Abstract
Octopamine functions as a neuromodulator, neurotransmitter, and neurohormone in insect nervous systems. Octopamine has a prominent role in influencing multiple physiological events: (a) as a neuromodulator, it regulates desensitization of sensory inputs, arousal, initiation, and maintenance of various rhythmic behaviors and complex behaviors such as learning and memory; (b) as a neurotransmitter, it regulates endocrine gland activity; and (c) as a neurohormone, it induces mobilization of lipids and carbohydrates. Octopamine exerts its effects by binding to specific proteins that belong to the superfamily of G protein-coupled receptors and share the structural motif of seven transmembrane domains. The activation of octopamine receptors is coupled with different second messenger pathways depending on species, tissue source, receptor type and cell line used for the expression of cloned receptor. The second messengers include adenosine 3′,5′-cyclic monophosphate (cAMP), calcium, diacylglycerol (DAG), and inositol 1,4,5-trisphosphate (IP3). The cAMP activates protein kinase A, calcium and DAG activate protein kinase C, and IP3 mobilizes calcium from intracellular stores. Octopamine-mediated generation of these second messengers is associated with changes in cellular response affecting insect behaviors. The main objective of this review is to discuss significance of octopamine-mediated neuromodulation in insect sensory systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Octopamine, p-hydroxyethanolamine was first discovered in the salivary glands of octopus vulgaris [1]. Octopamine (in invertebrates) can be distinguished from norepinephrine (noradrenaline, a structural analog of octopamine in vertebrates) by the absence of a hydroxyl group at position 3 of the phenol ring. Both phenolamines have functional significance in the central and peripheral nervous systems of invertebrates and vertebrates. Based on similarities in their action, it has been suggested that noradrenergic system in vertebrates is functionally substituted by octopaminergic system in invertebrates [2]. Octopamine is present in high concentration in the peripheral nervous system, central nervous system and various other insect tissues [3–4]. Octopamine serves as a neurotransmitter and neuromodulator in both peripheral and central nervous systems and also functions as a circulating neurohormone in insects [3–5]. In the peripheral nervous system, octopamine modulates the activity of flight muscles, peripheral organs (such as fat body, oviduct, and hemocytes), and almost all sense organs. In the central nervous system, octopamine is essential for regulation of motivation, desensitization of sensory inputs, initiation, and maintenance of various rhythmic behaviors and complex behaviors including learning and memory. Octopaminergic system is mainly functional in invertebrates because its receptors are not found in vertebrates. Therefore, octopamine receptors may be a suitable target for the development of highly specific insecticides. Collective evidence [2, 6–24] strongly suggests that octopamine plays a major neuromodulatory role in insect behaviors (Table 1).
Signals triggered by insect sensory receptors travel to the brain or ventral nerve cord resulting in appropriate behavioral responses mediated by neuromodulatory systems. Pharmacologically, as a neuromodulator, octopamine alters insect’s sensory behaviors by increasing or decreasing receptor sensitivity, receptor density or affecting neurotransmitter release by acting on presynaptic receptors [25]. Physiologically, as a neuromodulator, octopamine facilitates an excitatory post-synaptic potential and exacerbates an inhibitory post-synaptic potential evoked by the neurotransmitters such as acetylcholine. Octopamine may produce its modulatory effect by modifying neuronal membrane resistance, firing rate or bursting properties, the dynamics of adaptation, the strength of synaptic outputs and even the shape of the action potential [26]. However, its neuromodulatory effect on the intensity or duration of the response may occur after evokening of a short and intense response mediated by the neurotransmitter [26].
After the discovery of octopamine in insects, most of the octopamine-mediated studies have been performed using biochemical, pharmacological and physiological techniques [2–5, 26, 27]. In recent years, RNA-mediated interference (RNAi) technique has been successfully used to reduce the quantity of the proteins encoded by targeted genes. Studies on silencing of octopamine receptor gene by injection of octopamine receptor dsRNA in the specific region of the honeybee Apis mellifera brain have provided new insights on the functional roles of octopamine receptors in olfactory learning and memory [9, 28]. Several investigators have used RNAi technique for silencing specific gene functions such as post-mating behavior [29], addiction [30], odor perception and locomotor activity in fruit fly Drosophila melanogaster [31], axon path finding and synaptic target recognition in cockroach Periplaneta Americana [32], and gustatory response in honeybee A. mellifera [33]. Therefore, it is expected that future studies of RNAi in combination with behavior pharmacology, physiology, and immunocytochemistry analyses may help in shedding more light on the underlying molecular mechanisms of octopamine-mediated sensory behaviors. Molecular genetic studies performed in Drosophila can also potentially offer insights for the octopamine role by analyzing mutants for the genes required in the octopamine biosynthesis [34], and genes involved in cAMP signaling pathways [10, 35, 36], but such studies can not be recapitulated in every insect specie due to lack of mutational analysis. Therefore, RNAi is the best technology today to persue reverse genetics in other insects.
