Abstract
Background Drug use in pregnancy is often reason of concern for mothers and their physicians. However, only few studies investigated predictors of drug use in pregnancy. Objectives To examine maternal characteristics as predictors of medication use in the 6 months before pregnancy and during the first 6 months of pregnancy. To examine whether prescription and over-the-counter (OTC) medication use in the 6 months before pregnancy had an impact on medication use in pregnancy. Setting Six maternity care units and five community pharmacies. Method Data were collected using a specially designed self-reported questionnaire during the period March 2009–March 2010. Logistic regression was used to identify factors associated with medication use. Main outcome measure Adjusted odds ratios (aOR) and 95 % confidence intervals (CI) were used as association measures. Results A total of 236 women were included in the analysis. After controlling for maternal characteristics, parity of more than one was associated with lower prescription medication use in pregnancy (aOR 0.46; 95 % CI 0.22–0.93), higher household income with higher OTC medication use before pregnancy (aOR 3.13; 95 % CI 1.22–8.00), and miscarriage with higher C and D Food and Drug Administration (FDA) risk category medication use in pregnancy (aOR 3.65; 95 % CI 1.30–10.25). Prescription medication use before pregnancy was associated with higher prescription medication use in pregnancy (aOR 2.49; 95 % CI 1.12–5.52), OTC medication use before pregnancy with higher OTC medication use in pregnancy (aOR 35.95; 95 % CI 7.95–162.49), and C and D FDA risk category medication use before pregnancy with the same category medication use in pregnancy (aOR 3.54; 95 % CI 1.23–10.17). Conclusion Different maternal characteristics were shown as predictors of medication use before and during pregnancy. However, medication use before pregnancy was shown as the most important predicting factor for the medication use in pregnancy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Impact of findings on practice
-
Health care personnel should be aware of the impact of obstetric history and prior medication use on medication use in pregnancy.
-
Potentially harmful medications used before pregnancy should, sometimes, be avoided or substituted with medications with lower fetal risk.
-
Preconception counseling of women who need pharmacotherapeutic treatment may lead to a safer use of prescribed and OTC medications during pregnancy.
Introduction
Pregnancy is a unique period in the life of a woman when most women become highly motivated to avoid unhealthy substances to protect their unborn child from harm. This is, however, also time when illnesses and discomforts may occur—sometimes requiring pharmacological treatment. Studies have shown that three to nine out of ten pregnant women use prescribed medications [1]. Despite the fact that over-the-counter (OTC) medications use in pregnancy was less studied, reports showed that two to nine out of ten pregnant women used OTC medications [2, 3]. The majority of these medication usage studies have been conducted in the West-European countries, and North-America, and less is known about the East-European countries and the rest of the world.
In addition to wide variations in medication use during pregnancy across different countries, different maternal characteristics also may influence medication use during pregnancy. However, results from previous studies examining the impact of maternal characteristics on the use of medications during pregnancy are inconsistent. Some studies have identified wide range of factors related to medication use during pregnancy as follows: a high level of education, smoking, higher age and being single [4–6]. In contrast, other studies did not find any association between medication use during pregnancy and maternal socio-demographic and life-style characteristics [7–9]. Most of these studies investigated associations between prescription medication use and maternal characteristics, while OTC medications were not included in the analyses. OTC medications were usually used and selected by pregnant women in self-medication process, and their use was not controlled by physician. Accordingly, OTC medication use could be highly impacted by maternal characteristics. It would be clinically useful to determine potential predictors of both, prescription and OTC medication use in pregnancy.
Aim of the study
Due to insufficient and inconsistent data regarding predictors of medication use during pregnancy and a lack of data for Serbia, the main aim of this study was to examine maternal socio-economic and health characteristics as predictors of medication use (prescription and OTC) in 6 months before pregnancy and during the first 6 months of pregnancy. The aim was also to examine whether prescription and OTC medication use in the 6 months before pregnancy had an impact on medication use in pregnancy.
