Abstract
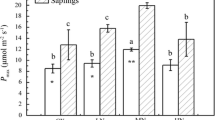
In juvenile trees growing at the rainforest understory, light is the most limiting factor for growth. It has been assumed that stomata quickly respond to light irrespective of the physical conditions prevailing before leaf illumination. Nevertheless, so far this issue has not been addressed for saplings of Amazonian tree species. The aim of this study was to determine how stomatal conductance (g s) and photosynthetic parameters of Amazonian saplings respond to diurnal variation in the physical environment and to rainfall seasonality. Light-saturated net photosynthetic rate (P Nmax) and g s at light saturation (g smax) were measured in the dry (August) and rainy (January) season of 2008 in saplings of 10 Amazonian tree species (Minquartia guianensis, Myrcia paivae, Protium apiculatum, Guatteria olivacea, Unonopsis duckei, Rinorea guianensis, Dicypellium manausense, Eschweilera bracteosa, Gustavia elliptica, and Tapura amazonica). At the forest understory, variables of the physical environment were measured. Rainfall seasonality did not affect P Nmax and g smax, nor was the effect of species on P Nmax and g smax significant (p>0.05). The g s and P Nmax increased as the forest understory became brighter and warmer; as a result, P Nmax and g smax were higher at midday than early in the morning or in the afternoon. However, contrary to expectations, neither changes in air vapor pressure deficit nor air CO2 concentration at the forest understory affected stomatal opening. More investigation is needed to elucidate the role of environmental factors in modulating stomatal movements in juvenile trees growing beneath the dense canopy of tropical rainforests.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- C i :
-
intercellular CO2 concentration
- Chl:
-
chlorophyll
- [CO2]air :
-
air CO2 concentration
- FSV:
-
fraction of sky visible
- g s :
-
stomatal conductance
- g smax :
-
stomatal conductance at light saturation
- L T :
-
fresh leaf thickness
- PARcan :
-
PAR above the canopy
- PARinst :
-
instantaneous PAR recorded during gas-exchange measurements
- PARund :
-
estimated daily PAR at the forest understory
- P N :
-
net photosynthetic rate
- P Nmax :
-
light-saturated net photosynthetic rate
- R:FR:
-
red to far-red ratio
- RH:
-
air relative humidity
- SLA:
-
specific leaf area
- SPAD:
-
values from Minolta chlorophyll meter
- T air :
-
air temperature
- VPDund :
-
understory air vapor pressure deficit
References
Aliniaeifard, S., van Meeteren, U.: Can prolonged exposure to low VPD disturb the ABA signalling in stomatal guard cells? — J. Exp. Bot. 64: 3551–3566, 2013.
Boyer, J.S., Wong, S.C., Farquhar, G.D.: CO2 and water vapor exchange across leaf cuticle (epidermis) at various water potentials. — Plant Physiol. 114: 185–191, 1997.
Buck, A.L.: New equations for computing vapor-pressure and enhancement factor. — J. Appl. Meteorol. 20: 1527–1532, 1981.
Buckley, T.N.: The control of stomata by water balance. — New Phytol. 168: 275–291, 2005.
Caird, M.A., Richards, J.H., Donovan, L.A.: Nighttime stomatal conductance and transpiration in C3 and C4 plants. — Plant Physiol. 143: 4–10, 2007.
Camargo, M. A. B., Marenco, R.A.: Growth, leaf and stomatal traits of crabwood (Carapa guianensis aubl.) in central Amazonia. — Rev. Arvore 36: 7–16, 2012.
Capers, R.S., Chazdon, R.L.: Rapid assessment of understory light availability in a wet tropical forest. — Agr. Forest Meteorol. 123: 177–185, 2004.
Casal, J.J.: Photoreceptor signaling networks in plant responses to shade. — Annu. Rev. Plant Biol. 64: 403–427, 2013.
Daley, M.J., Phillips, N.G.: Interspecific variation in nighttime transpiration and stomatal conductance in a mixed New England deciduous forest. — Tree Physiol. 26: 411–419, 2006.
