu et al. 2008), also shown by flow cytometry.
The use of molecular techniques has enabled scientists to evaluate changes in gene expression in microorganisms after exposure to terpenes. The exposure of S. aureus to subinhibitory concentrations of eugenol (Fig. 1) reduces the production of several virulence factors including haemolysin, toxic shock syndrome toxin 1 and staphylococcal enterotoxins A and B (Qiu et al. 2010). Whilst these results were determined by conventional phenotypic assays, they were also confirmed by real-time PCR which showed that transcription levels of the associated genes were reduced. Two further studies have also found reductions in virulence factors after terpene exposure using conventional phenotypic assays. The essential oil of Origanum vulgare, containing predominantly carvacrol, reduced lipase activity and coagulase production in S. aureus (Carneiro de Barros et al. 2009). There is also evidence that subinhibitory levels of the monoterpenes pulegone, α-terpineol, citral, citronellol and geraniol (Fig. 1) inhibit the virulence-associated factors swarming motility and haemolysin production in Proteus mirabilis (Echeverrigaray et al. 2008). Molecular techniques have also been used to examine protein expression after terpene exposure. Analysis of protein expression using two-dimensional polyacrylamide gel electrophoresis (2-D PAGE) in Salmonella enterica serovar Thompson grown with 0.1% thymol showed significant changes when compared to a control culture (Di Pasqua et al. 2010). Proteins with significantly altered expression were cytoplasmic (n = 37) and outer membrane proteins (n = 7), with an additional uncharacterised protein (Di Pasqua et al. 2010). Analysis of the altered proteins indicated that metabolism was markedly altered and that thymol exposure had elicited a stress-like response in the Salmonella test strain. Analysis of protein expression using the related technique of sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS–PAGE) revealed changes in the expression of proteins by P. acnes treated with clove oil (Fu et al. 2009). In particular, two proteins of 18 and 33 kDa, respectively, were absent in cells treated with 10 and 20 mg/ml of oil. It was postulated that these downregulated proteins may correspond to a lipase (GehA) and heat shock protein (PPA737) of the same size and that their absence may render cells less virulent (Fu et al. 2009). SDS–PAGE has also been used to demonstrate the loss of intracellular constituents from Salmonella typhi after eugenol treatment (Devi et al. 2010).
The use of defined genetic mutants has also provided significant insight into the bioactivity of terpenes. Efflux pumps are a well-known mechanism employed by microorganisms to pump out harmful substances. By examining the susceptibility of Pseudomonas aeruginosa mutants lacking defined efflux pump components it was demonstrated that efflux-deficient mutants were more susceptible than their efflux-competent counterparts to Melaleuca alternifolia (tea tree) essential oil and several monoterpene components (Papadopoulos et al. 2008). These data show unequivocally that efflux is used by bacteria to counteract the deleterious effects of monoterpenes. Defined bacterial mutants were also used in the study mentioned previously which examined the frequencies of resistance to tea tree oil (Hammer et al. 2008). This study examined S. aureus mutants with inactivated mutS and mutL genes, meaning that they were unable to repair their DNA and were therefore hypermutable (Hammer et al. 2008). These hypermutable isolates had increased frequencies of resistance to the antibiotic rifampicin compared to the non-hypermutable parent strain but did not differ in resistance frequencies to tea tree oil (Hammer et al. 2008).
In conclusion, recent studies have demonstrated insights into the antimicrobial activity of terpenes that significantly enhance our understanding of these natural compounds. In particular, the use of newer technologies has enabled researchers to visualise cells populations not readily evident by traditional techniques and to assess changes induced by terpenes at the level of gene transcription and expression.
This review has highlighted some of the latest developments in the field of terpene bioactivity. Although these studies have significantly advanced our understanding of the actions of terpenes, further research is required to continue the elucidation of mechanisms of action and activity.