Methods
Study design and data collection
Broad study among pregnant women in Serbia was performed during the period March 2009–March 2010. In total, six maternity care units (Central region: 3, Western region: 2 and Northern region: 1) and five community pharmacies (Central region: 3, Western region: 1 and Northern region: 1) participated in the study. Maternity care units and pharmacies enrolled covered 185117 female inhabitants of reproductive age (15–49 years old), equivalent to approximately 10 % of the Serbian female inhabitants of reproductive age [10].
A sample for this study was extracted from a total of 750 participants who accepted to participate in a broad study. Exclusion criterion was pregnancy less than 6 months since we have aimed to analyze predictors of drug use in the 6 months before pregnancy and during the first 6 months of pregnancy (218 women excluded). Other exclusion criterion was missing data regarding socio-economic, and health related characteristics, or medication use (230 women excluded). As we aimed to analyze predictors of medication use separately for prescription and OTC medications, participants who used both prescription and OTC medications were excluded (17 women). Although they had accepted to participate, forty-nine women returned empty questionnaires. Finally, there were 236 women with complete data in the study cohort. Participation in the study was voluntary, anonymous and confidential. After filling up the questionnaire, each woman had to give it back to nurse or pharmacist. The main researcher collected all questionnaires from all maternity care units and pharmacies.
A specially designed self-reported questionnaire was used as data source. Questionnaires were distributed by trained nurses and pharmacists. Questions about age and parity were in the form of open-ended questions. Multiple-choice questions were used in data collection regarding education (primary school (8 years), high school (11–12 years), graduate studies (15–18 years), postgraduate studies (19–23 years)) as well as household income per family member (<50/50–99/100–149/150–299/300–449/450–700/>700 EUR). Women’s health status regarding chronic disease such as asthma, diabetes mellitus, hypothyreosis, hypertension, heart diseases or any other chronic diseases, was inquired about and participants needed to mark diseases which they had been diagnosed with. There was an open-ended question regarding the presence of any other disease. Data regarding any eventual previous prenatal deaths [induced abortion and miscarriage (yes/no)] were also collected.
Participants were asked to write down names of medications which they had used 6 months before or during pregnancy for treatment of anxiety/depression, pain, hypertension, nausea and vomiting, cold and cough, or any other medication. They also had to mark the trimester when medication was used. The usage of at least one dose of medication was considered as a woman was exposed to particular medication.
National formularies were used for medication classification regarding its prescription and OTC status [11]. The Food and Drug Administration (FDA) pregnancy risk classification system was used for medication classification according to the risk to a fetus [12, 13].
Variables
Dependent variables were: (1) prescription medication use, (2) OTC medication use and (3) C/D FDA risk category medication use. The variables were coded “yes” or “no” (reference category).
The C/D FDA risk category medications included prescription and OTC medications which belonged to C or D FDA risk categories. If one OTC medication consisted of several active substances, the highest risk category of substances was considered as the risk category of that medication. X category medications were not included in our analysis as no women used category X medication during pregnancy.
The following independent variables were considered as potential predictors of receiving at least one prescription, OTC or C/D FDA risk category medication:
-
socio-economic characteristics: age (<25 (reference category)/25–29/≥30 years), education (≤12 (reference category)/>12 years), household income per family capita (≤300 (reference category)/>300 EUR), parity (1 (reference category)/≥2)
-
health related characteristics: presence of chronic diseases (no (reference category)/yes), previous prenatal death (no (reference category)/induced abortion/miscarriage)
-
medication use in the 6 months before pregnancy classified as: (1) prescription, (2) OTC and (3) C/D FDA risk category medication use in the period of the 6 months before pregnancy [yes/no (reference category)]; these categories of independent variables were used only in analysis which investigated medication use in pregnancy.
Statistical analysis
A McNemar’s test was performed to assess differences in prescription, OTC and C/D FDA risk category medication use before and during pregnancy.
A univariate analysis was used to analyze univariate associations between potential explanatory variables and prescription, OTC and C/D FDA risk category medication use before and during pregnancy. Crude odds ratio with 95 % confidence interval was used to explain the association of explanatory variable and a medication use.
Variables found to be associated with medication use in the univariate analysis, or shown in the literature to be associated with medication use, were included in multivariate logistic regression models (age, education, household income per family capita, parity, chronic disease, previous prenatal death and prescription medication use 6 months before pregnancy) [4, 8, 14, 15]. The impact of variables on medication use was estimated as an adjusted odds ratio (aOR) with 95 % confidence interval (CI).
