Abstract
A lifecourse perspective is key for understanding and interpreting racial and ethnic patterns in neuropsychological test performance. In this article, we discuss contextual factors that shape the environmental conditions encountered by racial and ethnic minorities in the United States, in particular African-Americans. These conditions include geographic segregation at the level of regions, metropolitan areas, and neighborhoods; intra- and inter-national migration patterns; socioeconomic position, including financial resources, and occupational and educational opportunities; discrimination; and group resources. Each of these exposures sets in course a cascade of individual mediators that ultimately manifest in neuropsychological outcomes. The physiological and behavioral consequences of these pathways likely accumulate across the lifecourse. We focus on cognitive aging, although the processes discussed here begin in infancy and likely influence cognitive outcomes throughout life from childhood into old age. A lifecourse framework can help inform clinical encounters, neuropsychological research, and surveillance regarding the population prevalence of cognitive impairments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Performance on neuropsychological tests is shaped by environmental experiences across the lifecourse, from the prenatal period to old age. Although adult social circumstances, e.g., financial resources or occupational hazards, have perhaps received more attention in neuropsychological research, the effects of early life environmental conditions on cognitive aging are increasingly recognized. This evidence has great relevance for neuropsychologists in both clinical and research settings. Recognizing the social context and the social patterning of deleterious exposures is valuable for interpreting racial and ethnic differences in cognitive test performance, for uncovering the causes of patterns in the prevalence and incidence of dementia across racial groups, and for predicting the future burden of illness as the population ages. Considering only current risk-factor status probably vastly understates the differences in exposure across racial and ethnic groups. In order to identify the most promising approaches to reducing racial disparities, it is valuable to recognize that physical and likely neurocognitive health may have already begun to diverge by childhood. Early life conditions may become physiologically embedded in ways that directly influence old-age cognitive risk or change the trajectory of exposures experienced later in life and thus exert indirect influence. These processes should be considered when assessing risk for individual patients or populations.
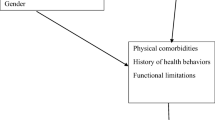
In this article we focus primarily on US blacks, with some discussion of evidence on Hispanics/Latinos and Asians, and consider how the lifecourse experiences of current cohorts of elderly minority populations may influence performance on neuropsychological tests. Figure 1 illustrates our conception of the primary mediators in this process, moving from very broad social patterns such as migration and concluding with individual level mediators, including physical health (e.g., cardiovascular disease) and health behaviors (e.g., exercise). We distinguish between cognitive outcomes and diagnoses of these outcomes because race/ethnicity may influence either. Measurement tools may operate differently for racial and ethnic minorities than for majority white groups, potentially resulting in either differential sensitivity or specificity. Issues relating specifically to measurement, and statistical approaches to eliminating differential item functioning, are discussed more fully in Pedraza and Mungas in this issue. We focus here on substantive social exposures that may lead to true disparities in neurocognitive outcomes, with some discussion of how these exposures may also compromise measurement.
In the first section, we introduce core concepts in lifecourse epidemiology and briefly review the rationale for believing that early life conditions are relevant to cognitive function, the development and maintenance of cognitive skills, cognitive aging and the onset and expression of neurodegenerative diseases. We then consider how racially patterned social exposures (termed socially patterned mediators in Fig. 1), including migration, socioeconomic position (SEP), and discrimination, may influence cognitive aging and are relevant to interpreting clinical findings. Drawing on Link and Phelan’s theory of fundamental social causes, we focus here on social conditions because of the power of social conditions and resources to set in motion a cascade of more proximal exposures (Link and Phelan 1996). We also briefly summarize evidence for the relevance of individual mediators hypothesized in Fig. 1, in particular cognitive engagement and stress. These constructs do not follow a strict hierarchy; the processes are dynamic and interactive. Our goal is to indicate how macro-level social conditions cascade down to individual level environments and resources which become manifest in individual behavior, individual exposures, medical care, physical health, and ultimately cognitive outcomes. We focus here primarily on cognitive aging, but many of the issues are relevant to understanding how social conditions may affect the onset or course of psychopathological conditions at any point in life. Cognitive function is a developmental trajectory, and harmful exposures may influence the likelihood of impairments in old age by derailing the maturation trajectory, promoting pathological processes, or restricting compensation or resilience after pathological events. All of these processes are relevant for understanding the determinants of cognitive disorders throughout life. We focus on social exposures and cognitive functioning in the elderly, in part for practical reasons. Elderly individuals have accumulated the effects of a lifetime of exposure and impairments are common, and thus it is often easier from a research perspective to observe the harm induced by the exposure of interest. On the other hand, an important disadvantage of using cognitive aging as a model is the difficulty of identifying the effects of specific exposures at specific points in the life course. Many of the exposures that influence cognitive aging presumably also increase the risk of cognitive disorders earlier in life. Better understanding of the relationship between early life conditions and cognitive function in specific domains, critical periods of influence, and valid measurement of early life exposures would facilitate testing many hypotheses regarding the links between social conditions and cognitive outcomes.
In many cases, the evidence on how lifecourse social exposures may influence cognitive aging is incomplete. For some exposures, there has been much more research on physical health outcomes, such as cardiovascular conditions. Although this is not a focus of this review, we must acknowledge prior work finding a strong link between cognitive health and physical health (Whitfield et al. 2000), especially vascular functioning. (Snowdon et al. 1997; Cerhan et al. 1998; Kilander et al. 1998; de la Torre 2000; Gregg et al. 2000; Hachinski and Munoz 2000; Hébert et al. 2000; Fontbonne et al. 2001) Recent work adopting a “brain at risk” approach highlights the value of considering neuropathological processes in the context of physical health, in particular cardiovascular health (Hachinski 2007), consistent with a broader lifecourse perspective on neurodegenerative diseases (Whalley et al. 2006). When appropriate, we therefore present the evidence for lifecourse effects on overall health and examine the implications of this work for cognitive outcomes.
Lifecourse Epidemiology Models and their Relevance to Understanding Racial Differences in Neuropsychological Test Performance
Much epidemiologic work focuses on either the influence of stable personal characteristics (e.g., genotype) or recent exposures (e.g., diet) on health outcomes. In the 1990s, a resurgence of research demonstrated that more complex etiologic pathways may not fit these simple temporal patterns. Exposures throughout life appear to influence both the incidence and course of chronic diseases, sometimes in ways that depend on the timing of exposure (Kuh and Ben-Shlomo 1997). Work by Barker and colleagues on the maternal and fetal origins of cardiovascular and other chronic diseases has provided a major stimulus for renewed attention to biological programming models (Barker and Martyn 1992). Although cognitive aging has not been a primary theme in lifecourse epidemiology, results from animal models (Meaney et al. 1988; Diamond et al. 1993; Meaney et al. 1996) and a growing group of human studies indicate that early life exposures and environmental conditions throughout life strongly influence cognitive risks in old age. The models elaborated for other diseases may therefore be useful for understanding lifecourse risk factors for cognitive decline and dementia.
Whalley et al. advocate a lifecourse approach to understand dementia and to disentangle genetic and environmental risk factors (Whalley et al. 2006). This framework may be crucial to understanding racial and ethnic disparities across a broad range of neuropsychological skills and impairments. Some possible models of how the timing of exposures relates to their disease-causing potential are listed below (Power and Hertzman 1997; Wadsworth 1997):
-
1.
Immediate risk models assume a very short etiologic period in which the exposure has instantaneous or short-term effects on the outcome. The key to this model is that once the risk factor is removed, risk declines, and possibly returns to the baseline (unexposed) level. For example, smoking cessation results in an almost immediate reduction in risk of cardiovascular events, and the risk declines to that of never-smokers after about 10–15 years. (US Department of Health and Human Services 2004) Delirium in hospitalized elderly is often a consequence of drug exposures and frequently resolves after changes in drug regimens, conforming to an immediate risk etiologic model (Gray et al. 1999).
-
2.
Cumulative biological models assume that each period of exposure to a risk factor wreaks some biological harm that increases risk of later developing illness, even if the individual is subsequently removed from exposure. For example, smoking increases risk of lung cancer, and the absolute risk does not decline after smoking cessation (although the risk does not increase as it would with continued smoking; US Department of Health and Human Services 2004). Similarly, lead exposure appears to have a cumulative biological effects on cognitive functioning in the elderly (Weisskopf et al. 2004). Embedded within cumulative processes, there may be critical events that alter an individual’s underlying biological vulnerability, for example early first birth may reduce breast cancer risk (Key et al. 2001). Geronimus attributed many racial and ethnic disparities in health to “weathering”, the accumulated consequences of exposure to economic and social adversity (Geronimus et al. 2006).
-
3.
Latency models apply if there is a window of time during which exposure to the risk factor puts one at risk for development of disease much later in life. Effects may not be manifest for years after exposure is removed. In a latency model, exposures encountered after the critical period ends do not influence risk. Latency models typically focus on early life experiences that do not have a cumulative effect. For example, Meaney has demonstrated that handling of rodent pups in the first weeks of life permanently alters the animals’ stress response, regardless of subsequent exposures (Meaney et al. 1985a, b, 1996). Fetal programming models, in which prenatal exposures change metabolic patterns or other physiologic characteristics, are another common example (Barker 1999; Leon 2001; Cutler et al. 2007).
-
4.
Social trajectory models describe factors whose effects are primarily mediated by a succession of harmful social exposures. Early deprivation may not cause immediate physical harm, but rather put one “at risk of risk.” For example, poverty early in life may reduce educational access, thereby increasing the likelihood of adult poverty, which may increase the risk of depression in late life and lead to diagnosis of cognitive impairment. Early life poverty did not directly induce a physiological change, but set in motion a trajectory of risk factors that changed risk much later in life. Such models may incorporate trajectory “elbow” or “lever” periods—moments when the direction of future social experiences can be more easily changed, either for better or worse. Potentially crucial points might be early schooling years or the period following widowhood or divorce. The important feature of trajectory models is that the early exposure must induce subsequent toxic exposures in order to harm later health.
These models are not mutually exclusive. Many exposures may affect cognitive disorders through multiple pathways, each fitting a different one of the above models.
Although lifecourse models were primarily developed in the context of research on cardiovascular diseases and cancer, there is substantial evidence indicating a lifecourse perspective is likely to be important in understanding cognitive aging. A number of studies show that various individual characteristics and exposures measured early in life predict cognitive outcomes in old age. These studies face many methodological challenges, but the strength and replicability of the relationships provides compelling evidence for the importance of lifecourse processes in shaping cognitive aging. As detailed in subsequent sections, parental or early life SEP, childhood IQ, measures of early growth, educational attainment, occupational characteristics, and various measures of social integration have all been linked to cognitive function and neurocognitive disorders in adulthood and old age. Furthermore, the breadth of evidence linking prenatal, childhood, and midlife exposures to cardiovascular functioning in old age suggests these exposures will also influence cognitive impairment and dementia via vascular mechanisms. (Lauer and Clark 1989; Barker 1990; Bao et al. 1995; Cheung et al. 2000; Godfrey and Barker 2000; Ravelli et al. 2000; Eriksson et al. 2001). These pathways are likely to play an important role in explaining racial disparities in neurocognitive conditions, because, as discussed below, social conditions for different racial and ethnic groups diverge very early in life in the US. Before considering evidence for proximal mechanisms linking early life conditions and cognitive aging, we review the ways in which lifecourse social experiences are distinctively patterned by racial and ethnic group.
Distal Socially Patterned Mediators
Racially Patterned Residential Histories
We begin by considering how the residential histories of elderly members of racial/ethnic minority groups in the United States (US) differ from those typical of elderly US-born whites. These patterns change across time and thus may differ for current cohorts of elderly compared to previous birth cohorts or cohorts yet to come. Residential and social segregation has been a persistent feature of life in the US, but the exact ways in which this segregation is manifested, and the likely consequences for cognitive function, may evolve over time.
The Great Migration and Regional Differences in Cognitive Test Performance
In the “Great Migration” that occurred between roughly 1910 and 1970, approximately three million African-Americans left the South for Northern, industrial cities such as New York, Chicago, and Detroit. The forces driving this exodus are thought to include flight from the segregation and violence that characterized the Jim Crow South; economic pulls to the North associated with the World Wars, especially after the 1924 immigration restrictions reduced the supply of low-cost labor from international migrants; and economic pushes as the sharecropper system in the South disintegrated with the invention of the mechanical cotton picker (Lemann 1991; Tolnay 2003).
Therefore, regardless of current residence, most elderly African Americans in the US were born and raised in the South. Table 1 shows the numbers and proportions of US blacks and whites cross-classified by Southern current residence and Southern birth based on the 2000 census. The experiences of sharecropper childhoods, education and early life in the Jim Crow South, and uprooting for migration to Northern industrial cities comprise essential elements of the early life social conditions for a large number of elderly African-Americans.
In general, migrants may differ from non-migrants for three reasons: the people who choose to move may differ from the non-movers (selection), the experience of dislocation itself may affect the well-being of the migrants, or the effects of exposure to the receiving community may differ from the effects of exposure to the native community. For African Americans who participated in the Great Migration, it is this last factor that is likely to be the most important.
Although evidence is somewhat mixed (Klineberg 1935), Tolnay used Public Use Microsample data from the Census to argue that that African-Americans who left the South were more likely to be literate and had higher education compared to those who remained behind; on the other hand, migrants had less education than the Northern-born African-Americans they joined (Tolnay 1998). The dislocation per se may not have had consequences as profound as those for international migrants because many individuals went back and forth between the North and South, and many social networks remained intact because of the high frequency of migration.
However, the social conditions of the rural South were extremely different than those in the urban north. Blacks attended de jure segregated schools, with starkly lower school quality than whites. Voter suppression was a core element of the Jim Crow structure. In 1947, for example, around 12% of voting age Southern blacks were registered to vote (Alt 1994), (Pg 374). McMillen estimated that in 1940 only 2,000 black Mississippians were registered to vote, which was 0.4% of the voting age black population (McMillen 1990), (pg 36). Extreme voter suppression continued in much of the South prior to the Voting Rights Act of 1965.
The widespread social inequalities in the South then and now may play a role in explaining the broad health disadvantage of Southerners regardless of race/ethnicity. Results from the Third National Health and Nutritional Examination Survey examined the prevalence of hypertension as a function of residence in the South, sex, ethnicity, and level of urbanization (Obisesan et al. 2000). Non-Hispanic white men and African American men and women living in rural southern regions had higher rates of hypertension than residents of non-southern locations. Geographic variation in the prevalence of cardiovascular risk factors may be related to regional dietary patterns, including differences in sodium, fiber, fatty acid, and cholesterol consumption (Hajjar and Kotchen 2003).
Migrants who leave the South do not entirely escape the health effects of Southern residence. For example, excess stroke rates among white and black residents of the Southern “stroke belt” have long been recognized, but it now appears that individuals who live in the South have excess stroke rates even if they migrate out of the stroke belt (Glymour et al. 2007). The consequences of these conditions probably vary depending on the age of immigration, but even young migrants are probably not entirely immune. Even, individuals who migrated as infants with their parents may have had prenatal exposures that influence their long term health.
Among residents of New York City, blacks born in Southern states were reported to have higher rates of mortality due to cardiovascular disease, particularly coronary heart disease, than their Northeastern and Caribbean-born counterparts (Fang et al. 1996). In fact, the significantly higher rate of mortality associated with cardiovascular risk factors among African Americans in NYC was accounted for primarily by location of birth, as Northeastern and Caribbean-born African Americans had similar or better rates of mortality than Northeastern-born whites. Similarly, national circulatory disease mortality rates for blacks indicate that the Southern born have excess mortality compared to blacks born in other regions, regardless of their region of death (Greenberg and Schneider 1992; Schneider et al. 1997).
Data from epidemiological studies of health status and mortality have suggested important interactions between race and geographical location. Kington and colleagues found that self-rated health and functional limitations for both Southern-born whites and blacks were worse than those of Northern-born whites and blacks. However, blacks who left the South had health similar to that of Northern born blacks, while whites who left the South did not have better health than whites who remained (Kington et al. 1998).
There is some evidence that the Southern disadvantage is not limited to physical health and health behaviors, but extends to cognitive outcomes. Most research on regional differences in cognitive test performance has been conducted among children and uses IQ measures as the key outcome variables. One study found that in the standardization sample of the Wechsler Preschool and Primary Scale of Intelligence-Revised, geographic-region (Northeast, Midwest, South, and West) effects on scores were small but significant (Sellers et al. 2002). Furthermore, there were a number of region-by-ethnicity interactions on FSIQ. In the West and Northeast, scores of African Americans were not significantly lower than those of whites, whereas in the South and Midwest disparities were evident. Among adults, we know that test developers continue to sample by region in their standardization cohorts (Wechsler 1997). Furthermore, a widely used formula applied in the estimation of premorbid IQ on the WAIS-R included geographic region, and urban vs. rural residence as two of the predictor variables, along with age, sex, years of education, and occupation (Barona et al. 1984). Factors that account for the secular trend of increasing IQ scores (Flynn 1984) may also contribute to the geographic differences in cognitive scores, such as nutrition (Lynn 1998), occupational status (Schooler 1998), exposure to technology and urbanization (Wheeler 1970), and socialization practices that improve performance on cognitive measures. Residence in urban areas in the North and West could be associated with increased exposure in schools to “teaching to the test”, and increased test sophistication(Williams 1998), such as guessing skills and persevering through difficult items (Brand 1987).
The South has historically been less urbanized than the Northeastern states, but it is unclear if rurality, per se, translates into effects on cognitive aging. Rural residence has been reported to have a modest effect on risk for developing AD in some studies outside the US (Rocca 1990; Prince et al. 1994; Hall et al. 1998; Liu et al. 1998; Tsolaki et al. 2005). We know of only one study of African Americans that reported childhood rural residence, along with low education, as a risk factor for prevalence of AD among elders in Indianapolis (Hall et al. 2000).
Regional differences in cognitive test performance may be primarily driven by regional differences in the quality and quantity of schooling. Educational experience may thus be an important pathway in which Southern childhoods harm cognitive trajectories of black elders. African Americans in most Southern states attended de jure segregated schools, until legally imposed desegregation in 1954. School quality was markedly lower than in schools attended by comparable whites. Segregated states frequently mandated minimum term lengths in schools for white children that were 50–100% longer than the term lengths mandated for schools for black children. As a result of these differences in school term length, a black child born in South Carolina in 1925 who attended school every day it was open for 10 years would have attended school the equivalent of 2.5 years less than a comparable South Carolinean white child who attended school every possible day for 10 years. Accounting for differential attendance rates, a white South Carolinean from the 1925 birth cohort who attended 10 years of school averaged 2.6 years more school than a black South Carolinean, but 1.0 years less than a comparable New York child. The school quality differences extended to every feature of schooling: physical infrastructure, textbooks and curriculum, teacher training and consistency, class size, hours in the school day. As an example, Fig. 2 compares the average school term length (days school was held) for segregated black and white schools in selected states. As the age of de jure school segregation drew to a close, the gaps in quality diminished (Card and Krueger 1996) but most elderly blacks raised in the South experienced very different schooling regimes than elderly whites. Even among whites, the low quality of schooling likely had cognitive consequences. whites born in states with fewer years of mandated compulsory schooling had worse memory scores decades later compared to whites born under more demanding state schooling regimes (Glymour et al. 2008). This study could not be conducted in blacks because compulsory schooling laws were not enforced for blacks for the relevant decades (Lleras-Muney 2002). This very non-enforcement, however, likely affected both educational attainment and memory performance of elderly blacks.
