Abstract
We investigated the effects of pyridoxine (vitamin B6) on cell death, cell proliferation, neuroblast differentiation, and the GABAergic system in the mouse dentate gyrus. We administered pyridoxine (350 mg/kg intraperitoneally) to 8 week old mice twice a day for 14 days and sacrificed them at 10 weeks of age. Pyridoxine treatment did not induce neuronal death or activate microglia in the dentate gyrus, while glial fibrillary acidic protein (GFAP)-positive cells were significantly increased in the subgranular zone of the dentate gyrus. The increase in GFAP-positive cells was confirmed to be due to proliferating cells based on double immunofluorescence staining. GFAP-positive cells, which were also labeled with Ki67, a marker for cell proliferation, and doublecortin, a marker for neuroblast differentiation, were significantly increased in the pyridoxine-treated group compared to those in the vehicle-treated group. Pyridoxine treatment also increased the protein levels of glutamic acid decarboxylase (GAD) 67, an enzyme for GABA synthesis, and pyridoxal 5′-phosphate (PNP) oxidase, an enzyme for pyridoxal phosphate synthesis, in the dentate gyrus. These results suggest that pyridoxine treatment distinctly increases cell proliferation, neuroblast differentiation, and upregulated the GABAergic system, as revealed by the increases of GAD67 and PNP oxidase in the mouse dentate gyrus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is widely accepted that newborn neurons are continuously produced in certain brain regions of adult mammals including humans. In the hippocampus, which is one of the neurogenic regions, progenitor cells residing in the subgranular zone of the dentate gyrus (DG) proliferate to form post-mitotic daughter cells, which migrate into the granule cell layer and appear to play a role in network activity and in some hippocampal-dependent behaviors [1–3].
There are many factors that regulate adult neurogenesis in the hippocampus. Recently, the amino acid γ-aminobutyric acid (GABA) has emerged as a key regulator that controls multiple phases of adult neurogenesis. GABAergic mechanisms regulate differentiation and the time of synaptic integration in the subgranular zone of the DG [4, 5].
GABA is synthesized by glutamic acid decarboxylase (GAD) and absolutely requires pyridoxal phosphate (PLP) as a cofactor [6]. The final step of pyridoxine synthesis is the oxidation of pyridoxal 5′-phosphate (PNP) to PLP, which is catalyzed by PNP oxidase (PNPO); this reaction also serves as a critical constituent in the recycling of PLP in mammals that require pyridoxine [7, 8]. In particular, pyridoxine and its active form PLP have been reported to promote neuronal survival and improve cognitive functions like memory [9, 10]. In addition, many neurotransmitters such as dopamine, norepinephrine, GABA, serotonin and taurine are synthesized by PLP-dependent enzymes [11]. Pyridoxine deficiency leads to a reduction of GABA concentration in the whole brain and people with this deficiency exhibit a range of neurological symptoms, including tremors and seizures [12]. In contrast, pyridoxine attenuates glutamate-induced neurotoxicity [13], and protects neurons from ischemia and vincristine- and glutamate-induced neuronal damage [14–19].
Although there are many reports on the effects of pyridoxine against neuronal damage, no study on its effects on the plasticity or neuronal death in the DG has been conducted. Therefore, we investigated the effects of pyridoxine on cell death, gliosis, cell proliferation, and neuroblast differentiation in the mouse DG. In addition, we observed the protein levels of the GABA synthesizing enzyme, GAD67, and its cofactor enzyme, PNPO, in the DG after pyridoxine treatment.
Experimental Procedures
Experimental Animals
Male C57BL/6J mice (7 weeks of age) were purchased from the Jackson Laboratory Co. Ltd (Bar Harbor, ME). The animals were housed in a conventional state under adequate controlled temperature (22°C), humidity (55%), 12 h light/12 h dark cycle, and provided with free access to food and tap water. The procedures for the care and handling of animals conformed to guidelines that are in compliance with current international laws and policies (NIH Guide for the Care and Use of Laboratory Animals, NIH Publication No. 85-23, 1985, revised 1996). All of the experiments were conducted in a way as to minimize the number of animals used and the suffering caused by the procedures used in the present study.
