Abstract
Chronic neuroinflammation is a pathological feature of neurodegenerative diseases. Inhibition of microglia-mediated neuroinflammation might be a potential strategy for neurodegeneration. Matairesinol, a dibenzylbutyrolactone plant lignan, presents in a wide variety of foodstuffs. It has been found to possess anti-angiogenic, anti-oxidative, anti-cancer and anti-fungal activities. In the present study, we investigated the anti-neuroinflammation effects of matairesinol on lipopolysaccharide (LPS)-induced BV2 microglia cells and the related molecular mechanisms. The results showed that matairesinol inhibited microglia activation by reducing the production of nitric oxide, the expression of inducible nitric oxide synthase and cyclooxygenase-2 in a concentration-dependent manner (6.25, 12.5, 25 μM). In the molecular signaling pathway, LPS-induced nuclear factor-kappa B (NF-κB) transcriptional activity and translocation into the nucleus were remarkably suppressed by matairesinol through the inhibition of the extracellular signal-regulated kinase (ERK)1/2 signal transduction pathways, but not p38 MAPK or c-jun N-terminal kinase (JNK). Meanwhile, matairesinol also blocked LPS-mediated microglia migration and this was associated with inhibition of LPS-induced Src phosphorylation as well as Src expression in a concentration-dependent manner. Taken together, these results suggest that matairesinol inhibited inflammatory response and migration in LPS-induced BV2 microglia, and the mechanisms may be associated with the NF-κB activation and modulation of Src pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Microglia are resident macrophages that undertake an indispensable function for maintaining the homeostasis in central nervous system (CNS). As sensors of pathological changes, microglia quickly transfer from a resting to an activated status, and thus restore the homeostasis of the CNS [1]. The activated microglia characterized by sustained release of cytotoxic molecules including inducible nitric oxide synthase/nitric oxide (iNOS/NO), cyclooxygenase-2 (COX2) and tumor necrosis factor alpha (TNF-α) will be incurred, which usually results in chronic neuroinflammation. This is a common phenomenon observed in many neurological disorders such as stroke, Parkinson’s disease, and Alzheimer’s disease (AD). Therefore, pharmacological interference with microglial activation and production of pro-inflammatory mediators and cytokines is considered to be a promising strategy in the onset and progression of neurodegenerative diseases.
Lipopolysaccharide (LPS), as major pathogenic component of the Gram-negative bacteria, is one of the most potent stimuli that induce microglial activation [2, 3]. Once being primed by LPS, microglia gives rise to an array of inflammatory biomarkers like iNOS/NO, COX2 and cytokines [4, 5]. Nuclear factor-κB (NF-κB), a vital activator in inflammatory processes, can regulate the expression of inflammatory cytokines and mediators in many cell types, including microglia to ignite relevant downstream signaling pathways. Under normal condition of cells, NF-κB exists in an inactive form in the cytoplasm. A large number of stimuli such as LPS can lead to the activation of NF-κB, then NF-κB translocates into the nucleus and regulates the transcription of inflammation-associated genes such as target genes TNF-α, iNOS and COX2 [6]. The mitogen-activated protein kinases (MAPKs) are known to play an important role in cellular processes including differentiation, stress response and inflammation process. LPS activates MAPK signaling pathways, which can promote pro-inflammatory cytokines production and expression [7]. In addition, adenosine 5′-monophosphate-activated protein kinase (AMPK) is a key regulator of energy homeostasis and metabolic stress [8]. In particular, recent studies reveal that AMPK might also be involved in modulating inflammatory response [9, 10]. It has been demonstrated that AMPK activation (p-AMPK) suppressed LPS-induced proinflammatory secretion and NF-κB activation in glial cells [11]. Therefore, both NF-κB and MAPK signaling pathways are vital approaches to regulate inflammatory responses, and are also regarded as suitable targets for screening anti-inflammatory compounds.
On the other hand, in response to stimulation, microglia displayed enhanced motility from one place to the next which is a hallmark of proinflammatory and chronic activation during the early phases of neurodegeneration. Src family kinases (SFKs), a group of nonreceptor protein tyrosine kinases, promote inflammatory processes under various pathological conditions. Src is the prototypic member of SFKs that comprises Src, Yes, Fyn, Lck, Lyn, Fgr, Hck, Blk, and Yrk [12]. Most importantly, Src activation is involved in cell adhesion and motility. When the C-terminal tyrosine (Tyr416) of Src is phosphorylated, it is located in the activation loop of the kinase domain, increases enzyme activity [13, 14]. Overall SFKs represent key components making up the microglial activation cascade.
Matairesinol (MAT, Fig. 1a), a dibenzylbutyrolactone lignan, has been reported to possess diverse therapeutic potential as anti-oxidative, estrogenic, or anti-estrogenic activities [15, 16]. Up to date, there is no evidence on the protective effect of MAT against microglia activation caused by LPS and the molecular mechanism. Our results confirmed the beneficial impact of MAT in LPS-induced BV2 microglia cells and showed that MAT could effectively inhibit the pro-inflammatory mediators production and cells migration, which possibly correlates with the inhibition of NF-κB activation and modulation of Src pathway, but there was no significant effect on AMPK activation, suggesting MAT is a powerful potential neuroprotective agent.
