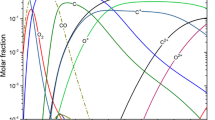
The composition and transport properties of CO2, CO, CH4, CO + Ar (50 vol%), CO + Fe (50 vol%) have been calculated at constant volume assuming local thermodynamic equilibrium (LTE). Except at low temperature (T < 3000 K), when the formation of condensed species or more complex molecules can occur, pressure increases with temperature at constant volume. For example, for 1 mol of CH4 starting at 0.1 MPa and 298 K the pressure can reach 40 MPa at 20,000 K. The consequence is a shift to higher temperatures of dissociation and ionization. The electrical conductivity σ e at constant volume increases drastically relative to that obtained at 0.1 MPa over 15,000 K, in spite of the decrease of the electron density n e This is due to the increase in the neutral species density. n i , with a much lower electron-neutral species collision cross section σ ei (σ e is inversely proportional to n i ,σ ei ). The viscosity always exhibits a maximum when the ionization degree increases over 1–30%, but this maximum is shifted to higher temperatures and its peak value is higher. The thermal conductivity peaks due to dissociation and ionization are shifted to higher temperatures and their values are reduced compared to those obtained at constant pressure.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D. Neuschütz (1998) J. High Temp. Mater. Process 2 261
D. Neuschütz (2000) J. High Temp. Mater. Process 5 IssueID1 127
P. Fauchais M.F. Elchinger J. Aubreton (2000) J. High Temp. Mater. Process 5 IssueID1 21
W.B. White S.M. Johnson G.B. et Dantzig (1958) J. Chem. Phys 28 IssueID5 751 Occurrence Handle10.1063/1.1744264
Hirschfelder J.O., Curtiss C.F., and Byron B.R., Molecular Theory of Gases and Liquids. (Pub.) John Wiley and Sons, second printing, New York 1964, pp. 151, 163, 209–219, 949–950, 988, 1147–1151, 1114–1115.
R.C. Reid J.M. Prausnitz B.E. Poling (1987) The Properties of Gases & Liquids EditionNumber4 (Pub) McGraw-Hill Book Company New York 658–664
Allen C.W., Astrophysical Quantities, (Pub.) Athlone Press, 1964.
Handbook of Chemistry and Physics, 67th Edition, (Pub.) CRC-Press, 1997–1998.
Benjamin C., Calculation of Transport Properties and Study of the Switching Effi-ciency of the Mixture SF6-CF4 or C2F6 and SF6–Cu Vapor (in French), PhD thesis, University Paul Sabatier, Toulouse III, No. 1997, 28/03/1995.
Koalaga Z., Contribution to the Experimental and Theoretical Study of Laminated Plasma Produced by d.c. Arcs (in French), PhD thesis, Clermont-Ferrand University, France, 1991.
L.V. Gurvich et al., Thermodynamic Properties of Individual Substances, (Pub.) Hemisphere, NY 4th 1996.
M. Chase et al., JANAF Thermochem. Tables, 3rd edn. J. Phys. Chem. Ref. Data, 1985. 14 (Supp. 1).
I. Barin (1993) Thermochemical Data of Pure Substances Springer Verlag Berlin
B. Pateyron M.F. Elchinger G. Delluc P. Fauchais (1992) Plasma Chem. Plasma Processing 12 IssueID4 421 Occurrence Handle10.1007/BF01447253
S. Chapman T. Cowling (1952) The Mathematical Theory of Non Uniform Gases Cambridge University Press New York
R.S. Devoto (1973) Phys. Fluids 16 616 Occurrence Handle10.1063/1.1694396
J. N. Butler R.S. Brokaw (1957) J. Chem. Phys 26 1636 Occurrence Handle10.1063/1.1743596
F. J. Smith R.J. Munn (1964) J. Chem. Phys 41 3560 Occurrence Handle10.1063/1.1725768
R.S. Brokaw (1961) Phys. Fluids 8 944 Occurrence Handle10.1063/1.1706443
L. Moonchick (1959) Phys. Fluids 2 695 Occurrence Handle10.1063/1.1705974
T. Kihara M.H. Taylor J.O. Hirschfelder (1960) Phys. Fluids 3 IssueID5 715 Occurrence Handle10.1063/1.1706115
J. Aubreton (1985) Study of the Thermodynamic and Transport Properties in Equilibrium and Non Equilibrium Thermal Plasmas: Application to Ar-H2 et Ar-O2 Mixtures (in French), State thesis Limoges University France
Y. Itikawa (1974) Data Nucl. Data Tables 14 1
Y. Itikawa (1978) Data Nucl. Data Tables 21 69 Occurrence Handle10.1016/0092-640X(78)90004-9
E.A. Mason R.J. Munn F.J. Smith (1967) Phys. Fluids 10 IssueID8 1827 Occurrence Handle10.1063/1.1762365
Gallagher J.W., JILA Data Center, Campus Box 440, Colorado University, Boulder, Colorado 80309, USA.(Also ftp://jila.colorado.edu/collision data/electron.txt).
M.F. Elchinger B. Pateyron G. Delluc P. Fauchais (1990) J. de Physique colloque C5 51 IssueID185 C5.3
M.I. Boulos P. Fauchais E. Pfender (1994) Thermal Plasmas NumberInSeries1 (Pub.) Plenum Press NY
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pateyron, B., Delluc, G. & Fauchais, P. Chemical and Transport Properties of Carbon–Oxygen–Hydrogen Plasmas in Isochoric Conditions. Plasma Chem Plasma Process 25, 485–502 (2005). https://doi.org/10.1007/s11090-005-4994-1
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11090-005-4994-1