Abstract
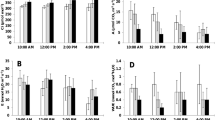
Excess solar radiation under hot climate can lead to decline in photosynthetic activity with detrimental effects on growth and yield. The aim of this study was to evaluate the use of a transparent plastic roof as shading for diurnal changes in photosynthetic gas exchange, chlorophyll fluorescence, fruit set and quality of mango (Mangifera indica L.) cv. ‘Nam Dok Mai’ growth in the field conditions. Fully expanded leaves were examined either shaded by the plastic roof or sunlit under natural conditions. Leaf temperature and leaf-to-air vapour pressure deficit of the shaded leaves measured on the clear day were lowered compared to those of the sunlit leaves. It resulted in increased stomatal conductance and photosynthetic rates of the shaded leaves compared to those of the sunlit leaves, especially from the morning to midday. Furthermore, the reversible decrease of the maximal quantum yield of PSII was more pronounced in the sunlit leaves than that in the shaded ones. Shading increased the total fruit number; the shaded fruits developed better external color than that of the sun-exposed fruits. Our results indicated that shading could maintain the high photosynthetic activity by reducing stomatal limitations for carbon supply and was effective in alleviating the photoinhibitory damage to PSII during bright and clear days with excessive radiation. Finally, shading could increase the number of fruits and improve mango peel color.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- b*:
-
yellowness
- Chl:
-
chlorophyll
- ETR:
-
electron transport rate
- Fv/Fm :
-
maximal quantum yield of PSII
- g s :
-
stomatal conductance
- L*:
-
brightness
- NPQ:
-
nonphotochemical quenching
- P N :
-
net photosynthetic rate
- qp:
-
photochemical quenching coefficient
- RH:
-
relative humidity
- Tair :
-
air temperature
- Tleaf :
-
leaf temperature
- TA:
-
titratable acidity
- TSS:
-
total soluble solid
- VPDair :
-
air vapour pressure deficit
- VPDleaf-air :
-
leaf-to-air vapour pressure deficit
- WUEi :
-
intrinsic water-use efficiency
References
Alarcón J.J., Ortuño M.F., Nicolás E. et al.: Improving water-use efficiency of young lemon trees by shading with aluminisedplastic nets.–Agr. Water Manage. 82: 387–398, 2006.
Aliakbarpour H., Rawi C.S.: Diurnal activity of four species of thrips (Thysanoptera: Thripidae) and efficiencies of three nondestructive sampling techniques for thrips in mango inflorescences.–J. Econ. Entomol. 103: 631–640, 2010.
Baker N.R., Rosenqvist E.: Application of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities.–J. Exp. Bot. 55: 1607–1621, 2004.
Barron-Gafford G.A., Grieve K.A., Murthy R.: Leaf- and standlevel responses of a forested mesocosm to independent manipulations of temperature and vapour pressure deficit.–New Phytol. 174: 614–625, 2007.
Björkman O., Demmig B.: Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 k among vascular plants of diverse origins.–Planta 170: 489–504, 1987.
Chamchaiyaporn T., Jutamanee K., Kasemsap P. et al.: Selection of the most appropriate coating particle film for improving photosynthesis in mango.–Kasetsart J. 47: 323–332, 2013.
Einhorn K.S., Rosenqvist E., Leverenz J.W.: Photoinhibition in seedlings of fraxinus and fagus under natural light conditions: implications for forest regeneration?–Oecologia 140: 241–251, 2004.
Flexas J., Bota J., Cifre J. et al.: Understanding down-regulation of photosynthesis under water stress future prospects and searching for physiological tools for irrigation management.–Ann. Appl. Biol. 144: 273–283, 2004.
Flowers M.D., Fiscus E.L., Burkey K.O. et al.: Photosynthesis, chlorophyll fluorescence, and yield of snap bean (Phaseolus vulgaris L.) genotypes differing in sensitivity to ozone.–Environ. Exp. Bot. 61: 190–198, 2007.
Genty B., Briantais J.M., Baker N.R.: The relationship between quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence.–Biochim. Biophys. Acta 990: 87–92, 1989.
Goh C.H., Ko S.M., Koh S. et al.: Photosynthesis and environments: photoinhibition and repair mechanisms in plants.–J. Plant Biol. 55: 93–101, 2012.
Goudriaan J., van Laar H.H.: Modelling Potential Crop Growth Processes. Pp. 238. Kluwer Academic Publ., Dordrecht–Boston–London 1994.
Ibrahim M.H., Jaafar H.Z.E.: Photosynthetic capacity, photochemical efficiency and chlorophyll content of three varieties of Labisia pumila Benth. exposed to open field and greenhouse growing conditions.–Acta Physiol. Plant. 33: 2179–2185, 2011.
Jifon J.L., Syvertsen J.P.: Moderate shade can increase net gas exchange and reduce photoinhibition in citrus leaves.–Tree Physiol. 23: 119–127, 2003.
