Abstract
Brain-derived neurotrophic factor (BDNF) is involved in neuronal survival and synaptic plasticity of the central and peripheral nervous system. BDNF appears to modulate nociceptive sensory inputs and pain hypersensitivity and has been studied in pathological situations, including chronic pain conditions and major depression. Increased serum BDNF levels have been recently reported in fibromyalgia (FM). In the present study, we assessed plasma BDNF levels in patients with FM and controls. Plasma BDNF was measured from 30 female patients with FM and 30 healthy age- and gender-matched volunteers using an enzyme immunoassay. FM patients showed higher levels of BDNF (FM = 167.1 ± 171.2 pg/mL) when compared with the control group (control = 113.8 ± 149.6 pg/mL) (P = 0.049; Mann–Whitney test). Six out of 30 controls presented superior values to the medium (15/15) of the patients with fibromyalgia (129 pg/mL) (P = 0.029, Fisher exact test). There was no correlation between plasma BDNF levels and age, disease duration, pain score, number of pain points and HAM-D score. Our results confirm previous findings of increased plasma BDNF levels in patients with FM, suggesting that BDNF may be involved in the pathophysiology of Fibromyalgia, despite high levels of depression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fibromyalgia (FM) is a common condition with a complex clinical pattern of unknown origin, consisting of diffuse musculoskeletal pain and muscle tenderness, accompanied by sleep disturbance, fatigue, anxiety and by other clinical manifestations such as depression, gastrointestinal symptoms and headache [1]. FM affects at least 2% of the adult population, females being significantly more often affected than males [2]. FM is usually diagnosed by application of the American College of Rheumatology (ACR) criteria. These criteria require the concurrent presence of widespread pain of at least 3 months’ duration and tenderness on palpation in at least 11 of 18 tender points sites, which are characterized by decreased pressure-pain thresholds that result in hyperalgesia or allodynia [3].
A sensitization process causing exaggerated perception of painful stimuli (hyperalgesia), and/or a perception of innocuous stimuli as painful (allodynia) may be involved in the generation of referred pain and hyperalgesia across multiple spinal segments in FM [4]. Several biochemical abnormalities that may contribute to this scenario have been reported in the cerebrospinal fluid of FM patients, including low concentrations of the metabolites of serotonin (5HT) and noradrenaline (NA), high concentrations of substance P and of nerve growth factors, suggesting a pathogenesis of central origin [5, 6].
Some studies have identified that pain thresholds in FM vary according to emotional contexts and it is frequently comorbid with psychiatric disorders such as major depression, dysthymia, anxiety and somatoform disorders [7, 8]. In fact, existing data suggest that the influence of psychological variables such as depression, pain, and fatigue must be considered as factors contributing to clinical presentation of FM [9].
Brain-derived neurotrophic factor (BDNF) enhances the growth and maintenance of several neuronal systems and may be a neurotransmitter modulator [10]. BDNF also acts as a regulator of neuronal excitability and a modulator of synaptic plasticity in the CNS [11]. BDNF is transported anterogradely to the central terminals of sensory neurons in the dorsal horn and the released BDNF binds to trkB receptors. Current evidence suggest that BDNF modulates both pre- and postsynaptic mechanisms [11]. Recently, the role of BDNF in pain states has received more attention as a neuromediator of hyperalgesia and spinal central sensitization [12, 13]. Moreover, BDNF induced hyperalgesia is dependent on an NMDA receptor-mediated mechanism [14].
There is growing evidence indicating that BDNF also plays a role in major depressive disorder (MDD) and that antidepressant treatment increases serum BDNF levels [15–18]. Moreover, in animal and human studies, antidepressant treatments could increase central as well as peripheral BDNF levels [19, 20].
Recent results have shown that patients with FM have increased levels of BDNF in serum and cerebrospinal fluid [8, 21]. In this study, our aim was to investigate plasma BDNF levels in patients with FM in comparison with a strictly matched control group.
Materials and Methods
Subjects
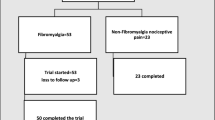
Patients with FM (n = 30, all female, age 46.3 ± 9.3 years) were recruited from the Physical Therapy Clinic of Feevale University. Diagnosis of FM was made according to the American College of Rheumatology (ACR) Criteria [3]. The variables evaluated in the patient group were: illness duration, pain intensity measured with the Visual Analogue Scale (VAS) from (no pain) to 10 (worst imaginable pain), number of tender points, a careful exploration of the medications history, severity of depressive symptoms assessed with the Hamilton Rating Scale for Depression (HDRS) [22]. Exclusion criteria were: (a) neurologic illness; (b) psychotic or other serious psychiatric conditions; (c) low cognitive performance; (d) pregnancy. Healthy subjects were matched (n = 30, all female, age 46.3 ± 9.3 years) and selected through an interview including a medical history. Exclusion criteria were: (a) past or current chronic physical or mental diseases; (b) regular medication intake; (c) pregnancy; (d) chronic pain. The study was approved by the Institutional Review Board and written informed consent was obtained from each individual.