Treatment with Pyridoxine
The mice were divided into two groups (n = 12 in each group): vehicle (physiological saline)- and 350 mg/kg pyridoxine hydrochloride (Sigma, St. Louis, MO)-treated groups. Vehicle or pyridoxine was administered to mice at 8 weeks of age twice a day for 2 weeks intraperitoneally and the animals were sacrificed 2 h after the last pyridoxine treatment. These schedules were adopted because doublecortin (DCX) is exclusively expressed in immature neurons from only 1 to 28 days of cell age [20, 21].
Tissue Processing
For histology, vehicle (n = 7)- and pyridoxine (n = 7)-treated animals were anesthetized with 30 mg/kg Zoletil 50 (Virbac, Carros, France) and perfused transcardially with 0.1 M phosphate-buffered saline (PBS, pH 7.4) followed by 4% paraformaldehyde in 0.1 M phosphate-buffer (PB, pH 7.4). The brains were removed and postfixed in the same fixative for 4 h. The brain tissues were cryoprotected by infiltration with 30% sucrose overnight. The 30-μm-thick brain sections in coronal plane were serially cut using a cryostat (Leica, Wetzlar, Germany). The sections were collected into six-well plates containing PBS for further process.
Fluoro-Jade B (F-J B) Histofluorescence Staining
F-J B, a marker for neuronal degeneration, histofluorescence staining procedures were conducted according to the method by Candelario-Jalil et al. [22]. In brief, the sections were first immersed in a solution containing 1% sodium hydroxide in 80% alcohol, and followed in 70% alcohol. They were then transferred to a solution of 0.06% potassium permanganate, and transferred to a 0.0004% F-J B (Histochem, Jefferson, AR) staining solution. After washing, the sections were placed on a slide warmer (approximately 50°C), and then examined using an epifluorescent microscope (Carl Zeiss, Göttingen, Germany) with blue (450–490 nm) excitation light and a barrier filter. With this method, neurons that undergo degeneration brightly fluoresce in comparison to the background [23].
Immunohistochemistry for NeuN, GFAP, Iba-1, Ki67, DCX and GAD67
To obtain the accurate data for immunohistochemistry, the free-floating sections were carefully processed under the same conditions. The tissue sections were selected between −1.46 and −2.46 mm posterior to the bregma in reference to the mouse atlas [24] for each animal. The sections were sequentially treated with 0.3% hydrogen peroxide (H2O2) in PBS and 10% normal horse serum in 0.05 M PBS. They were next incubated with mouse anti-neuronal nuclei (NeuN, diluted 1:1,000, Chemicon International, Temecula, CA) for neurons, rabbit anti-glial fibrillary acidic protein (GFAP, diluted 1:1,000, Chemicon International) for astrocytes, rabbit anti-ionized calcium-binding adapter molecule 1 (Iba-1, diluted 1:500, Wako, Osaka, Japan) for microglia, diluted rabbit anti-Ki67 antibody (1:1,000, Abcam, Cambridge, UK) for cell proliferation, goat anti-DCX antibody (1:50, SantaCruz Biotechnology, Santa Cruz, CA) for neuroblast differentiation and mouse anti-GAD67 (1:50, Chemicon International) for GABAergic neurons overnight, and subsequently exposed to biotinylated rabbit anti-goat, goat anti-mouse or goat anti-rabbit IgG (diluted 1:200, Vector, Burlingame, CA) and streptavidin peroxidase complex (diluted 1:200, Vector). Then, the sections were visualized by reaction with 3,3′-diaminobenzidine tetrahydrochloride (Sigma).
