ABSTRACT
Purpose
FDA’s bioequivalence recommendation for Zolpidem Tartrate Extended Release Tablets is the first to use partial AUC (pAUC) metrics for determining bioequivalence of modified-release dosage forms. Modeling and simulation studies were performed to aid in understanding the need for pAUC measures and also the proper pAUC truncation times.
Methods
Deconvolution techniques, In Vitro/In Vivo Correlations, and the CAT (Compartmental Absorption and Transit) model were used to predict the PK profiles for zolpidem. Models were validated using in-house data submitted to the FDA. Using dissolution profiles expressed by the Weibull model as input for the CAT model, dissolution spaces were derived for simulated test formulations.
Results
The AUC0–1.5 parameter was indicative of IR characteristics of early exposure and effectively distinguished among formulations that produced different pharmacodynamic effects. The AUC1.5-t parameter ensured equivalence with respect to the sustained release phase of Ambien CR. The variability of AUC0–1.5 is higher than other PK parameters, but is reasonable for use in an equivalence test.
Conclusions
In addition to the traditional PK parameters of AUCinf and Cmax, AUC0-1.5 and AUC1.5-t are recommended to provide bioequivalence measures with respect to label indications for Ambien CR: onset of sleep and sleep maintenance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
In August 2009, the FDA published the Draft Guidance on Zolpidem Tartrate Extended Release (ER) Tablets with bioequivalence (BE) recommendations that included partial area-under-the-curve (pAUC) metrics (1). The use of the pAUC as a measure for assessment of early exposure is described in the 2003 General BA/BE guidance (2), but is limited to immediate-release (IR) drug products. The Draft Guidance on Zolpidem is the first guidance which includes recommendations for pAUCs as pharmacokinetic (PK) parameters for BE assessment for modified-release (MR) drug products.
For two products to be considered bioequivalent, they must be equivalent in the rate and extent of absorption of the active moiety, which are usually measured by Cmax (the maximum drug concentration) and AUC (the area under the concentration-time curve), respectively. MR dosage forms are designed to achieve a particular plasma concentration profile, which in turn produces a specific therapeutic profile. For some drug products which exhibit multiphasic PK behavior (showing more than one peak/shoulder in the concentration profile), the traditional metrics of AUC and Cmax may not be sufficient to ensure BE. In these cases, the total extent of exposure may be equivalent for two products, but the rate or extent of exposure during a clinically relevant time interval may not be equivalent (3). An additional PK metric, such as a pAUC to assess early exposure, may be necessary to quantify potential differences in therapeutic equivalence.
The regulatory decision to recommend pAUC metrics for zolpidem has been the subject of extensive internal research, review, and discussion with industry and scientific communities. The pAUC for zolpidem was discussed at a workshop on establishing scientific and regulatory standards for assuring therapeutic equivalence of MR products sponsored by the American Association of Pharmaceutical Scientists, the International Pharmaceutical Federation, and the Product Quality Research Institute on October 2009 (4). At the Pharmaceutical Science and Clinical Pharmacology Advisory Committee Meeting on April 2010, the topic on the use of pAUCs for the evaluation of products with complex PK profiles, including zolpidem ER tablets, was reviewed (5). The use of pAUCs for MR dosage forms, and for zolpidem ER tablets in particular, received broad support at both of these meetings.
Ambien CR® (zolpidem tartrate extended release tablets) is formulated to exhibit biphasic absorption characteristics by incorporating an IR layer and a controlled release (CR) layer (6). Ambien CR® is indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance. To produce these therapeutic effects, the IR layer releases drug quickly to produce sleep onset and the CR layer releases drug over a sustained period of time for sleep maintenance.
Several modeling strategies were used to explore the need for pAUC measures and also to identify the proper pAUC truncation times for zolpidem ER tablets. A physiologically-based model (the Compartmental Absorption and Transit model; CAT), In Vitro/In Vivo Correlations (IVIVC), and deconvolution techniques were utilized and the approach implemented for each is described below.