In summary, the majority of African American elderly in the US today lived parts of their lives in the South. There is extensive evidence that Southern residence is associated with poor social, physical, and cognitive outcomes. This is not race-specific; Southern residence appears to be associated with worse outcomes for both blacks and whites. The harm may be disproportionate for elderly blacks, however, because the functional goal of social organization in the Jim Crow era was to privilege whites at the expense of blacks. Furthermore, a much greater fraction of African-American than white elderly was exposed to Southern life. A tremendous amount of research must be done to understand the consequences of these exposures on domain-specific cognitive functions. Because nationally representative samples with comprehensive neuropsychological assessments are rare, it has been difficult to examine such contextual risk factors except in highly selected samples.
International Migration
The effects of migration on individuals born outside the United States may be entirely different from intra-national migration and offers a window of opportunity to researchers for distinguishing the underlying causes of the relationship between social conditions and cognitive status. For example, in some cases, contrasting the experiences of non-US born blacks to US born blacks illuminates the caustic consequences of American culture on the health and well-being of US blacks (Read and Emerson 2005).
Immigrants to the US generally have better health than comparable non-immigrants. The immigrant advantage is especially large among blacks (David and Collins 1997; Singh and Siahpush 2002; Read and Emerson 2005) as compared to Asian and Latino immigrants. Most non- US born blacks living in the US are either Caribbean or African. Many Caribbean countries have racially diverse populations, but race is often conceptualized very differently in these countries than in the United States. Contrasting the experiences of black, white, or Hispanic immigrant streams arising from the same countries can illustrate the ways the American experience differs by race. After immigration, the social and health trajectories of immigrants of different racial/ethnic groups tend to diverge (Reitz and Sklar 1997), even for immigrants from the same country of origin (Zavodny 2003). In particular, Waters’ study of West Indian black immigrants to New York found that Caribbean immigrants are socially advantaged in multiple respects compared to US-born blacks. Immigrants’ attitudes and expectations are shaped by experiences in their country of origin, in which white-on-black racism is less frequent. Thus, Waters argues that West Indian immigrants are less likely to anticipate racist acts and more likely to resist structural discrimination. Because the work opportunities in their country of origin may be even more restricted than jobs available for comparably trained African Americans, they may evaluate low-level US jobs more positively. As a result of personal interactions less fraught with mistrust and a perceived better “attitude” regarding work and earnings, white employers may prefer Caribbean employees over US-born African Americans. However, these social advantages appear to fade for the children of black immigrants, who generally do not have accents and thus are no longer as easily identifiable as ethnically distinct from African Americans (Waters 2000). Recent black Caribbean immigrants are markedly healthier than US-born African-Americans, but over time immigrant health converges with population patterns of African-Americans (Portes and Zhou 1993; Portes 1995; Perlmann and Waldinger 1997).
There is a limited but interesting cluster of papers examining differences in cognitive test performance among Caribbean-born blacks in the United States and in the United Kingdom (Richards et al. 2000; Stewart et al. 2002; Byrd et al. 2005; González et al. 2007). Although far from conclusive, due to either small sample sizes or limited test batteries, these studies indicate that although quality of education among Caribbean-born black immigrants in the US is higher than that among US-born blacks, there are no mean differences in scores on cognitive tests.
Similarly, recent Hispanic immigrants from Mexico, Central, and South America tend to have better health than their socioeconomically matched US-born counterparts, but this advantage is smaller or nonexistent among immigrants who have lived in the United States for a long period of time (Cho et al. 2004; Lara et al. 2005). The precise explanation for the declining immigrant advantage is unclear but includes social pressures such as acculturative stress and racism, changes in health behavioral (such as diet and exercise) and environmental exposures.
Age of migration is especially important to consider for international migrants from non-English speaking countries. Functioning in an English-speaking community may pose cognitive challenges, but also may serve to isolate adult or elderly migrants who do not have the same opportunities or demands for mastering English as younger immigrants. Increases in gray matter density in the left inferior parietal cortex associated with bilingualism are greater for those who learned their second language earlier in life rather than later (Mechelli et al. 2004). English fluency is less likely if older adults immigrate to ethnic enclaves where it may be less critical for a member of the household to speak English. Although these households are defined as “linguistically isolated” by the US Census (Siegel et al. 2001), there are many neighborhoods in the United States where English is not needed to communicate with neighbors, get health care, work, or shop. Even among individuals who have gained considerable fluency or proficiency in English, performance on neuropsychological tests may be better in their first language (Ardila et al. 1994, 2000). Age of migration also influences the extent to which typically “American” behavioral patterns, governing, for example, smoking, substance use, sexual behaviors, and diet, are adopted. All of these behaviors may have consequences for the development of neurologic disorders.
Migration has also marked the experiences of many elderly Asians (Parker Frisbie et al. 2001). The consequences of migration may markedly differ for these groups, however, depending on the forces that prompted migration (for example, economic concerns, political turmoil, or family reunification); the characteristics of the community left behind (e.g. language, educational availability, health behavior norms); characteristics of the receiving community (presence of ethnic enclaves, discrimination, structural safety nets to facilitate integration); and age of immigration. Parker et al. note substantial diversity among Asian populations, with Vietnamese and Pacific Islanders in general having worse health outcomes compared to other Asian subgroups.
The Ni-Hon-San study of Japanese-ethnicity men compared cardiovascular risk for those who born and raised in Japan, those who immigrated to the US, those born in the US who spent some of their childhoods in Japan, and those born and raised in the US This study found that although US-born men overall had much higher coronary heart-disease risk than Japanese born men, those men who were US born but spent 10 or more years in Japan did not have elevated risk (Reed and Yano 1997).
International immigration is likely to have a number of cognitively beneficial consequences, such as the need to navigate very novel environments and the cognitive demands of functioning in a second language. Bilingualism has previously been linked to improved elderly cognitive outcomes (Bialystok et al. 2004, 2006; Kave et al. 2008). This research has many methodological limitations, because people are not randomly assigned to become bilingual but rather bilingualism is influenced by social resources, acculturation, cognitive skills, and age of exposure, any of which may themselves influence cognitive outcomes. Nonetheless, if this finding is valid it may benefit immigrants as they age. This topic is discussed at length in this issue (Rivera-Mindt, Gollan et al.)
The possible benefits associated with immigration, multiculturalism, and bilingual skills may be offset by the social stressors encountered as a result of cultural change and minority status and detrimental behavioral norms prevailing in the US Despite the apparent advantage of immigrants in some areas of health, other research suggests higher incidence of AD among Caribbean Hispanics living in Northern Manhattan (Tang et al. 2001), and dementia prevalence among Japanese-American men living in Hawaii similar to that of European ethnicity Americans, but higher than rates for Japanese men (White et al. 1996). As with migration and residential histories in the US, the cognitive consequences of international migration are not well understood. Biases that result from selection present even more challenges for interpretation of cognitive and other health outcomes among international immigrants, however, and the wide range of sending communities make simple summaries inadequate.
Neighborhood Effects
Despite modest improvements in black–white segregation in recent decades, residential segregation along racial lines remains quite severe in the US (Charles 2003). In landmark work on residential segregation, Massey and Denton proposed measuring metropolitan racial residential segregation along five dimensions: evenness of residence across the city; exposure of minority group members to majority group members residing in the same neighborhood; clustering of minority dominated neighborhoods into one large, contiguous enclave; centralization around an urban core, and concentration within a small geographic area (Massey and Denton 1988). They argued that the uniquely extreme segregation on at least four dimensions (termed hypersegregation) faced by African-Americans in many urban areas played an important role in constraining socioeconomic opportunities (Massey et al. 1987; Massey and Denton 1989). For example, in the 2000 Census, the average metropolitan white lived in a neighborhood that was 80% white and only 7% black. In contrast, the average black lived in a neighborhood that was 33% white and 51% black (Logan 2001).
Residential segregation for other minority populations is generally not so extreme; indeed, complete isolation is not possible if the minority population does not constitute a large fraction of the total population. In other words, people of Philippine descent can hardly avoid living in a neighborhood with a great number of non-Filipinos, simply as a function of the number of Filipinos versus non-Filipinos in the US Segregation of Latinos and Asians has generally increased as these populations have grown, but is still not nearly as extreme as that for blacks.
In theory, neighborhood of residence could profoundly affect both physical and cognitive health. Neighborhoods influence schools, grocery stores, parks, job opportunities, safety from crime, determinants of healthy behaviors (or “lifestyles”), medical access, cognitive environment, and stress (Kawachi and Berkman 2003). Racial residential segregation has played an enormous role in maintaining school segregation, even after the policy of separate schools was ended by the courts (Logan et al. 2002). Several studies implicate neighborhood disadvantage in physical health impairment, but the results have been somewhat inconsistent, and there are a number of methodological concerns (Kawachi and Subramanian 2007; Subramanian et al. 2007). The most troubling methodological concern in studies attempting to demonstrate the effect of neighborhood on health is the difficulty of disentangling the effects of individual SEP from community characteristics. Even multilevel models that purport to examine neighborhood effects while controlling for individual SEP rarely include comprehensive measures of individual SEP.
Few studies of neighborhood effects have focused on cognitive aging. Elderly individuals living in neighborhoods with a lower average educational level in the US (Wight and Aneshensel 2006) or lower socioeconomic deprivation in England (Lang et al. 2007) have been found to have better cognitive function than elderly living in neighborhoods with higher average socioeconomic conditions, independently of their own, individual educational level. The disadvantage of Mexican-American versus European-American elderly on Mini-Mental Status Examination (MMSE) scored cognitive impairment was explained by neighborhood-type in a study in San Antonio (Espino et al. 2001). This has not yet been extended to longitudinal studies of cognitive change, and many of the methodologic difficulties with neighborhood effects research pertain to these studies. For example, in the Wight et al. study, the cognitive outcome had a low ceiling (ceiling bias is discussed further in the article by Glymour, Weuve, and Chen in this issue). As a result, the association between individual SEP and the cognitive measure is likely to be incorrectly estimated, potentially producing a spurious relationship between community average education and the cognitive measure.
Further evidence for the role of neighborhoods in cognitive development emerges from work on child development. Longstanding results show that children living in affluent neighborhoods appear to have better cognitive function compared to children living in economically deprived neighborhoods, independent of their own family’s SEP.(Duncan et al. 1994). Sampson et al. recently showed that living in disadvantaged neighborhoods impairs the trajectory of growth of verbal skills for young black children in Chicago. In this study, they were not able to compare black and white children because there were almost no white children living in neighborhoods as disadvantaged as those where nearly all black children resided (Sampson et al. 2007). Nonetheless, black children who lived in comparable neighborhoods at baseline but subsequently moved to more advantaged neighborhoods (or whose neighborhoods improved around them) fared better on verbal-skills development than their compatriots whose neighborhoods remained the same or declined. Further disentangling how residential segregation plays out to constrain socioeconomic and cognitive training trajectories across the lifecourse, Charles et al. find that college students from highly segregated neighborhoods have higher family stress arising from violence and disorder in their home neighborhoods. Obligations to family members living in poor and segregated neighborhoods demand more time and attention, compromising students’ academic performance even after the student no longer lives in the segregated community (Charles et al. 2004)
Despite the growing body of observational research indicating pervasive harmful effects of residence in a disadvantaged neighborhood, experimental evidence is almost nonexistent. The best designed trial to randomize individuals to varying neighborhood contexts found mixed and generally modest results on adult economic self-sufficiency and physical health, with some benefits for mental health (Liebman et al. 2004). Cognitive function was not assessed in this study.
Socioeconomic Position
In addition to geographic and residential histories, socioeconomic position is a social exposure that differs drastically across racial and ethnic groups. We follow Krieger’s definition of SEP to indicate “an aggregate concept that includes both resource-based and prestige-based measures, as linked to both childhood and adult social class position.” (Krieger et al. 1997) (pg 345). Aspects of SEP have been linked to an array of old age cognitive outcomes. Education predicts prevalent cognitive function (Brayne and Calloway 1990), cognitive impairment (Launer et al. 1993), all-cause dementia (Fratiglioni et al. 1991; Katzman 1993; Prencipe et al. 1996), AD (Mortel et al. 1995; De Ronchi et al. 1998; Raiha et al. 1998; Gatz et al. 2001), vascular dementia (Mortel et al. 1995; De Ronchi et al. 1998), cognitive change (Schaie 1989; Albert et al. 1995; Farmer et al. 1995; Butler et al. 1996; Lyketsos et al. 1999), and incident dementia (Stern et al. 1994) (Evans et al. 1997; Karp et al. 2004). Occupational characteristics (Lindesay 1989; Kroger et al. 2008) and income have similarly been demonstrated to predict cognitive outcomes in the elderly. The association between SEP and cognitive aging is important because of the profound social stratification by race that stretches back across generations in the US. Below we discuss the dimensions of SEP, the reasons it may be relevant to cognitive trajectories across the lifecourse, and review racial disparities in socioeconomic resources.
Dimensions of SEP
SEP is often measured using educational credentials, income, or occupational class, but these three measures represent distinct exposures. They are only moderately correlated with one another and are themselves not comprehensive assessments of SEP (Berkman and Macintyre 1997; Krieger et al. 1997; Braveman et al. 2005; Galobardes et al. 2006). To illustrate the challenge of comprehensively capturing SEP, it is helpful to consider the pathways via which conventional SEP measures are likely to influence health and elderly cognitive outcomes. Link and Phelan have hypothesized that facets of SEP are “fundamental causes” of health and disease, in that they are flexible tools or assets that allow an individual to attain health promoting resources in a wide variety of contexts, no matter what the major threats to health may be (Link and Phelan 1995). They argue that SEP triggers multiple health-promoting pathways, and these protect high-SEP individuals from threats as varied as infectious diseases or chronic conditions or unintentional injuries. Individual SEP may affect cognitive aging or diagnoses through any of the four individual exposure pathways identified in Fig. 1: material conditions, psychosocial conditions (e.g., status), direct cognitive stimulation, or test-taking skills. Although direct cognitive stimulation is not typically considered in discussions of SEP and health, it is likely to be highly relevant when the health outcome of interest involves cognitive function.
Financial resources ensure access to health-enhancing material conditions such as healthy housing, high-quality food, safe working conditions, and ready access to high-quality medical care. These exposures influence both physical and mental health and thus directly and indirectly shape patterns of cognitive aging. Wealth and income may play distinct roles with respect to material conditions. Although income allows people to purchase goods, wealth is, in general, a broader measure of financial resources and safety nets (Berkman and Macintyre 1997; Braveman et al. 2005). Even with a high income, individuals with low wealth are much more vulnerable than those with comfortable bank accounts: short spells of unemployment or illness can be disastrous for those without wealth. For example, Himmelstein et al., found that medical conditions contributed to approximately half of bankruptcies (Himmelstein 2005). Wealth may be of special relevance for elderly people, who may have limited post-retirement income but whose material resources are determined by the wealth accumulated prior to retirement. Though income and wealth are the direct markers of financial resources, both education and occupation are also relevant because they are the primary determinants of income and a major determinant of wealth.
SEP is also important as a marker of social position and status. Social status arises from many personal characteristics and resources. Although income and wealth are often considered the most direct markers of status, occupation and education are also important markers. For example, individuals with high levels of education or income are treated with esteem and deference in their daily lives. To the extent that social stature allows one to avoid a multitude of small humiliations and stressors, it likely has substantial consequences for cognitive aging.
Many alternative occupational classifications have been developed and these have been demonstrated to predict a variety of health outcomes (Krieger et al. 1997; Lynch and Kaplan 2000). Occupations, indexed against the US census categorizations, have also been ranked by prestige (Hauser and Warren 1997). For example, with a possible range from 0 (lowest) to 100 (highest), postmasters received a prestige score of 53, architects 73, store sales clerks 32, and filling station attendants 21 (Nakao and Treas 1994). Blacks are systematically employed in lower prestige occupations than whites (Conley and Yeung 2005).
Conventional occupationally based social class indicators may distinguish managers/owners from employees; or white-collar jobs from blue-collar jobs; or skilled from unskilled jobs. Karasek and Theorell have promulgated the demand/control categorizations of work environments, theorizing that it is the conflict between high demand accompanied by low control that can be health-compromising (Theorell 2000). Alternative characterizations that may be especially relevant to cognitive outcomes relate to the complexity of interactions with data, objects, or people demanded in specific jobs (Kroger et al. 2008).
These alternative classifications do not necessarily correspond with one another: low prestige jobs may offer high control and blue-collar jobs may offer substantially higher prestige than many white-collar jobs. The relationships between any of these categorizations and cognitive aging have not been thoroughly investigated, so it is not possible to assign a preference of one over the other, or determine if the various job features have independent influences. Many of the occupational categorizations described here are likely to influence health via either material deprivation or stress pathways, but jobs, of course, often entail exposure to direct physical toxins. Such exposures are clearly relevant to understanding cognitive aging, and we do not explore this except to note that toxic occupational conditions tend to be concentrated among low-status jobs.
Finally, education has special relevance to understanding racial disparities in cognitive aging for several reasons. Education is perhaps the social exposure most frequently linked to cognitive aging [see, for example, the review in (Anstey and Christensen 2000)]. The benefits of education accumulate across life because schooling initiates a trajectory of socioeconomic conditions (Mirowsky and Ross 2005), and the cognitive training or engagement pathways stimulated by education may be crucial in linking many other social exposures to cognitive aging. Finally, education has historically been extraordinarily racially stratified in the US, and this stratification continues, albeit in less legally formalized patterns. As a result, to the extent that education influences cognitive aging, we expect this to create racial disparities in outcomes.
Education can be measured along several dimensions, such as credentials (e.g., high-school diploma), years of completed schooling, or quality of schooling (Krieger et al. 1997). Of two individuals completing 12 years of school, only one may have a high-school diploma, and there may be highly divergent educational experiences if they attended schools of different quality. The competing hypotheses about the pathways via which schooling may influence cognitive aging have substantive implications for which dimensions of education are most important. For example, we expect educational credentials (e.g., degrees) to be the most relevant dimension of education if education’s effects operate primarily through material hardship. Credentials are probably most relevant for influencing labor market opportunities; potential employers usually have little ability to distinguish “quality” of school and probably do not regard 11 years of school as substantively superior to 10 years of school. On the other hand, if cognitive skills or cognitive engagement are the relevant features of education, then years of education and quality of school are likely to be more important than credentials. Receiving a degree does not change the cognitive skills acquired during schooling, and an additional year of school entails additional cognitive demands even if a degree is not received for the additional year.
Efforts to tease out these influences for non-cognitive health conditions suggest that relationships may differ somewhat depending on the health outcome of interest, but these studies generally emphasize the importance of years of education as opposed to quality of school or credential (Ross and Mirowsky 1999). To date, this question has not been carefully examined for most outcomes of relevance in cognitive aging research or across domains of neuropsychological performance.
Any of the above dimensions of SEP may be measured at different points in the lifecourse. Characteristics of parents’ SEP are used to characterize children’s social conditions. The resources available to parents, including money, knowledge, and social connections, are invaluable to children in advancing education, insuring against spells of poor health or unemployment, and maximizing opportunities for health and well-being. Parental differences are likely to continue to redound in the socioeconomic, and therefore health and cognitive, trajectories of children, and have been shown to predict impairment on dementia screens among the elderly (Hayward and Gorman 2004; Luo and Waite 2005).
