Abstract
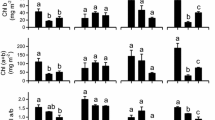
The invasion of Spartina alterniflora along the coasts of China has allowed this C4 grass to outcompete often much of the native, salt marsh vegetation, such as Phragmites australis (C3 grass), in the Yangtze Estuary. In this study, native grass, P. australis, and non-native grass, S. alterniflora, were grown in fresh and saline water (moderate salinity of 15‰ and high salinity of 30‰) to compare the effects of salinity on photosynthetic and biochemical parameters in combination with measurement temperatures. The C4 grass, S. alterniflora, showed a greater CO2 assimilation rate than P. australis, across the tested temperatures. The net photosynthetic rate declined significantly with increasing salinity as a result of inhibited stomatal conductance together with a greater decrease in the maximum rate of electron transport (J max). In P. australis, salt treatments shifted the optimum temperatures for the maximum rate of carboxylation by Rubisco (V cmax) and J max to lower temperatures. S. alterniflora showed a greater salt tolerance to moderate stress than that of the native grass, with lower sensitivity of V cmax, J max, and the maximum rate of phosphoenolpyruvate carboxylation. Both moderate and high stress decreased significantly stomatal conductance of S. alterniflora; high salinity reduced significantly photosynthetic efficiency and J max. Our findings indicated that the combination of stomatal conductance, enzyme activity, and electron transport affected the photosynthetic performance of the plants in response to salt treatments. The success of S. alterniflora could be probably attributed to its C4 photosynthetic pathway and the tolerance to moderate salinity. In this study, a modified parameterization of the photosynthetic model was suggested to support a more reasonable simulation of photosynthesis under salt stress.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- C a :
-
ambient CO2 concentration
- C c :
-
chloroplast CO2 concentration
- C i :
-
intercellular CO2 concentration
- C m :
-
CO2 concentration in the mesophyll cell
- C s :
-
CO2 concentration at the carboxylation site of Rubisco in the bundle-sheath
- g bs :
-
bundle sheath cell conductance
- g m :
-
mesophyll conductance
- g s :
-
light-saturated stomatal conductance
- H a :
-
enthalpy of activation
- H d :
-
enthalpy of deactivation
- HS:
-
high salinity
- J, J t :
-
rate of electron transport for C3 and C4 plants
- J max :
-
maximum rate of electron transport
- K c, K o :
-
Rubisco Michaelis constants for CO2 and O2
- K p :
-
Michaelis-Menten constants for PEP carboxylation
- MS:
-
moderate salinity
- O :
-
O2 concentration
- PEP:
-
phosphoenolpyruvate
- PEPC:
-
phosphoenolpyruvate carboxylase
- P N :
-
net photosynthetic rate
- P Nsat :
-
light-saturated net photosynthetic rate
- R :
-
molar gas constant
- R D :
-
dark respiration
- RH:
-
relative humidity
- R m :
-
mitochondrial respiration in the mesophyll
- RuBP:
-
ribulose-1,5-bisphosphate
- ΔS :
-
entropy of the desaturation equilibrium
- S c/o :
-
reciprocal of Rubisco specificity
- T L :
-
the leaf temperature
- V c :
-
rate of Rubisco carboxylation
- V cmax :
-
maximum rate of carboxylation by Rubisco
- V p :
-
rate of PEP carboxylation
- V pmax :
-
maximum rate of PEP carboxylation
- α:
-
quantum efficiency
- α1 :
-
photosynthetically active irradiance absorbed by PSII
- γ*:
-
half of S c/o
- θ:
-
curvature of the light response curve
- Γ*:
-
CO2 compensation point (absence of dark respiration)
- χ:
-
partitioning factor of electron transport
References
Agastian, P., Kingsley, S.J., Vivekanandan, M.: Effect of salinity on photosynthesis and biochemical characteristics in mulberry genotypes. — Photosynthetica 38: 287–290, 2000.
Bernacchi, C.J., Singsaas, E.L., Pimentel, C. et al.: Improved temperature response functions for models of Rubisco-limited photosynthesis. — Plant Cell Environ. 24: 253–259, 2001.
Bernacchi, C.J., Portis, A.R., Nakano, H. et al.: Temperature response of mesophyll conductance. Implications for the determination of Rubisco enzyme kinetics and for limitations to photosynthesis in vivo. — Plant Physiol. 130: 1992–1998, 2002.
Burdick, D.M., Buchsbaum, R., Holt, E.: Variation in soil salinity associated with expansion of Phragmites australis in salt marshes. — Environ. Exp. Bot. 46: 247–261, 2001.
