Abstract
Purpose.
To determine the optimal polyethylene glycol (PEG)-conjugate of octreotide by evaluating the effects of PEGylation chemistry on the biological activity and pharmacokinetic properties.
Methods.
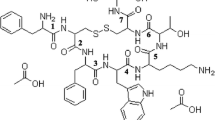
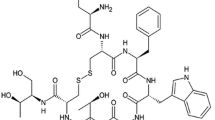
Octreotide was chemically modified by reaction with succinimidyl propionate monomethoxy PEG (SPA-mPEG, molecular weight 2000) or succinimidyl butyraldehyde-mPEG (ALD-mPEG, molecular weight 2000 and 5000). The structural conformation of PEG-octreotides was evaluated by circular dichroism (CD), the biological activity was assessed by measuring the decrease of serum insulin-like growth factor-I levels in rats, and a pharmacokinetic study was performed after subcutaneous administration in rats. The stability against acylation was investigated with poly(d,l-lactide-co-glycolide) (PLGA).
Results.
ALD-mPEG was site-specific in PEGylating octreotide at the N-terminus. The mono-PEG-octreotides prepared with ALD-mPEG (mono-ALDPEG-octreotide), which alkyl bond preserves the amine’s positive charge, showed complete preservation of biological activity, whereas the PEG-octreotides prepared with SPA-mPEG showed lower activity. In the CD analysis, the spectra of the mono-ALDPEG-octreotides were nearly superimposable with that of native octreotide. The mono-ALDPEG-5K-octreotide showed significantly improved pharmacokinetic properties compared with mono-ALDPEG-2K-octreotide as well as native octreotide. Both mono-ALDPEG-2K- and mono-ALDPEG-5K-octreotides were stable against acylation by degrading PLGA.
Conclusions.
The mono-PEGylation of octreotide at N-terminus with ALD-mPEG produced a conjugate that is biologically and structurally active and stable against acylation by PLGA, and therefore it may serve as a candidate for somatostatin microsphere formulations.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
1. P. Marbach, M. Neufeld, and J. Pless. Clinical applications of somatostatin analogues. Adv. Exp. Med. 188:339–353 (1985).
2. S. W. Lamberts, A. J. van der Lely, W. W. de Herder, and L. J. Hofland. Octreotide. N. Engl. J. Med. 334:246–254 (1996).
3. S. J. Hunter, J. A. Shaw, K. O. Lee, P. J. Wood, A. B. Atkinson, and J. S. Bevan. Comparison of monthly intramuscular injections of Sandostatin LAR with multiple subcutaneous injections of octreotide in the treatment of acromegaly; effects on growth hormone and other markers of growth hormone secretion. Clin. Endocrinol. (Oxf.) 50:245–251 (1999).
4. M. van de Weert, W. E. Hennink, and W. Jiskoot. Protein instability in poly(lactic-co-glycolic acid) microparticles. Pharm. Res. 17:1159–1167 (2000).
5. A. Brunner, K. Mäder, and A. Göpferich. pH and osmotic pressure inside biodegradable microspheres during erosion. Pharm. Res. 16:847–853 (1999).
6. K. Fu, D. W. Pack, A. M. Klibanov, and R. Langer. Visual evidence of acidic environment within degrading poly(lactic-co-glycolic acid) (PLGA) microspheres. Pharm. Res. 17:100–106 (2000).
7. A. Lucke, J. Kiermaier, and A. Göpferich. Peptide acylation by poly(α-hydroxy esters). Pharm. Res. 19:175–181 (2002).
8. A. Lucke, E. Fustella, J. Teßmar, A. Gazzaniga, and A. Göpferich. The effect of poly(ethylene glycol)-poly(D,L-lactic acid) diblock copolymers on peptide acylation. J. Control. Rel. 80:157–168 (2002).
9. D. H. Na, Y. S. Youn, S. D. Lee, M. W. Son, W. B. Kim, P. P. DeLuca, and K. C. Lee. Monitoring of peptide acylation inside degrading PLGA microspheres by capillary electrophoresis and MALDI-TOF mass spectrometry. J. Control. Rel. 92:291–299 (2003).
10. S. B. Murty, J. Goodman, B. C. Thanoo, and P. P. DeLuca. Identification of chemically modified peptide from poly(D,L-lactide-co-glycolide) microspheres under in vitro release conditions. AAPS PharmSciTech. 4:article 50 (2003).