It is hoped that this commentary would initiate more studies not only on the molecular aspects of octopamine receptors in various insects, but also on the identification of various second messengers associated with octopamine-mediated signal transduction processes and their interplay and cross-talk with different sensory receptors and other biogenic amine receptors during sensory processings in insect brain. Although several reports have been recently published on octopamine, octopaminergic neurons, and octopaminergic receptor classification in insects [2, 27, 37–41] but they do not deal with neurochemical aspects of octopamine-mediated processes involved in insect sensory behaviors.
Classification and distribution of octopaminergic neurons
Based on octopamine immunoreactive labeling studies in insects, octopaminergic-like immunoreactive neurons as clusters of cell bodies and parikarya within the cell body in the brain and subesophageal ganglion can be classified into ten groups (Table 2). Groups I–VII (G0–G6) include single group or clusters of groups of octopamine immunoreactive cell bodies in the brain whereas groups VIII–X include octopamine immunoreactive parikarya within the cell body rind of supra- and subesophageal ganglia (unpaired and paired mid-line neurons) providing immunoreactive neurites and processes in the circumscribed regions of the neuropils [40]. The anatomical distribution of octopamine-like immunoreactive neurons in different insect species has been described by several groups of investigators [39–44]. According to these studies, several well-distinguished clusters of lateral cell bodies in the brain and many mid-line perikarya provide octopamine-like immunoreactive processes to circumscribed regions of subesophageal ganglion, antennal lobes, optic lobes, and protocerebrum neuropils in different insect species. The locations and projection patterns of octopamine-immunoreactive neurons in the brain neuropils and subesophageal ganglion of different insects show some overlap with distinct differences in their distribution suggesting common as well as highly specific targets in different species [39–44]. I will mainly focus on ventral unpaired median (VUM, group VIII), protocerebral medulla (PM, cluster 3a in group IV), dorsal unpaired median (DUM, group IX) and ventral paired median (VPM, group X) neurons, which are putatively related to insect senses (Table 2).
Unpaired neurons situated at the ventral mid-line or dorsal mid-line of subesophageal, thoracic and abdominal ganglia are called as VUM or DUM neurons in insects [45, 46]. A subpopulation of subesophageal DUM/VUM neurons innervates most parts of insect brain and is octopaminergic [45, 46]. Some of these are peripherally projecting octopaminergic neurons, which have roles in neuromuscular transmission. For example, in the extensor-tibiae neuromuscular preparation of locust Schistocerca gregaria, an identified octopaminergic DUM neuron (DUMETi) modulates neuromuscular transmission from the slow motoneuron to this muscle [47]. In abdominal muscle preparation of fruit fly D. melanogaster, octopamine presynaptically inhibits nerve-evoked excitatory junctional currents and post-synaptically reduces the decay time constant (mean open time) of miniature excitatory junctional currents. Octopamine does not modify the mean open time of extrajunctional glutamate receptors suggesting the possibility that it post-synaptically inhibits synaptic transmission by acting on junctional glutamate receptor channels [48]. These studies demonstrate that octopamine modulates neuromuscular transmission by affecting both pre- and post-synaptic mechanisms.