MATERIALS AND METHODS
In clinical studies that supported the approvals of Ambien® and Ambien CR®, most subjects (>90%) fell asleep 1.5 h after dosing. For both Ambien® and Ambien CR®, the difference in the rate of falling asleep between the drug and placebo was also greatest at 1.5 h. At later times (such as 2.0 h), all subjects were asleep, while at earlier times (such as 1.0 h), there was still a growing difference in the rate of falling asleep compared to placebo. Based on this clinically relevant time point, the pAUC truncated through 1.5 h (AUC0–1.5) was chosen for investigation in modeling and simulation studies.
During drug product development of Ambien CR, several formulations with varying ratios of IR and ER components were investigated and tested in pharmacodynamic studies (7). Three of these formulations tested, C, E, and G, were similar to Ambien CR, but contained different ratios of zolpidem in IR to ER components, and their in-vitro release profiles differed. For all 3 formulations, the active ingredient was completely released by 4 h post-dose. Formulation C had a larger amount of IR zolpidem and immediately released 80% of the drug followed by a slow release of the remaining 20% over the following 4 h. Formulation E produced an immediate dose of 60% followed by sustained release of the remaining 40%; formulation G released an immediate dose of 40% followed by sustained release of the remaining 60%. In a comparative pharmacodynamic study of formulations C, E, and G, differences in duration of sleep and residual post-awakening effects were observed among the formulations. Formulation E was selected for further clinical development, and its in-vivo drug plasma concentration profile was determined from subsequent pharmacokinetic studies. However, in the comparative pharmacodynamic study that led to the selection of formulation E, the drug plasma concentrations of the various investigational formulation prototypes, including formulations C and G, were not measured at adequate time intervals to construct a PK profile. We used a series of modeling approaches (IVIVC, deconvolution, and CAT) to estimate the pharmacokinetic profiles of these prototype formulations, and to evaluate if the traditional bioequivalence parameters of AUC and Cmax would distinguish among prototypes C, E, and G that differed in their pharmacodynamic effects. The models were subsequently used to evaluate the sufficiency of the traditional PK parameters in determining BE, and the proper pAUC metrics for zolpidem ER. Simulations were performed using MATLAB® (MathWorks, Inc.) and Octave (www.octave.org).
Deconvolution
To compare the amount of systemic absorption of zolpidem from the IR and CR formulations, deconvolution by point-area method was performed on the mean PK data obtained from a pharmacokinetic study (8) submitted in support of approval of Ambien CR. The study included dosing with intravenous (IV) zolpidem, Ambien IR and Ambien CR. The mean PK profile of an IV bolus was simulated based on PK parameters from a two-compartment model of the IV infusion data. To predict the pharmacokinetic profiles for formulations C, E, and G, a deconvolution of the IR and bilayer formulations against IV data provided the independent contributions of the IR and ER components of the formulation by subtraction. The IR and ER components were then recombined based on the IR/ER ratio of the formulations C, E, and G. This deconvolution approach can only make predictions for formulations that have the same IR and ER components (in varying ratios) as formulations C, E, and G.
IVIVC
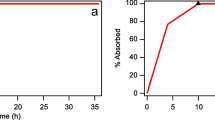
A standard two-stage approach (convolution and deconvolution) was used to establish a Level A IVIVC between the in vitro dissolution profiles and the in vivo release profiles of formulation E under fasting conditions. The dissolution profiles were measured with USP Apparatus 1 (basket) at 100 rpm with 500 mL aqueous Hydrochloric acid (pH = 2.2) at 37°C for 6 h. A two-compartment model with elimination from the central compartment was selected as it better described the IV infusion PK data (8) than a one-compartment model for most subjects. The mean PK profile after giving an IV bolus was simulated based on the two-compartment model PK parameters obtained by fitting the IV infusion data. Then the simulated mean IV bolus profile was used as the reference for all deconvolution and convolution calculations. The mean in vivo release profiles were obtained by deconvolution (by point-area method) using the mean plasma PK profiles of the zolpidem modified release (MR) formulation under two different doses. In order to correlate the in vitro and in vivo release profiles, a Hill model (1) was used with a lag time correction for the in vitro release profile:
where y is the % released in vivo, x is the % release in vitro, and a, b, and c are parameters obtained by fitting. The fitted values of a, b, and c were 73.75, 5.14, and 69.69, respectively. A plot of the in vitro-in vivo relationship is shown in Fig. 1.