References
Anderson L, Schmieder GJ, Werschler WP et al (2009) Randomized, double-blind, double-dummy, vehicle-controlled study of ingenol mebutate gel 0.025% and 0.05% for actinic keratosis. J Am Acad Dermatol 60:934–943
Bakkali F, Averbeck S, Averbeck D et al (2008) Biological effects of essential oils: a review. Food Chem Toxicol 46:446–475
Burt S (2004) Essential oils: their antibacterial properties and potential applications in foods: a review. Int J Food Microbiol 94:223–253
Calcabrini A, Stringaro A, Toccacieli L et al (2004) Terpinen-4-ol, the main component of Melaleuca alternifolia (tea tree) oil inhibits the in vitro growth of human melanoma cells. J Invest Dermatol 122:349–360
Carneiro de Barros J, Lúcia da Conceição M, Gomes Neto NJ et al (2009) Interference of Origanum vulgare L. essential oil on the growth and some physiological characteristics of Staphylococcus aureus strains isolated from foods. LWT Food Sci Technol 42:1139–1143
Challacombe JM, Suhrbier A, Parsons PG et al (2006) Neutrophils are a key component of the antitumor efficacy of topical chemotherapy with ingenol-3-angelate. J Immunol 177:8123–8132
Chaudhary SC, Alam MS, Siddiqui MS et al (2009) Perillyl alcohol attenuates Ras-ERK signaling to inhibit murine skin inflammation and tumorigenesis. Chem Biol Interact 179:145–153
Clark SS (2006) Perillyl alcohol induces c-Myc-dependent apoptosis in Bcr/Abl-transformed leukemia cells. Oncology 70:13–18
Cox SD, Mann CM, Markham JL et al (2000) The mode of antimicrobial action of the essential oil of Melaleuca alternifolia (tea tree oil). J Appl Microbiol 88:170–175
da Fonseca CO, Schwartsmann G, Fischer J et al (2008) Preliminary results from a phase I/II study of perillyl alcohol intranasal administration in adults with recurrent malignant gliomas. Surg Neurol 70:259–267
da Fonseca CO, Simao M, Lins IR et al (2010) Efficacy of monoterpene perillyl alcohol upon survival rate of patients with recurrent glioblastoma. J Cancer Res Clin Oncol 137:287–293
Devi KP, Nisha SA, Sakthivel R et al (2010) Eugenol (an essential oil of clove) acts as an antibacterial agent against Salmonella typhi by disrupting the cellular membrane. J Ethnopharmacol 130:107–115
Di Pasqua R, Mamone G, Ferranti P et al (2010) Changes in the proteome of Salmonella enterica serovar Thompson as stress adaptation to sublethal concentrations of thymol. Proteomics 10:1040–1049
Echeverrigaray S, Michelim L, Delamare APL et al (2008) The effect of monoterpenes on swarming differentiation and haemolysin activity in Proteus mirabilis. Molecules 13:3107–3116
Elegbede JA, Flores R, Wang RC (2003) Perillyl alcohol and perillaldehyde induced cell cycle arrest and cell death in BroTo and A549 cells cultured in vitro. Life Sci 73:2831–2840
Fernandes J, Da Fonseca CO, Teixeira A et al (2005) Perillyl alcohol induces apoptosis in human glioblastoma multiforme cells. Oncol Rep 13:943–947
Fisher K, Phillips C (2009) The mechanism of action of a citrus oil blend against Enterococcus faecium and Enterococcus faecalis. J Appl Microbiol 106:1343–1349
Fu YJ, Chen LY, Zu YG et al (2009) The antibacterial activity of clove essential oil against Propionibacterium acnes and its mechanism of action. Arch Dermatol 145:86–88
Greay SJ, Ireland DJ, Kissick HT et al (2010a) Inhibition of established subcutaneous murine tumour growth with topical Melaleuca alternifolia (tea tree) oil. Cancer Chemother Pharmacol 66:1095–1102
Greay SJ, Ireland DJ, Kissick HT et al (2010b) Induction of necrosis and cell cycle arrest in murine cancer cell lines by Melaleuca alternifolia (tea tree) oil and terpinen-4-ol. Cancer Chemother Pharmacol 65:877–888
