Abstract
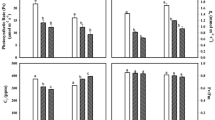
The current concentrations of O3 have been shown to cause significant negative effects on crop yield. The present levels of ozone may not induce visible symptoms in most of plants, but can result in substantial losses in reproductive output. This paper considers the impact of ambient O3 on gas exchange, photosynthetic pigments, chlorophyll (Chl) fluorescence and carbohydrate levels in the flag leaf of wheat plants during various stages of reproductive development using open-top chambers. Mean O3 concentration was 45.7 ppb during wheat growth and 50.2 ppb after flag leaf development. Reproductive stage showed higher exceedence of O3 above 40 ppb compared to the vegetative stage. Diurnal variations in net photosynthetic rate (P N) and stomatal conductance (g s), intercellular CO2 concentration (C i), Fv/Fm ratio, photosynthetic pigments, soluble sugars, and starch were measured at 10, 30, and 50 days after flag leaf expansion (DAFE). The results showed reductions in P N, g s, Fv/Fm ratio, photosynthetic pigments and starch, and increases in C i, F0, and soluble sugars in nonfiltered chambers (NFCs) compared to filtered chambers (FCs). Maximum changes in measured parameters were observed at 50 DAFE (i.e. grain filling and setting phase). Diurnal variation in P N showed double peaked curve in both FCs and NFCs, but delayed peak and early depression in NFCs. Stomatal conductance was significantly lower in NFCs. The study suggests that higher prevalence of ambient O3 during reproductive development led to significant alteration in physiological vitality of wheat having potential negative influence on yield.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- C i :
-
intercellular CO2 concentration
- Chl:
-
chlorophyll
- Fm :
-
maximal fluorescence of dark-adapted state
- Fv :
-
variable fluorescence
- F0 :
-
initial fluorescence of dark-adapted state
- FCs:
-
filtered chambers
- g s :
-
stomatal conductance
- NFCs:
-
nonfiltered chambers
- OPs:
-
open plots
- OTCs:
-
open-top chambers
- P N :
-
net photosynthetic rate
- PAR:
-
photosynthetically active radiation
- PSII:
-
photosystem II
References
Biswas, D.K., Xu, H., Li, Y.G., Sun, J.Z., Wang, X.Z., Han, X.G., Jiang, G.M.: Genotypic differences in leaf biochemical, physiological and growth responses to ozone in 20 winter wheat cultivars released over the past 60 years. — Global Change Biol. 14: 46–59, 2008.
Calatayud, A., Barreno, E.: Response to ozone in two lettuce varieties on chlorophyll a fluorescence, photosynthetic pigments and lipid peroxidation. — Plant Physiol. Biochem. 42: 549–555, 2004.
Calatayud, A., Barreno, E.: Chlorophyll a fluorescence, antioxidant enzymes and lipid peroxidation in tomato in response to ozone and benomyl. — Environ. Pollut. 115: 283–289, 2001.
Clark, A.J., Landolt, W., Bucher, J.B., Strasser, R.J.: Beech (Fagus sylvatica) response to ozone exposure assessed with a chlorophyll a fluorescence performance index. — Environ. Pollut. 109: 501–507, 2000.
Dubois, M., Gilles, K.A., Hamilton, J.K., Roberts, P.A., Smith, F.: Colorimetric method for determination of sugars and related substances. — Anal. Chem. 28: 350–356 1956.
Duxbury, A.C., Yentsch, C.S.: Plankton pigment monographs. — J. Mar. Res. 15: 19–101, 1956.
Feng, Z.Z., Kobayashi, K., Ainsworth, E.A.: Impact of elevated ozone concentration on growth, physiology, and yield of wheat (Triticum aestivum L.): a meta-analysis. — Global Change Biol. 14: 2696–2708, 2008.
Feng, Z.Z., Kobayashi, K., Wang, X.K., Feng, Z.W.: A metaanalysis of responses of wheat yield formation to elevated ozone concentration. — Chin. Sci. Bull. 54: 249–255, 2009.
Feng, Z.Z., Pang, J., Kobayashi, K., Zhu, J.G., Orts, D.R.: Differential resposnes in two varieties of winter wheat to elevated ozone concentration under fully open-air field conditions. — Global Change Biol. 17: 580–591, 2011.
Feng, Z.-Z., Yao, F.-F., Chen, Z., Wang, X.-K., Zheng, Q-W., Feng, Z.-W.: Response of gas exchange and yield components of field-grown Triticum aestivum L. to elevated ozone in China. — Photosynthetica 45: 441–446, 2007.
Fiscus, E.L., Booker, F.L., Burkey, K.O.: Crop loss responses to ozone: uptake, mode of action, carbon assimilation and partitioning. — Plant Cell Environ. 28: 997–1011, 2005.
Geiger, D.R., Servaites, J.C.: Diurnal regulation of photosynthetic carbon metabolism in C3 plants. — Annu. Rev. Plant Physiol. Plant Mol. Biol. 45: 235–256, 1994.
Guidi, L., Degl’Innocenti, E.: Ozone effects on high light induced photoinhibition in Phaseolus vulagris. — Plant Sci. 174: 590–596, 2008.
Guidi, L., Nali, C., Lorenzini, G., Filippi, F., Soldatini, G.F.: Effect of chronic O3 fumigation on the photosynthetic process of poplar clones showing different sensitivity. — Environ. Pollut. 113: 245–254, 2001.
Heath, R.L.: Alterations of plant metabolism by ozone exposure. — In: Alscher, R.G., Wellburn, A.R. (ed.): Plant Responses to the Gaseous Environment. Pp. 121–146. Chapman and Hall, London 1994.
