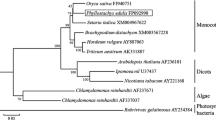
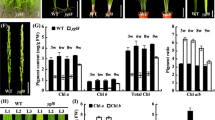
Abstract
One of the least understood enzymatic steps in chlorophyll biosynthesis is the formation of isocyclic ring, which is catalyzed by the Mg-protoporphyrin IX monomethyl ester (MgPME) cyclase that is involved in the conversion of MgPME to protochlorophyllide. Several genes encoding part of this enzyme have been identified and functional analysis of them has been performed. The enzyme plays important roles in higher plants and photosynthetic bacteria. The review focuses on the current knowledge of MgPME cyclase coding genes, with emphasis on their organization, expression pattern, and functional analysis obtained from mutants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ALA:
-
δ-aminolevulinic acid
- Bchl:
-
bacteriochlorophyll
- Chl:
-
chlorophyll
- Chlide a :
-
chlorophyllide a
- LHC1:
-
light-harvesting complex 1
- LHC2:
-
light-harvesting complex 2
- MgP:
-
Mg-protoporphyrin IX
- MgPME:
-
Mg-protoporphyrin IX monomethyl ester
- Pchlide:
-
protochlorophyllide
- PS:
-
photosystem
- SAM:
-
S-adenosyl-L-methionine
- WT:
-
wild type
References
Alawady, A.E., Grimm, B.: Tobacco Mg protoporphyrin IX methyltransferase is involved in inverse activation of Mg porphyrin and protoheme synthesis.-Plant J. 41: 282–290, 2005.
Bange, F.C., Vogel, U., Flohr, T., Kiekenbeck, M., Denecke, B., Bottger, E.C.: IFP 35 is an interferon-induced leucine zipper protein that undergoes interferon-regulated cellular redistribution.-J. biol. Chem. 269: 1091–1098, 1994.
Beale, S.I.: Enzymes of chlorophyll biosynthesis.-Photosynth. Res. 60: 43–73, 1999.
Bollivar, D.W.: Recent advances in chlorophyll biosynthesis.-Photosynth. Res. 90: 173–194, 2006.
Bollivar, D.W., Beale, S.I.: Formation of the isocyclic ring of chlorophyll by isolated Chlamydomonas reinhardtii chloroplasts.-Photosynth. Res. 43: 113–124, 1995.
Bollivar, D.W., Beale, S.I.: The chlorophyll biosynthetic enzyme Mg-protoporphyrin IX monomethyl ester (oxidative) cyclase. Characterization and partial purification from Chlamydomonas reinhardtii and Synechocystis sp. PCC 6803.-Plant Physiol. 112: 105–114, 1996.
Bollivar, D.W., Suzuki, J.Y., Beatty, J.T., Dobrowolski, J.M., Bauer, C.E.: Directed mutational analysis of bacteriochlorophyll a biosynthesis in Rhodobacter capsulatus.-J. mol. Biol. 237: 622–640, 1994.
Chereskin, B.M., Castelfranco, P.A.: Effects of iron and oxygen on chlorophyll biosynthesis. II. Observations on the biosynthetic pathway in isolated etiochloroplasts.-Plant Physiol. 69: 112–116, 1982.
Chereskin, B.M., Wong, Y.-S., Castelfranco, P.A.: In vitro synthesis of the chlorophyll isocyclic ring. Transformation of magnesium-protoporphyrin IX and magnesium-protoporphyrin IX monomethyl ester into magnesium-2,4-divinyl pheoporphyrin A5.-Plant Physiol. 70: 987–993, 1982.
De Vetten, N.C., Lu, G., Feri, R.J.: A maize protein associated with the G-box binding complex has homology to brain regulatory proteins.-Plant Cell 4: 1295–1307, 1992.
Eckhardt, U., Grimm, B., Hortensteiner, S.: Recent advances in chlorophyll biosynthesis and breakdown in higher plants.-Plant mol. Biol. 56: 1–14, 2004.