CAT Model
The CAT model simulates the effect of physiological conditions on drug absorption as it transits through successive gastrointestinal (GI) compartments (9–11). The system of equations includes different drug states (unreleased, undissolved, dissolved). The total amount of absorbed material is summed over the integrated amounts being absorbed from each compartment. This model requires input parameters of physicochemical drug properties and PK data. In this modeling study, data on zolpidem solubility at various pH, volume of distribution (Vd), and clearance (CL) were obtained from the New Drug Application (NDA) submission of Ambien CR and from population PK analysis of the NDA studies (Vd/F = 67.7 L and 39.5 L for males and females, respectively; and CL/F = 15.8 L/hr and 8.83 L/hr for males and females, respectively). The bioavailability, F, was set to 0.68. The permeability (Peff = 7.5 × 10−4 cm/sec) was obtained by a fit to data from the IR formulation. The physiological parameters for the GI tract are summarized in Table I.
Prediction of plasma concentration profiles (without any other adjustable parameters) is made by inputting the in vivo dissolution as described by a Weibull model (2):
where t is time and t lag is the lag time which was considered to be 0. The a parameter has units of time and is related to the dissolution rate with a larger a indicating slower dissolution. The b parameter is a dimensionless shape parameter with b = 1 being an exponential release, b > 1 representing an “S” shaped profile and b < 1 representing a profile with faster than exponential release. By using the CAT model together with the Weibull model, it is assumed that the in vitro dissolution profile suitably matches the in vivo dissolution profile.
Mapping the Bioequivalence Region
The advantage of using the Weibull model in conjunction with the CAT model is that the Weibull model can describe the drug release characteristics of the Reference (R) product, the Test (T) formulations used in the NDA product development, and the drug release space of all submitted Abbreviated New Drug Applications (ANDAs) for generic zolpidem CR. Therefore, by using appropriate a and b adjustable parameters, the Weibull model can be used to comprehensively describe the entire possible space of zolpidem drug release profiles that could be submitted as putative generic versions of zolpidem CR products in ANDAs.
The CAT model was then used to make predictions of Test to Reference (T/R) ratios for all PK parameters of interest including AUC, Cmax, AUC0–1.5, AUC1.5-t, AUC3–6, and AUC6−∞ over the entire dissolution space that could be potentially used by ANDA applicants. Test products were simulated with different values of a and b in the Weibull model. For each Test product, the in vivo dissolution was calculated for input into the CAT model, which predicted the resulting PK profiles. The T/R ratio was calculated for each PK parameter. Products were considered to be bioequivalent when the ratio was between 0.9–1.11, as most generic products differ by less than 10% in AUC from that of the innovator product (12).
Evaluation of the Variability of Partial AUCs
We further evaluated the relationship between the AUC T/R ratios with various cutoff times for partial AUCs (0–0.5, 0–1.0, 0–1.5, 0–2.0, 0–2.5, 0–3.0, 0–4.0, 0–6.0, and 0-tlast). The residual variability was also calculated for the above partial AUCs from all in-house BE studies to evaluate the variability of partial AUCs. Because BE criteria are based on confidence intervals, understanding the residual variability would be important for sample size calculation and study design.
RESULTS
Comparison of IR and CR Absorption
The cumulative systemic absorption of Ambien IR and CR formulations were obtained from deconvolution of data from a PK study in the NDA submission. The profiles of the total amount absorbed were almost superimposable up to 2 h (Fig. 2). This is consistent with the label claims which state that Ambien CR is designed to provide initial plasma concentrations comparable to Ambien IR.
CAT Model: Fitting and Validation
The in vivo permeability of zolpidem is unknown. To find the appropriate permeability, a parameter scan was performed. Figure 3a shows that with increasing permeability, Cmax increased and Tmax decreased. However, the changes in Cmax and Tmax became less sensitive to changes in permeability when permeability was higher than 4.0 × 10−4 cm/sec. The permeability (7.5 × 10−4 cm/sec) was obtained by fitting the CAT model to the mean PK profile of Ambien IR (Fig. 3b).
Effects of permeability on PK profiles using the CAT model. (a) Comparison of simulated PK profiles using different permeability values with the observed PK profile after dosing withgiving Ambien IR; (b) Comparison of the simulated PK profile using Peff = 7.5 × 10−4 cm/sec and the observed PK profile after giving dosing with Ambien IR.