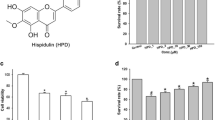
Structure and inhibitory effect of MAT on NO production and iNOS and COX2 expression in LPS-stimulated microglia cells. a Chemical structure of MAT. b Cell viability of BV2 cells exposed to MAT (10–100 μM) alone for 24 h was measured by MTT assay. BV2 microglia cells were pretreated with various concentrations of MAT for 4 h and then stimulated by LPS (1 μg/ml) for 24 h. c The production of NO were analyzed in LPS-stimulated BV2 and primary microglia cells as described in “Materials and Methods” section. d, e iNOS and COX2 expression were measured by western blot, GAPDH was used as loading control. Data represents mean ± SE # p < 0.05 vs. control; ^ p < 0.05 vs. LPS treated cells. Data were from at least three independent experiments. f BV2 microglial cells were immunostained with antibodies against iNOS and COX2 followed by FITC-labeled secondary antibodies and mounted with fluorescence mounting medium with DAPI. Antibody staining intensity was observed under a Zeiss fluorescence microscope for FITC and DAPI. Representative photomicrographs showed iNOS (red) and COX2 (green) protein expression in LPS-stimulated BV2 (magnification ×200). (Color figure online)
Materials and Methods
Reagents and Materials
MAT was provided by Hu’s group (Department of Natural Products Chemistry, School of Pharmacy, Fudan University) and the purity is over 99% determined by high performance liquid chromatography. MAT was dissolved in dimethyl sulfoxide (DMSO) and then diluted with DMEM to the desired concentration, the final concentration of DMSO was less than 0.1%. 3-(4, 5-dimetrylthiazol)-2, 5-diphenyltetrazolium bromide (MTT), DMSO and LPS (E. coli 055:B5) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Dulbecco’s modified Eagle’s medium was purchased from HyClone. Penicillin–streptomycin and fetal bovine serum (FBS) were purchased from GIBCO-BRL (USA). Antibodies for western blotting against total- and phospho (p)- c-Jun N-terminal kinase 1/2 (JNK1/2) (Thr183/Tyr185), total- and p-p38 (Thr180/Tyr182), total- and p-ERK1/2 (Thr202/Tyr204), NF-κB p65 and p-p65 (Ser563), COX2, Src, p-Src (Tyr416), AMPK, p-AMPK (Thr172) and FAK were purchased from Cell Signaling Technology (Danvers, MA, USA) and iNOS, HCK from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Antibodies for immunofluorescence staining against COX2, NF-κB p65 were from were purchased from Cell Signaling Technology (Danvers, MA, USA) and iNOS from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Primary Cultures of Microglial Cells
All animals and the experimental protocol conformed to the Animal Welfare Act Guide for Use and Care of Laboratory Animals, and was approved by Institutional Animal Care and Use Committee (IACUC),School of Pharmacy, Fudan University, China. Microglia cultures were prepared as previously described [17]. Transferred the intact whole brain from neonatal mice to a petri dish containing cold DMEM, and then removed the meningeal layer from the brain. Isolated cortex was cut into fine pieces and homogenized in 0.125% Trypsase/EDTA (Gibco, USA) for 8 min at 37 °C. The suspension was centrifuged for 5 min to 1500 rpm, discarded the supernatant by aspiration and suspended the pellet. The cell suspension was seeded in Poly-D-Lysine covered 75 cm2 flasks with 10 ml/each and incubated at 37 °C in water saturated 5% CO2:95% air atmosphere for about 7 days, without changing the culture medium. After that time, patted the flasks for 15 min and cells were then harvested at day 7. Centrifuged the cell suspension for 5 min to 1500 rpm, discarded the supernatant and suspended the pellet. Seeded the cells in plate and exchanged the culture medium to purify microglia cells. Between 90 and 95% of the isolated cells stained positive for CD11b.
BV-2 Cell Culture
BV-2 microglia cells were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured in DMEM supplemented with 10% FBS, 100 units/ml penicillin–streptomycin at 37 °C in a humidified atmosphere under 5% CO2.
Cell Viability Assay
Cell viability was evaluated by 3-(4, 5-dimetrylthiazol)-2, 5-diphenyltetrazolium bromide (MTT) assay. However, this method can be influenced by various conditions including acidic pH, polyphenols, pyruvate analog, and nanomaterials. BV2 cells were plated in a 96-well plate at a density of 2 × 104 cells/well. After serum starved overnight, BV-2 microglia cells were exposed to a gradient concentrations of MAT for 24 h. The results were expressed as fold changes by normalizing the data to the control values.