The DUM/VUM neurons are characterized by the location of their somata in the ventral nerve cord and subesophageal ganglion [45]. One particular octopaminergic neuron, which is one of a group of VUM neurons in the maxillary neuromere 1 (VUMmx1) has been identified by cobalt injection in honeybee A. mellifera brain [49]. VUMmx1 originates in the suboesophageal ganglion as the primary neurite that projects dorsally from the ventral median soma and bifurcates beyond the oesophagus [49]. It innervates brain neuropils (such as glomeruli of the antennal lobes, lateral protocerebrum, lips, and basal rings of mushroom body’s calyces) involved in odor processing. VUMmx1 mediates an interaction between an odor-sensitive (CS) and sucrose sensitive (US) pathways in the antennal lobe [49]. It releases octopamine into all or most glomeruli of the antennal lobe and provides the reinforcing properties of the US (equally effective as sucrose) in associative olfactory learning in honeybees [7]. Comparable VUM projection has been identified by performing octopamine immunohistochemistry in honeybee brains [40]. However, in moth Menduca sexta octopaminergic fibers, which connect calyces and the antennal lobes project through the inner antenno-cerebral tract [39] rather than going through the medial antenno-glomerular tract as in honeybees [40]. Thus, unlike VUMmx1, these fibers do not first innervate lateral protocerebrum and then proceed to the mushroom bodies in moth suggesting that these neurons may not be similar to VUMmx1 found in honeybees [39]. In some insects, octopamine-immunoreactive cell bodies of VUM neurons have been found in distinct clusters whereas in other insects they occur individually [39–41]. The organization of VUMmx1 is similar to VUM neurons (group VIII, Table 2).
In addition to octopaminergic VUM neurons, there are other octopamine immunoreactive neurons found in several insect species. These neurons originate from cluster G3 (perikarya of the bilateral cluster G3a, Table 2) lying medial to each antennal lobe and are called as PM neurons. They project posteriorly along the foramen to the ipsilateral protocerebrum, then laterally toward the optic lobes in various insects [39, 42–45]. In the locust Locusta migratoria brain, PM neurons arborize extensively in the medulla and lobula where they release octopamine, which results in an arousal mechanism evoking dishabitutaion in movement-detection system [23, 43]. The presence of octopamine immunoreactive processes in the honeybee optic lobes suggests the involvement of octopamine in regulation of vision [50]. Moreover, injecting octopamine in the lobula of honeybee A. mellifera has resulted in a marked increase in direction-specific antennal response to moving stripes [24]. In the American cockroach P. americana, antennal projection response has been used to test learning performance by associating a visual cue with an odor stimulus [51]. These studies suggest that octopamine released from PM neurons has divergent roles within visual pathways.
A similarity in location of DUM neurons as well as their projection patterns in insects suggest comparable modulatory roles among different insect species [39, 52–56] (group IX, Table 2). A large octopaminergic DUM neuron of the locust has been shown to innervate in the extensor tibiae muscle. However, soma of this neuron makes dense core vesicles, but not in exact vesicular form [57]. Electron microscopical studies suggest that DUM neurons of locust metathoracic ganglion are not associated with dense core vesicles [58]. Thus, the possible mechanism of octopamine release by exocytosis in the synaptic cleft remains unclear. Certain DUM neurons provide ascending processes to the brain, where they extend to the central complex. For example, in locust Locusta Schistocerca, one of the ascending neurons reaches the calyx, whereas another sends processes into the mushroom body lobes and a separate neuron innervates the antennal lobes [53]. This contrasts with honeybee, where VUMmx1 supplies octopamine to all three neuropils (antennal lobes, lateral protocerebrum, and mushroom bodies) associated with olfactory processing.
Ventral paired median neurons located ventrally in the subesophageal ganglion are paired mid-line neurons (group X, Table 2). In honeybee A. mellifera, VPM neurons arborize in the lateral and medial protocerebral neuropils [41]. However, in cockroach P. americana, each neuromere is associated with a cluster of dorsal somata consisting of paired as well as single mid-line neurons [41]. Thus, a single pair of ventrolateral immunoreactive somata gets associated with the second subesophageal neuromere; whereas, somata of these neurons arborize within subesophageal ganglion, tritocerebrum as well as with deuto and protocerebrum. This study indicates that clusters of octopaminergic lateral cell bodies in the brain and many mid-line perikarya are structurally comparable but have discrete arborizations in neuropils of different insects.