Juntamanee K., Onnom S., Yingjajaval S. et al.: Leave photosynthesis and fruit quality of mango growing under field or plastic roof condition.–Acta Hortic. 975: 415–420, 2013.
Kamaluddin M., Grace J.: Photoinhibition and light acclimation in seedlings of Bischofia javanica, a tropical forest tree from Asia.–Ann. Bot.-London 69: 47–52, 1992.
Kitao M., Lei T.T., Koike T. et al.: Susceptibility to photoinhibition of three deciduous broadleaf tree species with different successional traits raised under various light regimes.–Plant Cell Environ. 23: 81–89, 2000.
Krall J.P., Edwards G.E.: Relationship between photosystem II activity and CO2 fixation in leaves.–Physiol. Plantarum 86: 180–187, 1992.
Maxwell K., Johnson G.N.: Chlorophyll fluorescence - a practical guide.–J. Exp. Bot. 51: 659–668, 2000.
Medina C.L., Souza R.P., Machado E.C. et al.: Photosynthetic response of citrus grown under reflective aluminized polypropylene shading nets.–Sci. Hortic.-Amsterdam 96: 115–125, 2002.
Montanaro G., Dichio B., Lang A. et al.: Internal versus external control of calcium nutrition in kiwifruit.–J. Plant Nutr. Soil Sci. 177: 819–830, 2014.
Montanaro G., Dichio B., Lang A. et al.: Fruit calcium accumulation coupled and uncoupled from its transpiration in kiwifruit.–J. Plant Physiol. 181: 67–74, 2015.
Montanaro G., Dichio B., Xiloyannis C.: Shade mitigates photoinhibition and enhances water use efficiency in kiwifruit under drought.–Photosynthetica 47: 363–371, 2009.
Montanaro G., Dichio B., Xiloyannis C., Celano G.: Light influences transpiration and calcium accumulation in fruit of kiwifruit plants (Actinidia deliciosa var. deliciosa).–Plant Sci. 170: 520–527, 2006.
Monteith J.L.: A reinterpretation of stomatal responses to humidity.–Plant Cell Environ. 18: 357–364, 1995
Nicolás E., Torrecillas A., Dell Amico J., Alarcón J.J.: Sap flow, gas exchange, and hydraulic conductance of young apricot trees growing under a shading net and different water supplies.–J. Plant Physiol. 162: 439–447, 2005.
Pettigrew W.T., Hesketh J.D., Peters D.B. et al.: A vapour pressure deficit effect on crop canopy photosynthesis.–Photosynth. Res. 24: 27–34, 1990.
Phattarlerphong J.: [Photosynthetic Light Response at Various Leaf Ages in Two Mango Cultivars.]–MSc. Thesis, Kasetsart University, Thailand 1997. [In Thai]
Shirke P.A., Pathre U.V.: Influence of leaf-to-air vapour pressure deficit (VPD) on the biochemistry and physiology of photosynthesis in Prosopis juliflora.–J. Exp. Bot. 55: 2111–2120, 2004.
Snelgar W.P., Hopkirk G.: Effect of overhead shading on yield and fruit-quality of kiwifruit (Actinidia deliciosa).–J. Hortic. Sci. 63: 731–742, 1988
Syvertsen J.P., Goñi C., Otero A.: Fruit load and canopy shading affect leaf characteristics and net gas exchange of ‘spring’ navel orange trees.–Tree Physiol. 23: 899–906, 2003.
Tezara W., Martínez D., Rengifo E. et al.: Photosynthetic responses of the tropical spiney shrub Lycium nodosum (Solanaceae) to drought, soil salinity and salt spray.–Ann. Bot.-London 92: 757–765, 2003.
Tucci M.L.S, Erismann N.M., Machado E.C. et al.: Diurnal and seasonal variation in photosynthesis of peach palms grown under subtropical conditions.–Photosynthetica 48: 421–429, 2010.
Vasilev S., Wiebe S., Bruce D.: Non-photochemical quenching of chlorophyll fluorescence in photosynthesis. 5-hydroxy-1, 4- naphthoquinone in spinach thylakoids as a model for antenna based quenching mechanisms.–Biochim. Biophys. Acta 1363: 147–156, 1998.
Veres S., Tóth V.R. Láposi R. et al.: Carotenoid composition and photochemical activity of four sandy grassland species.–Photosynthetica 44: 255–261, 2006.
Zhou S.B., Liu K., Zhang D. et al.: Photosynthetic performance of Lycoris radiata var. radiate to shade treatments.–Photosynthetica 48: 241–248, 2010.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: The present work was supported by Kasetsart University, Thailand. We also would like to thank Prof. Dr. Suntaree Yingjajaval from Kasetsart University for allowing the use of photosynthesis instruments and Prof. Dr. Ian Bennett at Edith Cowan University, Australia for criticism of the manuscript.
Rights and permissions
About this article
Cite this article
Jutamanee, K., Onnom, S. Improving photosynthetic performance and some fruit quality traits in mango trees by shading. Photosynthetica 54, 542–550 (2016). https://doi.org/10.1007/s11099-016-0210-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-016-0210-1