Measurement of Plasma BDNF Concentration
Peripheral venous blood samples (10 ml) were collected in anticoagulant tubes using vacutainer system from all patients and controls between 10:00 and 12:00 am. Samples were centrifuged for 2,000×g for 15 min and plasma was stored at −18°C until analysis. Plasma levels of BDNF were measured using enzyme immunoassay—the commercial available BDNF immunoassay system kit (Chemikine by Chemicon, Temecula, CA, USA). All samples and standards were measured in duplicates and the coefficient of variation was less than 5%.
Statistical Analysis
For comparisons of BDNF levels between the FM patients and control group two-tailed Mann–Whitney U-test was used given the non-symmetric distribution of the data. Fisher exact test was used to evaluate the frequency of BDNF values in relation to the median of FM patients. The correlation analysis between BDNF levels and demographic characteristics of patients were analyzed by means of Spearman’s correlation coefficient. To compare the three groups according to antidepressant treatment, a nonparametric Kruskal–Wallis analysis was used. Significance for the results was set as P < 0.05. The statistic package SPSS 15.0 was used.
Results
Thirty female patients with FM and thirty female healthy volunteers matched for age were included (46.3 ± 9.3 years for both groups). In the FM group, illness duration was 6.3 ± 5.7 years, pain scores (0–10) were 8.1 ± 1.6, number of tender points (maximum 18) was 16.6 ± 2.3 and Hamilton-D score mean 30.4 ± 9.1. These results indicate that this group is severely ill both in terms of pain and depressive symptoms.
As shown in Fig. 1, plasma BDNF concentration in FM patients were significantly higher than the control group (FM patients = 167.1 ± 171.2 pg/mL and healthy controls = 113.8 ± 149.6 pg/mL; P = 0.049, Mann–Whitney test). In a secondary analysis, only 6 of 30 healthy controls showed values higher than the median (15/15) of FM patients (129 pg/mL) (P = 0.029, two-tailed Fisher exact test).
We found no significant correlation between plasma BDNF levels and age (r = −0.07, P = 0.71), illness duration (r = −0.05, P = 0.79), pain intensity by the VAS (r = −0.12, P = 0.50), number of tender points (r = −0.02, P = 0.89) or HAM-D score (r = −0.14, P = 0.44).
In order to evaluate the role of antidepressant treatments, we stratified patients in 3 groups: (1) antidepressant-naïve group (n = 6); (2) FM patients receiving low or only analgesic doses of tricyclic antidepressants (≤50 mg, n = 9); (3) patients with antidepressants at therapeutic doses for depression (amitriptyline ≥ 75 mg, fluoxetine ≥ 20 mg or equivalent—n = 15). As shown in Fig. 2, there were no significant differences in BDNF levels between these three groups (in pg/mL: group 1 = 130.4 ± 106.2; group 2 = 146.6 ± 137.9; group 3 = 194.0 ± 210.3, P = 0.81, Kruskal–Wallis test).
Serum BDNF in patients with Fibromyalgia stratified in 3 groups: (1) antidepressant-naïve group (n = 6) (no ADP); (2) FM patients receiving analgesic doses of tricyclic antidepressants (≤50 mg/day, n = 9) (low ADP); (3) patients with antidepressant doses (n = 15) (high ADP). There were no significant differences in BDNF levels between the three groups (BDNF group 1 = 130.4 ± 106.2; group 2 = 146.6 ± 137.9; group 3 = 194.0 ± 210.3, P = 0.81, Kruskal–Wallis test)
Discussion
In the present study, plasma BDNF levels were increased in FM patients as compared to age- and gender matched healthy controls. These findings are in line with recent evidence in serum and CSD of FM patients [8, 21]. Plasma BDNF levels were not correlated to the demographic data or FM clinical features such as age, gender, time of diagnosis, pain scores, number of tender points and levels of depressive symptoms.