Reflecting on the alternative lifecourse models considered earlier, changes in economic fortunes may immediately change health, alter physiology in ways that do not become clinically manifest for years to come, or start the individual on a long term trajectory of poverty and financial insecurity which much later is translated from a social condition to a physiological condition and only then compromises health. These timing effects need not be mutually exclusive, and similarly, material and psychosocial consequences of SEP may both be important. Both material and psychosocial pathways may trigger physiological and behavioral changes that influence health. For example, diet may be compromised by insufficient money to purchase high-quality nutritious foods, inadequate availability of nutritious foods, or by depression or other psychological states that make it difficult to devote extra time and attention to planning and preparing healthy meals. Although material and psychosocial pathways are depicted separately in Fig. 1, in many contexts the material and psychosocial pathways reinforce one another, and it is the coalescence of material and psychological obstacles that undermines healthy behaviors or promotes unhealthy behaviors. These behaviors, such as alcohol-use, smoking, nutrition, exercise, and management of chronic diseases such as diabetes, will ultimately shape neuropsychological trajectories of development and risk of incidence of many cognitive disorders, including Parkinson’s disease, Alzheimer’s disease (AD), and vascular dementia (Launer et al. 1996; McDowell 2001; Cotman and Berchtold 2002; Weuve et al. 2004).
Finally, it is possible that aspects of socioeconomic conditions that do not influence outcomes among whites may be relevant to outcomes for minority groups. For example, Gump et al. found that both neighborhood and family SEP influenced cardiovascular reactivity for African American children, but only family SEP was relevant for white children (Gump et al. 1999). Whites, by virtue of advantaged positions, may be buffered from social exposures that threaten blacks or other racial/ethnic minorities. On the other hand, Conley and Yeung report that parental occupational status predicted children’s verbal and math scores for white families but not black families (Conley et al. 2005).
Disparities in SEP and their Relevance to Cognitive Function
The relationships between dimensions of SEP and cognitive aging are relevant to understanding racial patterns of cognitive aging because of the stark racial disparities in SEP in the United States. The depth and breadth of racial disparities in SEP are easily underestimated. Figure 3 shows the population distribution of wealth for non-institutionalized US blacks and whites aged 50 and older, based on data from the nationally representative 1998 Health and Retirement Study. The 75th percentile of the black wealth distribution corresponds to just the 35th percentile of the white wealth distribution. Adjusting for age, years of attained education and current income, black households average over $100,000 less wealth than white households. In other words, within levels of each measured socioeconomic variable, blacks are on average more disadvantaged than whites on other socioeconomic variables. This phenomenon occurs for every period in the lifecourse.
A similar phenomenon applies for education. Extreme differences in educational level are often found between ethnic minorities and whites. Illiteracy rates in the United States are highest among people aged 65 and over, but are especially elevated among ethnic minority elders (Kirsch et al. 1993). Attempts to “equate” ethnic groups on years of education across ethnic and racial groups have not fully controlled for the effects of educational experience on cognitive test performance. Along with several other authors, (Loewenstein et al. 1994; Kaufman et al. 1997; Whitfield and Baker-Thomas 1999), we argue that due to the disparities in quality of education, matching on quantity of formal education does not necessarily mean that the quality of education received by each racial group is comparable. Systematic differences between African Americans and whites in quality of school (Welch 1966, 1973; Smith and Welch 1977; Smith 1984; Margo 1985; Margo 1990) result in persistence of racial differences in cognitive test performance despite matching groups on years of education. This presents a tremendous challenge to cross-cultural researchers seeking to find measures that are sensitive and specific to cognitive impairment across very heterogeneous educational backgrounds.(Ratcliff et al. 1998).
Childhood SEP measured, for example, as parental education or occupation, is on average much lower for blacks than whites. Childhood SEP appears to have a major effect on adult SEP, but also likely directly influences adult health through pathways not mediated by measured features of adult SEP. As mentioned above, any comprehensive SEP measure relevant to cognitive test performance among adults would include assessment of parental resources.
In summary, racial and ethnic differences in unmeasured variables, such as parental wealth or educational quality, are likely to be just as great as the differences in measured variables. The implication of this is that studies purporting to adjust for or match on socioeconomic position are probably nonetheless comparing whites with greater socioeconomic advantage to blacks with less socioeconomic advantage. Adjusting for socioeconomic position with just one or two rough indicators of adult SEP is very unlikely to adequately control for racially patterned differences in material resources or social status (Braveman et al. 2005).
Maintenance of SEP Disparities
In order to have a deeper understanding of racial differences in cognitive test performance, neuropsychologists must be familiar with the scholarly work that examines the processes that establish and maintain racial disparities in SEP. First, there is substantial SEP stickiness in the US across generations, so that the SEP of parents strongly influences the SEP of children. Initial econometrics research vastly underestimated the correlation between earnings of fathers and sons, but recent data indicate that for every 10% change in the earnings of the father, a 6% change in the earnings of the son is anticipated. With this level of stability, it would average five to six generations for descendants of a family at the poverty level to be within 5% of the national-average earnings level (Mazumder 2005; Haskins et al. 2008). There is substantial intergenerational transmission of not only individual SEP but also neighborhood average SEP, so children raised in highly impoverished neighborhoods are themselves very likely to live in similarly impoverished neighborhoods in adulthood (Sharkey 2008). Thus, the historical SEP disadvantage faced by African-Americans is likely to persist across generations.
This disadvantage is reinforced by processes such as racial residential segregation, which limits educational and job opportunities. Explicit discrimination in the labor market probably also contributes to lower earnings of blacks compared to whites. Several audit studies have shown that when black and white applicants matched on relevant qualifications apply for the same job, the white applicants are received much more favorably. The design of these studies provides compelling evidence for discrimination. Based on name frequencies from birth certificates, fabricated resumes were randomly assigned typically “white” names (e.g., Emily or Laurie) or typically “black” names (e.g., Aisha or Tamika) and these resumes submitted in response to job postings. On average, the results indicated that an applicant with a stereotypically black name would have to submit 15 resumes before receiving a call back for an interview, while an applicant with a stereotypically white name would only have to submit 10 letters. In other words, white applicants had a 50% higher response rate than black applicants, based on no difference but their names (Bertrand and Mullainathan 2004). Similar field experiments using matched auditors have documented pervasive race-based discrimination in the housing, car sales, and auto insurance markets.(Riach and Rich 2002).
In addition to interpersonal discrimination, social policies at the federal, state, and local level have helped create racial disparities in wealth. The history of home ownership provides an especially potent example. Home ownership is a primary vehicle of wealth accumulation for Americans. After World War II, the federal government implemented a highly successful policy to promote home ownership, based on providing mortgage insurance, allowing banks to make more loans at lower interest rates. Brodkin estimates that about half of suburban housing in the 1950s and 1960s was financed by loans from either the Federal Housing Authority (FHA) or the Veterans Administration (VA). The FHA explicitly promulgated racial restrictions, and the VA based their policies on the previously established FHA standards. The FHA refused to insure mortgages in racially integrated communities. This policy relegated black families to home ownership in racially segregated neighborhoods and motivated white homeowners to implement racial exclusion policies (Brodkin 2004). Housing values in predominantly black neighborhoods did not increase nearly as quickly as housing values in predominately white neighborhoods, and the consequences for accumulation of wealth in black families have been significant. Although the Fair Housing Act helped end explicitly discriminatory policies such as this, there is evidence that patterns of lending and insurance practices continue in ways that disadvantage black homeowners (Williams et al. 2005).
Blacks have been excluded from other social policies intended to promote economic progress. Although the “GI Bill” is generally heralded as an engine of economic opportunity for World War II veterans, the benefits accrued largely to white veterans. Black veterans were much less likely to be able to take advantage of the programs; for example, the college tuition benefit was irrelevant to the majority of blacks who had not graduated from high school or those who lived in regions of the country with very few seats for black students in the still-segregated colleges. Black veterans who returned to the South encountered job counselors who systematically routed them to low-wage jobs, even if these jobs corresponded to a much lower skill level than their training and work in the military (Turner and Bound 2003).
Standardized indicators of the level of discrimination and stigmatization are not available, but a sense of the power of the social divide is illustrated by the white response to school desegregation. In some states, when integration was legally mandated, state governments suspended compulsory schooling laws for white children. These mandates had by that point been in place for decades, but the state governments nonetheless would not insist white parents send their children to racially integrated schools. Survey questions from the General Social Survey (GSS) give a sense of more recent trends. The fraction of people espousing views such as whites have a right to keep blacks out of their neighborhoods or supporting laws against interracial marriage declined substantially from 1972 to 1996 (Valentino and Sears 2005). Bobo reported that in the 1990 GSS “considerably more than 50% of whites rated blacks and Hispanics as less intelligent [than whites]”(Bobo 2000)(pg 277). By 2006, when asked to rate the average intelligence of blacks, and separately, the average intelligence of whites, only approximately 30% of white GSS respondents rated blacks on average as less intelligent than whites (calculations from authors).
Psychological Consequences of Discrimination
Experiences of discrimination also have psychological consequences and we turn to these now. In addition to residential patterns and SEP disparities, the psychological consequences of discrimination may be a primary pathway via which racialized experiences influence lifecourse cognitive trajectories and cognitive aging. We consider here three categories of psychological consequences: stress resulting from experiences of discrimination; self or “internalized” stereotypes; and stereotype threat. Each of these can influence cognitive function and cognitive test performance acutely, and thus shape diagnoses of cognitive aging. They can also have accumulating effects over the lifecourse that influence the trajectory of cognitive aging.
Discrimination and Stress
Experiences of discrimination or unfair treatment may trigger a number of physiological responses related to stress or anger, as well as direct psychological experiences such as depression or anxiety (Mays et al. 2007; McNeilly et al. 1995; Clark et al. 1999; Harrell et al. 2003). Immediate physiologic responses to perceived racism have been demonstrated in laboratory settings (Clark et al. 1999; Harrell et al. 2003; Bennett et al. 2004; Merritt et al. 2006). Although the epidemiologic research on this topic is in its nascence, numerous studies now show that blacks who report experiencing higher levels of race-based discrimination also have higher levels of a range of indicators of psychological distress. For example, individuals with prior experiences of racism have greater cardiovascular responses to anger recall tasks (Richman et al. 2007) The evidence linking discrimination and physical health (e.g., blood pressure) is somewhat mixed, but regarding mental health outcomes the result is fairly strong (Williams and Williams-Morris 2000; Paradies 2006). To date, we know of no research linking cognitive aging with experience of racial discrimination.
Measurement of experiences of discrimination is a persistent challenge in such research (Krieger 1999). Challenges include determining whether individuals recognize and report discriminatory experiences, what types of experiences are most likely to influence health, and the most important time-scale for discrimination. It is unclear how frequently individuals are able or willing to identify experiences of racism as such. Many racist acts are committed without the knowledge of the target. For example, a job applicant who does not receive a response to a resume cannot know if racism was involved. Furthermore, ambiguous interactions may not be framed by the potential target of the racist act as stemming from race-based discrimination, but rather attributed to everyday rudeness or misfortune. A growing body of psychology research suggests that people who deny racist attitudes nonetheless have negative associations with race that influence their behaviors and interactions (Dovidio et al. 2002). This is sometimes termed “implicit racism”, although related work suggests the interpretation of the standard tests is ambiguous (Frantz et al. 2004; Goff 2005). Some researchers speculate that denying the racist intent behind unfair encounters may itself be psychologically harmful, perhaps because recognition of racism allows a set of healthy coping skills to be invoked (Krieger 1996). It is difficult to test this hypothesis, and it has largely been motivated by ambiguous evidence from epidemiological studies. Racism in certain contexts, such as medical settings or social interactions, may be more harmful than in others. Interestingly, self-reports of discrimination related to daily hassles may predict psychological outcomes better than self-reports of acute traumatic events. More relevant for the current discussion, research on discrimination and health has not adequately addressed the relevant etiologic period. Although there are short-term stress responses to discriminatory acts, it is also possible that the longer term, accumulative effects are most important.
It is sometimes difficult to disentangle the effects of structural disadvantage from the psychologically mediated effects. For example, Whitfield studied the effects of school desegregation on the health of elderly blacks (Whitfield and Wiggins 2003). Unfortunately, desegregation involved several simultaneous changes, any of which might have had profound health consequences: changing to a school of different quality (although the previously “white” schools may have had more resources, it is unclear if they were actually higher quality for the incoming black students); exposure to both acute and chronic discriminatory experiences as black children were heckled and berated for entering white schools; disruption of the network of relationships that previously functioned within the segregated black school; and differential treatment and expectations of white teachers towards black students (Hanushek et al. 2002; Ferguson 2003).
The effects of interpersonal discrimination on cognitive function in particular, and health in general, is an area where further evidence is much needed. As measures of discriminatory experiences expand in both quality and breadth, and research moves beyond the cross-sectional designs that have predominated so far, hopefully we will begin to understand at least the short term consequences of such experiences.
Self-Stereotypes Compared to Stereotype Threat
Racial socialization may affect cognitive test performance via self-stereotypes. Self-stereotypes are stereotypes that the individual believes describe his or her own skills or characteristics. Self-stereotypes have been shown to influence cardiovascular reactivity and gait speed among the elderly (Hausdorff et al. 1999; Levy 2003a). Self-stereotypes have the potential to influence the performance of elders on neuropsychological exams, and, more profoundly, to influence the trajectory of social conditions and cognitive engagement throughout life; however, there are no published studies that have examined the relevance of self-stereotypes in explaining racial differences in cognitive test performance.
Self-stereotypes are distinct from stereotype threat. Stereotype threat occurs when an individual is aware of a negative stereotype others hold about his or her group, and fears confirming this stereotype through his or her own actions. The potential harm and impaired performance resulting from self-stereotyping is distinct from the harm of stereotype threat, and the appropriate remedies differ (Levy 2003b). Stereotype threat cannot occur unless the victim of the stereotype is aware of the stereotype, and believes others likely to hold this stereotype. Self-stereotyping may be essentially unconscious and does not necessarily involve expectations about what other people may think or expect. Stereotype threat can be induced in laboratory settings by making the stereotype salient; for example, by providing a newspaper article about the poor academic performance of blacks or memory impairments among the elderly. Another approach for inducing stereotype threat is to frame a particular task as an assessment of a skill that is well known to be stereotyped; conversely, stereotype threat can be ameliorated by describing a task in a way that seems unrelated to the skill associated with the stereotype. Stereotype threat has been shown to compromise, for example, academic test performance among blacks (Steele and Aronson 1995), math test performance among women (Spencer et al. 1999), and memory among elderly (Hess et al. 2003).
Although it is not always straightforward to distinguish stereotype threat from self-stereotypes, Hess found that reinforcing the aging stereotype induced a greater decrement in memory performance among people who most valued memory. This suggests a threat process, rather than just internalized negative stereotypes. Although the behavioral consequences of stereotype threat have largely been assessed in short-term laboratory settings, Steele emphasizes the potential consequences of lifelong experiences with stereotype threat. Individuals may persistently avoid contexts in which they are likely to face stereotype threat, thus, over the long run, skills required to perform in that context may atrophy. The stereotype creates a self-fulfilling prophecy. Under this hypothesis, longstanding social stereotypes of blacks as bad at academic challenges may lead to disengagement from contexts that are framed as cognitively demanding, especially if performance is assessed. This describes most neuropsychological testing situations quite well. In less formal settings, but potentially comparable in terms of consequences, disengaging from cognitive challenges and engagement in daily tasks could ultimately compromise cognitive function and performance in old age.
Group Resources and Social Ties
Social networks influence health, including risk of both physical and cognitive impairments, via several avenues, for example by providing material assistance, emotional support, access to sources of social or political influence, and advice. Social contacts can also be detrimental, for example through excessive demands, contagious diseases or unhealthy behavioral norms (Berkman and Glass 2000).
Accumulating evidence links social interactions and features of social contacts to cognitive outcomes among the elderly (Bassuk and Glass 1999; Fratiglioni et al. 2000; Seeman et al. 2001; Barnes et al. 2004; Wilson et al. 2007; Ertel et al. 2008) Few of these studies included enough racial or ethnic diversity to examine differences in the effects across groups, but Barnes’ findings suggested the association may be stronger for whites than blacks (Barnes et al. 2004), although there was little difference in effect sizes as reported by Ertel (Ertel et al. 2008). There is little general consensus on either the precise components of social contact that are most important (Berkman 2000) or the specific domains of cognitive functioning most affected. The primary outcomes in these studies have usually been either a global, combined measure of cognition, or dementia diagnosis. Ertel’s analysis used a brief memory assessment as the outcome, but did not compare how associations differed with other cognitive assessments. Wilson found that loneliness predicted expedited declines in semantic memory, perceptual speed and visuospatial ability, but was not significantly associated with rate of decline in episodic memory or working memory (Wilson et al. 2007). Bennett et al. found that social network size modified the relationship between AD-like pathology and cognitive performance, such that those with larger networks were less likely to manifest neuropathological damage in cognitive tests. The interactions between network size and disease pathology were specifically evident for semantic and working memory (Bennett et al. 2006).
To fully understand how the association between social ties and cognition may contribute to racial disparities in cognitive aging, however, we need a more comprehensive understanding of how social networks differ across racial and ethnic groups. Social networks for blacks and Hispanics are often assumed to be larger and more supportive than those for whites, but the mixed empirical evidence suggests that the patterns vary across geographic or social contexts (Silverstein and Waite 1993; Peek and O’Neill 2001; Small 2007).
Ethnic Identity
Racial and ethnic identity may also benefit cognitive aging through various pathways. An enduring puzzle in psychiatric epidemiology is the lower prevalence of many psychiatric conditions and indicators of psychological distress among members of racial and ethnic minorities compared to majority-white populations despite exposure to discrimination. For example, blacks have higher levels of self-esteem, lower rates of suicide, and lower prevalence of many psychiatric disorders (Williams et al. 1992; Hoyert et al. 2001; Breslau et al. 2004, 2005). After controlling for sociodemographics and physical health, middle-aged African-Americans had lower rates of major depression than comparable whites in a national sample.(Dunlop et al. 2003). This finding is especially surprising because African-Americans are disadvantaged with respect to whites on a broad range of physical health measures and overall life expectancy.
Some of these differences may be due to diagnostic issues. For example, there is evidence that blacks and whites presenting with similar symptoms are frequently diagnosed with different conditions. However, it is also hypothesized to result from resilience or protection offered by membership in a group with a distinct identity. For example, models of pride and resistance are likely to be stress-reducing. In some contexts, recognizing and naming discrimination and stigma, and responding with “righteous anger” or indignation may be healthful (Corrigan 2002; Mossakowski 2003; Sellers et al. 2003). To our knowledge, the possible effects of ethnic identification and conscious resistance toward discriminatory exposures on cognitive aging have not been explicitly investigated. The association with offsetting the mental health consequences of discriminatory experiences, however, suggests that it could be relevant. Others hypothesize that social traditions such as church affiliation (Taylor et al. 1996) may be protective of health (Chatters 2000). If there are such benefits, it is unclear how much they might also benefit cognitive aging.