11. K. C. Lee, S. C. Moon, M. O. Park, J. T. Lee, D. H. Na, S. D. Yoo, H. S. Lee, and P. P. DeLuca. Isolation, characterization and stability of positional isomers of mono-PEGylated salmon calcitonins. Pharm. Res. 16:818–823 (1999).
12. D. H. Na, Y. S. Youn, E. J. Park, J. M. Lee, O. R. Cho, K. R. Lee, S. D. Lee, S. D. Yoo, P. P. DeLuca, and K. C. Lee. Stability of PEGylated salmon calcitonin in nasal mucosa. J. Pharm. Sci. 93:256–261 (2004).
13. D. H. Na, S. B. Murty, K. C. Lee, B. C. Thanoo, and P. P. DeLuca. Preparation and stability of PEGylated octreotide for application to microsphere delivery. AAPS PharmSciTech. 4:article 72 (2003).
14. D. H. Na and P. P. DeLuca. PEGylation of octreotide: I. Separation of positional isomers and stability against acylation by poly(d,l-lactide-co-glycolide). Pharm. Res. 22:736–742 (2005).
15. M. J. Roberts, M. D. Bentley, and J. M. Harris. Chemistry for peptide and protein PEGylation. Adv. Drug Deliv. Rev. 54:459–476 (2002).
16. M. Morpurgo, C. Monfardini, L. J. Hofland, M. Sergi, P. Orsolini, J. M. Dumont, and F. M. Veronese. Selective alkylation and acylation of alpha and epsilon amino groups with PEG in a somatostatin analogue: tailored chemistry for optimized bioconjugates. Bioconjug. Chem. 13:1238–1243 (2002).
17. O. B. Kinstler, D. N. Brems, S. L. Lauren, A. G. Paige, J. B. Hamburger, and M. J. Treuheit. Characterization and stability of N-terminally PEGylated rhG-CSF. Pharm. Res. 13:996–1002 (1996).
18. D. H. Na, M. O. Park, S. Y. Choi, Y. S. Kim, S. S. Lee, S. D. Yoo, H. S. Lee, and K. C. Lee. Identification of the modifying sites of mono-PEGylated salmon calcitonins by capillary electrophoresis and MALDI-TOF mass spectrometry. J. Chromatogr. B 754:259–263 (2001).
19. D. H. Na and K. C. Lee. Capillary electrophoretic characterization of PEGylated human parathyroid hormone with matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal. Biochem. 331:322–328 (2004).
20. D. H. Na, Y. S. Youn, and K. C. Lee. Optimization of the PEGylation process of a peptide by monitoring with matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 17:2241–2244 (2003).
21. S. S. Wong. Chemistry of Protein Conjugation and Cross-linking, CRC Press, Boca Raton, FL, 1991.
22. S. G. Chang, K. D. Choi, S. H. Jang, and H. C. Shin. Role of disulfide bonds in the structure and activity of human insulin. Mol. Cells 16:323–330 (2003).
23. E. Pohl, A. Heine, and G. M. Sheldrick. Structure of octreotide, a somatostatin analogue. Acta Crystallogr. D51:48–59 (1995).
24. G. Brabant. Insulin-like growth factor-I: marker for diagnosis of acromegaly and monitoring the efficacy of treatment. Eur. J. Endocrinol. 148:S15–S20 (2003).
25. G. Weckbecker, U. Briner, I. Lewis, and C. Bruns. SOM230: A new somatostatin peptidomimetic with potent inhibitory effects on the growth hormone/insulin-like growth factor-I axis in rats, primates, and dogs. Endocrinology 143:4123–4130 (2002).
26. P. Caliceti and F. M. Veronese. Pharmacokinetic and biodistribution properties of poly(ethylene glycol)-protein conjugates. Adv. Drug Deliv. Rev. 55:1261–1277 (2003).
27. M. J. Knauf, D. P. Bell, P. Hirtzer, Z. P. Luo, J. D. Young, and N. V. Katre. Relationship of effective molecular size to systemic clearance in rats of recombinant interleukin-2 chemically modified with water-soluble polymers. J. Biol. Chem. 263:15064–15070 (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Na, D., Lee, K. & DeLuca, P. PEGylation of Octreotide: II. Effect of N-terminal Mono-PEGylation on Biological Activity and Pharmacokinetics. Pharm Res 22, 743–749 (2005). https://doi.org/10.1007/s11095-005-2590-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-2590-y