Prediction of PK Profiles for Zolpidem ER
Three different approaches, IVIVC, deconvolution, and CAT modeling, were used to predict the PK profiles for zolpidem ER. The IVIVC for formulation C unrealistically predicted that formulation C would release faster than the IR Ambien product (Fig. 4a). Thus, the IVIVC was not valid for prediction of formulation C. Predicted PK profiles are shown in Fig. 4 and the calculated PK parameters are summarized in Table II for each modeling approach. The Weibull values obtained from fitting Ambien CR (a = 0.72 and b = 0.54) were used to simulate the PK profiles in Fig. 4c. Figure 4d further supports that the IVIVC approach is not accurate for the IR components as it does not accurately predict the results from a pure IR product.
Using formulation E as the reference formulation, the deconvolution method and the CAT model predicted similar T/R ratios for AUCinf, Cmax, and AUC3–6. For AUC0–1.5, the T/R ratios predicted by all models indicated that it was a sensitive measure of the formulation differences (Table II).
The CAT model is the most physiologically consistent approach of the three attempted because it includes realistic gastric emptying and GI transit times. The IVIVC and deconvolution methods are purely empirical methods with no link to the underlying physiology. However, it is of interest to note that the deconvolution method gave comparable results to the CAT model. Another advantage of using the CAT model is that PK profiles were predicted from dissolution profiles expressed by the Weibull model. By simulating the PK profiles from all possible combinations of the a and b parameters in the Weibull model, the dissolution region that passes the BE criteria can be mapped. This modeling approach can be useful in implementing a quality by design approach to formulation development.
The CAT Model Reasonably Predicted the Observed T/R Ratios of AUCinf, Cmax, Tmax and AUC0–1.5
We further validated the CAT model by comparing the predicted T/R ratios of AUCinf, Cmax, Tmax and AUC0–1.5 with the observed T/R ratios. The a and b parameters for 10 different Test formulations were obtained by fitting the in vitro dissolution profiles (obtained from 10 ANDA submissions) measured in 0.01N HCl at 100 rpm in USP Apparatus I. The predicted versus observed values of the T/R ratios for the PK parameters are shown in Fig. 5. Additionally, the tabulated prediction errors of this model were calculated per the FDA IVIVC Guidance (Table III) (13). The prediction errors for AUC and Cmax parameters were for the most part within the error margin of <10% for establishing the external predictitability of an IVIVC, even though some formulations had dramatically different dissolution rates. The higher observed errors in the predictability of the Tmax and AUC0–1.5 are expected as these parameters have higher variability.
Bioequivalence Region Predictions
Test products were then simulated with different values of a and b, reflecting the differing array of possible zolpidem release rates in proposed generic products. Contour plots were then generated based upon point estimate ratios for the various BE parameters, including AUCinf, Cmax, AUC0–1.5, AUC1.5-t, AUC3–6, and AUC6-inf as a function of zolpidem release rates using a and b parameters from the Weibull model (Fig. 6). The bioequivalence region is shown in white (the colored regions denote BE parameters outside the 0.9–1.11 ratio).
From this analysis, a wide range of zolpidem CR products with vastly differing zolpidem release rates, may be deemed to be equivalent to Ambien CR based upon the AUCinf parameter (Fig. 6a). This analysis also showed that only a small subset of these zolpidem CR products designed with limited ranges of zolpidem release rates would be equivalent to Ambien CR, based on the Cmax or AUC0–1.5 parameter (Fig. 6b and c). The passing regions for Cmax and AUC0–1.5 are orthogonal to each other, with a limited overlapping area. Based upon this analysis, one would expect that only zolpidem CR formulations designed with a narrow range of possible zolpidem release rates will yield product that is equivalent to Ambien CR in AUCinf, Cmax or AUC0–1.5 parameters, and this space is defined in Fig. 7.