Classification and distribution of octopaminergic receptors and octopamine-mediated signal transduction
Classification of insect octopamine receptors has been extensively reviewed by several investigators [27, 37, 59–65]. The initial classification scheme was first proposed by Evans (1981), which was based on pharmacological profiles of a range of physiological responses to octopamine in the extensor-tibiae muscle preparation of locust [59]. In this preparation, octopamine receptors were divided into two classes: (1) OCTOPAMINE1 receptors (Oct1), which modulate a myogenic rhythm of contraction; and OCTOPAMINE2 receptors (Oct2), which are responsible for modulation of neuromuscular transmission [59]. Oct1 receptors mediate their effects via a mechanism that increases intracellular calcium [Ca2+]i levels whereas Oct2 receptors modulate neuromuscular transmission via a mechanism that increases intracellular cAMP [cAMP]i [59–61] (Fig. 1a). Based on pharmacological differences, Oct2 class was initially divided into two subclasses (A and B). Oct2A receptors are located on presynaptic terminals of the slow motoneuron, which modulate transmitter release whereas Oct2B receptors are located post-synaptically on the muscle and modulate the relaxation rate of twitch tension [59]. Subsequently, a third class of receptor OCTOPAMINE3 (Oct3) was pharmacologically characterized in the locust brain [27, 62–64], which was different from peripheral octopamine receptors (1, 2A and 2B) by its rank order of affinities for the selected antagonists, and its distribution in the insect brain. Oct3 class was later called as Oct2C (a subclass of Oct2) because similar to Oct2A and Oct2B, it is coupled to increase in cAMP levels (Fig. 1a). This classification was based on whole tissue responses.
Classification schemes of octopamine receptors: (a) Original scheme is based on whole tissue responses. Octopamine receptors are classified into two main groups: Oct1 and Oct2. Activation of Oct1 receptors increases levels of intracellular calcium, whereas activation of Oct2 increases levels of intracellular cAMP due to activation of adenylyl cyclase (AC) activity. Oct2 class is divided into three subclasses termed as Oct2A, Oct2B, and Oct2C, each having distinct pattern of affinities for agonists and antagonists. (b) New scheme is based on newly cloned octopamine receptors. Octopamine receptors are classified into three main groups: OctαR, OctβR, and Oct/Tyr or TyrR. The activation of OctαR increases levels of intracellular Ca2+ as well as shows a relatively small increase in levels of intracellular cAMP. This group has a higher affinity for octopamine than tyramine. The activation of OctβR increases levels of intracellular cAMP but not calcium. This group also has a higher affinity for octopamine than tyramine. The OctβR class has three subclasses, which can be pharmacologically distinguished from each other. The Oct/Tyr or Tyr class is coupled with decrease in intracellular cAMP or increase in intracellular Ca2+ depending upon the preference for agonist (tyramine or octopamine) for receptor activation, coupling with specific G protein and cell line utilized for receptor expression
Molecular cloning studies have eased the identification of genes, which code for octopamine receptors in insects [66–79]. A new classification recently proposed by Evans and Maqueira [65] is based on similarities in structural and signaling properties of cloned fruit fly D. melanogaster octopamine receptors with vertebrate adrenergic receptors [65]. This classification has grouped insect octopamine receptors into three classes: (1) α-adrenergic-like octopamine receptors (OctαR), (2) β-adrenergic-like octopamine receptors (OctβR) and (3) octopamine/tyramine or tyraminergic receptors (TyrR) [65] (Fig. 1b). Table 3 shows a comprehensive classification of all known cloned octopamine receptors based on old and new schemes. The OctαR class shows sequence homology with vertebrate α-adrenergic receptors. These receptors have higher affinity for octopamine than tyramine and coupled to increase in calcium (e.g., A. mellifera octopamine receptor, AmOA1; D. melanogaster octopamine receptor splice variants, AmOA1A/AmOA1B). They also produce induction in [cAMP]i levels but at a very high-octopamine concentration [74, 77]. Other octopamine receptors in this class [e.g., D. melanogaster, octopamine receptor protein (OAMB); P. americana, Paoa1 and Bombyx mori, BmOAR1] have sequence homology with α-adrenergic receptors and show both increase in cAMP as well as Ca2+ levels in response to octopamine when expressed in human embryonic kidney (HEK-293) cells [72, 76, 78] (Table 3). Since cAMP increase by octopamine is independent of the increase in calcium therefore it raises