Measurement of Nitric Oxide (NO) Production
NO production from activated BV-2 microglia cells and primary microglial was quantified by nitrite accumulation in the culture medium using the Griess reaction. BV2 (4 × 104 cells/well) or primary microglial cells (1.5 × 104 cells/well) were seeded on a 96 well plate and treated with 1 μg/ml LPS for 24 h in the presence or absence of different concentrations of MAT. Then the collected supernatant was mixed with an equal volume of Griess reagent (Beyotime Biotechnology, China). NaNO2 was used to generate a standard curve, and nitrite production was determined by measuring the optical density at 540 nm by a microplate reader (M1000 PRO, TECAN, Austria GmbH, Austria).
Preparation of Whole Cell Lysates and Cytoplasmic and Nuclear Extractions
For western blot analysis, cells were seeded in a 6-well plate at a density of 6 × 105 cells/well and stimulated with or without LPS (1 μg/ml) in the presence or absence of MAT for various durations of time. BV-2 cells were treated for 24 h for the detection of iNOS and COX2, 15–90 min for detection of MAPKs, 3 h for detection of p65 translocation. Cultured cells were washed with ice-cold PBS and lysed in cold RIPA buffer (Sigma-Aldrich) supplemented with protease inhibitor cocktail (Roche Diagnostics, Rotkreuz, Switzerland). NE-PER Nuclear and Cytoplasmic Extraction kit was used for the nuclear protein extraction (Pierce, Rockford, IL, USA) according to the manufacturer’s instructions. The extract was centrifuged at 10,000×g for 15 min at 4 °C to remove cell debris before samples were prepared for western blotting analysis.
Western Blotting
Sample concentration was determined using the BCA protein assay (Pierce, Rockford, IL). Western blotting was performed as previously described [18]. Primary antibodies as p-JNK1/2 (Thr183/Tyr185), p-p38 (Thr180/Tyr182), p-ERK1/2 (Thr202/Tyr204), p-AMPK (Thr172), AMPK, JNK1/2, ERK1/2, p38, COX2, NF-κB p65 and p-p65 (All in 1:1000 dilution), iNOS (1:500), Lamin A/C (1:2000) and GAPDH (1:10,000) were performed overnight at 4 °C. The corresponding HRP-conjugated secondary antibodies (1:5000) are incubated for 2 h at room temperature. Protein levels are determined by enhanced chemiluminescence system (ThermoFisher Scientific, Shanghai, China) and quantified with the software Alpha Imager (Alpha Innotech Corp, San Leandro, CA). The results were determined as fold changes by normalizing the data to the control values.
Immunofluorescence Staining
BV-2 microglia cells were grown on cover slips in 24-well plates. Then, the cells were fixed using a 4% paraformaldehyde solution for 30 min at room temperature. The primary antibodies were applied overnight at 4 °C. Alexa Fluor 488-conjugated goat anti-rabbit or 568-conjugated goat anti-mouse IgG (1:200; Invitrogen) were applied according to the manufacturer’s instructions, and the cells were counterstained for nuclei with 4′,6-diamidino-2-phenylindole (DAPI). All multicolor fluorescence images were recorded from an Axio Scope.A1 microscope (Carl Zeiss Imaging Systems) with 20×/0.45 objective and processed by AxioVision Version 4.6.3.0 (Carl Zeiss Imaging Systems).
Cell Migration Assay
The migration behavior of BV2 microglia was tested using a modified Boyden chamber. Cells were pretreated with MAT (25 μM) for 4 h, then total of 2 × 105 cells were counted and loaded under serum-starved conditions in the upper chamber with polycarbonate filter (8 μm pore size). The inserts were placed into the bottom a 24-well plate that was filled with DMEM medium FBS free in the presence or absence of LPS. The chamber was incubated at 37 °C for 24 h to initiate migration, and then cells were fixed with 4% formaldehyde for 3 min and non-migrating cells were removed from the upper surface of the filter. The migrated cells on the under surface of the filter were stained with viola crystalline and measured by counting the number from five randomly chosen fields under a light microscope (Carl Zeiss Imaging Systems).
Statistical Analysis
Data are shown as mean ± standard error of the mean (SE) from at least three independent experiments. Statistical software used was Prism version 5 (GraphPad Software, San Diego, CA). Differences between mean values of multiple groups were analyzed by ANOVA with Dunnett’s test for post hoc comparisons and Tukey’s or Dunn’s test were used for parametric and nonparametric data respectively. Significance was defined as p < 0.05.
Results
Effect of MAT on Cell Viability of BV2 Microglia
The cytotoxic effect of a gradient concentration MAT (10, 20, 40, 80 and 100 μM) on BV2 microglia was evaluated 24 h after the treatment by MTT-based viability assay. Figure 1b indicated that MAT at the concentration up to 100 μM didn’t produce any cytotoxic effect on BV2 microglia. Considerably nontoxic lower concentrations of MAT (6.25, 12.5, 25 μM) were used for the subsequent experiments in further study.