As previously discussed, first generation immigrants are healthier than socioeconomically comparable US-born individuals. The explanations for this are controversial, but in some cases, immigrants bring with them health promoting social resources and health behaviors. Ethnic identity among black immigrants may provide protective resources with respect to attitudes about interpersonal and structural discrimination, ambitions and anticipation of life chances (Waters 2000). These identities may provide social and socioeconomic advantages that translate into health and cognitive advantages. Racial and ethnic identities may coexist. Although black race is often the predominant identity for blacks in the US because of the salient role of race in organizing daily life, blacks may simultaneously adhere to ethnic identities associated with national origin that provide alternative frames for encountering and interpreting white-dominated American society.
A large body of sociological research debates the potentially positive roles of segregated neighborhoods, particularly for recent immigrants, in creating social capital, fostering economic development, and preserving valued cultural traditions (Marcuse 1997). The possibility that ethnic enclaves are health-promoting has been one explanation for the so-called Latino-paradox. Recent studies find that Latinos living in neighborhoods with a high percentage Latino population have lower prevalence of asthma among children, and lower morbidity and all-cause mortality among elderly compared to Latinos living in neighborhoods with a lower fraction of Latinos (Eschbach et al. 2004; Cagney et al. 2007).
Individual Pathways
We now move downstream from the realm of contextual and social conditions to individual pathways that may directly link these environments and resources to cognitive aging. Research attempting to identify the specific exposures that are most important face many serious methodologic difficulties. A primary problem is that lifecourse studies are generally compromised by inadequate, retrospective measures of social conditions in early life.(De Stavola et al. 2006). Furthermore, many crucial social exposures are likely to be correlated. For example, the same children are frequently subject to multiple social disadvantages, such as inadequate prenatal nutrition, restricted early childhood education, and adolescent exposure to violence. As a result, it is difficult to precisely identify the most relevant exposures because alternatives cannot be fully tested against one another (Glymour 2007). Similar difficulties beset efforts to precisely weight hypothesized proximal mechanisms linking various early conditions to cognitive aging.
Material Adversity
The potentially harmful consequences of material adversity for cognitive aging require little explication. The links between poverty and physical health, medical access, and health behaviors are well established. In particular, we emphasize the relevance of unhealthy nutrition, poor housing, inadequate transportation, and limited resources for caregiving or caregiving relief. These factors may have direct neurologic consequences as well as trigger pathways that influence cognitive outcomes, such as onset and poor control of diabetes, cardiovascular conditions, and mental health. Material privation also constrains the opportunities for continued, active involvement with the broader environment, especially after onset of age-related physical disabilities. These pathways all appear to influence the likelihood of vascular dementia and may either exacerbate or expedite cognitive decline associated with Alzheimer’s dementia.
Poor nutrition is among the best elaborated pathways potentially linking material deprivation to cognitive impairments. Substantial research has focused on the relation between cognitive aging and growth measures, perhaps because these measures are often available. One approach to the challenge of retrospective measures of childhood conditions has been to use measures of growth, such as height or cranial size, thought to remain stable in adulthood and reflect childhood nutritional status. Growth measures are sometimes conceptualized as indicators of general deprivation or, alternatively, as posing a direct physiological constraint on neurologic development. Early life nutritional deprivation may restrict neuronal growth, impair neuronal organization, or distort the process of neural remodeling that is a crucial component of development. Nutritional deprivation may alternatively limit cranial growth, which in turn restricts brain development through purely physical means (Brown and Pollittt 1996; Ivanovic et al. 2000).
Several independent studies have found an association between measures of brain or head size, and late life cognitive function (Katzman et al. 1988; Schofield et al. 1995; Graves et al. 1996; Schofield et al. 1997; Gale et al. 2003). Katzman et al. found that women with neuropathological changes characteristic of AD but no clinical impairment had larger brain volume than did women with similar neuropathological changes exhibiting clinical impairments. Two subsequent studies, using larger samples, found an association between cognitive-function scores and head circumference (Graves et al. 1996; Schofield et al. 1997). Gale et al. attempted to distinguish the importance of prenatal and early childhood nutritional exposures by comparing the predictive value of head circumference at birth to that of head circumference in adulthood. She found that head circumference at birth was independent of old-age intelligence tests and logical memory tests but adult head circumference predicted the intelligence test (AH4) and change in Wechsler Logical Memory scores over a three-year follow-up (Gale et al. 2003).
Results have been mixed, however, with some research finding no association between measures of head size and subsequent dementia or cognitive impairment (Jenkins et al. 2000; Edland et al. 2002). Some of the null results have arisen from relatively prosperous populations. Although the jury is still out, it seems possible that among privileged populations, there is limited variability in cranial size. Cranial size in these populations may therefore not reflect nutritional deprivation, but rather naturally occurring genetic variability. If so, the finding that cranial size fails to predict late life cognitive outcomes in these populations suggests that cranial size is important not because it constrains brain growth, but rather as a marker of early nutritional deprivation that itself influences brain development. Graves reported that head circumference predicted incident Alzheimer’s only in apoe-e4 carriers (Graves et al. 2001). One explanation is that head circumference is a marker for something that protects against expression of pathological insults associated with AD, but this reserve is only relevant if the individual is at higher risk of incurring AD lesions in the first place.
A number of other growth measures have been linked to cognitive aging. Abbott (Abbott et al. 1998) found an association between height and late-life cognitive function and dementia risk in a sample of Japanese-American men. Abbott interpreted height as a marker of nutrition in early life. This population included first- or second-generation Japanese immigrants to Hawaii. In related research, Huang found an association between arm span, leg length, and dementia prevalence in the Cardiovascular Health Study participants (Huang et al. 2008).
Richards et al. (2001) used the 1946 British birth cohort to show that birthweight predicted cognitive function at ages 11, 15, and 26. At age 43, there was weak evidence of a gradient in cognitive function across birthweight even when cognitive test values were conditioned on prior assessments.
Early nutrition may directly influence cognitive function and/or may be merely a marker for SEP. Childhood SEP may influence cognitive function via environmental stimulation if poverty is associated with restrictive, less stimulating environments in infancy, altering brain or cognitive development (Greenough and Black 1992; Rosenzweig and Bennett 1996; Guo and Harris 2000). In addition to the research on growth measures and cognitive outcomes, a separate body of work focuses on measures of SEP, per se, and old-age cognition. A sample of Finnish men also documented a correlation between parental SEP, particularly father’s occupation and mother’s education, and cognitive function at ages 58 and 64 (Kaplan et al. 2001). Moceri used US census records to show that area of residence at birth and number of children in the family are associated with risk of AD (Moceri et al. 2001).
Although the nutritional pathway emphasizes that exposure in early life is crucial and adult exposures may be less important, a cumulative process may also be relevant. Geronimus proposed the “weathering” hypothesis to explain patterns of black-white disparities in birth outcomes, and specifically the observation that infant mortality associated with teen pregnancies was much larger for white infants than black infants (Geronimus 1992). She argued that cumulative experiences of social and economic adversity, including discrimination and material privation, accelerate aging in African American women and that, as a result, first birth occurs at an earlier (and also lower risk) age. The weathering concept has been extended more broadly to health conditions beyond perinatal outcomes (Geronimus et al. 2006). Weathering and cognitive decline have rarely been explicitly investigated. Lynch et al. found that economic hardship at multiple points in life predicted self-report of cognitive difficulties, but Karp found that adult occupational measures did not predict incident dementia independently of education (Karp et al. 2004). Additional work, with a broader range of racially heterogeneous samples, more comprehensive measures of lifecourse cumulative SEP, and more detailed cognitive assessments are needed. Because the weathering theory specifically relates to the idea of accelerated aging, this might be a promising framework for understanding racial disparities in dementia and cognitive impairment in the elderly.
Psychological/Psychosocial Stressors
Many of the social disparities discussed above may be mediated by psychological or stress-related pathways. How do these link to cognitive aging? Primate studies show that social hierarchies influence stress hormones and ultimately induce hippocampal damage among low status animals, especially in the context of social instability (Sapolsky et al. 1990). Chronic psychosocial stress predicts reduced hippocampal cell proliferation, and this effect was greatest in older animals.(Simon et al. 2005).
Stress also appears to influence cognitive aging in humans. People who have experienced extreme stress, such as those with post-traumatic stress disorder, have been shown to have reduced hippocampal volume (Kitayama et al. 2005). It has also been shown that chronic life stress among healthy women prospectively predicts reductions in gray matter volume in the hippocampus (Gianaros et al. 2007) and cortisol levels correlate with hippocampal volume (Lupien et al. 1998). Chronic stress has thus been hypothesized to play a crucial role in development of dementia (Magri et al. 2006). Gallo and Matthews hypothesize that SEP effects on health are partially mediated by the influence of negative emotions such as hostility, hopelessness, depression or anxiety (Gallo and Matthews 2003). It is unclear how experiences of daily life stressors associated with discrimination or low SEP may link to this, but Matthews et al. have shown that even brief manipulations of status have short-term neurohormonal consequences.
McEwen and others have adopted the idea of allostatic load to represent the cumulative physiological changes that occur when the body constantly attempts to maintain homeostasis under stressful circumstances. Stress triggers the autonomic nervous system and the hypothalamic-pituitary-adrenal (HPA) axis. Repeated elevations can cause dysregulations that manifest in physiological changes such as persistently elevated cortisol levels and increased cardiovascular reactivity. Standard operationalized measures of allostatic load include blood pressure, waist-to-hip ratio, and glycosylated hemoglobin. McEwen notes the brain’s crucial role in and vulnerability to the stress response and argues for a strong link between allostatic load and neurologic health (McEwen 2000). Allostatic load is hypothesized to play a key role in Geronimus’ weathering hypothesis, discussed above in the context of material adversity (Geronimus et al. 2006).
If chronic stress and the body’s reaction to stressors influences cognitive aging, some individuals may be primed from childhood for heightened stress responses (Mirescu et al. 2004; Weaver et al. 2004). A promising model for this link between early life conditions, and subsequent stress response and cognitive aging has been developed for a rodent model. Meaney et al. have found that handling rats in infancy leads to changes in HPA responsiveness to stress and reductions in age-related cognitive impairments (Meaney et al. 1988, 1991). This group has hypothesized that it was not the rat handling, per se, that caused the changes, but the maternal response to removal of the pup (Liu et al. 1997, 2000). Mothers of handled pups engaged in more licking and grooming during nursing. Although handled pups did not differ from non-handled pups in basal glucocorticoid levels, the glucocorticoid response to stress differed in the two groups. Handled pups had a somewhat lower glucocorticoid response to stress and, perhaps more importantly, a faster recovery to basal levels. These changes appeared to be the result of differences in glucocorticoid receptor density in the hippocampus, mediated by the pituitary–thyroid system in infancy and persisting throughout the rat’s life. Handled rats were less vulnerable to age-related memory loss. These changes are now believed to be mediated by epigenetic modifications of a hippocampus-specific promoter for the glucocorticoid receptor. These modifications can be induced in early infancy by early life maternal interactions but then remain fairly stable through the life of the animal. (Weaver et al. 2004). The human cognate of rat pup handling is not obvious, although reduced levels of plasma cortisol have been noted in preterm infants after massage (Acolet et al. 1993).
Another potentially important pathway via which stress may influence cognitive function and cognitive aging is via sleep. Lauderdale showed that sleep latency (time from bed to sleep) is longer and sleep interruptions more frequent for blacks than whites, and for black men in particular. As a result, black men in her study averaged an hour and a half less sleep per night than white women (Lauderdale et al. 2006). Others report lighter sleep and lower quality in blacks and American Indians compared to whites, although differences were smaller than those found by Lauderdale (Redline et al. 2004). Sleep quality and sleepiness have in turn been linked to short term cognitive deficits and, in elderly populations, to risk of cognitive impairment.(Ohayon and Vecchierini 2002; Lee and Shinkai 2005).
Cognitive Engagement
The robust association between education and dementia, cognitive impairment, and cognitive decline in the elderly has prompted extensive hypothesizing and debate about the possible role of cognitive stimulation on brain health in the elderly. Given the broad differences in educational opportunities across racial and ethnic groups in the US, understanding the education–cognition association may be critical to understanding racial disparities. Many competing explanations for the link between education and dementia have been offered. Some explanations of this association posit bias, rather than causal sources. We conceptualize these alternatives as: confounding, assessment bias, brain battering, cognitive reserve, or brain exercise.
Confounding
Under the “confounding” explanation, the association between education and cognitive outcomes arises because individuals with pre-existing social or cognitive advantages choose to pursue more education. The advantages that led to extended schooling also protect against cognitive aging, and the association between schooling and improved cognitive outcomes is attributable to this confounding. The evidence on the relationship between early life IQ or cognitive skills, and old-age cognitive outcomes supports this plausible explanation. Confounding is extremely difficult to either prove or rule out in the absence of intervention studies. No matter how early in life a child takes the cognitive test used to predict cognitive aging, he or she has almost certainly already been influenced by social and schooling experiences prior to the test. On the other hand, regardless of the early childhood cognitive measures available, it is hard to be confident that attained schooling is not influenced by other, unmeasured, cognitive skills that themselves influence cognitive aging. This concern is especially troubling for advocates of primarily genetic explanations for racial and social disparities in cognitive outcomes (Gottfredson 2005). Although the hereditarian approach has rarely been extended to discussions of racial disparities in cognitive aging, it is a small extension from the growing debates about IQ and physical health (Deary and Batty 2007) to incorporating cognitive outcomes among the elderly.
This hypothesis is supported by studies that show that cognitive skills established very early in life predict outcomes in old age. Snowdon’s famous nun study demonstrated an association between cognitive skills in early adulthood (average age 22 years old), as measured by idea density in a writing sample, and subsequent risk of Alzheimer’s disease (Snowdon et al. 1996). Snowdon’s study is especially interesting because the participating nuns presumably shared environment with respect to diet, environmental toxins, and so forth through their adulthood and old age. This suggests that some determining exposure had already occurred by young adulthood, when the women became nuns. Other studies have found correlations between measures of adult IQ and cognitive function in old age (Plassman et al. 1995) or incident dementia (Schmand et al. 1997). The earliest cognitive measures are from the Scottish Mental Status Surveys, given nationwide to 11-year olds in 1932. Linking selected records to adult dementia findings, Whalley demonstrated that low test scores at age 11 predicted senile but not pre-senile dementia risk (Whalley et al. 2000). Manly et al. found that better literacy skills, typically established by early adulthood and thought to change little in old age, predict slower memory loss in old age. Effects were similar for white and non-white sample members (Manly et al. 2003). In combination, these studies demonstrate that across a broad range of populations, early conditions predict the trajectory of cognitive performance, even if clinical consequences do not emerge for decades.
One approach to disentangling this Gordian knot is to find natural experiments in which individuals were exposed to more or less education for reasons having nothing to do with their own cognitive skills. Several nations conducted such natural experiments during the 20th century by changing their mandatory schooling policies. In Sweden and the United Kingdom, these changes have been shown to predict socioeconomic outcomes (Meghir and Palme 2005; Oreopoulos 2006). In the US, where education policy is largely set at the state level, compulsory schooling changed in different states at different time points, permitting a large number of comparisons. This design has been exploited to show that increases in state compulsory schooling laws predicted increased education (Lleras-Muney 2002), reduced mortality (Lleras-Muney 2005), and improved old age memory (Glymour et al. 2008). This research is still limited, but it does suggest confounding is unlikely to entirely explain the associations between education and cognitive aging.
Assessment Bias
Studies in adults indicate that schooling improves performance on a wide range of cognitive assessments (Heaton et al. 1986; Heaton and Miller 2004). These findings are likely to extend to assessments used to screen for dementia and cognitive impairment in the elderly. Thus, it is possible that lower rates of dementia associated with greater education are attributable to assessment bias. Psychometric studies indicate that for many common cognitive assessments, education predicts performance on many items independently of ability level, so that two people with the same level of true function but different levels of education were likely to score differently on the cognitive assessment. This frequently favors the highly educated (Teresi et al. 2000; Jones and Gallo 2002; Crane et al. 2004). The contribution of assessment bias to racial and ethnic disparities in neuropsychological test scores is discussed further below and is reviewed in much greater detail in the Pedraza & Mungas article in this issue.
Although differential item functioning is routinely present, the bias introduced by this does not appear to be large enough to account for educational differences in neuropsychological test scores. The weight of the evidence from longitudinal studies also suggests that although most assessment instruments over-diagnose impairments among individuals with low education, assessment bias cannot entirely explain the differences in cognitive aging between those with more or less education (Stern et al. 1994; Callahan et al. 1996; Stern et al. 1999; Qiu et al. 2001; Snowdon et al. 1989; Albert et al. 1995; Farmer et al. 1995; Butler et al. 1996; Christensen et al. 1997; Snowdon et al. 2000; Whalley et al. 2000; Wilson et al. 2004).
Brain Battering
Kilander et al. (1997) found evidence in the Uppsala cohort that education may increase risk of impaired cognitive function in old age by influencing vascular risk factors such as smoking and obesity (Kilander et al. 1997). The brain-battering hypothesis draws on these associations to explain the link between education and dementia (Del Ser et al. 1999). This hypothesis essentially states that individuals with low education are at greater risk for vascular damage, which increases the overall dementia risk. All of the material deprivation and stress pathways discussed above would be implicated under the brain-battering hypothesis. Stern has proposed that differences in brain or cognitive “reserve” may account for the differential rates of dementia among those with more education. Among the several variants of reserve, passive models of brain reserve would fit with the brain-battering hypothesis. Brain battering may reduce the reserve available and bring an individual closer to the threshold of damage at which a clinical impairment is manifested (Stern 2002). Neurologic damage may also result directly from stress hormones, along pathways not mediated by vascular health.(McEwen 2001, 2002)
One implication of the brain-battering hypothesis is that the effects should be very broad. Racial and SES differences in cognitive functioning would be expected to manifest across many or all cognitive domains, rather than in specific areas. Thus, evidence for the competing explanations of the education-cognition association may be gathered as we move toward more sophisticated, domain-specific studies.
Brain Exercise
Brain exercise refers to the idea that cognitive engagement and stimulation may have direct neurologic benefits. The “use it or lose it” hypotheses fit in this category, although such hypotheses unnecessarily seem to posit immediate benefits from cognitive engagement, or immediate harm from cognitive disengagement. The timing of possible effects is uncertain: brain exercise during a critical early life period could theoretically have benefits even for someone who subsequently ceased to “use it.”
The role of cognitive engagement and cognitive challenges in promoting long term trajectories and preventing impairments is key to understanding not only the association between education or SEP, and dementia, but also to understanding racial patterns in cognitive test performance. As discussed above, opportunities for schooling in the US have historically been extremely different across racial and ethnic groups, with African-Americans’ facing especially restricted options. School may be a key arena for cognitive engagement and development of cognitive skills, but this disadvantage in opportunities continues throughout life via occupational stratification that reinforces the educational disadvantages for many racial and ethnic minority groups.
Direct pathways from cognitive activity to protection of brain function have been posited, but evidence rests primarily on animal models and brain-injury studies. Researchers have struggled to explicitly define the physiological processes by which education might directly influence risk of cognitive injury, resilience to such injury, or recovery after injury. The evidence is fairly well established in animal research regarding cognitive engagement and environmental enrichment. Environmental enrichment, in the context of animal research, means living or spending time in an “enriched” cage, with more space, other animals, toys, mazes, etc. If cognitive stimulation directly protects against cognitive decline, this could account for the education-dementia relationship.