The importance of this analysis is that this modeling approach may be used to define the space of possible zolpidem release rates that will result in generic zolpidem formulations that are equivalent to Ambien CR in AUC1.5-t, Cmax and AUC0–1.5 parameters. Once this possible space of zolpidem release rates is known, one can use this information to predict whether these formulations having these release rates, will be equivalent to Ambien CR with respect to other pAUC parameters such as AUC3–6 and AUC6-inf. The ratios for AUC3–6 and AUC6-inf that will occur in the white region of Fig. 7 (the region that is bioequivalent with respect to AUC1.5-t, Cmax and AUC0–1.5) are summarized in Table IV. One can conclude from this modeling analysis that requiring that generic zolpidem formulations be equivalent to Ambien CR in AUC1.5-t, Cmax and AUC0–1.5 will result in a generic zolpidem ER product that is equivalent to Ambien CR with respect to the second sustained release phase of zolpidem including other late pAUC measures such as AUC3–6, and AUC6-inf.
The formulations considered in this model all provided for continuous release. Formulations with untraditional designs, such as those which sequester the drug and provide for rapid release, may also be considered by a similar modeling approach.
Variability of AUC0−1.5 Higher than Other PK Parameters but Reasonable for Use in Equivalence Test
As expected, the discrimination of partial AUCs decreased with the increase in partial AUC truncation time (Fig. 8). Most studies had T/R ratios for AUC0–1.5 outside the range of 0.8–1.25. However, all studies had T/R ratios for AUC0-t within the range of 0.8–1.25. The residual variability (% CV) also decreased with the increasing value of AUC (Fig. 9). The residual variability in the crossover studies is the best available estimate of the within-subject variability. In the IR ANDAs, the % CV for the partial AUC ranged from 16% to 53% with a mean of 32.5%. The % CV in Ambien CR ANDAs ranged from 18% to 61% with a mean of 39.4% (Fig. 9). The variabilities of AUC and Cmax are less than that of AUC0–1.5 for all products shown in Figs. 8 and 9. For highly variable products (CV ≥ 30%), the number of subjects needed can be reduced by using the reference-scaled method recommended for highly variable drugs that have been recently proposed (14–16). For example, Haidar, et al. (15) showed that even at 60% CV, study power greater than 90% could be achieved using the reference-scaled approach with 36 subjects.
Relationship between the AUC T/R ratios with various the cutting off truncation time points for partial AUCs in bioequivalence studies comparing of the 10 tTest formulations of zolpidem ER to the reference product (ANDA vs. RLD) and in normalized dose proportionality studies and bioequivalence studies comparing formulations from the same manufacturer (RLD vs. RLD). RLD: Reference Listed Drug (Ambien CR).
DISCUSSION
The Guidance on Zolpidem Extended Release Tablet recommends bioequivalence testing based on two partial AUC measures, AUC0–1.5 and AUC1.5-t, in addition to the traditional PK parameters of AUCinf and Cmax. These pAUC metrics were recommended to ensure that a generic version of Ambien CR® will produce the same therapeutic effect per label indications. Modeling and simulation studies were an integral part of the decision making process for recommendations for the zolpidem Guidance.
Insufficiency of AUC and Cmax in Determining Bioequivalence
Using the CAT model, test products were simulated with different values of a and b, reflecting the differing array of possible zolpidem release rates in proposed generic products. Simulations of the test product showed that formulations with significantly different Weibull parameters could be deemed bioequivalent to Ambien CR when evaluated by AUC and Cmax.
The results of the simulations were verified using in-house data from generic drug applications. As anticipated, ANDA formulations exhibiting differing formulation designs and differing release profiles from Ambien CR gave rise to varying in vivo plasma concentration profiles. For example, ANDA formulations with a predominantly IR component gave rise to zolpidem in vivo drug plasma concentration profiles shifted to the left when compared with Ambien CR. Conversely, ANDA formulations with only an ER component in their formulation design gave rise to zolpidem in vivo drug plasma concentration profiles shifted to the right when compared with Ambien CR. This was verified by the observed differences in Tmax, amongst the various formulations. While the median Tmax of Ambien CR is approximately 1.5 h, the median Tmax of generic formulations varied from as early as 0.5 h (for formulations having a predominantly IR component) to as late as 3.0 h (for formulations having a predominantly ER component). The in vivo differences in Tmax and shape of the PK profiles raised concerns that some of the potential generic products may not provide the same therapeutic effect as the RLD. Nevertheless, all ANDAs were bioequivalent to the PK parameters of AUC and Cmax.