MAT Reduced NO Release in LPS-Stimulated BV2 and Primary Microglial Cells
To investigate the anti-inflammatory effects of MAT, the effects of MAT on LPS-induced NO production were detected firstly. As shown in Fig. 1c, compared to the control group, the production of NO was dramatically elevated in LPS-treated group. As expected, MAT suppressed LPS-induced NO production in a concentration-dependent manner. MAT at 25 μM concentration significantly inhibited the production of NO compared with LPS-stimulated BV2 cells (4.96 ± 0.14 vs. 17.32 ± 0.4, p < 0.05). Therefore, MAT at a concentration range from 6.25 to 25 μM was used in the following experiments. Meanwhile, 25 μM MAT significantly inhibited the production of NO compared with LPS-stimulated primary microglial cells (3.91 ± 0.54 vs. 11.49 ± 1.54, p < 0.05).
MAT Reduced Pro-inflammatory Mediators Production in LPS-Stimulated BV2
Furthermore, since the production of NO in microglia is regulated primarily by the iNOS enzymes, we performed western blot analysis to determine whether MAT affected the expression of iNOS. The results showed MAT significantly inhibited protein expression of iNOS (Fig. 1d) in BV2 and primary microglia cells, similarly MAT decreased the NO release in LPS-stimulated microglia. As COX2 plays an important role in inflammatory regulation, we also investigated the effects of MAT on COX2 expression. The results showed that MAT significantly suppressed protein expression of COX2 (Fig. 1e). Consistent with western blotting, immunofluorescence staining in LPS-single treated and MAT pretreated (25 μM) in BV-2 microglia indicated that MAT markedly reduced the iNOS and COX2 expression (Fig. 1f).
MAT Alleviated the LPS-Induced ERK1/2 Activation in BV2 Microglia
It is well known that MAPKs (JNK, ERK and p38) signaling pathways play critical roles in the regulation of inflammatory response. In order to investigate the mechanism of MAT inhibiting inflammatory response of BV2 microglia induced by LPS, the effects of MAT on activation of MAPK pathways were determined. As shown in Fig. 2a, exposure of BV2 cells to LPS from 15 min resulted in increased phosphorylation of p38, ERK1/2, JNK, continued to 90 min. Interestingly, MAT (25 μM) selectively inhibited phosphorylation of ERK1/2 in a time-dependent manner. On contrast, MAT didn’t exert inhibitory effects on the phosphorylation of p38 and JNK.
MAT inhibited the phosphorylation of ERK1/2 in time-dependent manner in LPS-stimulated BV2 microglia cells. BV2 cells were pretreated with or without MAT (25 μM) for 4 h and then were stimulated with LPS (1 µg/ml) for 0, 15, 30, 60, 90 and 120 min. Cell lysates were prepared and blotted with total or phosphospecific antibodies to ERK1/2, p38 MAPK, and JNK. a Representative images of Western blotting for the phosphorylation of MAPKs. The band intensity of p-ERK1/2 (b), p-p38 (c), and p-JNK (d) was normalized to p38, JNK, and ERK1/2, respectively. The ratio of the control group band was set to 1.00. Data represents mean ± SE # p < 0.05 vs. control; ^ p < 0.05 vs. LPS treated cells. Data were from at least three independent experiments
Subsequently, BV2 cells were stimulated with LPS for 45 min to define the concentration-related inhibitory effects of MAT on MAPKs. Accordingly, MAT concentration-dependently attenuated LPS increased the phosphorylation of ERK1/2 rather than JNK and p38 (Fig. 3). These revealed that MAT specifically suppressed LPS-mediated ERK activation in BV2 microglia cells and ERK-activated inflammatory responses in time and concentration-dependent manner.
MAT inhibited the phosphorylation of ERK1/2 in concentration-dependent menner in LPS-stimulated BV2 microglia cells. BV2 microglia cells were pretreated with various concentrations of MAT for 4 h and then stimulated by LPS (1 µg/ml) for 45 min. The activation of ERK, JNK, and p38 was analyzed by western blotting, with specific anti-p-ERK, p-p38, and p-JNK antibodies, respectively. The band intensity of p-ERK1/2 (a), p-JNK (b), and p-p38 (c) was normalized to ERK1/2, JNK, and p38, respectively. The ratio of the control group band was set to 1.00. Data represents mean ± SE # p < 0.05 vs. control; ^ p < 0.05 vs. LPS treated cells. Data were from at least three independent experiments
MAT Blocked LPS-Mediated NF-κB Activation and p65 Nuclear Translocation, But Didn’t Affect AMPK Activation
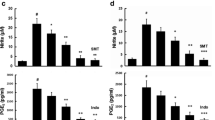
NF-κB is a critical transcriptional factor, which is implicated in the regulation of many genes that encode mediators of innate immunity and acute and chronic inflammatory responses, such as iNOS and COX2. As shown in Fig. 4a, NF-κB p65 was phosphorylated after LPS treatment (1 μg/ml) for 45 min. However, pre-treatment with MAT for 4 h, at concentrations ranging from 6.25 to 25 μM, markedly inhibited LPS-induced NF-κB p65 phosphorylated. In order to confirm the effects of MAT on the activation of NF-κB, we further explored whether MAT blocked LPS-induced nuclear translocation of NF-κB p65 subunit. As shown in Fig. 4b, the level of nuclear p65 in BV2 cells was increased after the cells were stimulated with LPS for 3 h; while the levels were decreased in response to the pretreatment of the LPS-stimulated cells with MAT. In line with this observation, immunofluorescence staining result also exhibited MAT diminished p65 nuclear translocation (Fig. 4c). Furthermore, AMPK also is involved in modulating inflammatory response, so we examined the effects of MAT on the AMPK signaling. However, pre-treatment with MAT didn’t affect p-AMPK signaling at various times and concentrations (Fig. 4d). These findings indicated that the anti-neuroinflammatory effect of MAT is associated, at least in part, with the suppression of NF-κB activation.