A large body of research, dating back to the 1970s, has demonstrated experience and behavior-dependent plasticity in the adult rodent brain, especially with respect to synaptic structure (Diamond et al. 1993; Benuskova et al. 1994; Lebedev et al. 2000). Kempermann et al. found that, although neurogenesis decreased with increasing age, housing aged mice in an enriched environment led to increased survival of neuron progenitor cells (Kempermann et al. 1998; Kempermann and Gage 1999a, b; Horner et al. 2000; van Praag et al. 2000). Environmental enrichment also reduces apoptosis and protects against insult-induced cell death, perhaps by promoting upregulation of neurotrophins (Young et al. 1999). Environmental enrichment thus influences synaptic density, synaptic structure, formation and modification of cortical maps, neurogenesis, insult-induced cell death, and apoptosis. These results hold even if mice are put into enriched cages in very late life. Parallel research indicates that keeping animals in environmentally impoverished circumstances compromises brain structure.
Extensions of this research to humans have conceptualized environmental complexity or cognitive demands as arising from educational or occupational exposures. Education may change one’s “cognitive environment” both during school and/or after the completion of schooling. People with more education may be more likely to engage in cognitively challenging activities in old age, such as reading, bridge, etc., or to find themselves in more cognitively challenging situations, compared to people with less education. Many of the research techniques used to demonstrate plasticity in animals cannot be applied directly to humans. Nonetheless, recent evidence from imaging studies indicates that adult humans also have extensive brain plasticity that responds to environmental demands and opportunities, suggesting that many of the animal results are likely to extend to humans. For example, Maguire et al. found that the posterior hippocampus—an area of the brain responsible for spatial representation and thus navigation—is larger in London taxi drivers compared to controls (Maguire et al. 2000). Remarkably, dedicated attention to navigation may have costs in terms of other cognitive skills. For example, taxi drivers performed worse than bus drivers (who follow fixed routes) on various visuospatial tests (Maguire et al. 2006). Bilinguals have greater gray matter density in the left inferior parietal cortex compared to monolinguals. This density is greater for early bilinguals than those who acquired a second language later in life and is positively correlated with performance in the second language (Mechelli et al. 2004). Studies that incorporate a longitudinal design following interventions are especially compelling. Juggling training induced localized gray matter enlargements, and subsequent neglect of juggling was associated with loss of the new volume.(Draganski et al. 2004). Adults who receive cochlear implants have been shown to recruit new brain areas (outside the typical language-processing regions) to make sense of sounds.(Giraud et al. 2001). Stroke recovery is mediated by such plasticity (Cramer 1999; Cramer et al. 2000; Nelles et al. 2001). Among blind individuals who learn Braille, areas of the brain typically devoted to visual interpretation are recruited to assist with tactile information processing. (Hamilton and Pascual-Leone 1998). More directly related to the exposures and outcomes on which we focus here, there is also some evidence drawn from humans that schooling-related skills, such as literacy acquisition, influence the functional architecture of the brain (Petersson et al. 2001).
Epidemiologic evidence also suggests that leisure activities or complexity of activities protects against cognitive decline in humans (Scarmeas et al. 2001; Schooler and Mulatu 2001). Kroger found that job complexity predicted reductions in risk of vascular dementia (Kroger et al. 2008), and Smyth found similar results in a case-control study (Smyth et al. 2004). Friedland showed greater diversity of activities and intensity of intellectual activities in midlife among controls compared to patients with AD (Friedland et al. 2001), but the direction of causation in this cross-sectional study is unclear. The association may occur because cognitive stimulation protects against AD or because a disease process that reduces activity levels may already be operating prior to midlife. Overall, it is difficult to demonstrate a causal relationship for this question using observational epidemiologic studies.
Because of the strong influence of early life educational experiences on subsequent cognitive exposures (via for example, occupational stratification, social norms about leisure time, financial conditions), observed associations between education and cognitive changes in old age provide very little information about whether and how the timing of exposures matters. There is biological evidence for neural plasticity in early life, but adult exposure extending through old age may also affect reserve and resilience.
Friedland has suggested several possible pathways by which education might directly counteract pathological processes in aging brains; for example, increased cerebral blood flow due to neurological activity, changes in the production of brain kinases associated with learning and memory leading to changes in phosphorylation patterns of proteins that create neurofibrillary tangles, and reduction in the stress response due to chronic stimulation of the hypothalamus (Friedland 1993).
The idea of active brain or cognitive reserve fits with the brain exercise hypothesis. Brain plasticity may increase resilience to neurologic injury, whether due to ischemic injuries or Alzheimer-related processes. Acute brain injury, for example in the context of stroke, is frequently followed by extensive recovery (Cramer 2000; Cramer et al. 2000). This recovery is probably the result of synaptic reshaping and use of surviving tissue for new cognitive tasks. For example, fMRI studies of patients with stroke damage to left-hemisphere language areas have documented that language control shifts to the contralateral, undamaged hemisphere over hours to months as recovery from aphasia occurs (Thulborn et al. 1999). Neural injuries, such as cortical lesions, are followed by synaptic proliferation—probably reflecting both more intense use of non-injured brain areas and response to trophic factors released after injury. Animal models suggest that both synaptic proliferation and neurogenesis play important roles in recovery of function after injury (Kernie et al. 2001).
Individuals with more education may have higher levels of neurotrophins or denser alternative neural networks enabling them to more quickly and effectively recruit new brain areas to accomplish a given cognitive task. Greater past exposure to cognitive engagement may facilitate recruitment of multiple alternative networks to accomplish the same task, thus reducing the clinical manifestations of any given injury. (Stern 2002; Habeck et al. 2003; Stern 2006). Stern et al. (1992) attempted to investigate this issue using measures of cerebral blood flow (CBF) (Stern et al. 1992). They theorized that, if the cognitive reserve hypothesis were true, dementia patients matched on clinical severity would have different levels of underlying pathology. They used CBF as a measure of this underlying pathology and found that AD patients with more education had poorer cerebral perfusion than AD patients of similar clinical severity with less education. The finding is bolstered by the subsequent observation that decline among dementia patients occurs more quickly in highly educated individuals (Stern et al. 1999). This approach is analogous to Snowdon’s attempt to test the cognitive reserve hypothesis based on density of NFT’s at autopsy (Snowdon et al. 1996). The validity of this approach depends on the assumption that NFTs measure the pathologic insult (as opposed to the symptoms of that insult).
Brain-exercise hypotheses suggest that there should be some specificity between the environmental exposure and the domain affected. The cognitive consequences of work as an accountant should be very different from the cognitive consequences of work as a cab driver or an attorney.
The best tests of the brain-exercise hypotheses will come from intervention studies. For example, the Advanced Cognitive Training for Independent and Vital Elderly (ACTIVE) trial randomized elderly people to a cognitive training intervention. Although this trial succeeded in improving the trained cognitive skills, transfer to other cognitive domains and to maintenance of daily function was modest (Ball et al. 2002; Willis et al. 2006; Wolinsky et al. 2006). The Experience Corps trial is another innovative trial that may lend insight into the effects of cognitive engagement (Fried et al. 2004).
Proximal Mediators
Physical Health
Disparities in Childhood Health
Racial disparities in health emerge literally at birth and persist throughout life (Gortmaker and Wise 1997; Hummer et al. 2004). Population-level surveillance data on child illness and mortality can be used to give a sense of the racial disparities. Throughout the 20th century, black infant mortality rates hovered at two to three times white infant mortality. The major causes of infant and under-five mortality are diarrheal illnesses, pneumonia, and unintentional injuries. The disparities in diarrheal diseases and pneumonia suggest that black children have historically been substantially more vulnerable to infections than white children. There is also evidence for much more severe exposure to nutritional deficiencies. More recent studies have demonstrated that racial disparities in cardiovascular health are established very early in life. Before adolescence, black children have diverged from white children in indicators of cardiovascular health (Morrison et al. 1992; Berenson et al. 2006). These childhood disparities likely continue to influence health over subsequent years, because childhood health directly and indirectly (via adult conditions) influences both physical and cognitive health.(Luo and Waite 2005; Haas 2007a, b).
Disparities in Adult Cardiovascular Health and their Implications for Cognitive Function
The prevalence of several cardiovascular risk factors is higher in African Americans than in whites at midlife and among the elderly 65 to 85 years of age. These risk factors may be related to higher rates of cognitive decline and risk for dementia, as well as increased morbidity and mortality from cardiovascular and cerebrovascular disease (Anonymous 2005). Among African-Americans 85 years and older, the prevalence of cardiovascular risk factors is comparable to that of whites, suggesting a healthy-survivor effect. Data from the 2001–2002 NHIS show that the prevalence of hypertension and diabetes is higher in middle-aged and elderly (50+ years) African Americans compared with whites, while the prevalence of stroke is comparable. An increased prevalence of hypertension and diabetes among elderly African Americans was also observed in NHANES III (Sundquist et al. 2001). Among adults 65 to 84 years old who were examined in NHANES III, both African American and Mexican American women had a significantly higher prevalence of type II diabetes than white women, after adjustment for age and SEP (Sundquist et al. 2001).
Data from the third National Health and Nutrition Examination Survey show that, among men without diabetes, fasting insulin is highest in Mexican Americans compared to African Americans and whites, and among women without diabetes, both African Americans and Mexican Americans have higher fasting insulin levels than whites (Harris et al. 2002). The differences for C-peptide are similar, but black men and women have lower levels of C-peptide, suggesting a defect in insulin clearance that is different than in whites and Hispanics.
Survey data from the 2001–2003 Behavioral Risk Factor Surveillance System (BRFSS), a state-based surveillance system, also document that the prevalence of multiple risk factors for cardiovascular disease and stroke is higher among African-American elderly than among white elderly (2005).
Medical Access
Medical access differences may arise from cultural or language barriers, financial disadvantages, geographic or transportation difficulties and, in some cases, preferences or distrust of potential patients of the medical system. Severe racial and ethnic inequalities in access to medical care and the quality of medical care received persist (Smedley et al. 2003; Williams et al. 2005). Restricted access to medical care probably contributes substantially to impaired health and exacerbates the consequences of disease after its onset (Williams et al. 2005).
Medical access may differ for minority groups because of direct inter-personal discrimination or due to other factors, such as differences in resources, or geographic context. Direct discrimination is difficult to prove, but in an ingenious study using actors of different races reading identical “patient” scripts and randomly assigned to physicians, Schulman et al. showed that, on average, physician responses differed based on the race of the patient. This study was criticized for some aspects of the statistical analysis, but the key point holds that even in a highly controlled environment, physicians with identical information regarding a patient had different treatment patterns depending on the patient’s race (Schulman et al. 1999).
Despite the overall benefits of medical access, it is worth noting that medical care can often have iatrogenic consequences and is not always desirable or health promoting. The results of the Women’s Health Initiative trial, showing that hormone replacement therapy, previously thought to be beneficial for physical and cognitive health, was net harmful. Indeed, estrogen appeared to induce substantial increases in Alzheimer’s disease risk (Shumaker et al. 2003).
Health Behaviors
The evidence linking most health behaviors and cognitive aging is somewhat equivocal, but exercise and physical activity have repeatedly been shown to influence cognitive function and protect against cognitive deterioration (Laurin et al. 2001; Stewart et al. 2001; Cotman and Berchtold 2002; Kramer et al. 2003; Richards et al. 2003; Weuve et al. 2004). A handful of studies indicate that neighborhood characteristics predict physical activity, especially walking (King et al. 2000; Ross 2000; Stimpson et al. 2007). Some of these results indicate the importance of neighborhoods may be especially significant for elderly individuals, who are likely to spend more of their time in the neighborhood (rather than at work or elsewhere). As mentioned earlier, results on the importance of neighborhoods for frequency of physical activity are not uniform (Ross 2000).
Although the direct contribution of health behaviors to racial disparities in most health outcomes is controversial, there are substantial racial disparities in important health behaviors. For example, the prevalence of sedentarism and obesity tends to be higher among racial and ethnic minorities than whites (Bolen et al. 2000). The prevalence of any alcohol use or binge drinking is lower among blacks than whites in most states in the BRFSS (Bolen et al. 2000). White adults, especially women and those with less education, are more likely to smoke than blacks or Mexican-Americans (Mensah et al. 2005).
Diagnosis
Observed differences in the prevalence of cognitive impairments may reflect either true differences or differential ability to accurately diagnose impairments in different racial or ethnic groups. This problem is closely linked to profound differences in educational histories between ethnic minorities and whites in the US Presence of poor literacy skills among elders is a particularly relevant issue for neuropsychologists attempting to accurately detect dementia using cognitive measures. Reading level is a very powerful predictor of cognitive test performance, independent of years of education, age, or ethnicity (Lecours et al. 1987; Ardila and Rosselli 1989; Rosselli et al. 1990; Rosselli 1993; Weiss et al. 1995; Manly et al. 1999).
Discrepancies in quality of education (as measured by reading level) have been found to explain differences in cognitive test score between African American and whites matched on years of education (Manly et al. 2002; Ryan et al. 2005). This suggests that years of education is an inadequate measure of the educational experience among multicultural adults, and that adjusting for quality of education may improve the specificity of certain neuropsychological measures across racial groups. The full extent of discrepancies in educational experience between African Americans and whites are not captured by a simple “highest grade attained” variable, and thus residual confounding may explain findings of “persistent” race effects after matching groups on years of education. Nevertheless, despite the clear improvement in specificity that is provided by adjusting cognitive test scores for differences in educational experience across ethnic groups, some researchers caution against controlling for educational variables in studies of dementia, since low education may itself be a risk factor for disease.
Many other difficulties in developing comparable assessments are discussed in the paper by Pedraza & Mungas in this issue.
Implications
Clinically, life histories of elderly blacks are relevant for interpreting neuropsychological measures. Poor schooling may cause elderly blacks who are cognitively high-functioning to perform badly on the standard cognitive screens used in clinical settings. Residential segregation and the related experiences of social segregation may isolate elderly blacks from social and cognitive interactions that would facilitate performance on such tests. Interpersonal experiences of discrimination may lead individuals to have special dread or mistrust of medical settings or of cognitive or neuropsychological assessments.
With respect to research, profound differences in social conditions across the lifecourse make black-white comparisons very difficult, and childhood exposures probably both influence and confound estimated associations between adult exposures and cognitive outcomes. Understanding the key pathways, and the most important time periods of exposure, would be invaluable in designing and prioritizing efforts to reduce racial disparities in cognitive impairments. Neuropsychological research focused on distinguishing effects in specific cognitive domains and life periods could help elucidate these pathways.
This topic is also important to understanding and anticipating the population burden of cognitive dysfunction. Increased investments in education and improved conditions for US-born blacks may reduce the incidence of dementia and disabling cognitive impairment, offsetting increased burden expected as a consequence of population aging (Freedman et al. 2001; Freedman et al. 2002). Although Jim Crow laws have been eliminated and overt racial discrimination is on the decline, experiences of discrimination and inequality remain common among racial and ethnic minorities in the US. Disparities linked to these life-long conditions are therefore likely to persist as large cohorts, such as the baby boomers, reach ages when cognitive declines become prevalent.
References
Anonymous (2005). Regional and racial differences in prevalence of stroke–23 states and District of Columbia, 2003. MMWR Morb Mortal Wkly Rep, 54(19), 481–484.
Abbott, R., White, L., et al. (1998). Height as a marker of childhood development and late-life cognitive function: the Honolulu-Asia Aging Study. Pediatrics, 102(3 Pt 1), 602–609.
Acolet, D., Modi, N., et al. (1993). Changes in plasma cortisol and catecholamine concentrations in response to massage in preterm infants. Archives of Disease in Childhood, 68(1 Spec No), 29–31.
Albert, M. S., Jones, K., et al. (1995). Predictors of cognitive change in older persons: MacArthur studies of successful aging. Psychology and Aging, 10(4), 578–589.
Alt, J. E. (1994). The impact of the voting rights act on black and white voter registration in the South. In C. Davidson, and B. Grofman (eds.), Quiet Revolution in the South: The Impact of the Voting Rights Act, 1965–1990 (pp. 351–377). Princeton University Press.
Anstey, K., & Christensen, H. (2000). Education, activity, health, blood pressure and apolipoprotein E as predictors of cognitive change in old age: a review. Gerontology, 46(3), 163–177.
Ardila, A., & Rosselli, M. (1989). Neuropsychological characteristics of normal aging. Developmental Neuropsychology, 5, 307–320.
Ardila, A., Rosselli, M., et al. (1994). Neuropsychological Evaluation of the Spanish Speaker. New York: Plenum.
Ardila, A., Rosselli, M., et al. (2000). Syntactic comprehension, verbal memory, and calculation abilities in Spanish-English bilinguals. Applied Neuropsychology, 7, 3–16.
Ball, K., Berch, D. B., et al. (2002). Effects of cognitive training interventions with older adults—A randomized controlled trial. Jama—Journal of the American Medical Association, 288(18), 2271–2281.
Bao, W. H., Srinivasan, S. R., et al. (1995). The relation of parental cardiovascular-disease to risk-factors in children and young-adults—the Bogalusa Heart-Study. Circulation, 91(2), 365–371.
Barker, D. J. (1990). The fetal and infant origins of adult disease. BMJ, 301(6761), 1111.
Barker, D. J. (1999). Fetal origins of cardiovascular disease. Annals of Medicine, 31(Suppl 1), 3–6.
Barker, D. J., & Martyn, C. (1992). The maternal and fetal origins of cardiovascular disease.” Journal of Epidemiology and Community Health, 46, 8–11.
Barnes, L. L., de Leon, C. F. M., et al. (2004). Social resources and cognitive decline in a population of older African Americans and whites. Neurology, 63(12), 2322–2326.
Barona, A., Reynolds, C. R., et al. (1984). A demographically based index of premorbid intelligence for the Wais-R. Journal of Consulting and Clinical Psychology, 52(5), 885–887.
Bassuk, S. S., Glass, T. A., et al. (1999). Social disengagement and incident cognitive decline in community-dwelling elderly persons. Annals of Internal Medicine, 131(3), 165–173.
Bennett, D. A., Schneider, J. A., et al. (2006). The effect of social networks on the relation between Alzheimer’s disease pathology and level of cognitive function in old people: a longitudinal cohort study. Lancet Neurology, 5(5), 406–412.
Bennett, G. G., & Merritt, M. M., et al. (2004). Perceived racism and affective responses to ambiguous interpersonal interactions among African American Men. American Behavioral Scientist, 47, 963.
Benuskova, L., Diamond, M. E., et al. (1994). Dynamic synaptic modification threshold: computational model of experience-dependent plasticity in adult rat barrel cortex. Proceedings of the National Academy of Sciences of the United States of America, 91(11), 4791–4795.
Berenson, G., Srinivasan, S., et al. (2006). Racial (Black–White) contrasts of risk for hypertensive disease in youth have implications for preventive care: The Bogalusa Heart Study. Ethnicity & Disease, 16(3), S2–S9.
Berkman, L. F. (2000). Which influences cognitive function: living alone or being alone?, editorial. The Lancet, 355, 1291–1292.
Berkman, L. F., & Glass, T. (2000). Social integration, social networks, social support, and health. In L. F. Berkman, & I. Kawachi (Eds.), Social Epidemiology (pp. 137–173). New York: Oxford University Press.
Berkman, L. F., & Macintyre, S. (1997). The measurement of social class in health studies: old measures and new formulations. IARC Scientific Publications (Lyon), 138, 51–64.
Bertrand, M., & Mullainathan, S. (2004). Are Emily and Greg more employable than Lakisha and Jamal? A field experiment on labor market discrimination. The American Economic Review, 94(4), 991–1013.