The poor discriminatory power of AUC was also demonstrated in a published study that showed that Ambien IR vs. Ambien CR had 90% conference intervals within 80–125% (8). The study was conducted as a three-way crossover study with intravenously administered zolpidem serving as a reference. The absolute bioavailabilities of Ambien IR and CR determined from the study were similar (66% and 68%, respectively). This indicates that a formulation which has release properties similar to Ambien IR may be deemed bioequivalent to Ambien CR in terms of AUC.
Early Partial AUC Measure
Ambien IR and CR have almost identical absorption time profiles from 0–1.5 h, indicating that the amount absorbed in vivo is similar for the IR and CR products at early times. In addition, all approved generics for Ambien IR meet the 90% confidence interval statistical criterion for AUC0–1.5. This suggests that AUC0–1.5 is indicative of IR characteristics which relate to early exposure. Ambien IR is indicated for the short-term treatment of insomnia characterized by difficulties with sleep initiation. As most subjects fall asleep within 1.5 h after dosing with zolpidem, the AUC0–1.5 metric represents the therapeutic effect of sleep onset.
CAT model predictions showed that formulations with various IR:ER ratios that produced undesirable PD effects (such as residual post-awakening effects and psychomotor impairment) could be deemed bioequivalent to Ambien CR when evaluated by AUC and Cmax. However, these formulations were not bioequivalent with respect to AUC0–1.5. While the undesirable PD effects did not occur during the 0–1.5 h time period (they occurred at least 5 h post-dose), detecting differences in early exposure (AUC0–1.5) can distinguish formulations that may produce different PD effects at later times.
Late Partial AUC Measure
To ensure therapeutic equivalence with respect to sleep maintenance, AUC1.5h-t was recommended in place of AUC0-t. The modeling approach in predicting bioequivalence regions is useful for defining the space of possible zolpidem release rates that will result in generic zolpidem formulations that are equivalent to Ambien CR in AUC1.5-t, Cmax and AUC0–1.5 parameters. Once this possible space of zolpidem release rates is known, one can use this information to predict whether these formulations having these release rates, will be equivalent to Ambien CR with respect to sleep maintenance. Other parameters to represent sleep maintenance have been proposed, such as AUC3–6 and AUC6−∞ (9). However, as evidenced from the bioequivalence regions, the areas of failure in either AUC1.5-t, Cmax, or AUC0–1.5 already encompass the failure regions for AUC3–6 and AUC6−∞. From the modeling analysis, one can conclude that requiring that generic zolpidem formulations be equivalent to Ambien CR in AUC1.5-t, Cmax and AUC0–1.5 parameters will result in a generic zolpidem CR product that is equivalent to Ambien CR with respect to the second sustained release phase of zolpidem, including other late pAUC parameters.
Results from the simulations indicate that only a small range of zolpidem release rates can yield a product that is bioequivalent to Ambien CR in all PK parameters, including the two pAUCs. Limiting the bioequivalence region to a select range of release rates is desirable, however, to ensure therapeutic equivalence. Expanding the bioequivalence region to include a wider range of release rates may permit formulations such as C and G (development formulations of Ambien CR which produced undesirable PD effects) to be deemed bioequivalent to the reference product.
CONCLUSIONS
The decision to include pAUC measures for bioequivalence assessment of zolpidem ER tablets was reached after thorough investigation and review by the FDA. Modeling and simulation studies played an important role in understanding the need for additional BE metrics and in the proper selection of truncation times for pAUC measures. Due to the close link between the drug concentration profile and clinical effect, FDA’s recommended pAUC measures are necessary to ensure the therapeutic equivalence of potential generics to Ambien CR.
MR drug products are designed to release drug in a controlled manner to achieve a particular efficacy and safety profile. With the increase in development of MR formulations, it is anticipated that consideration of additional measures of BE (such as pAUC), will be relevant to future generic products referencing brand products that employ complex release technologies. As a result, improved modeling and simulation approaches for predicting the therapeutic profile of MR products are expected to increasingly contribute to regulatory decisions concerning when a pAUC would be appropriate for use as a BE measure.