Centritto, M., Loreto, F., Chartzoulakis, K.: The use of low [CO2] to estimate diffusional and non-diffusional limitations of photosynthetic capacity of salt-stressed olive saplings. — Plant Cell Environ. 26: 585–594, 2003.
Chambers, R.M., Mozdzer, T.J., Ambrose, J.C.: Effects of salinity and sulfide on the distribution of Phragmites australis and Spartina alterniflora in a tidal saltmarsh. — Aquat. Bot. 62: 161–169, 1998.
Cousins, A.B., Ghannoum, O., von Caemmerer, S., Badger, M.R.: Simultaneous determination of Rubisco carboxylase and oxygenase kinetic parameters in Triticum aestivum and Zea mays using membrane inlet mass spectrometry. — Plant Cell Environ. 33: 444–452, 2010.
Crafts-Brandner, S.J., Salvucci, M.E.: Sensitivity of photosynthesis in a C4 plant, maize, to heat stress. — Plant Physiol. 129: 1773–1780, 2002.
Dadkhah, A.: Effect of salinity on growth and leaf photosynthesis of two sugar beet (Beta vulgaris L.) cultivars. — J. Agr. Sci. Tech. 13: 1001–1012, 2011.
Deng, Ch., Zhang, G., Pan, X.: Photosynthetic responses in Reed (Phragmites australis (CAV.) TRIN. ex Steud.) seedlings induced by different salinity-alkalinity and nitrogen levels. — J. Agr. Sci. Tech. 13: 687–699, 2011.
Desingh, R., Kanagaraj, G.: Influence of salinity stress on photosynthesis and antioxidative systems in two cotton varieties. — Gen. Appl. Plant Physiol. 33: 221–234, 2007.
Farquhar, G.D., von Caemmerer, S., Berry, J.A.: A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. — Planta 149: 78–90, 1980.
Farquhar, G.D., von Caemmerer, S.: Modelling of photosynthetic responses to environmental conditions. — In: Lange, O.L., Nobel, P.S., Osmond, C.B., Ziegler, H. (ed.): Physiological Plant Ecology. II. Encyclopedia of Plant Physiology. New Series. Vol. 12B. Pp. 548–577. Springer-Verlag, Berlin 1982.
Farquhar, G.D., Wong, S.C.: An empirical model of stomatal conductance. — Aust. J. Plant. Physiol. 11: 191–209, 1984.
Farquhar, G.D., von Caemmerer, S., Berry, J.A.: Models of photosynthesis. — Plant Physiol. 125: 42–45, 2001.
Flowers, T.J., Troke, P.F., Yeo, A.R.: The mechanism of salt tolerance in halophytes. — Annu. Rev. Plant Phys. 28: 89–121, 1977.
Ge, Z.M., Zhou, X., Wang, K. et al.: [Research methodology on carbon pool dynamics in the typical wetland of Yangtze River estuary.] — Acta Ecolog. Sin. 30: 1097–1108, 2010. [In Chinese]
Ge, Z.M., Zhou, X., Kellomäki, S. et al.: Acclimation of photosynthesis in a boreal grass (Phalaris arundinacea L.) under different temperature, CO2, and soil water regimes. — Photosynthetica 50: 141–151, 2012.
He, X.J., Chen, J.Q., Zhang, Z.G. et al.: Identification of saltstress responsive genes in rice (Oryza sativa L.) by cDNA array. — Sci. China Ser B 45: 477–484, 2002.
He, Y., Yu, C.L., Zhou, L. et al.: Rubisco decrease is involved in chloroplast protrusion and Rubisco-containing body formation in soybean (Glycine max) under salt stress. — Plant Physiol. Bioch. 74: 118–124, 2014.
Hichem, H., Naceur, El A., Mounir, D.: Effects of salt stress on photosynthesis, PSII photochemistry and thermal energy dissipation in leaves of two corn (Zea mays L.) varieties. — Photosynthetica 47: 517–526, 2009.
Huang, H.M., Zhang, L.Q.: A study on the population dynamics of Spartina alterniflora at Jiuduansha Shoals, Shanghai, China. — Ecol. Eng. 29: 164–172, 2007.
Kubien, D.S., von Caemmerer, S., Furbank, R.T., Sage, R.F.: C4 photosynthesis at low temperature. A study using transgenic plants with reduced amounts of Rubisco. — Plant Physiol. 132: 1577–1585, 2003.
Leuning, R.: Temperature dependence of two parameters in a photosynthesis model. — Plant Cell Environ. 25: 1205–1210, 2002.
Li, H.P., Zhang, L.Q., Wang, D.H.: A study on the distribution of an exotic plant Spartina alterniflora in Shanghai. — Biodivers. Sci. 14: 114–120, 2006.