MAT inhibited phosphorylation of NF-κB p65 and nuclear translocation p65 in LPS-induced BV2 microglial cells, but didn’t affect p-AMPK signaling. a BV2 microglia cells were pretreated with various concentrations (6.25–25 μM) of MAT compound for 4 h and then stimulated by LPS (1 μg/ml) for 45 min. Cell lysates were prepared and blotted with total or phosphospecific antibodies to NF-κB (p65). b BV2 microglia cells were pretreated with various concentrations of MAT compound for 4 h and then stimulated by LPS (1 μg/ml) for 3 h. The nucleus fraction was collected by a protein fraction enrichment kit. The location of NF-κB was analyzed by western blotting with specific anti-NF-κB (p65) antibody. The Lamin A/C proteins were detected as internal control for nucleus fraction. The ratio of the control group band was set to 1.00. Data represents mean ± SE # p < 0.05 vs. control; ^ p < 0.05 vs. LPS treated cells. Data were from at least three independent experiments. c BV2 microglial cells pretreated with or without 25 μM MAT were treated with LPS (1 μg/ml) for 3 h, followed by staining with specific p65 antibody. Representative photomicrographs showed p65 nuclear translocation in LPS-stimulated BV2 (magnification ×200). d BV-2 microglia cells were treated with MAT (25 μM) for 0, 1, 3, 6, 9 and 12 h, or with the different concentrations (3.125, 6.25, 12.5, 25 and 50 μM) of MAT for 1 h, cell lysates were prepared and blotted with total or phosphor specific antibodies to AMPK and p-AMPK
MAT Inhibited LPS-Induced Src Phosphorylation and Expression in BV2 Cells
LPS induces the activation of multiple tyrosine kinase families including the SFKs [19]. We therefore investigated whether MAT was able to influence HCK and Src, as the predominant SFKs in LPS signaling. Our results showed that BV2 cells by LPS stimulation for 45 min lead to significant increase in phosphorylation (Tyr416) of Src. Furthermore, MAT attenuated the Src phosphorylation in a concentration-dependent manner (Fig. 5a). Consistent with a previous study, LPS stimulation for 24 h markedly induced Src expression, which was also suppressed by MAT in a concentration-dependent manner (Fig. 5b). HCK is another dominant SFK family member in mediating LPS-induced inflammation response. Subsequently, we also detected the effect of MAT on HCK activation in LPS-stimulated BV2 microglia, as shown in Fig. 5c, HCK expression was significantly increased in LPS-stimulated BV2 microglia, but pretreatment with MAT for 4 h didn’t effect HCK expression. In contrast, the expression of FAK was almost unaltered before and after LPS exposure (Fig. 5d). Taken together, our results suggested MAT attenuated Src pathway activation in LPS-exposed microglia.
MAT inhibited Src pathway activation in LPS-induced BV2 microglia cells. a BV2 microglia cells were pretreated with various concentrations of MAT for 4 h and then stimulated by LPS (1 μg/ml) for 45 min. The activation of Src was analyzed by western blotting, with specific anti-p-Src antibodies. BV2 microglial cells were pretreated with various concentrations of MAT for 4 h and then stimulated by LPS (1 μg/ml) for 24 h, Src (b), HCK (c), and FAK (d) expression were measured by western blot, GAPDH was used as loading control. Data represents mean ± SE # p < 0.05 vs. control; ^ p < 0.05 vs. LPS treated cells. Data were from at least three independent experiments
MAT Impeded the Migration of LPS-Induced BV2 Microglia Cells
Apart from inflammatory mediators releasing, cell mobilization was also important for the amplification of inflammation response. Cell migration was measured using the matrigel-coated transwell chamber assay. Representative microscopic images clearly showed that MAT reduced the migratory potential of the LPS-stimulated BV-2 cells (Fig. 6a), while the statistical analysis of three independent experiments revealed a significantly reduced number of motile cells for the MAT-pretreated group relative to the LPS-treated group (4.37 ± 0.94 vs. 48.29 ± 3.59, p < 0.001) (Fig. 6b). The results indicated that MAT significantly reduced microglial movement in response to LPS-induced activation.