For MR products that exhibit a link between drug concentration and effect, modeling and simulation methods could be helpful in investigating the PK/PD relationship. Such methods can be employed as a tool to better understand the PK/PD relationship and to determine whether additional BE metrics (such as a pAUC) would be needed to ensure therapeutic equivalence.
REFERENCES
FDA. Guidance on Zolpidem Extended Release Tablet. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM175029.pdf.
FDA/CDER. Guidance for industry: bioavailability and bioequivalence studies for orally administered drug products - general considerations.
Kondra PM, Endrenyi L, Tothfalusi L. The need for additional metrics to assess therapeutic equivalence of some multiphasic modified-release products. Clin Ther. 2011;33:1214–9.
Chen ML, Shah VP, Ganes D, Midha KK, Caro J, Nambiar P, Rocci Jr ML, Thombre AG, Abrahamsson B, Conner D, Davit B, Fackler P, Farrell C, Gupta S, Katz R, Mehta M, Preskorn SH, Sanderink G, Stavchansky S, Temple R, Wang Y, Winkle H, Yu L. Challenges and opportunities in establishing scientific and regulatory standards for determining therapeutic equivalence of modified-release products: workshop summary report. Clin Ther. 2010;32:1704–12.
FDA. Briefing Information for the April 13, 2010 Meeting of the Pharmaceutical Science and Clinical Pharmacology Advisory Committee. http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/AdvisoryCommitteeforPharmaceuticalScienceandClinicalPharmacology/UCM209320.pdf.
FDA approved label for Ambien CR®. http://www.accessdata.fda.gov/drugsatfda_docs/label/2010/021774s010lbl.pdf.
Citizen Petition, Ambien CR (2007-P-0182). http://www.regulations.gov/#!documentDetail;D=FDA-2007-P-0182-0017.
Weinling E, McDougall S, Andre F, Bianchetti G, Dubruc C. Pharmacokinetic profile of a new modified release formulation of zolpidem designed to improve sleep maintenance. Fundam Clin Pharmacol. 2006;20:397–403.
Yu LX, Amidon GL. A compartmental absorption and transit model for estimating oral drug absorption. Int J Pharm. 1999;186:119–25.
Huang W, Lee SL, Yu LX. Mechanistic approaches to predicting oral drug absorption. AAPS J. 2009;11:217–24.
Zhang X, Lionberger RA, Davit BM, Yu LX. Utility of physiologically based absorption modeling in implementing Quality by Design in drug development. AAPS J. 2011;13:59–71.
Davit BM, Nwakama PE, Buehler GJ, Conner DP, Haidar SH, Patel DT, Yang Y, Yu LX, Woodcock J. Comparing generic and innovator drugs: a review of 12 years of bioequivalence data from the United States Food and Drug Administration. Ann Pharmacother. 2009;43:1583–97.
FDA/CDER. Guidance for Industry: Extended Release Oral Dosage Forms: Development, Evaluation, and Application of In Vitro/In Vivo Correlations; 1997.
Haidar SH, Davit B, Chen ML, Conner D, Lee L, Li QH, Lionberger R, Makhlouf F, Patel D, Schuirmann DJ, Yu LX. Bioequivalence approaches for highly variable drugs and drug products. Pharm Res. 2008;25:237–41.
Haidar SH, Makhlouf F, Schuirmann DJ, Hyslop T, Davit B, Conner D, Yu LX. Evaluation of a scaling approach for the bioequivalence of highly variable drugs. AAPS J. 2008;10:450–4.
Davit BM, Conner DP, Fabian-Fritsch B, Haidar SH, Jiang X, Patel DT, Seo PR, Suh K, Thompson CL, Yu LX. Highly variable drugs: observations from bioequivalence data submitted to the FDA for new generic drug applications. AAPS J. 2008;10:148–56.
ACKNOWLEDGMENTS & DISCLOSURES
This views presented in this article by the authors do not necessarily reflect those of the Food and Drug Administration (FDA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lionberger, R.A., Raw, A.S., Kim, S.H. et al. Use of Partial AUC to Demonstrate Bioequivalence of Zolpidem Tartrate Extended Release Formulations. Pharm Res 29, 1110–1120 (2012). https://doi.org/10.1007/s11095-011-0662-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-011-0662-8