Li, J.Y., Zhao, C.Y., Li, J. et al.: Growth and leaf gas exchange in Populus euphratica across soil water and salinity gradients. — Photosynthetica 51: 321–329, 2013.
Long, S.P., Ainsworth, E.H., Rogers, A., Ort, D.R.: Rising atmospheric carbon dioxide: plants face the future. — Annu. Rev. Plant Biol. 55: 591–628, 2004.
Maricle, B.R., Lee, R.W., Hellquist, C.E. et al.: Effects of salinity on chlorophyll fluorescence and CO2 fixation in C4 estuarine grasses. — Photosynthetica 45: 433–440, 2007.
Massad, R.S., Tuzet, A., Bethenod, O.: The effect of temperature on C4-type leaf photosynthesis parameters. — Plant Cell Environ. 30: 1191–1204, 2007.
Meinzer, F.C., Zhu, J.: Efficiency of C4 photosynthesis in Atriplex lentiformis under salinity stress. — Aust. J. Plant Physiol. 26: 79–86, 1999.
Munns, R., Tester, M.: Mechanisms of salinity tolerance. — Annu. Rev. Plant Biol. 59: 651–681, 2008.
Naz, N., Hameed, M., Ashraf, M. et al.: Relationships between gas-exchange characteristics and stomatal structural modifications in some desert grasses under high salinity. — Photosynthetica 48: 446–456, 2010.
Rout, N.P., Shaw, B.P.: Salt tolerance in aquatic macrophytes: Ionic relation and interaction. — Biol. Plantarum 55: 91–95, 2001.
Saha, A.K., Saha, S., Sadle, J. et al.: Sea level rise and South Florida coastal forests. — Climatic Change 107: 81–108, 2011.
Sharkey, T.D., Bernacchi, C.J., Farquhar, G.D., Singsaas, E.L.: Fitting photosynthetic carbon dioxide response curves for C3 leaves. — Plant Cell Environ. 30: 1035–1040, 2007.
Sudhir, P., Murthy, S.D.S.: Effects of salt stress on basic processes of photosynthesis. — Photosynthetica 42: 481–486, 2004.
Vasquez, E.A., Glenn, E.P., Guntenspergen, G.R. et al.: Salt tolerance and osmotic adjustment of Spartina alterniflora (Poaceae) and the invasive M haplotype of Phragmites australis (Poaceae) along a salinity gradient. — Am. J. Bot. 93:1784–1790, 2006.
von Caemmerer, S., Furbank, R.T.: Modeling C4 photosynthesis. — In: Sage, R.F., Monson, R.K. (ed.): C4 Plant Biology. Pp. 173–211. Academic Press, Toronto 1999.
Wang, H.M., Wang, W.J., Wang, H.Z. et al.: Effect of inland saltalkaline stress on C4 enzymes, pigments, antioxidant enzymes, and photosynthesis in leaf, bark, and branch chlorenchyma of poplars. — Photosynthetica 51: 115–126, 2013.
Wu, Z.H., Yang, C.W., Yang, M.Y.: Photosynthesis, photosystem II efficiency, amino acid metabolism and ion distribution in rice (Oryza sativa L.) in response to alkaline stress. — Photosynthetica 52: 157–160, 2014.
Yin, X.Y., Sun, Z.P., Struik, P.C. et al.: Using a biochemical C4 photosynthesis model and combined gas exchange and chlorophyll fluorescence measurements to estimate bundle-sheath conductance of maize leaves differing in age and nitrogen content. — Plant Cell Environ. 34: 2183–2199, 2011.
Yu, J.B., Wang, X.H., Ning, K. et al.: Effects of salinity and water depth on germination of Phragmites australis in coastal wetland of the Yellow River Delta. — Clean-Soil Air Water. 40: 1154–1158, 2012.
Zhang, L.G., Xing, D.: Rapid determination of the damage to photosynthesis caused by salt and osmotic stresses using delayed fluorescence of chloroplasts. — Photochem. Photobiol. Sci. 7: 352–360, 2008.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: This work was funded through the Natural Science Foundation of China (41201091), the Shanghai Pujiang Program (13PJ1402200) and the International Project (12230707500) of Shanghai Science & Technology Committee, the Global Change Scientific Research Program of China (2010CB951204), and the Project sponsored by SRF for ROCS, SEM. We thank the experts of Edanz Editing (Edanz Group Ltd.) for revising the language of this paper.
Rights and permissions
About this article
Cite this article
Ge, Z.M., Zhang, L.Q., Yuan, L. et al. Effects of salinity on temperature-dependent photosynthetic parameters of a native C3 and a non-native C4 marsh grass in the Yangtze Estuary, China. Photosynthetica 52, 484–492 (2014). https://doi.org/10.1007/s11099-014-0055-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-014-0055-4