Fuesler, T.P., Wong, Y.-S., Castelfranco, P.A.: Localization of Mg-chelatase and Mg-protoporphyrin IX monomethyl ester (oxidative) cyclase activities within isolated, developing cucumber chloroplasts.-Plant Physiol. 75: 662–664, 1984.
Fujita, Y.: Protochlorophyllide reduction: a key step in the greening of plants.-Plant Cell Physiol. 37: 411–421, 1996.
Gough, S.P., Petersen, B.O., Duus, J.O.: Anaerobic chlorophyll isocyclic ring formation in Rhodobacter capsulatus requires a cobalamin cofactor.-Proc. nat. Acad. Sci. USA 97: 6908–6913, 2000.
Heyes, D.J., Hunter, N.: Making light work of enzyme catalysis: protochlorophyllide oxidoreductase.-Trends biochem. Sci. 30: 642–649, 2005.
Ilag, L.L., Kumar, A.M., Söll, D.: Light regulation of chlorophyll biosynthesis at the level of 5-aminolevulinate formation in Arabidopsis.-Plant Cell 6: 265–275, 1994.
Kageyama, R., Pastan, I.: Molecular cloning and characterization of a human DNA binding factor that represses transcription.-Cell 59: 815–825, 1989.
Kovacevic, D., Dewez, D., Popovic, R.: Irradiation-induced in vivo re-localization of NADPH-protochlorophyllide oxidoreductase from prolamellar body to stroma of barley etioplast.-Photosynthetica 45: 105–109, 2007.
Liu, N., Yang, Y.T., Liu, H.H., Yang, G.D., Zhang, N.H., Zheng, C.C.: NTZIP antisense plants show reduced chlorophyll levels.-Plant Physiol. Biochem. 42: 321–327, 2004.
Mattheis, J.R., Rebeiz, C.A.: Chloroplast biogenesis. Net synthesis of protochlorophyllide from magnesium-protoporphyrin monoester by developing chloroplasts.-J. biol. Chem. 252: 4022–4024, 1977.
McCormac, A.C., Fischer, A., Kumar, A.M., Soll, D., Terry, M.J.: Regulation of HEMA1 expression by phytochrome and a plastid signal during de-etiolation in Arabidopsis thaliana.-Plant J. 25: 549–561, 2001.
Meskauskiene, R., Nater, M., Goslings, D., Kessler, F., op den Camp, R., Apel, K.: FLU: a negative regulator of chlorophyll biosynthesis in Arabidopsis thaliana.-Proc. nat. Acad. Sci. USA 98: 12826–12831, 2001.
Moseley, J., Quinn, J., Eriksson, M., Merchant, S.: The Crd1 gene encodes a putative di-iron enzyme required for photosystem I accumulation in copper deficiency and hypoxia in Chlamydomonas reinhardtii.-EMBO J. 19: 2139–2151, 2000.
Moseley, J.L., Page, M.D., Alder, N.P., Eriksson, M., Quinn, J., Soto, F., Theg, S.M., Hippler, M., Merchant, S.: Reciprocal expression of two candidate di-iron enzymes affecting photosystem I and light-harvesting complex accumulation.-Plant Cell 14: 673–688, 2002.
Nasrulhaq-Boyce, A., Griffiths, W.T., Jones, O.T.G.: The use of continuous assays to characterize the oxidative cyclase that synthesizes the chlorophyll isocyclic ring.-Biochem. J. 243: 23–29, 1987.
Ouchane, S., Steunou, A.S., Picaud, M., Astier, C.: Aerobic and anaerobic Mg-protoporphyrin monomethyl ester cyclases in purple bacteria: a strategy adopted to bypass the repressive oxygen control system.-J. biol. Chem. 279: 6385–6394. 2004.
Papenbrock, J., Grimm, B.: Regulatory network of tetrapyrrole biosynthesis — studies of intracellular signalling involved in metabolic and developmental control of plastids.-Planta 213: 667–681, 2001.