Hammer KA, Carson CF (2011) Antibacterial and antifungal activities of essential oils. In: Thormar H (ed) Lipids and essential oils as antimicrobial agents. Wiley, West Sussex
Hammer KA, Carson CF, Riley TV (2008) Frequencies of resistance to Melaleuca alternifolia (tea tree) oil and rifampicin in Staphylococcus aureus, Staphylococcus epidermidis and Enterococcus faecalis. Int J Antimicrob Agents 32:170–173
Inoue Y, Shiraishi A, Hada T et al (2004) The antibacterial effects of terpene alcohols on Staphylococcus aureus and their mode of action. FEMS Microbiol Lett 237:325–331
Lambert RJW, Skandamis PN, Coote PJ et al (2001) A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J Appl Microbiol 91:453–462
Li L, Shukla S, Lee A et al (2010) The skin cancer chemotherapeutic agent ingenol-3-angelate (PEP005) is a substrate for the epidermal multidrug transporter (ABCB1) and targets tumor vasculature. Cancer Res 70:4509–4519
Ogbourne SM, Suhrbier A, Jones B et al (2004) Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer Res 64:2833–2839
Oussalah M, Caillet S, Lacroix M (2006) Mechanism of action of Spanish oregano, Chinese cinnamon, and savory essential oils against cell membranes and walls of Escherichia coli O157:H7 and Listeria monocytogenes. J Food Prot 69:1046–1055
Papadopoulos CJ, Carson CF, Chang BJ et al (2008) Role of the MexAB-OprM efflux pump of Pseudomonas aeruginosa in tolerance to tea tree (Melaleuca alternifolia) oil and its monoterpene components terpinen-4-ol, 1, 8-cineole, and alpha-terpineol. Appl Environ Microbiol 74:1932–1935
Paparella A, Taccogna L, Aguzzi I et al (2008) Flow cytometric assessment of the antimicrobial activity of essential oils against Listeria monocytogenes. Food Control 19:1174–1182
Qiu J, Feng H, Lu J et al (2010) Eugenol reduces the expression of virulence-related exoproteins in Staphylococcus aureus. Appl Environ Microbiol 76:5846–5851
Siller G, Gebauer K, Welburn P et al (2009) PEP005 (ingenol mebutate) gel, a novel agent for the treatment of actinic keratosis: results of a randomized, double-blind, vehicle-controlled, multicentre, phase IIa study. Australas J Dermatol 50:16–22
Stratton SP, Alberts DS, Einspahr JG et al (2010) A phase 2a study of topical perillyl alcohol cream for chemoprevention of skin cancer. Cancer Prev Res (Phila) 3:160–169
Wiseman DA, Werner SR, Crowell PL (2007) Cell cycle arrest by the isoprenoids perillyl alcohol, geraniol, and farnesol is mediated by p21(Cip1) and p27(Kip1) in human pancreatic adenocarcinoma cells. J Pharmacol Exp Ther 320:1163–1170
Xu J, Zhou F, Ji BP et al (2008) The antibacterial mechanism of carvacrol and thymol against Escherichia coli. Lett Appl Microbiol 47:174–179
Yeruva L, Pierre KJ, Elegbede A et al (2007) Perillyl alcohol and perillic acid induced cell cycle arrest and apoptosis in non small cell lung cancer cells. Cancer Lett 257:216–226
Yeruva L, Hall C, Elegbede JA et al (2010) Perillyl alcohol and methyl jasmonate sensitize cancer cells to cisplatin. Anticancer Drugs 21:1–9
Acknowledgments
We thank Dr C. Locher (UWA) for providing the chemical structures. S. J. Greay and K. A. Hammer received financial support from Rural Industries Research and Development Corporation (RIRDC), Australia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Greay, S.J., Hammer, K.A. Recent developments in the bioactivity of mono- and diterpenes: anticancer and antimicrobial activity. Phytochem Rev 14, 1–6 (2015). https://doi.org/10.1007/s11101-011-9212-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-011-9212-6