Statistical significance in all analysis was deemed likely if computed probability value was <0.05.
Data analysis was performed using Statistical Package for Social Science (SPSS) software (SPSS 18.0 for Windows, SPSS Inc., Chicago, IL, USA).
Results
The mean age of the cohort was 27.8 ± 5.3 years (range 16–44 years). Socio-economic and health characteristics of pregnant women according to medication use are shown in Table 1. A total of 7.2 % of women reported chronic diseases, most commonly asthma (1.7 %), diabetes mellitus (1.7 %) and hypertension (1.3 %).
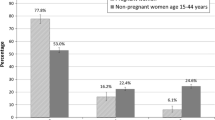
There was significant increase in prescription and C/D FDA risk categories medications use comparing periods before and during pregnancy, 14.8 versus 26.3 %, and 8.5 versus 18.2 %, respectively, and significant decrease in OTC medications use, 11.9 versus 6.4 %, p < 0.05.
Maternal characteristics associated with prescription medication use
The univariate analysis showed significant association between maternal age and prescription medication use both, before and during pregnancy. Women older than 25 years of age were more likely to use at least one prescription medication before their pregnancy as compared with women younger than 25 years. Similarly, women older than 29 were more likely to use at least one prescription medication in pregnancy compared to women younger than 25. Highly educated pregnant women and women who used at least one prescription medication before pregnancy were more likely to use at least one prescription medication in pregnancy (Table 2).
Univariate associations disappeared after multivariate analyses were performed regarding medication use before pregnancy. This analysis showed that a higher parity and prescription medication use before pregnancy were the only factors significantly associated with prescription medication use in pregnancy. Women, with parity higher than one, were less likely to use prescription medication in the first 6 months of pregnancy (aOR 0.46; 95 % CI 0.22–0.93). Women who used at least one prescription medication before pregnancy were more likely to use prescription medication in pregnancy (aOR 2.49; 95 % CI 1.12–5.52) (Table 2).
Maternal characteristics associated with OTC medication use
The univariate analysis revealed no significant associations between maternal characteristics and OTC medication use before pregnancy. In contrast, the same analysis showed that OTC medication use in pregnancy was associated with the parity and OTC medication use before pregnancy, but these associations were not confirmed in the multivariate analysis (Table 3).
The multivariate analysis revealed that women who had household income per family capita above 300 EUR were more likely to use OTC medications before pregnancy, aOR 3.13; 95 % CI 1.22–8.00. The only factor significantly associated with OTC medication use during the first 6 months of pregnancy in a multivariate analysis was OTC medication use before pregnancy (aOR 35.95; 95 % CI 7.95–162.49) (Table 3).
Maternal characteristics associated with a C/D FDA risk categories medication use
The univariate analysis revealed no significant associations between maternal characteristics and C/D FDA risk categories medication use before pregnancy. In contrast, the same analysis showed that highly educated women, women who had miscarriage or who used C/D FDA risk categories medications before pregnancy were more likely to use C/D FDA risk categories medications in the first 6 months of pregnancy (Table 4).
The multivariate analysis revealed that previous miscarriages and C/D FDA risk categories medication use before pregnancy were significantly associated with C/D FDA risk categories medications use in pregnancy, aOR 3.65; 95 % CI 1.30–10.25, and aOR 3.54; 95 % CI 1.23–10.17, respectively (Table 4).
Discussion
Many previous studies reported different patterns of medication use in the period before pregnancy compare to medication use in pregnancy [16–21]. However, to our knowledge, previous studies did not explore maternal characteristics associated with prescription and OTC medication use before and during pregnancy, neither the impact of medication use before pregnancy on medication use in pregnancy.
Several maternal socio-economic and life style characteristics have been reported previously as significantly associated with prescription medication use in pregnancy (e.g. higher level of education, higher household income, cohabitation status, place of birth, age) [7, 22]. However, Nordeng et al. [8] showed that most of associations between socio-economic and life style factors (age, parity, marital status, smoking habits, maternal education and occupation) became insignificant after adjusting for the presence of chronic disease. Accordingly, Nordeng et al. [8] concluded that the use of medication is driven by a need of pharmacological treatment, and not by maternal characteristics.