Papenbrock, J., Mock, H.P., Tanaka, R., Kruse, E., Grimm, B.: Role of magnesium chelatase activity in the early steps of the tetrapyrrole biosynthetic pathway.-Plant Physiol. 122: 1161–1169, 2000.
Pattanayak, G.K., Biswal, A.K., Reddy, V.S., Tripathy, B.C.: Light-dependent regulation of chlorophyll b biosynthesis in chlorophyllide a oxygenase overexpressing tobacco plants.-Biochem. biophys. Res. Commun. 326: 466–471, 2005.
Pinta, V., Picaud, M., Reiss-Husson, F., Astier, C.: Rubrivivax gelatinosus acsF (previously orf358) codes for a conserved, putative binuclear-iron-cluster-containing protein involved in aerobic oxidative cyclization of Mg-protoporphyrin IX monomethylester.-J. Bacteriol. 184: 746–753, 2002.
Porra, R.J.: Recent progress in porphyrin and chlorophyll biosynthesis.-Photochem. Photobiol. 65: 492–516, 1997.
Porra, R.J., Schafer, W., Katheder, I., Scheer, H.: The derivation of the oxygen atoms of the 13(1)-oxo and 3-acetyl groups of bacteriochlorophyll a from water in Rhodobacter sphaeroides cells adapting from respiratory to photosynthetic conditions: evidence for an anaerobic pathway for the formation of isocyclic ring E.-FEBS Lett. 371: 21–24, 1995.
Reinbothe, S., Reinbothe, C.: Regulation of chlorophyll biosynthesis in angiosperms.-Plant Physiol. 111: 1–7, 1996.
Reinbothe, S., Reinbothe, C., Lebedev, N., Apel, K.: PORA and PORB, two light-dependent protochlorophyllide-reducing enzymes of angiosperm chlorophyll biosynthesis.-Plant Cell 8: 763–769, 1996.
Ron, D., Habener, J.F.: CHOP, a novel developmentally regulated nuclear protein that dimerizes with transcription factors C/EBP and LAP and functions as a dominant-negative inhibitor of gene transcription.-Gene Develop. 6: 439–453, 1992.
Rudiger, W., Bohm, S., Helfrich, M., Schulz, S., Schoch, S.: Enzymes of the last steps of chlorophyll biosynthesis: modification of the substrate structure helps to understand the topology of the active centers.-Biochemistry 44: 10864–10872, 2005.
Rzeznicka, K., Walker, C.J., Westergren, T., Kannangara, C.G., Wettstein, D. von, Merchant, S., Gough, S.P., Hansson, M.: Xantha-l encodes a membrane subunit of the aerobic Mg-protoporphyrin IX monomethyl ester cyclase involved in chlorophyll biosynthesis.-Proc. nat. Acad. Sci. USA 102: 5886–5891, 2005.
Sofia, H.J., Chen, G., Hetzler, B.G., Reyes-Spindola, J.F., Miller, N.E.: Radical SAM, a novel protein superfamily linking unresolved steps in familiar biosynthetic pathways with radical mechanisms: functional characterization using new analysis and information visualization methods.-Nucleic Acids Res. 29: 1097–1106, 2001.
Spiller, S.C., Castelfranco, A.M., Castelfranco, P.A.: Effects of iron and oxygen on chlorophyll biosynthesis. I. In vivo observations on iron-and oxygen-deficient plants.-Plant Physiol. 69: 107–111, 1982.
Sun, D.S., Chang, A.C., Jenkins, N.A., Gilbert, D.J., Copeland, N.G., Chang, N.C.: Identification, molecular characterization, and chromosomal localization of the cDNA encoding a novel leucine zipper motif-containing protein.-Genomics 36: 54–62, 1996.
Suzuki, J.Y., Bollivar, D.W., Bauer, C.E.: Genetic analysis of chlorophyll biosynthesis.-Annu. Rev. Genet. 31: 61–89, 1997.