MAT inhibited LPS-induced migration of BV2 microglia cells. BV2 cells were pretreated with MAT (25 μM) for 4 h and then stimulated with LPS for 24 h. The migratory ability of each group was determined by using a Boyden chamber as described in “Materials and Methods” section. a Cells that migrated to the lower membrane were photographed (magnification ×200). b The percentage of migrated cells was quantified. To quantify migration, cells were counted in three microscopic fields per sample. The results are presented as the mean ± SE of three assays. # p < 0.05 vs. control; ^ p < 0.05 vs. LPS treated cells
Discussion
MAT, a dibenzylbutyrolactone lignan, has been reported to possess diverse biological activities. None of the reports, however, documented the anti-inflammatory activity of MAT against activated microglia cells. The present study demonstrated for the first time that the anti-inflammatory properties of MAT in microglia and their underlying molecular mechanisms. MAT attenuated LPS-stimulated NO production and the expression of proinflammatory mediators iNOS and COX2 in BV2 microglia. In addition, MAT inhibited LPS-induced BV2 microglia migration. Furthermore, MAT possessed anti-inflammatory properties by down-regulating NF-κB activation via the inhibition of ERK MAPK phosphorylation as well as Src pathway in LPS-stimulated BV2 microglia cells.
The inflammatory mediators are thought to be responsible for some of detrimental effects on brain and diseases including ischemia and AD, resulting in neuronal death [20, 21]. Uncontrolled activation of microglia which could result in quantitative release of various inflammatory mediators such as NO, a key inflammatory mediator, has been shown to be directly toxic to neurons [22]. The excessive productions of NO by enhanced protein expression of iNOS, are associated with various types of central nervous injuries and diseases. Besides iNOS, the excessive expression of COX2 from activated microglia contributes to uncontrolled inflammation in neurodegenerative diseases [23]. The previous studies have demonstrated that iNOS and COX2 inhibitors provide neuroprotective effects against LPS-induced neurotoxicity [24]. We herein showed that MAT has an ability to inhibit production of NO in LPS-stimulated BV2. Moreover, we provided evidence that the MAT-mediated inhibition of NO production is due to suppression of iNOS expressions at the protein levels, concurrent with the decreased COX2 expression. Thus, the present findings of anti-inflammatory effects of MAT, herein iNOS and COX2 inhibition, on the LPS-activated microglia cells address that MAT may has potential protective effects against neuroinflammation.
A variety of intracellular protein kinases, including JNK, p38 and ERK, are necessary for enhancing expression of genes, including iNOS, COX2, and pro-inflammatory cytokines via NF-κB activation [25, 26]. There are numerous reports demonstrating that inhibition of phosphorylation of JNK, p38, and ERK contributes to down-regulation of iNOS and COX2 in LPS-induced BV2 microglia cells [27, 28]. In our study, we have specifically shown that MAT inhibited the phosphorylation of ERK, but not the p38 or JNK pathway, in response to LPS in BV2 microglia cells, suggesting that ERK is additional target of MAT. Furthermore, we have shown that MAT inhibited NF-κB p65 phosphorylation as well as the nuclear translocation of p65 subunit in LPS-stimulated BV2 microglia cells. Accumulating evidences show that AMPK is a repressor of inflammation. Some AMPK activators such as metformin have also been demonstrated for their potential to modulate inflammation [29]. However, our results showed MAT as a feasible mean to attenuate LPS-induced inflammatory responses in BV2 cells, but didn’t affect AMPK activation. Considering roles of MAPKs in inflammatory gene expression, it is deduced that inhibition of the cellular levels of iNOS, COX2, and pro-inflammatory cytokines by MAT in LPS-treated BV2 cells is associated with the ability of MAT to inhibit NF-κB activation by blocking ERK phosphorylation.
It is well-recognized that cell migration is closely associated with activation of microglia cells, which leads to tissue damage and chronic inflammation [1, 30]. In this study, we first exhibited MAT significantly suppressed LPS-mediated migration of BV2 microglia. Src activation is involved in cell motility [31]. Concurrent with the decreased migration, MAT effectively inhibited phosphorylation of C-terminal tyrosine (Tyr416) of Src, it is located in the activation loop of the kinase domain, indicating reduction enzyme activity in LPS-stimulated microglia. However, FAK, as a substrate of Src, not altered by MAT in LPS-mediated BV2 microglia. The previous studies exhibited that the increase of Src expression and its activity formed a positive feedback loop with NF-κB in macrophages [32]. We also demonstrated that MAT was able to suppress Src expression, while the expression of another SFK family protein HCK was not obviously altered. The critical role of Src in microglia migration suggested that the inhibition of Src activation by MAT might contribute to prevention of LPS-mediated inflammation responses. Based on the observations from literatures and our findings, inhibition of Src activation by MAT, at least in part, contributes to inhibiting LPS-stimulated BV2 microglia migration.