After adjusting for women’s socio-economic and health status in our analysis (age, education, household income, parity, chronic disease status, previous prenatal death, prescription medication use before pregnancy), independent predictors of prescription medication use in pregnancy were parity and prescription medication use before pregnancy, and other maternal characteristics could be considered as confounders. Such results support the conclusion of Nordeng et al. since prescription medication use before pregnancy indicates the need of pharmacological treatment before pregnancy which may continue in pregnancy as well. Accordingly, history of medication use should be carefully examined by physician during the periconceptual care for woman. Thus, potentially harmful medications could be avoided or substituted with medications with lower fetal risk. Moreover, our results indicate that women who already had an experience of giving birth are less likely to use medications in pregnancy possibly because they are more informed and aware of potential harmful effects.
Few studies have examined factors related to OTC medication use in pregnancy. Despite of univariate associations between OTC medication use in pregnancy and the high education, place of birth and early initiation of prenatal care which was shown by Bercaw et al. [23], independent associations between socio-economic factors and OTC medication use were not found in multivariate modeling. Rubin et al. [24] found positive association between non-prescription medication use and multiple illnesses, higher socio-economic status score and a Caucasian origin, however control for potential confounders was not performed. Our results suggested that OTC medication use before pregnancy significantly impacts OTC medication use in pregnancy. Regardless of decrease in OTC medication use in pregnancy, this finding may indicate that women’s habits of OTC medication use change relatively little when becoming pregnant. Explanations may be related to underlying reason for OTC use (illness-related) or in the women’s attitudes (“OTC medications are sold without prescription and must consequently be mild”). Pregnant women’s attitudes and beliefs towards OTC medications warrant further investigation.
The fact that income had had an impact on OTC medication use before pregnancy could be explained by the fact that these medications have not been reimbursed in Serbia. Accordingly, a higher maternal economic status could have contributed to a higher OTC medication use by women.
Similarly to the prescription medication use, OTC medication use in the periconceptual period should be carefully examined and discussed with physician as well. However, Morgan et al. [25] reported that 14 % of obstetricians did not ask pregnant women about OTC medication use, while more obstetricians (86 %) were likely to ask women about OTC medication use during obstetric care visit than during the routine care visit (52 %). Counseling on an adequate medication use and its harmful potential for the fetus could be late if women already used harmful medications during the first weeks of pregnancy. Accordingly, information about potentially harmful medications should be distributed among women during their whole reproductive period and discussed during the routine care visits.
Previous studies from the United States (US), United Kingdom (UK), and Canada found that 2–4 % and 0.5–3 % of women used D and X FDA risk categories medications, respectively, in the first trimester [14, 17, 26, 27]. However, few studies have shown that the health status of women, e.g. complications of pregnancy, history of infertility or chronic health condition, unplanned pregnancy, and multiple pregnancy were associated with the use of category D and X medications [22]. Potentially, socio-economic and life style predictors of unsafe medications use in pregnancy were the place of residence, being single, being a smoker, unemployed, or nulliparous [4, 15]. Yang et al. [14] reported that women who were under 25 years of age, who had a parity of more than two and were included in a social assistance plan were at increased risk to be exposed to C, D and X FDA risk category medications. Our analysis confirmed the importance of women’s health status for C/D FDA risk categories medication use, and showed that women who had history of miscarriage, and women who used C/D FDA risk category medications before pregnancy had more chance to use these medications in pregnancy.
The need for a comprehensive public health approach with activities directed towards providing evidence-based information about medication use available to the public, and translating them into safe and effective health care was recognized by Lagoy et al. [28]. Determination of predictors of medication use in pregnancy could be useful in this public health approach, as it can help in identification of vulnerable groups of women who have more chance to be exposed to medications, and particularly unsafe medications, during pregnancy. Accordingly, it would be useful to continue with exploring such predictors in future studies.