Laske et al. [8] were the first to investigate the concentration of serum BDNF of 41 patients with FM compared to a control group with 45 subjects. Serum BDNF levels found in patients with FM (19.6 pg/mL) were significantly increased when compared to the control group (16.8 pg/mL), but BDNF levels were independent of age, gender, illness duration, preexisting recurrent major depression and treatment with low doses of antidepressants. Sarchielli et al. [21] measured BDNF levels in cerebrospinal fluid in three groups of subjects: 20 patients with FM, 20 with chronic migraine and 20 age-matched control subjects. BDNF values were significantly increased in patients with FM (40.4 ± 4.6 pg/mL), and in those with chronic migraine (39.4 ± 6.7 pg/mL) when compared to controls (11.3 ± 3.4 pg/mL). No correlation was found between BDNF levels and pain intensity and number of tender points examined in the migraine and FM groups. However, BDNF levels were significantly correlated with the duration of chronic headache and the duration of chronic pain in both groups. Thus, in general, our data are in agreement with these recent findings from the literature.
In this study we found the mean score of Hamilton-D scale of 30.4 ± 9.1 in patients with FM, which indicates that this group is severely depressed, despite treatment with antidepressants, with no significant correlation with serum BDNF levels. Shimizu et al. [18] showed that the serum BDNF levels in patients with depressive symptoms without treatment were significantly lower in relation to treated patients and the control group. Their results also indicated a significant negative correlation between HAM-D scores and serum BDNF levels. Low expression of BDNF levels are observed in cellular and molecular processes contributing to the development of chronic depression and in animals subjected to stress [23, 24]. Thus, it is interesting to note that the levels of BDNF in our patients with FM were increased despite the high level of depressive symptoms. Laske et al. [8] compared FM patients with and without major depression and found that BDNF levels were not significantly different between the two groups and the concentration of BDNF in patients with FM was independent of the pre-existence of major depression.
Our results showed no relationship between the use of antidepressants and plasma BDNF levels. Similarly, no significant differences were found in the concentration of BDNF in patients with FM in the groups with and without treatment with antidepressants in low doses [8]. Nibuya et al. [25] have shown that chronic administration of various types of antidepressants, including serotonin selective inhibitors, increases BDNF expression in the hippocampus of rodents. The therapeutic effects of antidepressant drugs have been attributed to increases in proliferation of neuronal progenitor cells through mechanisms involving up-regulation of hippocampal BDNF levels [26, 27]. Thus, we can not totally exclude the hypothesis that plasma BDNF levels in some of our FM patients were not affected by chronic use of antidepressants.
The expression of BDNF is known for its importance in synaptic plasticity and its action as a neuromodulator in the dorsal horn of the spinal cord has been proposed in various models of pain, including peripheral inflammation, axotomy, nerve damage and neuropathic pain [28, 29]. In our study, plasma BDNF levels in patients with FM were unrelated to pain intensity and number of tender points. Laske et al. [8] hypothesized that BDNF levels may be related to many modulatory mechanisms of pain involving the perception of pain and its association with emotional phenomena.
A positive characteristic of the current study was a strictly matched control group in regard to age and gender. However, the existence of higher levels of depression, as well as the variability in pharmacological treatments, in particular antidepressants, are limitations of our study. Thus, stratification of the data, as in the case of antidepressant use, significantly reduced statistical power and limited interpretation.
In summary, we have confirmed previous findings of higher BDNF in FM, suggesting that BDNF is involved in the pathophysiology of abnormal pain syndromes. However, the precise role of BDNF in the pathophysiology of FM and its connections with depression and use of antidepressants should be further investigated.