The measurement of NeuN-, Ki67- and DCX-positive cells in all of the groups was performed using an image analysis system equipped with a computer-based CCD camera (software: Optimas 6.5, CyberMetrics, Scottsdale, AZ). In addition, images of all of the DCX-immunoreactive structures in the DG were taken through a BX51 light microscope (Olympus, Tokyo, Japan) equipped with a digital camera (DP71, Olympus) connected to a PC monitor. The dendritic complexity of the DCX-positive cells was traced using camera lucida at 100× magnification (Neurolucida; MicroBrightField, Williston, VT). DCX-positive cells were separated into two categories according to dendritic complexity as demonstrated in supplement Fig. 1. The first category contained cells that lacked dendrites or had immature dendrites with primary or secondary branches which did not extend into the outer molecular layer. The second category contained cells that had mature dendrites with tertiary branches which extended into the outer molecular layer. The DCX-positive cells in the DG on each section were counted using Optimas 6.5 software (CyberMetrics). The cell counts from all of the sections of all of the rats were averaged. Counts are presented as a percentage value that is relative to the counts of the vehicle group.
Double Immunofluorescence Staining for DCX and GFAP
To confirm the proliferating neuroblast into double labeling with DCX and GFAP, the sections were processed by double immunofluorescence staining under the same conditions. Double immunofluorescence staining was conducted in goat anti-DCX (1:50)/rabbit anti-GFAP (1:200). The sections were incubated in the mixture of antisera overnight at room temperature. After washing three times for 10 min with PBS, they were then incubated in a mixture of both FITC-conjugated donkey anti-goat IgG (1:600; Jackson ImmunoResearch, West Grove, PA) and Cy5.5-conjugated donkey anti-rabbit IgG (1:600; Jackson ImmunoResearch) for 2 h at room temperature unifying the demonstration of figures. The immunoreactions were observed under the AxioM1 microscope attached with fluorescence lamp power supply (HBO100, Carl Zeiss), and confocal MS (LSM510 META NLO, Carl Zeiss).
Western Blot Analysis for DCX, PNP Oxidase and GAD67
To confirm the effects of pyridoxine on neuroblast differentiation and GABAergic transmission, five animals at each age were sacrificed and used for Western blot analysis. After sacrificing them and removing the brain, dentate gyrus was then dissected with a surgical blade. The tissues were homogenized in 50 mM PBS (pH 7.4) containing 0.1 mM ethylene glycol bis (2-aminoethyl Ether)-N,N,N′,N′ tetraacetic acid (EGTA) (pH 8.0), 0.2% Nonidet P-40, 10 mM ethylendiamine tetraacetic acid (EDTA) (pH 8.0), 15 mM sodium pyrophosphate, 100 mM β-glycerophosphate, 50 mM NaF, 150 mM NaCl, 2 mM sodium orthovanadate, 1 mM phenylmethylsulfonyl fluoride (PMSF) and 1 mM dithiothreitol (DTT). After centrifugation, the protein level was determined in the supernatants using a Micro BCA protein assay kit with bovine serum albumin as the standard (Pierce Chemical, Rockford, IL). Aliquots containing 20 μg of total protein were boiled in loading buffer containing 150 mM Tris (pH 6.8), 3 mM DTT, 6% SDS, 0.3% bromophenol blue and 30% glycerol. The aliquots were then loaded onto a polyacrylamide gel. After electrophoresis, the gels were transferred to nitrocellulose transfer membranes (Pall Crop, East Hills, NY). To reduce background staining, the membranes were incubated with 5% non-fat dry milk in PBS containing 0.1% Tween 20 for 45 min, followed by incubation with goat anti-DCX (1:100), mouse anti-PNP oxidase (1:50) [18, 25] or mouse anti-GAD67 (1:50) peroxidase-conjugated anti-goat IgG or anti-mouse IgG (Sigma) and an enhanced luminol-based chemiluminescent (ECL) kit (Pierce Chemical). The blot was densitometrically scanned for the quantification of relative optical density (ROD) of each band using Scion Image software (Scion Corp., Frederick, MD). These data were normalized against β-actin.