Doughty, C.E., Goulden, M.L., Miller, S.D., da Rocha, H.R.: Circadian rhythms constrain leaf and canopy gas exchange in an Amazonian Forest. — Geophys. Res. Lett. 33: 1–5, 2006, doi: 10.1029/2006GL026750.
Ferreira, S.J.F., Luizão, F.J., Mello-Ivo, W. et al.: [Soil physical properties after selective logging in Central Amazonia.] — Act. Amaz. 32: 449–466, 2002. [In Portuguese]
Grantz, D.A.: Plant response to atmospheric humidity. — Plant Cell Environ. 13: 667–679, 1990.
Heintzen, C., Melzer, S., Fischer, R. et al.: A light- and temperature-entrained circadian clock controls expression of transcripts encoding nuclear proteins with homology to RNAbinding proteins in meristematic tissue. — Plant J. 5: 799–813, 1994.
Hozain, M.I., Salvucci, M.E., Fokar, M., Holaday, A.S.: The differential response of photosynthesis to high temperature for a boreal and temperate Populus species relates to differences in Rubisco activation and Rubisco activase properties. — Tree Physiol. 30: 32–44, 2010.
Huang, J.: Ultrastructure of bacterial penetration in plants. — Annu. Rev. Phytopathol. 24: 141–157, 1986.
Ilan, N., Moran, N., Schwartz, A.: The role of potassium channels in the temperature control of stomatal aperture. — Plant Physiol. 108: 1161–1170, 1995.
Johnson, D. M., McCulloh, K. A., Woodruff, D. R, Meinzer, F. C.: Hydraulic safety margins and embolism reversal in stems and leaves: Why are conifers and angiosperms so different? — Plant Sci. 195: 48–53, 2012.
Jones, H. G.: Stomatal control of photosynthesis and transpiration. — J. Exp. Bot. 49: 387–398, 1998.
Kojima, S., Shingle, D.L., Green, C.B.: Post-transcriptional control of circadian rhythms. — J. Cell Sci. 124: 311–320, 2011.
Kumar, A.S., Lakshmanan, V., Caplan, J.L. et al.: Rhizobacteria Bacillus subtilis restricts foliar pathogen entry through stomata. — Plant J. 72: 694–706, 2012.
Lawson, T.: Guard cell photosynthesis and stomatal function. — New Phytol. 181: 13–34, 2009.
Magalhães, N.S., Marenco, R.A., Camargo, M.A.B.: Do soil fertilization and forest canopy foliage affect the growth and photosynthesis of Amazonian saplings? — Sci. Agr. 71: 58–65, 2014.
Mansfield, T.A., Hetherington, A.M., Atkinson, C.J.: Some current aspects of stomatal physiology. — Annu. Rev. Plant Phys. 41: 55–75, 1990.
Manzoni, S., Vico, G., Katul, G. et al.: Hydraulic limits on maximum plant transpiration and the emergence of the safety-efficiency trade-off. — New Phytol. 198: 169–178, 2013.
Mas, P., Yanovsky, M.J.: Time for circadian rhythms: plants get synchronized. — Curr. Opin. Plant Biol. 12: 574–579, 2009.
Melotto, M., Underwood, W., He, S.Y.: Role of stomata in plant innate immunity and foliar bacterial diseases. — Annu. Rev. Phytopathol. 46: 101–122, 2008.
Mendes, K.R., Marenco, R.A.: Leaf traits and gas exchange in saplings of native tree species in the Central Amazon. — Sci. Agr. 67: 624–632, 2010.
Mendes, K.R., Marenco, R.A., Magalhães, N.S.: [Growth and photosynthetic use efficiency of nitrogen and phosphorus in saplings of Amazonian tree species.] — Rev. Arvore 37: 707–716, 2013. [in Portuguese]
Mendgen, K., Hahn, M., Deising, H.: Morphogenesis and mechanisms of penetration by plant pathogenic fungi. — Ann. Rev. Phytopathol. 34: 367–386, 1996.
Miranda, E.J., Vourlitis, G.L., Priante, N. et al.: Seasonal variation in the leaf gas exchange of tropical forest trees in the rain forest-savanna transition of the southern Amazon Basin. — J. Trop. Ecol. 21: 451–460, 2005.