In this study, MAT showed the inhibition of NO production, iNOS and COX2 expression, and migration of microglia cells, which may partly arise from downstream activation of Src and MAPK/ERK pathways and subsequent inhibition of NF-κB p65 tanslocation. The findings presented herein suggest that MAT may be a potential chemopreventive agent of reducing neuroinflammation and deserve more study in the future.
Abbreviations
- MAT:
-
Matairesinol
- LPS:
-
Lipopolysaccharide
- NF-κB:
-
Nuclear factor-κB
- ERK:
-
Extracellular signal-regulated kinase
- JNK:
-
c-Jun N-terminal kinase
- CNS:
-
Central nervous system
- iNOS:
-
Inducible nitric oxide synthase
- NO:
-
Nitric oxide
- COX2:
-
Cyclooxygenase-2
- TNF-α:
-
Tumor necrosis factor alpha
- SFKs:
-
Src family kinases
- AMPK:
-
Adenosine 5′-monophosphate-activated protein kinase
References
Nimmerjahn A, Kirchhoff F, Helmchen F (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308:1314–1318
Bodea LG, Wang Y, Linnartz-Gerlach B, Kopatz J, Sinkkonen L, Musgrove R, Kaoma T, Muller A, Vallar L, Di Monte DA, Balling R, Neumann H (2014) Neurodegeneration by activation of the microglial complement-phagosome pathway. J Neurosci 34:8546–8556
Fan K, Li D, Zhang Y, Han C, Liang J, Hou C, Xiao H, Ikenaka K, Ma J (2015) The induction of neuronal death by up-regulated microglial cathepsin H in LPS-induced neuroinflammation. J Neuroinflamm 12:54
Yuste JE, Tarragon E, Campuzano CM, Ros-Bernal F (2015) Implications of glial nitric oxide in neurodegenerative diseases. Front Cell Neurosci 9:322
Chawla A, Nguyen KD, Goh YP (2011) Macrophage-mediated inflammation in metabolic disease. Nat Rev Immunol 11:738–749
Wan F, Anderson DE, Barnitz RA, Snow A, Bidere N, Zheng L, Hegde V, Lam LT, Staudt LM, Levens D, Deutsch WA, Lenardo MJ (2007) Ribosomal protein S3: a KH domain subunit in NF-kappaB complexes that mediates selective gene regulation. Cell 131:927–939
Sun LD, Wang F, Dai F, Wang YH, Lin D, Zhou B (2015) Development and mechanism investigation of a new piperlongumine derivative as a potent anti-inflammatory agent. Biochem Pharmacol 95:156–169
Hardie DG (2003) Minireview: the AMP-activated protein kinase cascade: the key sensor of cellular energy status. Endocrinology 144:5179–5183
Ayasolla KR, Giri S, Singh AK, Singh I (2005) 5-Aminoimidazole-4-carboxamide-1-beta-4-ribofuranoside (AICAR) attenuates the expression of LPS- and Aβ peptide-induced inflammatory mediators in astroglia. J Neuroinflamm 20:2–21
Zhao X, Zmijewski JW, Lorne E, Liu G, Park YJ, Tsuruta Y, Abraham E (2008) Activation of AMPK attenuates neutrophil proinflammatory activity and decreases the severity of acute lung injury. Am J Physiol Lung Cell Mol Physiol 295:L497–L504
Giri S, Nath N, Smith B, Viollet B, Singh AK, Singh I (2004) 5-Aminoimidazole-4-carboxamide-1-beta-4-ribofuranoside inhibits proinflammatory response in glial cells: a possible role of AMP-activated protein kinase. J Neurosci 24:479–487
Leu TH, Maa MC (2003) Functional implication of the interaction between EGF receptor and c-Src. Front Biosci 8:s28–38
Lowell CA (2011) Src-family and Syk kinases in activating and inhibitory pathways in innate immune cells: signaling cross talk. Cold Spring Harb Perspect Biol. doi:10.1101/cshperspect.a002352
Liu G, Vogel SM, Gao X, Javaid K, Hu G, Danilov SM, Malik AB, Minshall RD (2011) Src phosphorylation of endothelial cell surface intercellular adhesion molecule-1 mediates neutrophil adhesion and contributes to the mechanism of lung inflammation. Arterioscler Thromb Vasc Biol 31:1342–1350
Choi SW, Park KI, Yeon JT, Ryu BJ, Kim KJ, Kim SH (2014) Anti-osteoclastogenic activity of matairesinol via suppression of p38/ERK-NFATc1 signaling axis. BMC Complement Altern Med 14:35
Yamawaki M, Nishi K, Nishimoto S, Yamauchi S, Akiyama K, Kishida T, Maruyama M, Nishiwaki H, Sugahara T (2011) Immunomodulatory effect of (−)-matairesinol in vivo and ex vivo. Biosci Biotechnol Biochem 75:859–863
Saura J, Tusell JM, Serratosa J (2003) High-yield isolation of murine microglia by mild trypsinization. Glia 44:183–189