References
Mease P (2005) Fibromyalgia syndrome: review of clinical presentation, pathogenesis, outcome measures, and treatment. J Rheumatol Suppl 75:6–21
Gran JT (2003) The epidemiology of chronic generalized musculoskeletal pain. Best Pract Res Clin Rheumatol 17:547–561
Katz RS, Wolfe F, Michaud K (2006) Fibromyalgia diagnosis: a comparison of clinical, survey, and American College of Rheumatology criteria. Arthritis Rheum 54:169–176
Meeus M, Nijs J (2007) Central sensitization: a biopsychosocial explanation for chronic widespread pain in patients with fibromyalgia and chronic fatigue syndrome. Clin Rheumatol 26:465–473
Schwarz MJ, Spath M, Muller-Bardorff H (1999) Relationship of substance P, 5-hydroxyindole acetic acid and tryptophan in serum of fibromyalgia patients. Neurosci Lett 259:196–198
Julien N, Goffaux P, Arsenault P (2005) Widespread pain in fibromyalgia is related to a deficit of endogenous pain inhibition. Pain 114:295–302
Kassam A, Patten SB (2006) Major depression, fibromyalgia and labour force participation: a population-based cross-sectional study. BMC Musculoskelet Disord 7:4
Laske C, Stransky E, Eschweiler GW (2007) Increased BDNF serum concentration in fibromyalgia with or without depression or antidepressants. J Psychiatr Res 41:600–605
Suhr JA (2003) Neuropsychological impairment in fibromyalgia: relation to depression, fatigue, and pain. J Psychosom Res 55:321–329
Wu YJ, Kruttgen A, Moller JC, Len G, McAuliffe G, Ma C, Tai JP, Xu F (2004) Nerve growth factor, brain-derived neurotrophic factor, and neurotrophin-3 are sorted to dense-core vesicles and released via the regulated pathway in primary rat cortical neurons. J Neurosci Res 75:825–834
Malcangio M, Lessmann V (2003) A common thread for pain and memory synapses? Brain-derived neurotrophic factor and trkB receptors. Trends Pharmacol Sci 24:116–121
Delafoy L, Gelot A, Ardid D (2006) Interactive involvement of brain derived neurotrophic factor, nerve growth factor, and calcitonin gene related peptide in colonic hypersensitivity in the rat. Gut 55:940–945
Groth R, Aanonsen L (2002) Spinal brain-derived neurotrophic factor (BDNF) produces hyperalgesia in normal mice while antisense directed against either BDNF or trkB, prevent inflammation-induced hyperalgesia. Pain 100:171–181
Wu K, Len GW, McAuliffe G (2004) Brain-derived neurotrophic factor acutely enhances tyrosine phosphorylation of the AMPA receptor subunit GluR1 via NMDA receptor-dependent mechanisms. Brain Res Mol Brain Res 130:178–186
Yoshimura R, Mitoma M, Sugita A et al (2007) Effects of paroxetine or milnacipran on serum brain-derived neurotrophic factor in depressed patients. Prog Neuropsychopharmacol Biol Psychiatry 31:1034–1037
Karege F, Perret G, Bondolfi G (2002) Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res 109:143–148
Aydemir O, Deveci A, Taneli F (2005) The effect of chronic antidepressant treatment on serum brain-derived neurotrophic factor levels in depressed patients: a preliminary study. Prog Neuropsychopharmacol Biol Psychiatry 29:261–265
Shimizu E, Hashimoto K, Okamura N, Koike K, Komatsu N, Kumakiri C, Nakazato M, Watanabe H, Shinoda N, Okada S, Iyo M (2003) Alterations of serum levels of brain-derived neurotrophic factor (BDNF) in depressed patients with or without antidepressants. Biol Psychiatry 54:70–75
Piccinni A, Marazziti D, Catena M et al (2007) Plasma and serum brain-derived neurotrophic factor (BDNF) in depressed patients during 1 year of antidepressant treatments. J Affect Disord 105:279–283
Chen B, Dowlatshahi D, MacQueen GM (2001) Increased hippocampal BDNF immunoreactivity in subjects treated with antidepressant medication. Biol Psychiatry 50:260–265
Sarchielli P, Mancini ML, Floridi A et al (2007) Increased levels of neurotrophins are not specific for chronic migraine: evidence from primary fibromyalgia syndrome. J Pain 8:737–745
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62
Duric V, McCarson KE (2006) Effects of analgesic or antidepressant drugs on pain- or stress-evoked hippocampal and spinal neurokinin-1 receptor and brain-derived neurotrophic factor gene expression in the rat. J Pharmacol Exp Ther 319:1235–1243
Duric V, McCarson KE (2006) Persistent pain produces stress-like alterations in hippocampal neurogenesis and gene expression. J Pain 7:544–555
Nibuya M, Morinobu S, Duman RS (1995) Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci 15:7539–7547
Lee J, Duan W, Mattson MP (2002) Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J Neurochem 82:1367–1375
Sairanen M, Lucas G, Ernfors P (2005) Brain-derived neurotrophic factor and antidepressant drugs have different but coordinated effects on neuronal turnover, proliferation, and survival in the adult dentate gyrus. J Neurosci 25:1089–1094
Pezet S, Cunningham J, Patel J (2002) BDNF modulates sensory neuron synaptic activity by a facilitation of GABA transmission in the dorsal horn. Mol Cell Neurosci 21:51–62
Pezet S, Malcangio M, Lever IJ (2002) Noxious stimulation induces Trk receptor and downstream ERK phosphorylation in spinal dorsal horn. Mol Cell Neurosci 21:684–695
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haas, L., Portela, L.V.C., Böhmer, A.E. et al. Increased Plasma Levels of Brain Derived Neurotrophic Factor (BDNF) in Patients with Fibromyalgia. Neurochem Res 35, 830–834 (2010). https://doi.org/10.1007/s11064-010-0129-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-010-0129-z