Nascimento, H.C.S., Marenco, R.A.: Mesophyll conductance variations in response to diurnal environmental factors in Myrcia paivae and Minquartia guianensis in Central Amazonia. — Photosynthetica 51: 457-464, 2013.
Neilson, R.E., Jarvis, P.G.: Photosynthesis in sitka spruce (Picea sitchensis (Bong) Carr).VI: response of stomata to temperature. — J. Appl. Ecol. 12: 879–891, 1975.
Okamoto, M., Tanaka, Y., Abrams, S.R. et al.: High humidity induces abscisic acid 8′-hydroxylase in stomata and vasculature to regulate local and systemic abscisic acid responses in Arabidopsis. — Plant Physiol. 149: 825–834, 2009.
Peak, D., Mott, K.A.: A new vapour-phase mechanism for stomatal responses to humidity and temperature. — Plant Cell Environ. 34: 162–178, 2011.
Pearcy, R.W.: Sunflecks and photosynthesis in plant canopies. — Annu. Rev. Plant Phys. 41: 421–453, 1990.
Pearcy, R.W., Gross, L.J., He, D.: An improved dynamic model of photosynthesis for estimation of carbon gain in sun-fleck light regimes. — Plant Cell Environ. 20: 411–424, 1997.
Racker, E., Hinkle, P.C.: Effect of temperature on the function of a proton pump. — J. Membrane Biol. 17: 181–188, 1974.
Roth-Bejerano, N., Itai, C.: Involvement of phytochrome in stomatal movement: Effect of blue and red light. — Physiol. Plantarum 52: 201–206, 1981.
Shimazaki, K.I., Doi, M., Assmann, S.M., Kinoshita, T.: Light regulation of stomatal movement. — Annu. Rev. Plant. Biol. 58: 219–247, 2007.
Singsaas, E.L., Ort, D.R., DeLucia, E.H.: Diurnal regulation of photosynthesis in understory saplings. — New Phytol. 145: 39–49, 2000.
Song, X., Rampitsch, C., Soltani, B. et al.: Proteome analysis of wheat leaf rust fungus, Puccinia triticina, infection structures enriched for haustoria. — Proteomics 11: 944–963. 2011.
Stålfelt, M.G.: The effect of temperature on opening of the stomatal cells. — Physiol. Plantarum 15: 772–779, 1962.
Talbott, L.D., Zhu, J.X., Han, S.W., Zeiger, E.: Phytochrome and blue light-mediated stomatal opening in the orchid, Paphiopedilum. — Plant Cell Physiol. 43: 639–646, 2002.
Vialet-Chabrand, S., Dreyer, E., Brendel, O.: Performance of a new dynamic model for predicting diurnal time courses of stomatal conductance at the leaf level. — Plant Cell Environ. 36: 1529–1546, 2013.
Wang, W., Barnaby, J.Y., Tada, Y. et al.: Timing of plant immune responses by a central circadian regulator. — Nature 470: 110–114, 2011.
Warren, C.R.: Soil water deficits decrease the internal nocductance to CO2 transfer but atmospheric water deficits do not. — J. Exp. Bot. 59: 327–334, 2008.
Webb, A.A.R.: The physiology of circadian rhythms in plants. — New Phytol. 160: 281–303, 2003.
Wellburn, A.R.: The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. — J. Plant Physiol. 144: 307–313, 1994.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: To the Ministry of Science, Technology and Innovation and to the Research Foundation for the State of the Amazon (FAPEAM; grant number: UA 6203164.12) for financial support. We also thank the National Council for Scientific and Technological Development (CNPq) and the Coordination for the Improvement of Higher Education Personnel (CAPES) for scholarships.
Rights and permissions
About this article
Cite this article
Marenco, R.A., Nascimento, H.C.S. & Magalhães, N.S. Stomatal conductance in Amazonian tree saplings in response to variations in the physical environment. Photosynthetica 52, 493–500 (2014). https://doi.org/10.1007/s11099-014-0056-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-014-0056-3