Jia Y, Dong X, Zhou P, Liu X, Pan L, Xin H, Zhu YZ, Wang Y (2012) The synthesis and biological evaluation of novel Danshensu-cysteine analog conjugates as cardiovascular-protective agents. Eur J Med Chem 55:176–187
Boulet I, Ralph S, Stanley E, Lock P, Dunn AR, Green SP, Phillips WA (1992) Lipopolysaccharide- and interferon-gamma-induced expression of hck and lyn tyrosine kinases in murine bone marrow-derived macrophages. Oncogene 7:703–710
Meda L, Cassatella MA, Szendrei GI, Otvos L Jr, Baron P, Villalba M, Ferrari D, Rossi F (1995) Activation of microglial cells by beta-amyloid protein and interferon-gamma. Nature 374:647–650
Wilms H, Zecca L, Rosenstiel P, Sievers J, Deuschl G, Lucius R (2007) Inflammation in Parkinson’s diseases and other neurodegenerative diseases: cause and therapeutic implications. Curr Pharm Des 13:1925–1928
Kim YS, Choi DH, Block ML, Lorenzl S, Yang L, Kim YJ, Sugama S, Cho BP, Hwang O, Browne SE, Kim SY, Hong JS, Beal MF, Joh TH (2007) A pivotal role of matrix metalloproteinase-3 activity in dopaminergic neuronal degeneration via microglial activation. FASEB J 21:179–187
Choi Y, Lee MK, Lim SY, Sung SH, Kim YC (2009) Inhibition of inducible NO synthase, cyclooxygenase-2 and interleukin-1beta by torilin is mediated by mitogen-activated protein kinases in microglial BV2 cells. Br J Pharmacol 156:933–940
Sil S, Ghosh T (2016) Role of cox-2 mediated neuroinflammation on the neurodegeneration and cognitive impairments in colchicine induced rat model of Alzheimer’s Disease. J Neuroimmunol 291:115–124
Oh YT, Lee JY, Lee J, Kim H, Yoon KS, Choe W, Kang I (2009) Oleic acid reduces lipopolysaccharide-induced expression of iNOS and COX-2 in BV2 murine microglial cells: possible involvement of reactive oxygen species, p38 MAPK, and IKK/NF-kappaB signaling pathways. Neurosci Lett 464:93–97
Jang BC, Paik JH, Kim SP, Shin DH, Song DK, Park JG, Suh MH, Park JW, Suh SI (2005) Catalase induced expression of inflammatory mediators via activation of NF-kappaB, PI3K/AKT, p70S6K, and JNKs in BV2 microglia. Cell Signal 17:625–633
Lima-Rodrigues M, Lamas N, Valle-Fernandes A, Cruz A, Vieira A, Oliveira P, Pedrosa J, Castro AG, Reis RM, Baltazar F, Almeida A (2010) The selective COX-2 inhibitor Etoricoxib reduces acute inflammatory markers in a model of neurogenic laryngitis but loses its efficacy with prolonged treatment. Inflamm Res 59:743–753
Duggan KC, Walters MJ, Musee J, Harp JM, Kiefer JR, Oates JA, Marnett LJ (2010) Molecular basis for cyclooxygenase inhibition by the non-steroidal anti-inflammatory drug naproxen. J Biol Chem 285:34950–34959
Hattori Y, Suzuki K, Hattori S, Kasai K (2006) Metformin inhibits cytokine-induced nuclear factor kappaB activation via AMP-activated protein kinase activation in vascular endothelial cells. Hypertension 47:1183–1188
Dou Y, Wu HJ, Li HQ, Qin S, Wang YE, Li J, Lou HF, Chen Z, Li XM, Luo QM, Duan S (2012) Microglial migration mediated by ATP-induced ATP release from lysosomes. Cell Res 22:1022–1033
Maa MC, Chang MY, Hsieh MY, Chen YJ, Yang CJ, Chen ZC, Li YK, Yen CK, Wu RR, Leu TH (2010) Butyrate reduced lipopolysaccharide-mediated macrophage migration by suppression of Src enhancement and focal adhesion kinase activity. J Nutr Biochem 21:1186–1192
Maa MC, Chang MY, Chen YJ, Lin CH, Yu CJ, Yang YL, Li J, Chen PR, Tang CH, Lei HY, Leu TH (2008) Requirement of inducible nitric-oxide synthase in lipopolysaccharide-mediated Src induction and macrophage migration. J Biol Chem 283:31408–31416
Acknowledgements
This work was supported by grants from National Natural Science Foundation of China (No. 81470164), a key laboratory program of the Education Commission of Shanghai Municipality (No. ZDSYS14005).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Xu, P., Huang, MW., Xiao, CX. et al. Matairesinol Suppresses Neuroinflammation and Migration Associated with Src and ERK1/2-NF-κB Pathway in Activating BV2 Microglia. Neurochem Res 42, 2850–2860 (2017). https://doi.org/10.1007/s11064-017-2301-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-017-2301-1