No Heading
Purpose.
The effects and mechanisms of different antioxidants, methionine, glutathione, acetylcysteine, and ascorbic acid (AscH2), on the oxidation of methionine residues in granulocyte colony-stimulating factor (G-CSF) and human parathyroid hormone fragment 13-34 (hPTH 13-34) by hydrogen peroxide (H2O2) were quantified and analyzed.
Methods.
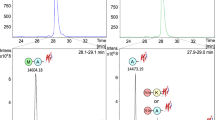
The rates of oxidation of methionine residues in G-CSF were determined by peptide mapping analyses, and the oxidation of methionine residue in hPTH 13-34 was quantified by reverse-phase HPLC.
Results.
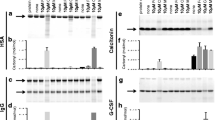
At pH 4.5, free methionine reduces, glutathione and acetylcysteine have no obvious effect on, and AscH2 promotes the rates of oxidation of methionine residues in G-CSF. The H2O2-induced oxidation rate constants for free methionine, acetylcysteine, and glutathione at pH 4.5 were measured to be 32.07, 1.00, and 1.63 M-1h-1, respectively, while the oxidation rate constant for Met1, the most readily oxidizable methionine residue in G-CSF, is 13.95 M−1h−1. Therefore, the different effects of free methionine, acetylcysteine, and glutathione on the rates of oxidation of methionine residues in G-CSF are consistent with their different reactivity toward oxidation by H2O2. By using hPTH 13-34, the effect of AscH2 on the H2O2-induced oxidation of methionine residue was quantified, and the mechanisms involved were proposed. Because of the presence of trace transition metal ions in solution, at low concentrations, AscH2 is prone to be a prooxidant, increasing the hydroxyl radical (⋅OH) production rate via Fenton-type reactions. In addition to peroxide oxidation, these radicals lead to the degradation of hPTH 13-34 to smaller peptide fragments. At high concentrations, AscH2 tends to act as an ⋅OH scavenger. EDTA inhibits ⋅OH production and thus eliminates the degradation of hPTH 13-34 by forming complexes with transition metal ions. However, the rate of oxidation of the methionine residue in hPTH 13-34 increases as the concentration of AscH2 is increased from 0 to 200 mM, and the reason for this is still not clear.
Conclusions.
Our results demonstrate that free methionine is an effective antioxidant to protect G-CSF against methionine oxidation at pH 4.5. Acetylcysteine and glutathione are not effective antioxidants at pH 4.5. Their oxidation rates at different pH values imply that they would be much more effective antioxidants than free methionine at alkaline conditions. AscH2 is a powerful electron donor. It acts as a prooxidant in the conditions in this study and is unlikely to prevent oxidation by H2O2 in protein formulation, whether or not EDTA is present.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
1. S. H. Li, C. Schöneich, and R. T. Borchardt. Chemical-instability of protein pharmaceuticals - mechanisms of oxidation and strategies for stabilization. Biotechnol. Bioeng. 48:490–500 (1995).
2. W. Wang. Instability, stabilization, and formulation of liquid protein pharmaceuticals. Int. J. Pharm. 185:129–188 (1999).
3. Y. Nabuchi, E. Fujiwara, K. Ueno, H. Kuboniwa, Y. Asoh, and H. Ushio. Oxidation of recombinant human parathyroid hormone: Effect of oxidized position on the biological activity. Pharm. Res. 12:2049–2052 (1995).
4. S. W. Griffiths and C. L. Cooney. Relationship between protein structure and methionine oxidation in recombinant human alpha 1-antitrypsin. Biochemistry 41:6245–6252 (2002).
5. S. M. Van Patten, E. Hanson, R. Bernasconi, K. Zhang, P. Manavalan, E. S. Cole, J. M. McPherson, and T. Edmunds. Oxidation of methionine residues in antithrombin - Effects on biological activity and heparin binding. J. Biol. Chem. 274:10268–10276 (1999).
6. E. T. Duenas, R. Keck, A. De Vos, A. J. S. Jones, and J. L. Cleland. Comparison between light induced and chemically induced oxidation of rhVEGF. Pharm. Res. 18:1455–1460 (2001).
7. J. L. Liu, K. V. Lu, T. Eris, V. Katta, K. R. Westcott, L. O. Narhi, and H. S. Lu. In vitro methionine oxidation of recombinant human leptin. Pharm. Res. 15:632–640 (1998).
8. H. S. Lu, P. R. Fausset, L. O. Narhi, T. Horan, K. Shinagawa, G. Shimamoto, and T. C. Boone. Chemical modification and site-directed mutagenesis of methionine residues in recombinant human granulocyte colony-stimulating factor: effect on stability and biological activity. Arch. Biochem. Biophys. 362:1–11 (1999).
9. J. L. Cleland and R. Langer. Formulation and delivery of proteins and peptides - design and development strategies. ACS Sym. Ser 567:1–19 (1994).
10. T. Osterberg and A. Fatouros. Oxygen-reduced aqueous solution of factor VIII. U.S. Patent No. 5,962,650 (1999)
11. T. Osterberg and A. Fatouros. Protein formulation comprising coagulation factor VIII or factor IX in an aqueous solution. U.S. Patent No. 5,919,908 (1999)
12. S. H. Li, C. Schöneich, and R. T. Borchardt. Chemical pathways of peptide degradation. VIII. Oxidation of methionine in small model peptides by prooxidant/transition metal ion systems: influence of selective scavengers for reactive oxygen intermediates. Pharm. Res. 12:348–355 (1995).