Statistical Analysis
The data shown here represent the means ± SE(M). Differences among the means were statistically analyzed by Student t test in order to elucidate the effects of pyridoxine on cell proliferation, neuroblast differentiation and changes in GABAergic systems in mice. Statistical significance was considered at P < 0.05.
Results
Neuronal Damage
In the vehicle-treated group, neurons positive for the Neuronal Nuclei (NeuN) protein, a neuronal specific nuclear protein, were easily detected in granule cells as well as hilar neurons in the DG (Fig. 1a). In the pyridoxine-treated group, the number of NeuN-immunoreactive neurons was similar to that in the vehicle-treated group (Fig. 1b, e). On the other hand, cells immunoreactive for Fluoro-Jade B (F-J B), a marker of neuronal degeneration, were not detected in the DG of both the vehicle- and pyridoxine-treated groups (Fig. 1c, d).
Immunohistochemistry for the protein Neuronal Nuclei (NeuN; a, b) and histofluorescence for Fluoro-Jade B (F-J B; c, d) in the dentate gyrus (DG) of the vehicle- (a, c) and pyridoxine-treated (b, d) groups. There are no significant changes in the number of NeuN- and F-J B-positive cells in the vehicle- and pyridoxine-treated groups. GCL, granule cell layer; ML, molecular layer; PL, polymorphic layer. Scale bar = 50 μm. E: The number of NeuN-positive cells in 1 mm2 area in the GCL of the DG in all of the groups (n = 7 per group). The bars indicate the standard errors of the mean (SEM)
Glial Changes
In the vehicle-treated group, glial fibrillary acidic protein (GFAP)-positive astrocytes were diffusely detected in the DG, and they had thread-like processes (Fig. 2a). In the pyridoxine-treated group, GFAP-positive astrocytes showed significant morphological changes in the DG. In particular, the GFAP-positive astrocytes had a hypertrophied cytoplasm in the subgranular zone of the DG (Fig. 2b).
Immunohistochemistry for glial fibrillary acidic protein (GFAP; a, b) and ionized calcium-binding adapter molecule 1 (Iba-1; c, d) in the DG of the vehicle- (a, c) and pyridoxine-treated (b, d) groups. GFAP-positive astrocytes (arrows) in the pyridoxine-treated group are significantly increased in the subgranular zone of the DG compared to that in the vehicle-treated group. However, the number of Iba-1-positive microglia in the pyridoxine-treated group is similar to that in the vehicle-treated group. GCL granule cell layer, ML molecular layer, PL polymorphic layer. Scale bar = 50 μm
Microglia positive for ionized calcium-binding adapter molecule 1 (Iba-1) in the vehicle-treated group were easily detected throughout the DG, and they had a small round cytoplasm and long processes (Fig. 2c). In the pyridoxine-treated group, Iba-1-immunoreactive microglia were similar to those in the vehicle-treated group (Fig. 2d).
Cell Proliferation
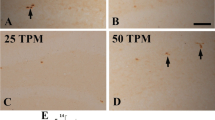
In the vehicle-treated group, nuclei positive for Ki67, a marker of cell proliferation, were observed in the DG, and these cells were mainly found in the subgranular zone (Fig. 3a). In the pyridoxine-treated group, the staining pattern of Ki67-positive nuclei was similar to that in the vehicle-treated group (Fig. 3b). However, the number of Ki67-positive nuclei was significantly increased by 1.10-fold in the pyridoxine-treated group compared to that in the vehicle-treated group (Fig. 3c).