Thimm, O., Blasing, O., Gibon, Y., Nagel, A., Meyer, S., Kruger, P., Selbig, J., Muller, L.A., Rhee, S.Y., Stitt, M.: MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes.-Plant J. 37: 914–939, 2004.
Tottey, S., Block, M.A., Allen, M., Westergren, T., Albrieux, C., Scheller, H.V., Merchant, S., Jensen, P.E.: Arabidopsis CHL27, located in both envelope and thylakoid membranes, is required for the synthesis of protochlorophyllide.-Proc. nat. Acad. Sci. USA 100: 16119–16124, 2003.
Vavillin, D.V. Vermaas, W.F.J. Regulation of the tetrapyrrole biosynthetic pathway leading to heme and chlorophyll in plants and cyanobacteria.-Physiol. Plant. 115: 9–24, 2002
Walker, C.J., Castelfranco, P.A., Whyte, B.J.: Synthesis of divinyl protochlorophyllide. Enzymological properties of the Mg-protoporphyrin IX monomethyl ester oxidative cyclase system.-Biochem. J. 276: 691–697, 1991.
Walker, C.J., Mansfield, K.E., Rezzano, I.N., Hanamoto, C.M., Smith, K.M., Castelfranco, P.A.: The magnesium-protoporphyrin IX (oxidative) cyclase system. Studies on the mechanism and specificity of the reaction sequence.-Biochem. J. 255: 685–692, 1988.
Wallar, B.J., Lipscomb, J.D.: Dioxygen activation by enzymes containing binuclear non-heme iron clusters.-Chem. Rev. 96: 2625–2658, 1996.
Wettstein, D. von, Gough, S., Kannangara, C.G.: Chlorophyll biosynthesis.-Plant Cell 7: 1039–1057, 1995.
Whyte, B.J., Castelfranco, P.A.: Further observations on the Mg-protoporphyrin IX monomethyl ester (oxidative) cyclase system.-Biochem. J. 290: 355–359, 1993.
Wong, Y.-S., Castelfranco, P.A.: Resolution and reconstitution of Mg-protoporphyrin IX monomethyl ester (oxidative) cyclase, the enzyme system responsible for the formation of the chlorophyll isocyclic ring.-Plant Physiol. 75: 658–661, 1984.
Wong, Y.-S., Castelfranco, P.A.: Properties of the Mg-protoporphyrin IX monomethyl ester (oxidative) cyclase system.-Plant Physiol. 79: 730–733, 1985.
Wong, Y.-S., Castelfranco, P.A., Goff, D.A., Smith, K.M.: Intermediates in the formation of the chlorophyll isocyclic ring.-Plant Physiol. 79: 725–729, 1985.
Xue, H., Yang, Y.T., Wu, C. A., Yang, G.D., Zhang, M.M., Zheng, C.C.: TM2, a novel strong matrix attachement region isolated from tabacco, increases transgene expression in transgenic rice calli and plants.-Theor. appl. Genet. 110: 620–627, 2005.
Yang, Y.T., Yang, G.D., Liu, S.J., Zheng, C.C.: Isolation and functional analysis of a stronger specific promoter in photosynthetic tissues.-Sci. Chin. (Ser. C) 46: 651–660, 2003.
Zheng, C.C., Porat, R., Lu, P., O’Neill, S.D.: PNZIP is a novel mesophyll-specific cDNA that is regulated by phytochrome and the circadian rhythm and encodes a protein with a leucine zipper motif.-Plant Physiol. 116: 27–35, 1998.
Zheng, C.C., Yang, G.D., Wang, D., Li, H.M.: Identification and tissue-specific expression of a full length cDNA encoding ZIP protein in Arabidopsis.-Acta biochem. biophys. sin. 34: 109–112, 2002.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, H.H., Zheng, C.C. MPEC: an important gene in the chlorophyll biosynthesis pathway in photosynthetic organisms. Photosynthetica 46, 321–328 (2008). https://doi.org/10.1007/s11099-008-0060-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-008-0060-6