Bialystok, E., Craik, F. I. M., et al. (2004). Bilingualism, aging, and cognitive control: Evidence from the Simon task. Psychology and Aging, 19(2), 290–303.
Bialystok, E., Craik, F. I. M., et al. (2006). Executive control in a modified antisaccade task: Effects of aging and bilingualism. Journal of Experimental Psychology–Learning Memory and Cognition, 32(6), 1341–1354.
Bobo, L. (2000). Racial attitudes and relations at the close of the twentieth century. In N. Smelser, W. Wilson, & F. Mitchell (Eds.), America Becoming: Racial Trends and Their Consequences (vol. 1, pp. 264–301). Washington DC: National Research Council, National Academy Press.
Bolen, J. C., Rhodes, L., et al. (2000). State-specific prevalence of selected health behaviors, by race and ethnicity—Behavioral Risk Factor Surveillance System, 1997. MMWR CDC Surveillance Summaries, 49(2), 1–60.
Brand, C. (1987). Intelligence-testing—Bryter Still and Bryter. Nature, 328(6126), 110–110.
Braveman, P. A., Cubbin, C., et al. (2005). Socioeconomic status in health research - One size does not fit all. Jama-Journal of the American Medical Association, 294(22), 2879–2888.
Brayne, C., & Calloway, P. (1990). The association of education and socioeconomic status with the Mini Mental State Examination and the clinical diagnosis of dementia in elderly people. Age & Ageing, 19(2), 91–96.
Breslau, J., Aguilar-Gaxiola, S., et al. (2005). Specifying race–ethnic differences in risk for psychiatric disorder in a USA national sample. Psychological Medicine, 36(01), 57–68.
Breslau, J., & Kendler, K. S., et al. (2004). Lifetime risk and persistence of psychiatric disorders across ethnic groups in the United States, Cambridge University Press. 35: 317–327.
Brodkin, K. (2004). How Did Jews Become White Folks? And What That Says About Race in America. New Brunswick, NJ: Routledge.
Brown, L., & Pollittt, E. (1996). Malnutrition, poverty and intellectual development. Scientific American, 274, 38–43.
Butler, S. M., Ashford, J. W., et al. (1996). Age, education, and changes in the Mini-Mental State Exam scores of older women: findings from the Nun Study. Journal of the American Geriatrics Society, 44(6), 675–681.
Byrd, D. A., Sanchez, D., et al. (2005). Neuropsychological test performance among Caribbean-born and U.S.-born African American Elderly: The role of age, education and reading level. Journal of Clinical and Experimental Neuropsychology, 27(8), 1056–1069.
Cagney, K. A., Browning, C. R., et al. (2007). The Latino Paradox in neighborhood context: The case of asthma and other respiratory conditions. American Journal of Public Health, 97(5), 919.
Callahan, C. M., & Hall, K. S., et al. (1996). Relationship of age, education, and occupation with dementia among a community-based sample of African Americans. Archives of Neurology, 53(2), 134–140.
Card, D., & Krueger, A. (1996). School resources and student outcomes: An overview of the literature and new evidence from North and South Carolina. Journal of Economic Perspectives, 10(4), 31–50.
Cerhan, J. R., Folsom, A. R., et al. (1998). Correlates of cognitive function in middle-aged adults. Gerontology, 44(2), 95–105.
Charles, C. Z. (2003). The dynamics of racial residential segregation. Annual Review of Sociology, 29(1), 167–207.
Charles, C. Z., Dinwiddie, G., et al. (2004). The continuing consequences of segregation: Family stress and college academic performance. Social Science Quarterly, 85(5), 1353–1373.
Chatters, L. M. (2000). Religion and health: Public health research and practice. Annual Review of Public Health, 21(1), 335–367.
Cheung, Y. B., Low, L., et al. (2000). Fetal growth and early postnatal growth are related to blood pressure in adults. Hypertension, 36(5), 795–800.
Cho, Y., Frisbie, W. P., et al. (2004). Nativity, duration of residence, and the health of Hispanic adults in the United States. International Migration Review, 38(1), 184–211.
Christensen, H., Korten, A. E., et al. (1997). Education and decline in cognitive performance: compensatory but not protective. International Journal of Geriatric Psychiatry, 12(3), 323–330.
Clark, R., & Anderson, N. B., et al. (1999). Racism as a Stressor for African Americans: A Biopsychosocial Model. The American Psychologist, 54, 805–816.
Conley, D., & Yeung, W. J. (2005). Black–white differences in occupational prestige—Their impact on child development. American Behavioral Scientist, 48(9), 1229–1249.
Corrigan, P. W. (2002). The Paradox of Self-Stigma and Mental Illness. Blackwell Synergy. 9: 35–53.
Cotman, C. W., & Berchtold, N. C. (2002). Exercise: a behavioral intervention to enhance brain health and plasticity. Trends in Neurosciences, 25(6), 295–301.
Cramer, S. C. (1999). Stroke recovery. Lessons from functional MR imaging and other methods of human brain mapping. Physical Medicine & Rehabilitation Clinics of North America, 10(4), 875–886, ix.
Cramer, S. C. (2000). Stroke recovery: how the computer reprograms itself. Neuronal plasticity: the key to stroke recovery. Kananskis, Alberta, Canada, 19–22 March 2000. Molecular Medicine Today, 6(8), 301–303.
Cramer, S. C., Moore, C. I., et al. (2000). A pilot study of somatotopic mapping after cortical infarct. Stroke, 31(3), 668–671.
Crane, P. K., Belle, G., et al. (2004). Test bias in a cognitive test: Differential item functioning in the CASI. Statistics in Medicine, 23(2), 241–256.
Cutler, D. M., Miller, G., et al. (2007). Economics of health and mortality special feature: Evidence on early-life income and late-life health from America’s Dust Bowl era. Proceedings of the National Academy of Sciences, 104(33), 13244–13249.
David, R. J., & Collins, J. W. (1997). Differing birth weight among infants of US-Born Blacks, African-Born Blacks, and US-Born Whites. New England Journal of Medicine, 337(17), 1209.
de la Torre, J. C. (2000). Critically attained threshold of cerebral hypoperfusion: can it cause Alzheimer’s disease? Annals of the New York Academy of Sciences, 903, 424–436.
De Ronchi, D., & Fratiglioni, L., et al. (1998). The effect of education on dementia occurrence in an Italian population with middle to high socioeconomic status. Neurology, 50(5), 1231–1238.
De Stavola, B. L., Nitsch, D., et al. (2006). Statistical issues in life course epidemiology. American Journal of Epidemiology, 163(1), 84–96.
Deary, I. J., & Batty, G. D. (2007). Cognitive epidemiology. J Epidemiol Community Health, 61(5), 378–384.
Del Ser, T., Hachinski, V., et al. (1999). An autopsy-verified study of the effect of education on degenerative dementia. Brain, 122(Pt 12), 2309–2319.
Diamond, M. E., Armstrong-James, M., et al. (1993). Experience-dependent plasticity in adult rat barrel cortex. Proceedings of the National Academy of Sciences of the United States of America, 90(5), 2082–2086.
Dovidio, J. F., & Kawakami, K., et al. (2002). Implicit and explicit prejudice and interracial interaction. Journal of Personality and Social Psychology, 82, 62–68.
Draganski, B., Gaser, C., et al. (2004). Neuroplasticity: Changes in grey matter induced by training. Nature, 427, 311–312.
Duncan, G. J., Brooksgunn, J., et al. (1994). Economic deprivation and early-childhood development. Child Development, 65(2), 296–318.
Dunlop, D. D., Song, J., et al. (2003). Racial/ethnic differences in rates of depression among preretirement adults. American Public Health Association, 93, 1945–1952.
Edland, S. D., Xu, Y., et al. (2002). Total intracranial volume: Normative values and lack of association with Alzheimer’s disease. Neurology, 59(2), 272–274.
Eriksson, J. G., Forsen, T., et al. (2001). Early growth and coronary heart disease in later life: longitudinal study. BMJ, 322(7292), 949–953.
Ertel, K., Glymour, M., et al. (2008). Effects of social integration on preserving memory function in a nationally representative US elderly population. American Journal of Public Health, 98(7), 1215–1220.
Eschbach, K., & Ostir, G. V., et al. (2004). Neighborhood context and mortality among older Mexican Americans: Is there a Barrio advantage? American Journal of Public Health, 94, 1807–1812.
Espino, D. V., Lichtenstein, M. J., et al. (2001). Ethnic differences in Mini-Mental State Examination (MMSE) scores: Where you live makes a difference. Journal of the American Geriatrics Society, 49(5), 538–548.
Evans, D. A., Hebert, L. E., et al. (1997). Education and other measures of socioeconomic status and risk of incident Alzheimer disease in a defined population of older persons. Archives of Neurology, 54(11), 1399–1405.
Fang, J., Madhavan, S., et al. (1996). The association between birthplace and mortality from cardiovascular causes among black and white residents of New York City. New England Journal of Medicine, 335(21), 1545–1551.
Farmer, M. E., Kittner, S. J., et al. (1995). Education and change in cognitive function. The epidemiologic catchment area study. Annals of Epidemiology, 5(1), 1–7.
Ferguson, R. F. (2003). Teachers’ perceptions and expectations and the black-white test score gap. Urban Education, 38(4), 460.
Flynn, J. R. (1984). The mean IQ of Americans—massive gains 1932 to 1978. Psychological Bulletin, 95(1), 29–51.
Fontbonne, A., Berr, C., et al. (2001). Changes in cognitive abilities over a 4-year period are unfavorably affected in elderly diabetic subjects: Results of the Epidemiology of Vascular Aging Study. Diabetes Care, 24(2), 366–370.
Frantz, C. M., Cuddy, A. J. C., et al. (2004). A threat in the computer: The race implicit association test as a stereotype threat experience. Personality and Social Psychology Bulletin, 30(12), 1611.
Fratiglioni, L., Grut, M., et al. (1991). Prevalence of Alzheimer’s disease and other dementias in an elderly urban population: relationship with age, sex, and education. Neurology, 41(12), 1886–1892.
Fratiglioni, L., & Wang, H.-X., et al. (2000). Influence of social network on occurrence of dementia: a community-based longitudinal study. The Lancet, 355, 1315–1319.
Freedman, V. A., Aykan, H., et al. (2001). Aggregate changes in severe cognitive impairment among older Americans: 1993 and 1998. Journals of Gerontology Series B, Psychological Sciences & Social Sciences, 56(2), S100–S111.
Freedman, V. A., Martin, L. G., et al. (2002). Recent trends in disability and functioning among in the United States - A systematic review. Jama-Journal of the American Medical Association, 288(24), 3137–3146.
Fried, L. P., Carlson, M. C., et al. (2004). A social model for health promotion for an aging population: Initial evidence on the experience corps model. Journal of Urban Health-Bulletin of the New York Academy of Medicine, 81(1), 64–78.
Friedland, R. P. (1993). Epidemiology, education, and the ecology of Alzheimer’s disease. Neurology, 43(2), 246–249.
Friedland, R. P., Fritsch, T., et al. (2001). Patients with Alzheimer’s disease have reduced activities in midlife compared with healthy control-group members. Proceedings of the National Academy of Sciences of the United States of America, 98(6), 3440–3445.
Gale, C. R., Walton, S., et al. (2003). Foetal and postnatal head growth and risk of cognitive decline in old age. Brain, 126, 2273–2278.
Gallo, L. C., & Matthews, K. A. (2003). Understanding the association between socioeconomic status and physical health: Do negative emotions play a role? Psychological Bulletin, 129(1), 10–51.
Galobardes, B., Shaw, M., et al. (2006). Indicators of socioeconomic position (part 1). Journal of Epidemiology and Community Health, 60(1), 7–12.
Gatz, M., Svedberg, P., et al. (2001). Education and the risk of Alzheimer’s disease: findings from the study of dementia in Swedish twins. Journals of Gerontology Series B-Psychological Sciences & Social Sciences, 56(5), 292–300.
Geronimus, A. T. (1992). The weathering hypothesis and the health of African-American women and infants: evidence and speculations. Ethnicity and Disease, 2(3), 207–221.
Geronimus, A. T., Hicken, M., et al. (2006). Weathering and age patterns of allostatic load scores among blacks and whites in the United States. AJPH, 96(5), 826–833.
Gianaros, P. J., Jennings, J. R., et al. (2007). Prospective reports of chronic life stress predict decreased grey matter volume in the hippocampus. Neuroimage, 35(2), 795–803.
Giraud, A. L., Price, C. J., et al. (2001). Functional plasticity of language-related brain areas after cochlear implantation. Brain, 124(Pt 7), 1307–1316.
Glymour, M. M. (2007). Commentary: Selected samples and nebulous measures: some methodological difficulties in life-course epidemiology. International Journal of Epidemiology, 36(3), 566–568.
Glymour, M. M., Avendano, M. P., et al. (2007). Is the stroke belt worn from childhood? Risk of first stroke and state of residence in childhood and adulthood. Stroke, 38(9), 2415–2421.
Glymour, M. M., Kawachi, I., et al. (2008). Does childhood schooling affect old age memory or mental status? Using state schooling laws as natural experiments. Journal of Epidemiology and Community Health, 62(6), 532–537.
Godfrey, K. M., & Barker, D. J. (2000). Fetal nutrition and adult disease. American Journal of Clinical Nutrition, 71(5 Suppl), 1344S–1352S.
Goff, P. A. (2005). The space between us: Stereotype threat for Whites in interracial domains. Journal of Personality and Social Psychology, Stanford University.
González, H. M., Whitfield, K. E., et al. (2007). Modified-symbol digit modalities test for African Americans, Caribbean Black Americans, and non-Latino Whites: Nationally representative normative data from the National Survey of American Life. Archives of Clinical Neuropsychology, 22(5), 605–613.
Gortmaker, S. L., & Wise, P. H. (1997). The first injustice: Socioeconomic disparities, health services technology, and infant mortality. Annual Reviews in Sociology, 23(1), 147–170.
Gottfredson, L. S. (2005). What if the hereditarian hypothesis is true? Psychology Public Policy and Law, 11(2), 311–319.
Graves, A. B., Mortimer, J. A., et al. (1996). Head circumference as a measure of cognitive reserve. Association with severity of impairment in Alzheimer’s disease. British Journal of Psychiatry, 169(1), 86–92.
Graves, A. B., Mortimer, J. A., et al. (2001). Head circumference and incident Alzheimer’s disease - Modification by apolipoprotein E. Neurology, 57(8), 1453–1460.
Gray, S. L., Lai, K. V., et al. (1999). Drug-induced cognition disorders in the elderly: Incidence, prevention and management. Drug Safety, 21, 101–122.
Greenberg, M., & Schneider, D. (1992). Region of birth and mortality of blacks in the United-States. International Journal of Epidemiology, 21(2), 324–328.
Greenough, W., & Black, J. (1992). Induction of brain structure by experience: substrates for cognitive development. In M. Gunnar, & C. Nelson (Eds.), Minnesota Symposia on Child Psychology: Developmental Neuroscience (vol. 24, pp. 155–200). Hillsdale, NJ: Erlbaum.
Gregg, E. W., Yaffe, K., et al. (2000). Is diabetes associated with cognitive impairment and cognitive decline among older women? Study of Osteoporotic Fractures Research Group. Archives of Internal Medicine, 160(2), 174–180.
Gump, B. B., Matthews, K. A., et al. (1999). Modeling relationships among socioeconomic status, hostility, cardiovascular reactivity, and left ventricular mass in African American and White children. Health Psychology, 18(2), 140–150.
Guo, G., & Harris, K. M. (2000). The mechanisms mediating the effects of poverty on children’s intellectual development. Demography, 37(4), 431–447.
Haas, S. (2007a). The long-term effects of poor childhood health: an assessment and application of retrospective reports. Demography, 44(1), 113–135.
Haas, S. (2007b). Trajectories of functional health: The ‘long arm’ of childhood health and socioeconomic factors. Social Science and Medicine.
Habeck, C., Hilton, H. J., et al. (2003). Relation of cognitive reserve and task performance to expression of regional covariance networks in an event-related fMRI study of nonverbal memory. Neuroimage, 20(3), 1723–1733.
Hachinski, V. (2007). Stroke and vascular cognitive impairment: A transdisciplinary, translational and transactional approach. Stroke, 38(4), 1396–1403.
Hachinski, V., & Munoz, D. (2000). Vascular factors in cognitive impairment—where are we now? Annals of the New York Academy of Sciences, 903, 1–5.
Hajjar, I., & Kotchen, T. (2003). Regional variations of blood pressure in the united states are associated with regional variations in dietary intakes: The NHANES-III data. Journal of Nutrition, 133(1), 211–214.
Hall, K., Gureje, O., et al. (1998). Risk factors and Alzheimer’s disease: a comparative study of two communities. Australian and New Zealand Journal of Psychiatry, 32(5), 698–706.
Hall, K. S., Gao, S., et al. (2000). Low education and childhood rural residence Risk for Alzheimer’s disease in African Americans. Neurology, 54(1), 95–95.
Hamilton, R. H., & Pascual-Leone, A. (1998). Cortical plasticity associated with Braille learning. Trends in Cognitive Sciences, 2(5), 168–174.
Hanushek, E. A., & Kain, J. F., et al. (2002). New evidence about brown v. board of education: The complex effects of school racial composition on achievement. NBER Working Paper.
Harrell, J. P., Hall, S., et al. (2003). Physiological responses to racism and discrimination: An assessment of the evidence. American Journal of Public Health, 93(2), 243–248.
Harris, M. I., Cowie, C. C., et al. (2002). Higher fasting insulin but lower fasting C-peptide levels in African Americans in the US population. Diabetes Metab Res Rev, 18, 149–155.
Haskins, R., & Isaacs, J., et al. (2008). Getting Ahead or Losing Ground: Economic Mobility in America. Economic Mobility Project (pp. 1–120). Washington DC: Brookings Institution.
Hausdorff, J. M., Levy, B. R., et al. (1999). The power of ageism on physical function of older persons: reversibility of age-related gait changes. Journal of American Geriatric Society, 47(11), 1346–1349.
Hauser, R. M., & Warren, J. R. (1997). Socioeconomic indexes for occupations: A review, update, and critique. Sociological Methodology, 27, 177–298.
Hayward, M. D., & Gorman, B. (2004). The long arm of childhood: the influence of early-life social conditions on men’s mortality. Demography, 41(1), 87–107.
Heaton, R. K., & Grant, I., et al. (1986). Differences in neuropsychological test performance associated with age, education, and sex. Neuropsychological assessment of neuropsychiatric disorders (vol. 1, pp. 100–120). New York: Oxford University Press.
Heaton, R. K., & Miller, S. (2004). Revised comprehensive norms for an expanded Halstead–Reitan battery: Demographically adjusted neuropsychological norms for African American and Caucasian Adults. Lutz, FL.
Hébert, R., Lindsay, J., et al. (2000). Vascular dementia: incidence and risk factors in the Canadian study of health and aging. Stroke, 31(7), 1487–1493.
Hess, T. M., Auman, C., et al. (2003). The impact of stereotype threat on age differences in memory performance. Journals of Gerontology Series B-Psychological Sciences and Social Sciences, 58(1), P3–P11.
Himmelstein, D. U. (2005). Illness and Injury as Contributors to Bankruptcy. Cambridge, MA: Harvard Medical School.
Horner, P. J., Power, A. E., et al. (2000). Proliferation and differentiation of progenitor cells throughout the intact adult rat spinal cord. Journal of Neuroscience, 20(6), 2218–2228.