Immunohistochemistry for Ki67 in the DG of the vehicle- (a) and pyridoxine-treated (b) groups. Ki67-positive cells (arrows) are more abundant in the pyridoxine-treated group than in the vehicle-treated group. GCL granule cell layer, ML molecular layer, PL polymorphic layer. Scale bar = 50 μm. c The number of Ki67-positive cells per section in all of the groups (n = 7 per group; * P < 0.05, significantly different from the vehicle group). The bars indicate the SEM
Neuroblast Differentiation
In both the vehicle- and pyridoxine-treated groups, neuroblasts positive for doublecortin (DCX), a marker for neuroblast differentiation, were easily observed in the subgranular zone of the DG (Fig. 4a–f). However, there were significant changes in the morphology and in the number of DCX-positive neuroblasts after pyridoxine treatment. In the pyridoxine-treated group, the number of DCX-positive neuroblasts with and without tertiary dendrites was significantly increased by 2.42 and 0.35-fold, respectively, in the subgranular zone of the DG compared to that in the vehicle-treated group (Fig. 4e). In addition, DCX-positive dendrites were more complex in the pyridoxine-treated group compared to that in the vehicle-treated group (Fig. 4c, d).
Immunohistochemistry for doublecortin (DCX) in the DG of the vehicle- (a, b) and pyridoxine-treated (c, d) groups. DCX-positive neuroblasts and their processes are significantly increased in the pyridoxine-treated group compared to the vehicle-treated group. GCL granule cell layer, ML molecular layer, PL polymorphic layer. Scale bar = 50 μm (a, c) or 25 μm (b, d). e The number of DCX-immunoreactive neuroblasts with and without tertiary dendrites per section in all of the groups (n = 7 per group; * P < 0.05, significantly different from the vehicle group). The bars indicate the SEM. f Western blot analysis of DCX in the DG in the vehicle- and pyridoxine-treated groups. The relative optical density of the immunoblot bands are demonstrated as percent values (n = 5 per group; *P < 0.05, indicates a significant difference compared to the vehicle-treated group). Bars indicate SE
Furthermore, DCX protein levels in the pyridoxine-treated group were also increased by 3.12-fold compared to that in the vehicle-treated group (Fig. 4f).
Proliferating Neuroblasts
In the vehicle-treated group, a few DCX and GFAP double-labeled cells were found in the subgranular zone of the DG (Fig. 5a–f). In the pyridoxine-treated group, DCX and GFAP double-labeled cells were increased compared to the vehicle-treated group, and in this group, DCX-immunoreactive neuroblasts were associated with the GFAP immunoreactivity (Fig. 5g–l).
Double immunofluorescence staining for DCX (green, a, d, g, j), GFAP (red, b, e, h, k), and a merged image (yellow, c, f, i, l) in the DG of the vehicle- (a–f) and pyridoxine-treated (g–l) groups. DCX and GFAP double-labeled cells (arrows and high magnification) in the pyridoxine-treated group are increased compared to that in the vehicle-treated group. Scale bar = 100 μm (a–c, g–i) or 50 μm (d–f, j–l) (Color figure online)
GABAergic Enzymes
In the vehicle-treated group, GAD67 immunoreactivity was detected in the polymorphic layer of the DG (Fig. 6a). In addition, a few GAD67-positive somata were observed in the granule cell layer (Fig. 6a). In the pyridoxine-treated group, the number of GAD67-positive neurons was significantly increased compared to that in the vehicle-treated group (Fig. 6b).
Immunohistochemistry for GAD67 in the DG of the vehicle- (a) and pyridoxine-treated (b) groups. GAD67 immunoreactivity is found in the neuropil of the polymorphic layer and in neurons (arrows) in both groups. GCL granule cell layer, PL polymorphic layer. Scale bar = 50 μm. c The number of GAD67-immunoreactive cells per section in all of the groups (n = 7 per group; * P < 0.05, significantly different from the vehicle group). Bars indicate means ± SEM. d Western blot analyses of GAD67 and PNP oxidase in the DG of the vehicle- and pyridoxine-treated groups. The relative optical density of the immunoblot bands are demonstrated as percent values (n = 5 per group; *P < 0.05, indicating a significant difference compared to the vehicle-treated group). Bars indicate means ± SE
In western blot analyses, GAD67 and PNP oxidase protein levels in the pyridoxine-treated group were increased by 0.38 and 1.75-fold, respectively, compared to that in the vehicle-treated group (Fig. 6c).