There are some limitations to this study that should be acknowledged. A small sample size could impact the statistical power to detect any association between maternal characteristics and medication use before and during pregnancy. For the same reason, a sub-analysis on specific medications could not be carried out. Moreover, due to a limited number of data in a relatively small sample size, few large confidence intervals were observed. In addition, although we recorded indication for use of each medication, we could not evaluate appropriateness of the individual medication use. Even the use of category D can in some cases be appropriate when no other alternative is judged to be adequate. Comparing characteristics of our cohort with the Serbian population of 20–44 years of age, lower prevalence of chronic diseases in studied cohort was observed, 7.2 versus 33.5 %, and more women in the cohort had education above 12 years, 30.9 versus 4.9 % [29]. The advantages lie in the ability to examine the impact of a preconceptual medication use on medication use after conception, and OTC and prescription medications use analysis separately. Moreover, an FDA categorization included OTC medications, which has not been done in any previous study which examined predictors of C, D and X medication use in pregnancy. Our results should be interpreted with these advantages and limitations in mind.
Conclusions
Several maternal characteristics were found to be associated with medication use before and during pregnancy. A higher household income was associated with OTC medications use before pregnancy, parity with prescription medication use in pregnancy, and miscarriage with categories C/D FDA medications use in pregnancy. Nevertheless, prior use of medication was shown as an important predicting factor of medication use during pregnancy, either for prescribed and OTC medications.
References
Daw JR, Hanley GE, Greyson DL, Morgan SG. Prescription drug use during pregnancy in developed countries: a systematic review. Pharmacoepidemiol Drug Saf. 2010;20(9):895–902.
Glover DD, Amonkar M, Rybeck BF, Tracy TS. Prescription, over-the-counter, and herbal medicine use in a rural, obstetric population. Am J Obstet Gynecol. 2003;188(4):1039–45.
Black RA, Hill AD. Over-the-counter medications in pregnancy. Am Fam Physician. 2003;67(12):2517–24.
Cleary BJ, Butt H, Strawbridge JD, Gallagher PJ, Fahey T, Murphy DJ. Medication use in early pregnancy-prevalence and determinants of use in a prospective cohort of women. Pharmacoepidemiol Drug Saf. 2010;19(4):408–17.
Donati S, Baglio G, Spinelli A, Grandolfo ME. Drug use in pregnancy among Italian women. Eur J Clin Pharmacol. 2000;56(4):323–8.
Bonassi S, Magnani M, Calvi A, Repetto E, Puglisi P, Pantarotto F, et al. Factors related to drug consumption during pregnancy. Acta Obstet Gynecol Scand. 1994;73(7):535–40.
Olesen C, Thrane N, Henriksen T, Ehrenstein V, Olsen J. Associations between socio-economic factors and the use of prescription medication during pregnancy. Eur J Clin Pharmacol. 2006;62(7):547–53.
Nordeng H, Eskild A, Nesheim BI, Aursnes I, Jacobsen G. Drug use during early pregnancy. Eur J Clin Pharmacol. 2001;57(3):259–63.
Buitendijk S, Bracken MB. Medication in early pregnancy: prevalence of use and relationship to maternal characteristics. Am J Obstet Gynecol. 1991;165(1):33–40.
Statistical Office of the Republic of Serbia. Population: census of population, households and dwellings. Sex and age, data by localities. Belgrade 2003. ISBN: 86-84433-01-7. Available at: http://webrzs.stat.gov.rs/WebSite/Public/PageView.aspx?pKey=164. Accessed 01 Dec 2012.
Djukic Lj. National formulary. Belgrade: Medicines and Medical Devices Agency of Serbia, 2010. ISSN: 1452-3337.
Food and Drug Administration. Available at: http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm. Accessed 05 Dec 2012.
Briggs GG, Freeman RK, Yaffe SJ. Drugs in pregnancy and lactation: a reference guide to fetal and neonatal risk. 8th ed. Philadelphia: Lippincott Wiliams & Wilkins; 2008. ISBN: 978-0-7817-7876-3.
Yang T, Walker MC, Krewski D, Yang Q, Nimrod C, Garner P, et al. Maternal characteristics associated with pregnancy exposure to FDA category C, D, and X drugs in a Canadian population. Pharmacoepidemiol Drug Saf. 2008;17(3):270–7.