Heck, W.W, Furiness, C.S., Cowling, E.B., Sims, C.K.: Effects of ozone on crop, forest and natural ecosystem: assessments of research needs. — In: Cowling, E.B., Furiness, C.S. (ed.): The State of Southern Oxidants Study. Air and Waste. Pp. 11–22. Managament Association, Pittsburgh 1998.
Ishii, S., Marshall, F.M., Bell, J.N.B.: Physiological and morphological responses of locally grown Malaysian rice cultivars (Oryza sativa L.) to different ozone concentrations. — Water Air Soil Pollut. 155: 205–221, 2004.
Kangasjarvi, J., Jaspers, P., Kollist, H.: Signalling and cell death in O3 exposed plants. — Plant Cell Environ. 28: 1021–1036, 2005.
Keutgen, N., Lenz, F.: Responses of strawberry to long-term elevated atmospheric ozone concentrations - I. Leaf gas exchange, chlorophyll fluorescence, and macronutrient contents. — Gartenbauwissenschaft 66: 27–33, 2001.
Köllner, B., Krause, G.H.M.: Changes in carbohydrates, leaf pigments and yield in potatoes induced by different ozone exposure regimes. — Agric. Ecosyst. Environ. 78: 149–158, 2000.
Lee, E.H., Tingey, D.T., Hogsett, W.E.: Evaluation of ozone exposure indices in exposure-response modeling. — Environ. Pollut. 53: 43–52, 1988.
Leitao, L., Delacôte, E., Dizengremel, P., Le Thiec, D., Biolley, J.-P.: Assessment of the impact of increasing concentrations of ozone on photosynthetic components of maize (Zea mays L.), a C4 plant. — Environ. Pollut. 146: 5–8, 2007.
Li, Q., Jacob, D.J., Bey, I., Palmer, P.J., Duncan, B.N., Field, B.D., Martin, R.V., Fiore, A.M., Yantosca, R.M., Parrish, D.D., Simmonds, P.G., Oltmans, S.J.: Transatlantic Transport of Pollution and its Effects on Surface Ozone in Europe and North America. — J. Geophys. Res. 107: 4166, 2002.
Lichtenthaler, H.K.: Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. — Methods Enzymol. 148: 351–382, 1987.
Maclachlan, S., Zalik, S.: Plastid structure, chlorophyll concentration and free amino acid composition of a chlorophyll mutant of barley. — Can. J. Bot. 41: 1053–1062, 1963.
Mc Cready, R.M., Goggolz, J., Silviera, V., Owens, H.S.: Determination of starch and amylose in vegetables. Application to peas. — Anal. Chem. 22: 1156–1158, 1950.
Meyer, U., Köllner, B., Willenbrink, J., Krause, G.H.M.: Physiological changes on agricultural crops induced by different ambient ozone exposure regimes. 1. Effects on photosynthesis and assimilate allocation in spring. — New Phytol. 136: 645–652, 1997.
Mills, G., Buse, A., Gimeno, B., Bermejo, V., Holland, M., Emberson, L., Pleijel, H.: A synthesis of AOT40-based response functions and critical levels of ozone for agricultural and horticultural crops. — Atmos. Environ. 41: 2630–2643, 2007.
Pleijel, H., Skärby, L., Wallin, G., Selldén, G.: A process- oriented explanation of the nonlinear relationship between grain yield of wheat and ozone exposure. — New Phytol. 131: 241–246, 1995.
Rai, R., Agrawal, M., Agrawal, S.B.: Assessment of yield losses in tropical wheat using open top chambers. — Atmos. Environ. 41: 9543–9554, 2007.
Reiling, K., Davison, A.W.: Effects of exposure to ozone at different stages in the development of Plantago major L. on chlorophyll fluorescence and gas exchange. — New Phytol. 128: 509–514, 1994.
Richards, B.L., Middleton, J.T., Hewitt, W.B.: Air pollution with reference to agronomic crops. — Agron. J. 50: 559–561, 1958.
Saitanis, C.J., Riga-Karandinos, A.N., Karandinos, M.G.: Effects of ozone on chlorophyll and quantum yield of tobacco (Nicotiana tabacum L.) varieties. — Chemosphere 42: 945–953, 2001.
Sarkar, A., Agrawal, S.B.: Identication of ozone stress in Indian rice through foliar injury and differential protein profile. — Environ. Monit. Assess. 161: 205–215, 2010.
Singh, E., Tiwari, S., Agrawal, M.: Effects of elevated ozone on photosynthesis and stomatal conductance of two soybean varieties: a case study to assess impacts of one component of predicted global climate change. — Plant Biol. 11: 101–108, 2009.
Vingarzan, R. A review of surface ozone background levels and trends. — Atmos. Environ. 38: 3431–3442, 2004.
Zadoks, J.C, Chang, T.T., Konzak, C.F.: A decimal code for the growth stages of cereals. — Weed Res. 415: 415–421, 1974.
Acknowledgements
The authors are thankful to the Head, Department of Botany for all the laboratory facilities and to the Department of Science and Technology, New Delhi for providing financial support to the work. Richa Rai is grateful to Council of Scientific and Industrial Research, New Delhi for awarding Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rai, R., Agrawal, M. & Agrawal, S.B. Effects of ambient O3 on wheat during reproductive development: Gas exchange, photosynthetic pigments, chlorophyll fluorescence, and carbohydrates. Photosynthetica 49, 285–294 (2011). https://doi.org/10.1007/s11099-011-0038-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-011-0038-7