Discussion
Pyridoxine, acting through PLP, is one of the vitamin B complex molecules important for maintaining or modulating normal brain functions. In this study, we observed the effects of pyridoxine on cell death and gliosis in the mouse DG, which is a very dynamic region in the brain as this is a region where neurogenesis occurs throughout the lifespan [26]. The repeated administration of pyridoxine for 2 weeks did not induce any neuronal damage or microgliosis in the mouse DG. However, pyridoxine treatment significantly increased the number of astrocytes in the subgranular zone of the DG. It is well known that GFAP-positive cells such as astrocytes located in the subgranular zone of the DG are neural stem cells [27, 28]. To confirm that proliferating cells were labeled with GFAP, we found that the DCX-positive cells were double-labeled with GFAP in the subgranular zone of the DG. This result is supported by previous studies that have shown that GFAP-positive cells in the subgranular zone of the DG can proliferate and generate both astrocytes [27, 29] and DCX-positive neuroblasts [30]. In the present study, we first examined if pyridoxine treatment induced an increase in the GFAP/DCX double-positive cells in the mouse DG.
Next, we confirmed the increase of cell proliferation and neuroblast differentiation by using Ki67 and DCX, respectively. Ki67 is a nuclear protein used to label whole cell cycles except for G0 and the early G1 phase and is therefore used as a marker of cell proliferation [31]. DCX is a cytoskeletal protein which is expressed in immature neurons and which is therefore used as a marker of neuroblast differentiation [32]. The administration of pyridoxine significantly increased the number of Ki67- and DCX-positive cells compared to the vehicle-treated group. In addition, pyridoxine treatment increased the complexity of the dendrites of the DCX-positive neuroblasts in the mouse DG. This result is supported by a previous study that showed that deficits of vitamin B6 in utero significantly reduced the number of total neurons and normal neurons in the neocortex, with an increase in the number of shrunken neurons (700–1,500% of controls) [33]. However, we did not observe any significant increase of NeuN-positive neurons because DCX was exclusively expressed in immature neurons from 1 to 28 days of cell age [20, 21].
In this study, we also observed the effects of pyridoxine on the GABAergic system in the mouse DG since, in a previous study, pyridoxine treatment and the development of seizure activity were associated with the decreased synthesis and secretion of GABA [34]. In addition, high equilibrium potential for Cl− in neuroblasts causes GABA, acting through GABAA receptors, to depolarize, rather than hyperpolarize, young neurons [35]. In our present study, pyridoxine treatment significantly increased GAD67 and PNP oxidase in the mouse DG. This result is supported by a report that pyridoxine decreases the intracellular levels of glutamate by increasing GAD activity and decreasing calcium influx through actions on cell surface calcium channels [17]. In addition, it was reported that adult-generated granule cells received exclusively GABAergic synaptic input [36–38] and these GABAergic synaptic events had immature characteristics including slow rise and decay phases and depolarized reversal potentials [5]. Similarly, pyridoxine inhibited depolarization-evoked glutamate release in rat cortical synaptosomes [39]. The depolarizing effects of GABA on neuroblasts and on immature neurons contribute strongly to the activity-dependent regulation of the different stages of adult neurogenesis, including the proliferation of progenitors and the differentiation of newborn neurons [5, 40]. Ge et al. [38] conducted an animal study using retroviral techniques and argued that GABA might regulate the synaptic integration of newly generated neurons in the adult brain.
In conclusion, pyridoxine significantly enhanced cell proliferation and neuroblast differentiation and increased the GABAergic system in the mouse DG.