Lee E, Maneno MK, Smith L, Weiss SR, Zuckerman IH, Wutoh AK, et al. National patterns of medication use during pregnancy. Pharmacoepidemiol Drug Saf. 2006;15(8):537–45.
Egen-Lappe V, Hasford J. Drug prescription in pregnancy: analysis of a large statutory sickness fund population. Eur J Clin Pharmacol. 2004;60(9):659–66.
Andrade SE, Gurwitz JH, Davis RL, Chan KA, Finkelstein JA, Fortman K, et al. Prescription drug use in pregnancy. Am J Obstet Gynecol. 2004;191(2):398–407.
Stephansson O, Granath F, Svensson T, Haglund B, Ekbom A, Kieler H. Drug use during pregnancy in Sweden—assessed by the prescribed drug register and the medical birth register. J Clin Epidemiol. 2011;3(1):43–50.
Engeland A, Bramness JG, Daltveit AK, Rønning M, Skurtveit S, Furu K. Prescription drug use among fathers and mothers before and during pregnancy. A population-based cohort study of 106,000 pregnancies in Norway 2004–2006. Br J Clin Pharmacol. 2008;65(5):653–60.
Bakker MK, Jentink J, Vroom F, Van Den Berg PB, De Walle HE, De Jong-Van Den Berg LT. Drug prescription patterns before, during and after pregnancy for chronic, occasional and pregnancy-related drugs in the Netherlands. BJOG. 2006;113(5):559–68.
Olesen C, Steffensen FH, Nielsen GL, De Jong-Van Den Berg L, Olsen J, Sørensen HT. Drug use in first pregnancy and lactation: a population-based survey among Danish women. Eur J Clin Pharmacol. 1999;55(2):139–44.
Riley EH, Fuentes-Afflick E, Jackson RA, Escobar GJ, Brawarsky P, Schreiber M, et al. Correlates of prescription drug use during pregnancy. J Womens Health. 2005;14(5):401–9.
Bercaw J, Maheshwari B, Sangi-Haghpeykar H. The use during pregnancy of prescription, over-the-counter, and alternative medications among Hispanic women. Birth. 2010;37(3):211–8.
Rubin JD, Ferencz C, Loffredo C. Use of prescription and non-prescription drugs in pregnancy. Clin Pharmacol. 1993;46(6):581–9.
Morgan MA, Cragan JD, Goldenberg RL, Rasmussen SA, Schulkin J. Management of prescription and nonprescription drug use during pregnancy. J Matern Fetal Med. 2010;23(8):813–9.
Hardy JR, Leaderer BP, Holford TR, Hall GC, Bracken MB. Safety of medications prescribed before and during early pregnancy in a cohort of 81,975 mothers from the UK general practice research database. Pharmacoepidemiol Drug Saf. 2006;15(8):555–64.
Andrade SE, Raebel MA, Morse AN, Davis RL, Chan KA, Finkelstein JA, et al. Use of prescription medications with a potential for fetal harm among pregnant women. Pharmacoepidemiol Drug Saf. 2006;15(8):546–54.
Lagoy CT, Joshi N, Cragan JD, Rasmussen SA. Medication use during pregnancy and lactation: an urgent call for public health action. J Womens Health. 2005;14(2):104–9.
Ministry of Health Republic of Serbia. National Health Survey. Key Findings. Belgrade: 2006. Available at: http://www.batut.org.rs/download/publikacije/National%20Health%20Survey%20Serbia%202006.pdf. Accessed 02 Dec 2012.
Acknowledgments
We are very grateful to the Associate Professor Natasa Bogavac Stanojevic, Department of Biochemistry, The University of Belgrade-Faculty of Pharmacy for her useful comments in the statistical analysis, and to Mrs. Zorica Bogdanovic for editing of the English.
Funding
This work is supported by the Ministry of Education and Science, Republic of Serbia, Project No. 41012, and Project No. 175064, 2011–2014.
Conflicts of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Odalovic, M., Vezmar Kovacevic, S., Nordeng, H. et al. Predictors of the use of medications before and during pregnancy. Int J Clin Pharm 35, 408–416 (2013). https://doi.org/10.1007/s11096-013-9750-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-013-9750-7