Hoyert, D. L., & S. National Center for Health (2001). Deaths: Final Data for 1999, Dept. of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics.
Huang, T. L., Carlson, M., Fitzpatrick, A. L., Kuller, L.H., Fried, L. P., Zandi, P. P. (2008). Knee height and arm span: A reflection of early life environment and risk of dementia. Neurology, 70, 1818–1826.
Hummer, R. A., & Benjamins, M. R., et al. (2004). Racial and ethnic disparities in health and mortality among the U.S. elderly population. In N. B. Anderson, R. A. Bulatao, and B. Cohen (Eds.), Critical perspectives on racial and ethnic differences in health in late life (pp. 53–94). National Academies Press.
Ivanovic, D. M., Leiva, B. P., et al. (2000). Long-term effects of severe undernutrition during the first year of life on brain development and learning in Chilean high-school graduates. Nutrition, 16(11–12), 1056–1063.
Jenkins, R., Fox, N. C., et al. (2000). Intracranial volume and Alzheimer disease - Evidence against the cerebral reserve hypothesis. Archives of Neurology, 57(2), 220–224.
Jones, R. N., & Gallo, J. J. (2002). Education and sex differences in the mini-mental state examination effects of differential item functioning. Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 57(6), 548–558.
Kaplan, G. A., Turrell, G., et al. (2001). Childhood socioeconomic position and cognitive function in adulthood. International Journal of Epidemiology, 30(2), 256–263.
Karp, A., Kareholt, I., et al. (2004). Relation of education and occupation-based socioeconomic status to incident Alzheimer’s disease. American Journal of Epidemiology, 159(2), 175.
Katzman, R. (1993). Education and the prevalence of dementia and Alzheimer’s disease. Neurology, 43(1), 13–20.
Katzman, R., Terry, R., et al. (1988). Clinical, pathological, and neurochemical changes in dementia: a subgroup with preserved mental status and numerous neocortical plaques. Annals of Neurology, 23(2), 138–144.
Kaufman, J. S., Cooper, R. S., et al. (1997). Socioeconomic status and health in blacks and whites: the problem of residual confounding and the resilience of race. Epidemiology, 8, 621–628.
Kave, G., Eyal, N., et al. (2008). Multilingualism and cognitive state in the oldest old. Psychology and Aging, 23(1), 70–78.
Kawachi, I., & Berkman, L. F. (2003). Neighborhoods and Health. New York: Oxford University Press, USA.
Kawachi, I., & Subramanian, S. V. (2007). Neighbourhood influences on health. Journal of Epidemiology and Community Health, 61(1), 3–4.
Kempermann, G., & Gage, F. H. (1999a). Experience-dependent regulation of adult hippocampal neurogenesis: effects of long-term stimulation and stimulus withdrawal. Hippocampus, 9(3), 321–332.
Kempermann, G., & Gage, F. H. (1999b). New nerve cells for the adult brain. Scientific American, 280(5), 48–53.
Kempermann, G., Kuhn, H. G., et al. (1998). Experience-induced neurogenesis in the senescent dentate gyrus. Journal of Neuroscience, 18(9), 3206–3212.
Kernie, S. G., Erwin, T. M., et al. (2001). Brain remodeling due to neuronal and astrocytic proliferation after controlled cortical injury in mice. Journal of Neuroscience Research, 66(3), 317–326.
Key, T. J., Verkasalo, P. K., et al. (2001). Epidemiology of breast cancer. The Lancet Oncology, 2(3), 133–140.
Kilander, L., Nyman, H., et al. (1997). Cognitive function, vascular risk factors and education. A cross-sectional study based on a cohort of 70-year-old men. Journal of Internal Medicine, 242(4), 313–321.
Kilander, L., Nyman, H., et al. (1998). Hypertension is related to cognitive impairment: a 20-year follow-up of 999 men. Hypertension, 31(3), 780–786.
King, A. C., Castro, C., et al. (2000). Personal and environmental factors associated with physical inactivity among different racial-ethnic groups of U.S. middle-aged and older-aged women. Health Psychology, 19(4), 354–364.
Kington, R., Carlisle, D., et al. (1998). Racial differences in functional status among elderly US migrants from the South. Social Science & Medicine, 47(6), 831–840.
Kirsch, I. S., Jungeblut, A., et al. (1993). Adult literacy in America: The National Adult Literacy Survey. National Center for Education Statistics, US Department of Education. Washington DC: US Government Printing Office.
Kitayama, N., Vaccarino, V., et al. (2005). Magnetic resonance imaging (MRI) measurement of hippocampal volume in posttraumatic stress disorder: A meta-analysis. Journal of Affective Disorders, 88(1), 79–86.
Klineberg, O. (1935). Race differences. New York: Harper & Brothers.
Kramer, A. F., Colcombe, S. J., et al. (2003). Enhancing brain and cognitive function of older adults through fitness training. Journal of Molecular Neuroscience, 20(3), 213–221.
Krieger, N. (1996). Racial discrimination and blood pressure: the CARDIA Study of young black and white adults. American Journal of Public Health, 86(10), 1370–1378.
Krieger, N. (1999). Embodying inequality: A review of concepts, measures, and methods for studying health consequences of discrimination. International Journal of Health Services, 29(2), 295–352.
Krieger, N., Williams, D., et al. (1997). Measuring social class in US public health research: concepts methodologies, guidelines. Annual Review of Public Health, 18, 341–378.
Kroger, E., Andel, R., et al. (2008). Is complexity of work associated with risk of Dementia?: The Canadian Study of Health and Aging. American Journal of Epidemiology, 167(7), 820–830.
Kuh, D., & Ben-Shlomo, Y. (Eds.) (1997). A lifecourse approach to chronic disease epidemiology: tracing the origins of ill-health from early to adult life. Oxford: Oxford University Press.
Lang, I. A., Llewellyn, D. J., et al. (2007). Neighborhood deprivation, individual socioeconomic status, and cognitive function in older people: Analyses from the English Longitudinal Study of Ageing. Journal of the American Geriatrics Society, 56(2), 191–198.
Lara, M., Gamboa, C., et al. (2005). Acculturation and Latino health in the United States: A Review of the Literature and its Sociopolitical Context. Annual Review of Public Health, 26(1), 367–397.
Lauderdale, D. S., Knutson, K. L., et al. (2006). Objectively measured sleep characteristics among early-middle-aged adults—The CARDIA study. American Journal of Epidemiology, 164(1), 5–16.
Lauer, R. M., & Clarke, W. R. (1989). Childhood risk factors for high adult blood pressure: the Muscatine Study. Pediatrics, 84(4), 633–641.
Launer, L. J., Dinkgreve, M. A., et al. (1993). Are age and education independent correlates of the Mini-Mental State Exam performance of community-dwelling elderly? Journal of Gerontology, 48(6), 271–277.
Launer, L. J., Feskens, E. J., et al. (1996). Smoking, drinking, and thinking. The Zutphen Elderly Study. American Journal of Epidemiology, 143(3), 219–227.
Laurin, D., Verreault, R., et al. (2001). Physical activity and risk of cognitive impairment and dementia in elderly persons. Archives of Neurology, 58(3), 498–504.
Lebedev, M. A., Mirabella, G., et al. (2000). Experience-dependent plasticity of rat barrel cortex: redistribution of activity across barrel-columns. Cerebral Cortex, 10(1), 23–31.
Lecours, A. R., Mehler, J., et al. (1987). Illiteracy and brain damage. 1. Aphasia testing in culturally contrasted populations (control subjects). Neuropsychologia, 25, 231–245.
Lee, Y., & Shinkai, S. (2005). Correlates of cognitive impairment and depressive symptoms among older adults in Korea and Japan. International Journal of Geriatric Psychiatry, 20, 576–586.
Lemann, N. (1991). The Promised Land: The Great Black Migration and how it changed America pp. 1–221. New York: AA Knopf.
Leon, D. A. (2001). Commentary: Getting to grips with fetal programming-aspects of a rapidly evolving agenda. International Journal of Epidemiology, 30(1), 96–98.
Levy, B. R. (2003a). Mind matters: Cognitive and physical effects of aging self-stereotypes. Journals of Gerontology Series B-Psychological Sciences and Social Sciences, 58(4), P203–P211.
Levy, B. R. (2003b). Mind matters: Cognitive and physical effects of aging self-stereotypes. Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 58(4), 203–211.
Liebman, J. B., Katz, L. F., et al. (2004). Beyond treatment effects: Estimating the relationship between neighborhood poverty and individual outcomes in the MTO experiment pp. 1–42. Princeton, NJ: Princeton Industrial Relations Section.
Lindesay, J., Briggs, K., et al. (1989). The Guy’s/Age Concern survey. Prevalence rates of cognitive impairment, depression and anxiety in an urban elderly community. British Journal of Psychiatry, 155, 317–329.
Link, B. G., & Phelan, J. (1995). Social conditions as fundamental causes of disease. Journal of Health & Social Behavior Spec, 35, 80–94.
Link, B. G., & Phelan, J. C. (1996) Understanding sociodemographic differences in health—the role of fundamental social causes. American Journal of Public Health, 86(4), 471–472.
Liu, D., Diorio, J., et al. (1997). Maternal care, hippocampal glucocorticoid receptors, and hypothalamic-pituitary-adrenal responses to stress. Science, 277(5332), 1659–1662.
Liu, D., Diorio, J., et al. (2000). Maternal care, hippocampal synaptogenesis and cognitive development in rats. Nature Neuroscience, 3(8), 799–806.
Liu, H. C., Fuh, J. L., et al. (1998). Prevalence and subtypes of dementia in a rural Chinese population. Alzheimer Disease & Associated Disorders, 12(3), 127–134.
Lleras-Muney, A. (2002). Were compulsory attendance and child labor laws effective? An analysis from 1915 to 1939. Journal of Law & Economics, 45(2), 401–435.
Lleras-Muney, A. (2005). The relationship between education and adult mortality in the US. Review of Economic Studies, 72(1), 189–221.
Loewenstein, D. A., Arguelles, T., et al. (1994). Potential cultural bias in the neuropsychological assessment of the older adult. Journal of Clinical and Experimental Neuropsychology, 16, 623–629.
Logan, J. R. (2001) Ethnic diversity grows, neighborhood integration lags. Albany, NY: Lewis Mumford Center. http://mumford.albany.edu/census/WholePop/WPreport/MumfordReport.pdf (accessed Feb. 11, 2008).
Logan, J. R., & Stowell, J., et al. (2002). Choosing segregation: Racial imbalance in American public schools, 1990–2000, Lewis Mumford Center for Comparative Urban and Regional Research, University at Albany, Business Administration B-10, Albany, NY 12222. Tel: 518-442-4656; Web site: http://www.albany. edu/mumford.
Luo, Y., & Waite, L. J. (2005). The impact of childhood and adult SES on physical, mental, and cognitive well-being in later life. Journals of Gerontology Series B-Psychological Sciences and Social Sciences, 60(2), S93–S101.
Lupien, S. J., de Leon, M., et al. (1998). Cortisol levels during human aging predict hippocampal atrophy and memory deficits. Nature Neuroscience, 1(1), 69–73.
Lyketsos, C. G., Chen, L. S., et al. (1999). Cognitive decline in adulthood: an 11.5-year follow-up of the Baltimore Epidemiologic Catchment Area study. American Journal of Psychiatry, 156(1), 58–65.
Lynch, J., & Kaplan, G. (2000). Socioeconomic position. In L. F. Berkman, & I. Kawachi (Eds.), Social Epidemiology (p. 391). New York: Oxford University Press.
Lynn, R. (1998). In support of the nutrition theory. In U. Neisser (Ed.), The rising curve: long-term gains in IQ and related measures (pp. 207–218). Washington DC: American Psychological Association.
Magri, F., Cravello, L., et al. (2006). Stress and dementia: the role of the hypothalamic–pituitary–adrenal axis. Aging Clinical and Experimental Research, 18(2), 167–170.
Maguire, E. A., Gadian, D. G., et al. (2000). Navigation-related structural change in the hippocampi of taxi drivers. Proceedings of the National Academy of Sciences of the United States of America, 97(8), 4398–4403.
Maguire, E. A., Woollett, K., et al. (2006). London taxi drivers and bus drivers: A structural MRI and neuropsychological analysis. Hippocampus, 16(12), 1091–1101.
Manly, J. J., Jacobs, D. M., et al. (1999). Effect of literacy on neuropsychological test performance in nondemented, education-matched elders. Journal of the International Neuropsychological Society, 5(3), 191–202.
Manly, J. J., Jacobs, D. M., et al. (2002). Reading level attenuates differences in neuropsychological test performance between African American and White elders. Journal of the International Neuropsychological Society, 8, 341–348.
Manly, J. J., Touradji, P., et al. (2003). Literacy and memory decline among ethnically diverse elders. Journal of Clinical and Experimental Neuropsychology, 25(5), 680–690.
Marcuse, P. (1997). The enclave, the citadel, and the ghetto: What has changed in the post-Fordist US city. Urban Affairs Review, 33(2), 228.
Margo, R. A. (1985). Disenfranchisement, school finance, and the economics of segregated schools in the United States south, 1980–1910. New York: Garland Publishing.
Margo, R. A. (1990). Race and schooling in the South, 1880–1950: An economic history. Chicago: University of Chicago Press.
Massey, D. S., Condran, G. A., et al. (1987). The effect of residential segregation on black social and economic well-being. Social Forces, 66(1), 29–56.
Massey, D. S., & Denton, N. A. (1988). The dimensions of residential segregation. Social Forces, 67(2), 281–315.
Massey, D. S., & Denton, N. A. (1989). Hypersegregation in US metropolitan areas: Black and Hispanic segregation along five dimensions. Demography, 26(3), 373–391.
Mays, V. M., Cochran, S. D., et al. (2007). Race, race-based discrimination, and health outcomes among African-Americans. Annual Review of Psychology, 58, 201–225.
Mazumder, B. (2005). Fortunate sons: New estimates of intergenerational mobility in the United States using social security earnings data. Review of Economics and Statistics, 87(2), 235–255.
McDowell, I. (2001). Alzheimer’s disease: Insights from epidemiology. Aging Clinical and Experimental Research, 13(3), 143–162.
McEwen, B. S. (2000). Effects of adverse experiences for brain structure and function. Biological Psychiatry, 48(8), 721–731.
McEwen, B. S. (2001). Plasticity of the hippocampus: Adaptation to chronic stress and allostatic load. In B. A. Sorg, & I. R. Bell (Eds.), The role of neural plasticity in chemical intolerance. Annals of the New York Academy of Sciences (vol. 933, pp. 265–277). New York, NY, US: New York Academy of Sciences, New York Academy of Sciences.
McEwen, B. S. (2002). Sex, stress and the hippocampus: allostasis, allostatic load and the aging process. Neurobiology of Aging, 23(5), 921–939.
McMillen, N. R. (1990). Dark Journey: Black Mississippians in the Age of Jim Crow p. 448. Urbana: University of Illinois Press.
McNeilly, M. D., Robinson, E. L., et al. (1995). Effects of racist provocation and social support on cardiovascular reactivity in African American Women. International Journal of Behavioral Medicine, 2(4), 321–338.
Meaney, M. J., Aitken, D. H., et al. (1988). Effect of neonatal handling on age-related impairments associated with the hippocampus. Science, 239(4841, Pt 1), 766–768.
Meaney, M. J., & Bhatnagar, S., et al. (1996). Early environment and the development of individual differences in the hypothalamic–pituitary–adrenal stress response. In C. R. Pfeffer (Ed.), Severe stress and mental disturbance in children (pp. 85–127). Washington, DC, USA, American Psychiatric Press, Inc.
Meaney, M. J., et al. (1985a). Early postnatal handling alters glucocorticoid receptor concentrations in selected brain regions. Behavioral Neuroscience, 99(4), 765–770.
Meaney, M. J., et al. (1985b). The effects of postnatal handling on the development of the glucocorticoid receptor systems and stress recovery in the rat. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 9(5–6), 731–734.
Meaney, M. J., Mitchell, J. B., et al. (1991). The effects of neonatal handling on the development of the adrenocortical response to stress: Implications for neuropathology and cognitive deficits in later life. Psychoneuroendocrinology, 16(1–3), 85–103.
Mechelli, A., Crinion, J. T., et al. (2004). Neurolinguistics: Structural plasticity in the bilingual brain. Nature, 431(7010), 757–757.
Meghir, C., & Palme, M. (2005). Educational reform, ability, and family background. American Economic Review, 95(1), 414–424.
Mensah, G. A., Mokdad, A. H., et al. (2005). State of disparities in cardiovascular health in the United States. Circulation, 111(10), 1233–1241.
Merritt, M. M., Bennett, G. G., et al. (2006). Perceived racism and cardiovascular reactivity and recovery to personally relevant stress. Health Psychology, 25(3), 364–369.
Mirescu, C., Peters, J. D., et al. (2004). Early life experience alters response of adult neurogenesis to stress. Nature Neuroscience, 7(8), 841–846.
Mirowsky, J., & Ross, C. E. (2005). Education, cumulative advantage, and health. Ageing International, 30(1), 27–62.
Moceri, V. M., Kukull, W. A., et al. (2001). Using census data and birth certificates to reconstruct the early-life socioeconomic environment and the relation to the development of Alzheimer’s disease. Epidemiology, 12(4), 383–389.
Morrison, J. A., Biro, F. M., et al. (1992). Obesity and cardiovascular disease—risk-factors in black-and-white girls—the NHLBI growth and health study. American Journal of Public Health, 82(12), 1613–1620.
Mortel, K. F., Meyer, J. S., et al. (1995). Education and occupation as risk factors for dementias of the Alzheimer and ischemic vascular types. Dementia, 6(1), 55–62.
Mossakowski, K. N. (2003). Coping with perceived discrimination: Does ethnic identity protect mental health? Journal of Health and Social Behavior, 44(3), 318–331.
Nakao, K., & Treas, J. (1994). Updating occupational presige and socioeconomic scores: how the new measures measure up. Sociological Methodology, 24, 1–72.
Nelles, G., Jentzen, W., et al. (2001). Arm training induced brain plasticity in stroke studied with serial positron emission tomography. Neuroimage, 13(6 Pt 1), 1146–1154.
Obisesan, T. O., Vargas, C. M., et al. (2000). Geographic variation in stroke risk in the United States - Region, urbanization, and hypertension in the Third National Health and Nutrition Examination Survey. Stroke, 31(1), 19–25.
Ohayon, M. M., & Vecchierini, M. F. (2002). Daytime sleepiness and cognitive impairment in the elderly population. Archives of Internal Medicine, 162(2), 201.
Oreopoulos, P. (2006). Estimating average and local average treatment effects of education when compulsory schooling laws really matter. American Economic Review, 96(1), 152–175.
Paradies, Y. (2006). A systematic review of empirical research on self-reported racism and health. International Journal of Epidemiology, 35(4), 888.
Parker Frisbie, W., Cho, Y., et al. (2001). Immigration and the health of Asian and Pacific islander adults in the United States. American Journal of Epidemiology, 153(4), 372–380.
Peek, M. K., & O’Neill, G. S. (2001). Networks in later life: An examination of race differences in social support networks. International Journal of Aging & Human Development, 52(3), 207–229.
Perlmann, J., & Waldinger, R. (1997). Second generation decline? Children of immigrants, past and present-a reconsideration. International Migration Review, 31, 893–922.