References
Saxe MD, Battaglia F, Wang JW et al (2006) Ablation of hippocampal neurogenesis impairs contextual fear conditioning and synaptic plasticity in the dentate gyrus. Proc Natl Acad Sci USA 103:17501–17506
Saxe MD, Malleret G, Vronskaya S et al (2007) Paradoxical influence of hippocampal neurogenesis on working memory. Proc Natl Acad Sci USA 104:4642–4646
Garthe A, Behr J, Kempermann G (2009) Adult-generated hippocampal neurons allow the flexible use of spatially precise learning strategies. PLoS One 4:e5464
Ge S, Pradhan DA, Ming GL, Song H (2007) GABA sets the tempo for activity-dependent adult neurogenesis. Trends Neurosci 30:1–8
Markwardt S, Overstreet-Wadiche L (2008) GABAergic signalling to adult-generated neurons. J Physiol 586:3745–3749
Chen C-H, Battahlioli G, Martin DL, Hobart SA, Colon W (2003) Distinctive interactions in the holoenzyme formation for two isoforms of glutamate decarboxylase. Biochim Biophys Acta 1645:63–71
Choi JD, Bowers-Komro M, Davis MD, Edmondson SE, McCormick DB (1983) Kinetic properties of pyridoxamine (pyridoxine)-5′-phosphate oxidase from rabbit liver. J Biol Chem 258:840–845
Kelly S, Zhao H, Hua Sun G et al (2004) Glycogen synthase kinase 3beta inhibitor Chir025 reduces neuronal death resulting from oxygen-glucose deprivation, glutamate excitotoxicity, and cerebral ischemia. Exp Neurol 188:378–386
Geng MY, Saito H, Katsuki H (1995) Effects of vitamin B6 and its related compounds on survival of cultured brain neurons. Neurosci Res 24:61–65
Araujo JA, Landsberg GM, Milgram NW, Miolo A (2008) Improvement of short-term memory performance in aged beagles by a nutraceutical supplement containing phosphatidylserine, Ginkgo biloba, vitamin E, and pyridoxine. Can Vet J 49:379–385
Dakshinamurti K, Paulose CS, Viswanathan M, Siow YL, Sharma SK, Bolster B (1990) Neurobiology of pyridoxine. Ann NY Acad Sci 585:128–144
Baxter P (2003) Pyridoxine-dependent seizures: a clinical and biochemical conundrum. Biochim Biophys Acta 1647:36–41
Kaneda K, Kikuchi M, Kashii S et al (1997) Effects of B vitamins on glutamate-induced neurotoxicity in retinal cultures. Eur J Pharmacol 322:259–264
Geng MY, Saito H, Nishiyama N (1997) Protective effects of pyridoxal phosphate against glucose deprivation-induced damage in cultured hippocampal neurons. J Neurochem 68:2500–2506
Yamashima T, Zhao L, Wang XD, Tsukada T, Tonchev AB (2001) Neuroprotective effects of pyridoxal phosphate and pyridoxal against ischemia in monkeys. Nutr Neurosci 4:389–397
Wang XD, Kashii S, Zhao L et al (2002) Vitamin B6 protects primate retinal neurons from ischemic injury. Brain Res 940:36–43
Dakshinamurti K, Sharma SK, Geiger JD (2003) Neuroprotective actions of pyridoxine. Biochim Biophy Acta 1647:225–229
Hwang IK, Yoo KY, Kim DH, Lee BH, Kwon YG, Won MH (2007) Time course of changes in pyridoxal 5′-phosphate (vitamin B6 active form) and its neuroprotection in experimental ischemic damage. Exp Neurol 206:114–125
Ozyurek H, Turker H, Akbalik M, Bayrak AO, Ince H, Duru F (2007) Pyridoxine and pyridostigmine treatment in vincristine-induced neuropathy. Pediatr Hematol Oncol 24:447–452
Brown JP, Couillard-Després S, Cooper-Kuhn CM, Winkler J, Aigner L, Kuhn HG (2003) Transient expression of doublecortin during adult neurogenesis. J Comp Neurol 467:1–10
Couillard-Despres S, Winner B, Schubeck S et al (2005) Doulecortin expression levels in adult brain reflect neurogenesis. Eur J Neurosci 21:1–14
Candelario-Jalil E, Alvarez D, Merino N, León OS (2003) Delayed treatment with nimesulide reduces measures of oxidative stress following global ischemic brain injury in gerbils. Neurosci Res 47:245–253