13. V. M. Knepp, J. L. Whatley, A. Muchnik, and T. S. Calderwood. Identification of antioxidants for prevention of peroxide-mediated oxidation of recombinant human ciliary neurotrophic factor and recombinant human nerve growth factor. PDA J. Pharm. Sci. Technol. 50(3):163–171 (1996).
14. X. M. Lam, J. Y. Yang, and J. L. Cleland. Antioxidants for prevention of methionine oxidation in recombinant monoclonal antibody HER2. J. Pharm. Sci. 86:1250–1255 (1997).
15. J. A. Cadee, M. J. van Steenbergen, C. Versluis, A. J. R. Heck, W. J. M. Underberg, W. den Otter, W. Jiskoot, and W. E. Hennink. Oxidation of recombinant human interleukin-2 by potassium peroxodisulfate. Pharm. Res. 18:1461–1467 (2001).
16. S. H. Li, C. Schöneich, G. S. Wilson, and R. T. Borchardt. Chemical pathways of peptide degradation. V. ascorbic-acid promotes rather than inhibits the oxidation of methionine to methionine sulfoxide in small model peptides. Pharm. Res. 10:1572–1579 (1993).
17. J. C. Deutsh. Ascorbic acid oxidation of hydrogen peroxide. Anal. Biochem. 255:1–7 (1998).
18. B. Bishop, D. C. Koay, A. C. Sartorelli, and L. Regan. Reengineering granulocyte colony-stimulating factor for enhanced stability. J. Biol. Chem. 276:33465–33470 (2001).
19. J.-W. Chu, J. Yin, D. I. C. Wang, and B. L. Trout. Molecular dynamics and oxidation rates of methionine residues of granulocyte colony-stimulating factor (G-CSF) at different pH values. Biochemistry 43:1019–1029 (2004).
20. A. L. Frelinger and J. E. Zull. Oxidized forms of parathyroid-hormone with biological-activity - separation and characterization of hormone forms oxidized at methionine-8 and methionine-18. J. Biol. Chem. 259:5507–5513 (1984).
21. Available at: http://www.rxlist.com/cgi/generic/filgrastim.htm (accessed 4/27/2004).
22. S. P. Wolff. Ferrous ion oxidation in presence of ferric ion indicator xylenol orange for measurement of hydroperoxides. Method. Enzymol. 233:182–189 (1994).
23. C. C. Winterbourn and D. Metodiewa. Reactivity of biologically important thiol compounds with superoxide and hydrogen peroxide. Free Radical Bio. Med. 27:322–328 (1999).
24. J. C. Deutsh. Oxygen-accepting antioxidants which arise during ascorbate oxidation. Anal. Biochem. 265:238–245 (1998).
25. J. C. Deutsh. Dehydroascorbic acid. J. Chromatogr. A. 881:299–307 (2000).
26. P. Wardman and L. P. Candeias. Fenton chemistry: an introduction. Radiat. Res. 145:523–531 (1996).
27. M. J. Burkitt and B. C. Gilbert. Model studies of the iron-catalysed Haber-Weiss cycle and the ascorbate-driven Fenton reation. Free Radical Res. Com. 10:265–280 (1990).
28. M. J. Zhao and L. Jung. Kinetics of the competitive degradation of deoxyribose and other molecules by hydroxyl radicals produced by the Fenton reaction in the presence of ascorbic-acid. Free Radic. Res. 23:229–243 (1995).
29. G. R. Buettner and B. A. Jurkiewicz. Catalytic metals, ascorbate and free radicals: combinations to avoid. Radiat. Res. 145:532–541 (1996).
30. J. E. Biaglow, Y Manevich, F. Uckun, and K.D. Held. Quantitation of hydroxyl radicals produced by radiation and copper-linked oxidation of ascorbate by 2-deoxy-D-ribose method. Free Radical Bio. Med. 22:1129–1138 (1997).
31. A. Rees and T. F. Slater. Ascorbic acid and lipid peroxidation: the cross-over effect. Acta Biochim. Biophys. 22:241–249 (1987).
32. L. Packer and J. Fuchs. Health and Disease, Marcel Dekker Inc., New York, 1997, pp. 75–94
33. G. R. Buettner. In the absence of catalytic metals ascorbate does not autoxidize at pH 7: ascorbate as a test for catalytic metals. J. Biochem. Biophys. Methods 16:27–40 (1988).
34. G. R. Buettner, T. P. Doherty, and L. K. Patterson. The kinetics of the reaction of superoxide radical with Fe(III) complexes of EDTA, DETAPAC and HEDTA. FEBS Lett. 158:143–146 (1983).
35. J. Butler and B. Halliwell. Reaction of iron EDTA chelates with the superoxide radical. Arch. Biochem. Biophys. 218:174–178 (1982).
36. K. D. Welch, T. Z. Davis, and S. D. Aust. Iron autoxidation and free radical generation: effects of buffers, ligands, and chelators. Arch. Biochem. Biophys. 397:360–369 (2002).
37. J. Van der Zee and P. J. A. Van den Broek. Determination of the ascorbate free radical concentration in mixtures of ascorbate and dehydroascorbate. Free Radical Bio. Med. 25:282–286 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, J., Chu, JW., Ricci, M. et al. Effects of Antioxidants on the Hydrogen Peroxide-Mediated Oxidation of Methionine Residues in Granulocyte Colony-Stimulating Factor and Human Parathyroid Hormone Fragment 13-34. Pharm Res 21, 2377–2383 (2004). https://doi.org/10.1007/s11095-004-7692-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-004-7692-4