Petersson, K. M., Reis, A., et al. (2001). Cognitive processing in literate and illiterate subjects: a review of some recent behavioral and functional neuroimaging data. Scandinavian Journal of Psychology, 42(3), 251–267.
Plassman, B. L., Welsh, K. A., et al. (1995). Intelligence and education as predictors of cognitive state in late life: a 50-year follow-up. Neurology, 45(8), 1446–1450.
Portes, A. (1995). The Economic Sociology of Immigration: Essays on Networks, Ethnicity and Entrepreneurship. In A. Portes (Ed.), Children of immigrants: Segmented assimilation and its determinants (pp. 248–280). New York: Russell Sage Foundation.
Portes, A., & Zhou, M. I. N. (1993). The new second generation: Segmented assimilation and its variants. The ANNALS of the American Academy of Political and Social Science, 530(1), 74–96.
Power, C., & Hertzman, C. (1997). Social and biological pathways linking early life and adult disease. British Medical Bulletin, 53, 210–222.
Prencipe, M., Casini, A. R., et al. (1996). Prevalence of dementia in an elderly rural population: effects of age, sex, and education. Journal of Neurology, Neurosurgery & Psychiatry, 60(6), 628–633.
Prince, M., Cullen, M., et al. (1994). Risk-factors for Alzheimers-disease and dementia—a case-control study based on the Mrc Elderly Hypertension Trial. Neurology, 44(1), 97–104.
Qiu, C., Backman, L., et al. (2001). The influence of education on clinically diagnosed dementia incidence and mortality data from the Kungsholmen Project. Archives of Neurology, 58(12), 2034–2039.
Raiha, I., Kaprio, J., et al. (1998). Environmental differences in twin pairs discordant for Alzheimer’s disease. Journal of Neurology, Neurosurgery & Psychiatry, 65(5), 785–787.
Ratcliff, G., Ganguli, M., et al. (1998). Effects of literacy and education on measures of word fluency. Brain & Language, 61, 115–122.
Ravelli, A. C., van der Meulen, J. H., et al. (2000). Infant feeding and adult glucose tolerance, lipid profile, blood pressure, and obesity. Archives of Disease in Childhood, 82(3), 248–252.
Read, J. G., & Emerson, M. O. (2005). Racial context, Black immigration and the U. S. Black/White health disparity. Social Forces, 84(1), 181–199.
Redline, S., Kirchner, H. L., et al. (2004). The effects of age, sex, ethnicity, and sleep-disordered breathing on sleep architecture. Archives of Internal Medicine, 164, 406–418.
Reed, D., & Yano, K. (1997). Racial and Ethnic Differences in the Health of Older Americans. In L. Martin, & B. Soldo (Eds.), Cardiovascular disease among elderly Asian Americans (pp. 270–284). Washington, DC: National Academy Press.
Reitz, J. G., & Sklar, S. M. (1997). Culture, race, and the economic assimilation of immigrants. Sociological Forum, 12, 233–277.
Riach, P. A., & Rich, J. (2002). Field experiments of discrimination in the market place. The Economic Journal, 112(483), 480–518.
Richards, M., Brayne, C., et al. (2000). Cognitive function in UK community-dwellings African-Caribbean and white elders: a pilot study. International Journal of Geriatric Psychiatry, 16, 518–527.
Richards, M., Hardy, R., et al. (2001). Birth weight and cognitive function in the British 1946 birth cohort: longitudinal population based study. BMJ, 322(7280), 199–203.
Richards, M., Hardy, R., et al. (2003). Does active leisure protect cognition? Evidence from a national birth cohort. Social Science & Medicine, 56(4), 785–792.
Richman, L. S., Bennett, G. G., et al. (2007). Discrimination, dispositions, and cardiovascular responses to stress. Health Psychology, 26(6), 675–683.
Rocca, W. A. (1990). Prevalence of clinically diagnosed Alzheimer’s disease and other dementing disorders: a door-to-door survey in Appignano, Macerata Province, Italy. Neurology, 40(4), 626–631.
Rosenzweig, M. R., & Bennett, E. L. (1996). Psychobiology of plasticity: effects of training and experience on brain and behavior. Behavioural Brain Research, 78(1), 57–65.
Ross, C. E. (2000). Walking, exercising, and smoking: does neighborhood matter? Social Science & Medicine, 51(2), 265–274.
Ross, C. E., & Mirowsky, J. (1999). Refining the association between education and health: The effects of quantity, credential, and selectivity. Demography, 36(4), 445–460.
Rosselli, M. (1993). Neuropsychology of illiteracy. Special issue: The origins of cognitive activity. Behavioural Neurology, 6, 107–112.
Rosselli, M., Ardila, A., et al. (1990). Neuropsychological assessment in illiterates. II. Language and praxic abilities. Brain and Cognition, 12, 281–296.
Ruggles, S., Sobek, M., Alexander, T., et al. (2004). Intergrated Public Use Microdata Series: Version 3.0 [Machine-readable database]. Minneapolis, MN: Minnesota Population Center.
Ryan, E. L., Baird, R., et al. (2005). Neuropsychological impairment in racial/ethnic minorities with HIV infection and low literacy levels: effects of education and reading level in participant characterization. Journal of the International Neuropsychological Society, 11(7), 889–898.
Sampson, R., & Sharkey, P., et al. (2007). Durable effects of concentrated disadvantage on verbal ability among African-American children. Proceedings of the National Academy of Sciences.
Sapolsky, R. M., Uno, H., et al. (1990). Hippocampal damage associated with prolonged glucocorticoid exposure in primates. Journal of Neuroscience, 10(9), 2897–2902.
Scarmeas, N., Levy, G., et al. (2001). Influence of leisure activity on the incidence of Alzheimer’s Disease. Neurology, 57(12), 2236–2242.
Schaie, K. W. (1989). The hazards of cognitive aging. Gerontologist, 29(4), 484–493.
Schmand, B., Smit, J. H., et al. (1997). The effects of intelligence and education on the development of dementia. A test of the brain reserve hypothesis. Psychological Medicine, 27(6), 1337–1344.
Schneider, D., Greenberg, M. R., et al. (1997). Region of birth and mortality from circulatory diseases among black Americans. American Journal of Public Health, 87(5), 800–804.
Schofield, P. W., Logroscino, G., et al. (1997). An association between head circumference and Alzheimer’s disease in a population-based study of aging and dementia. Neurology, 49(1), 30–37.
Schofield, P. W., Mosesson, R. E., et al. (1995). The age at onset of Alzheimer’s disease and an intracranial area measurement. A relationship. Archives of Neurology, 52(1), 95–98.
Schooler, C. (1998). Environmental complexity and the Flynn effect. In U. Neisser (Ed.), The rising curve: long-term gains in IQ and related measures (pp. 67–79). Washington DC: American Psychological Association.
Schooler, C., & Mulatu, M. S. (2001). The reciprocal effects of leisure time activities and intellectual functioning in older people: A longitudinal analysis. Psychology and Aging, 16(3), 466–482.
Schulman, K., Berlin, J., et al. (1999). The effect of race and sex on physicians’ recommendations for cardiac catheterization. The New England Journal of Medicine, 340(8), 618–626.
Seeman, T. E., Lusignolo, T. M., et al. (2001). Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur studies of successful aging. Health Psychology, 20(4), 243–255.
Sellers, A. H., & Burns, W. J., et al. (2002). Differences in Young Children's IQs on the Wechsler Preschool and Primary Scale of Intelligence-Revised as a Function of Stratification Variables, Lawrence Earlbaum. 9: 65–73.
Sellers, R. M., Caldwell, C. H., et al. (2003). Racial identity, racial discrimination, perceived stress, and psychological distress among African American young adults. Journal of Health and Social Behavior, 44(3), 302–317.
Sharkey, P. (2008). The intergenerational transmission of context. American Journal of Sociology, 113(4), 931–969.
Shumaker, S. A., Legault, C., et al. (2003). Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women. The Women’s Health Initiative Memory Study: A Randomized Controlled Trial. American Medical Association, 289, 2651–2662.
Siegel, P., & Martin, E., et al. (2001). Language Use and Linguistic Isolation: Historical Data and Methodological Issues. Statistical Policy Working Paper 32: 2000 Seminar on Integrating Federal Statistical Information and Processes. Washington DC: 167–190.
Silverstein, M., & Waite, L. J. (1993). Are blacks more likely than whites to received and provide social support in middle and old-age—yes, no, and maybe so. Journals of Gerontology, 48(4), S212–S222.
Simon, M., Czeh, B., et al. (2005). Age-dependent susceptibility of adult hippocampal cell proliferation to chronic psychosocial stress. Brain Research, 1049(2), 244–248.
Singh, G. K., & Siahpush, M. (2002). Ethnic–immigrant differentials in health behaviors, morbidity, and cause-specific mortality in the United States: An analysis of two national data bases. Human Biology, 74(1), 83–109.
Small, M. L. (2007). Racial differences in networks: Do neighborhood conditions matter? Social Science Quarterly, 88(2), 320–343.
Smedley, B. D., & Stith, A. Y., et al. (2003). Unequal treatment: Confronting racial and ethnic disparities in health care. National Academy Press.
Smith, J. P. (1984). Race and human capital. American Economic Review, 4, 685–698.
Smith, J. P., & Welch, F. (1977). Black-White male wage ratios: 1960–1970. American Economic Review, 67, 323–328.
Smyth, K. A., Fritsch, T., et al. (2004). Worker functions and traits associated with occupations and the development of AD. Neurology, 63(3), 498–503.
Snowdon, D. A., Greiner, L. H., et al. (1997). Brain infarction and the clinical expression of Alzheimer disease. The Nun Study. JAMA, 277(10), 813–817.
Snowdon, D. A., Greiner, L. H., et al. (2000). Linguistic ability in early life and the neuropathology of Alzheimer’s disease and cerebrovascular disease. Findings from the Nun Study. Annals of the New York Academy of Sciences, 903, 34–38.
Snowdon, D. A., Kemper, S. J., et al. (1996). Linguistic ability in early life and cognitive function and Alzheimer’s disease in late life. Findings from the Nun Study. Jama, 275(7), 528–532.
Snowdon, D. A., Ostwald, S. K., et al. (1989). Education, survival, and independence in elderly Catholic sisters, 1936–1988. American Journal of Epidemiology, 130(5), 999–1012.
Spencer, S. J., Steele, C. M., et al. (1999). Stereotype threat and women’s math performance. Journal of Experimental Social Psychology, 35(1), 4–28.
Steele, C. M., & Aronson, J. (1995). Stereotype threat and the intellectual test performance of African Americans. J Pers Soc Psychol, 69(5), 797–811.
Stern, Y. (2002). What is cognitive reserve? Theory and research application of the reserve concept. Journal of the International Neuropsychological Society, 8(03), 448–460.
Stern, Y. (2006). Cognitive reserve and Alzheimer disease. Alzheimer Disease & Associated Disorders, 20(2), 112–117.
Stern, Y., Albert, S., et al. (1999). Rate of memory decline in AD is related to education and occupation: cognitive reserve? Neurology, 53(9), 1942–1947.
Stern, Y., Alexander, G. E., et al. (1992). Inverse relationship between education and parietotemporal perfusion deficit in Alzheimer’s disease. Annals of Neurology, 32(3), 371–375.
Stern, Y., Gurland, B., et al. (1994). Influence of education and occupation on the incidence of Alzheimer’s disease. Jama, 271(13), 1004–1010.
Stewart, R., Johnson, J., et al. (2002). The distribution of Mini-Mental State Examination scores in an older African-Caribbean population compared to MRC CFA Study norms. International Journal of Geriatric Psychiatry, 17, 745–751.
Stewart, R., Richards, M., et al. (2001). Vascular risk and cognitive impairment in an older, British, African-Caribbean population. Journal of the American Geriatrics Society, 49(3), 263–269.
Stimpson, J. P., Ju, H., et al. (2007). Neighborhood deprivation and health risk behaviors in NHANES III. American Journal of Health Behavior, 31(2), 215–222.
Subramanian, S. V., & Glymour, M. M., et al. (2007). Identifying causal ecologic effects on health: a methodological assessment. Macrosocial determinants of health. S. Galea, Springer Media.
Sundquist, J., Winkleby, M. A., et al. (2001). Cardiovascular disease risk factors among older black, Mexican-American, and white women and men: an analysis of NHANES III, 1988–1994. JAGS, 49, 109–116.
Tang, M. X., Cross, P., et al. (2001). Incidence of AD in African-Americans, Caribbean Hispanics, and Caucasians in northern Manhattan. Neurology, 56(1), 49–56.
Taylor, R. J., Chatters, L. M., et al. (1996). Black and white differences in religious participation: a multisample comparison. Journal for the Scientific Study of Religion, 35(4), 403–410.
Teresi, J. A., Kleinman, M., et al. (2000). Modern psychometric methods for detection of differential item functioning: application to cognitive assessment measures. Statistics in Medicine, 19(11–12), 1651–1683.
Theorell, T. (2000). Working conditions and health. In L. F. Berkman, & I. Kawachi (Eds.), Social Epidemiology (pp. 95–117). New York: Oxford University Press.
Thulborn, K. R., Carpenter, P. A., et al. (1999). Plasticity of language-related brain function during recovery from stroke. Stroke, 30(4), 749–754.
Tolnay, S. E. (1998). Educational selection in the migration of southern blacks, 1880–1990. Social Forces, 77(2), 487–514.
Tolnay, S. E. (2003). The African American “Great Migration” and beyond. Annual Review of Sociology, 29, 209–232.
Tsolaki, M., Fountoulakis, K., et al. (2005). Risk factors for clinically diagnosed Alzheimer’s disease: A case-control study of a Greek Population. International Psychogeriatrics, 9(03), 327–341.
Turner, S., & Bound, J. (2003). Closing the gap or widening the divide: The effects’ of the GI Bill and World War II on the educational outcomes of black Americans. Journal of Economic History, 63(1), 145–177.
U.S. Department of Health and Human Services (2004). The Health Consequences of Smoking: A Report of the Surgeon General, Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health.
Valentino, N. A., & Sears, D. O. (2005). Old times there are not forgotten: Race and partisan realignment in the contemporary South. American Journal of Political Science, 49(3), 672–688.
van Praag, H., Kempermann, G., et al. (2000). Neural consequences of environmental enrichment. Nature Reviews Neuroscience, 1(3), 191–198.
Wadsworth, M. (1997). Health inequalities in the life course perspective. Social Science and Medicine, 44(6), 859–869.
Waters, M. C. (2000). Black Identities: West Indian Immigrant Dreams and American Realities, Harvard University Press.
Weaver, I. C. G., Cervoni, N., et al. (2004). Epigenetic programming by maternal behavior. Nature Neuroscience, 7(8), 847–854.
Wechsler, D. (1997). Wechsler Adult Intelligence Scale—III. San Antonio, TX: Psychological Corporation.
Weiss, B. D., Reed, R., et al. (1995). Literacy and performance on the Mini-Mental State Examination. Journal of the American Geriatric Society, 43, 807–810.
Weisskopf, M. G., Wright, R. O., et al. (2004). Cumulative lead exposure and prospective change in cognition among elderly men: The VA Normative Aging Study. American Journal of Epidemiology, 160(12), 1184–1193.
Welch, F. (1966). Measurement of the quality of education. American Economic Review, 56, 379–392.
Welch, F. (1973). Black–White differences in returns to schooling. American Economic Review, 63(5), 893–907.
Weuve, J., Kang, J. H., et al. (2004). Physical activity, including walking, and cognitive function in older women. Jama—Journal of the American Medical Association, 292(12), 1454–1461.
Whalley, L. J., Dick, F. D., et al. (2006). A life-course approach to the aetiology of late-onset dementias. Lancet Neurology, 5(1), 87–96.
Whalley, L. J., Starr, J. M., et al. (2000). Childhood mental ability and dementia. Neurology, 55(10), 1455–1459.
Wheeler, L. R. (1970). Cross-cultural studies of behavior. In I. Al-Issa, & W. Dennis (Eds.), A trans-decade comparison of the IQ’s of Tennessee mountain children (pp. 120–133). New York: Holt, Rinehart & Winston.
White, L., Petrovitch, H., et al. (1996). Prevalence of dementia in older Japanese-American men in Hawaii: The Honolulu-Asia Aging Study. Jama, 276(12), 955–960.
Whitfield, K. E., & Baker-Thomas, T. (1999). Individual differences in aging minorities. International Journal of Aging and Human Development, 48, 73–79.
Whitfield, K. E., Fillenbaum, G. G., et al. (2000). The effect of race and health-related factors on naming and memory. The MacArthur Studies of Successful Aging. Journal of Aging & Health, 12(1), 69–89.
Whitfield, K. E., & Wiggins, S. A. (2003). The impact of desegregation on cognition among older African Americans. Journal of Black Psychology, 29, 275–291.
Wight, R. G., Aneshensel, C. S., et al. (2006). Urban neighborhood context, educational attainment, and cognitive function among older adults. American Journal of Epidemiology, 163(12), 1071–1078.
Williams, D. R., & Jackson, P. B. (2005). Social sources of racial disparities in health. Health Affairs, 24(2), 325.
Williams, D. R., Takeuchi, D. T., et al. (1992). Socioeconomic status and psychiatric disorder among Blacks and Whites. Social Forces, 71(1), 179–194.
Williams, D. R., & Williams-Morris, R. (2000). Racism and mental health: The African American experience. Ethnicity & Health, 5(3), 243–268.
Williams, R., Nesiba, R., et al. (2005). The changing face of inequality in home mortgage lending. Social Problems, 52(2), 181–208.
Williams, W. (1998). Are we raising smarter children today? School and home influences on IQ. The rising curve: long-term gains in IQ and related measures. U. Neisser. Washington DC, American Psychological Association: 125–154.
Willis, S. L., Tennstedt, S. L., et al. (2006). Long-term effects of cognitive training on everyday functional outcomes in older adults. Jama—Journal of the American Medical Association, 296(23), 2805–2814.
Wilson, R. S., Krueger, K. R., et al. (2007). Loneliness and risk of Alzheimer disease. Archives of General Psychiatry, 64(2), 234–240.
Wilson, R. S., Li, Y., et al. (2004). Education and the course of cognitive decline in Alzheimer disease. Neurology, 63(7), 1198–1202.
Wolinsky, F. D., Unverzagt, F. W., et al. (2006). The ACTIVE cognitive training trial and health-related quality of life: Protection that lasts for 5 years. Journals of Gerontology Series a-Biological Sciences and Medical Sciences, 61(12), 1324–1329.
Young, D., Lawlor, P. A., et al. (1999). Environmental enrichment inhibits spontaneous apoptosis, prevents seizures and is neuroprotective. Nature Medicine, 5(4), 448–453.
Zavodny, M. (2003). Race, wages, and assimilation among Cuban immigrants. Population Research and Policy Review, 22, 201–219.
Acknowledgments
Maria Glymour was a Robert Wood Johnson Health and Society Scholar at Columbia University when this was written. The authors gratefully acknowledge funding from a Robert Wood Johnson Health and Society Program seed grant and the National Institute on Aging R01 AG016206.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Glymour, M.M., Manly, J.J. Lifecourse Social Conditions and Racial and Ethnic Patterns of Cognitive Aging. Neuropsychol Rev 18, 223–254 (2008). https://doi.org/10.1007/s11065-008-9064-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11065-008-9064-z