Schmued LC, Hopkins KJ (2000) Fluoro-Jade B: a high affinity fluorescent marker for the localization of neuronal degeneration. Brain Res 874:123–130
Franklin KBJ (1997) Paxinos G. The mouse brain in stereotaxic coordinates. Academic Press, San Diego
Bahn JH, Kwon OS, Joo HM et al (2002) Immunohistochemical studies of brain pyridoxine-5′-phosphate oxidase. Brain Res 925:159–168
van Praag H, Schinder AF, Christie BR, Toni N, Palmer TD, Gage FH (2002) Functional neurogenesis in the adult hippocampus. Nature 415:1030–1034
Ahn S, Joyner AL (2005) In vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nature 437:894–897
Alvarez-Buylla A, Lim DA (2004) For the long run: maintaining germinal niches in the adult brain. Neuron 41:683–686
Steiner B, Kronenberg G, Jessberger S, Brandt MD, Reuter K, Kempermann G (2004) Differential regulation of gliogenesis in the context of adult hippocampal neurogenesis in mice. Glia 46:41–52
Garcia AD, Doan NB, Imura T, Bush TG, Sofroniew MV (2004) GFAP-expressing progenitors are the principal source of constitutive neurogenesis in adult mouse forebrain. Nat Neurosci 7:1233–1241
Cooper-Kuhn CM, Kuhn HG (2002) Is it all DNA repair? Methodological considerations for detecting neurogenesis in the adult brain. Brain Res Dev Brain Res 134:13–21
Karl C, Couillard-Despres S, Prang P et al (2005) Neuronal precursor-specific activity of a human doublecortin regulatory sequence. J Neurochem 92:264–282
Kirksey A, Morré DM, Wasynczuk AZ (1990) Neuronal development in vitamin B6 deficiency. Ann N Y Acad Sci 585:202–218
Stephens MC, Havlicek V, Dakshinamurti K (1971) Pyridoxine deficiency and development of the central nervous system in the rat. J Neurochem 18:2407–2416
Pathania M, Yan LD, Bordey AA (2010) symphony of signals conducts early and late stages of adult neurogenesis. Neuropharmacology 58:865–876
Espósito MS, Piatti VC, Laplagne DA et al (2005) Neuronal differentiation in the adult hippocampus recapitulates embryonic development. J Neurosci 25:10074–10086
Overstreet Wadiche L, Bromberg DA, Bensen AL, Westbrook GL (2005) GABAergic signaling to newborn neurons in dentate gyrus. J Neurophysiol 94:4528–4532
Ge S, Goh EL, Sailor KA, Kitabatake Y, Ming GL, Song H (2006) GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 439:589–593
Yang TT, Wang SJ (2009) Pyridoxine inhibits depolarization-evoked glutamate release in nerve terminals from rat cerebral cortex: a possible neuroprotective mechanism? J Pharmacol Exp Ther 331:244–254
Marty A, Llano I (2005) Excitatory effects of GABA in established brain networks. Trends Neurosci 28:284–289
Acknowledgments
The authors would like to thank Mr. Seung Uk Lee and Mrs. Hyun Sook Kim for their technical help in this study, and Seung-Hae Kwon of the Korean Basic Science Institute Chuncheon Center for technical assistance with the confocal image analyses (LSM 510 META NLO). This work was supported by the National Research Foundation of Korea Grant funded by the Korean Government (MEST), Republic of Korea (2010-0007711).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
11064_2010_385_MOESM1_ESM.tif
sFig. 1. Micrographs of doublecortin (DCX) immunoreactive neuroblasts without (A) and with (B) tertiary dendrites. (TIFF 443 kb)
Rights and permissions
About this article
Cite this article
Yoo, D.Y., Kim, W., Kim, D.W. et al. Pyridoxine Enhances Cell Proliferation and Neuroblast Differentiation by Upregulating the GABAergic System in the Mouse Dentate Gyrus. Neurochem Res 36, 713–721 (2011). https://doi.org/10.1007/s11064-010-0385-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-010-0385-y