the possibility that octopamine may utilize more than one second messenger systems in vivo by coupling to specific G proteins present in the cell. The OctβR class has sequence similarities with vertebrate β-adrenergic receptors and specifically increases [cAMP]i levels in response to octopamine (e.g., D. melanogaster octopamine 2 receptor, DmOA2) [65, 77]. OctβR class is subdivided into 3 subclasses: (1) Octβ1R, (2) Octβ2R, and (3) Octβ3R [38, 65]. These receptors are coupled to increase in cAMP levels upon stimulation with octopamine but not to Ca2+ when expressed in Chinese hamster ovary (CHO) cells. Each subclass has a higher affinity for octopamine than tyramine and shows a unique pharmacological profile with synthetic octopaminergic agonists and antagonists [38]. They are preferentially expressed in Drosophila heads rather than bodies suggesting that OctβR class of receptors (DmOctβ1R, CG6919; DmOctβ2R, CG6989; and DmOctβ3R, CG7078) may participate in modulation of sensory processing in brain by cAMP signaling (Table 3). The TyrR class shows structural and pharmacological similarities with vertebrate α-adrenergic receptors (e.g., D. melanogaster, OAR_DROME) [66–67]. In most of the cases, these receptors have preference for tyramine over octopamine in inducing reduction in cAMP levels [67–69, 73], whilst in some preparations octopamine has been shown to be more or equally potent as tyramine in increasing [Ca2+]i release [67, 70, 71] (Table 3). These studies suggest that cloned receptors when expressed in different cell lines may be coupled to multiple effector pathways via different G proteins. A definitive designation for this class of receptors as “dual octopamine/tyramine or specific tyraminergic” (Fig. 1b) remains to be elucidated.
The distribution of insect neuronal octopamine receptors in insect nervous system has been demonstrated by using different techniques. Using radioligand-binding assay, a highest octopamine receptor density has been reported in the mushroom bodies and optic lobes, whereas, lowest octopamine receptor density observed in the suboesophageal ganglion, thoracic ganglia, antennal lobes and remainder of the brain in honeybee A. mellifera and locust S. gregaria [80]. An in situ hybridization study in honeybee A. mellifera brain has demonstrated abundant expression of Amoa1 gene that encodes octopamine receptor (AmOA1) in the mushroom body intrinsic neurons, optic lobes and deutocerebrum suggesting its involvement in the olfactory and visual processing [74]. Immunohistochemical analysis in fruit fly D. melanogaster brain has revealed highly enriched expression of OAMB in the mushroom body calyces, pedunculi, lobes (α, β, and γ), and ellipsoid body in the central brain [72]. However, octopamine-immunoreactive processes have been found in all brain neuropils and subesophageal ganglion except with some differences in mushroom bodies (Table 4). In honeybee A. mellifera, cockroach P. Americana, fruit fly D. melanogaster and hawk moth Manduca sexta, octopamine-like immunoreactivity has been observed only in calyces and γ lobes of mushroom bodies, omitting their other divisions [39, 40–41]. The γ lobes have been suggested to play a role in short-term memory in insects [81], but their role in other octopamine-mediated sensory behaviors still remains uncertain. Since α, β, and γ lobes, and pedunculus in blow fly Phaenicia sericata are octopamine-immunoreactive [41] therefore it raises a possibility that positive octopamine-immunoreactivity might be due to higher concentration of octopamine in blowfly mushroom bodies compared to other insects. The comparison between the distribution of octopamine-immunoreactive processes [39, 40–41, 44] and expression of octopamine receptors in insect brain neuropils and subesophageal ganglion [72, 74] shows an overlap in all brain neuropils except in mushroom bodies (Table 4). Collective studies suggest many similarities and some specific differences among the distribution of octopamine-immunoreactive processes and octopaminergic receptors in insect brain neuropils.
Interaction of octopamine with its specific receptors leads to a signal transduction cascade involving different second messenger systems (Fig. 2). These second messengers include: Ca2+, cAMP, inositol-1,4,5-trisphosphate (IP3), and diacylglycerol (DAG) [37, 82]. The activation of α-adrenergic-like octopamine receptors (OctαR) expressed in HEK-293 cells leads to elevation of both [Ca2+]i and [cAMP]i levels in response to octopamine [72, 76, 78]. The OctαR is coupled to phospholipase C (PLC) via the Gq protein [72]. PLC hydrolyzes inositol 4,5-bisphosphate into IP3 and DAG. IP3 binds to its receptor in the endoplasmic reticulum, which results in the opening of Ca2+ channels allowing Ca2+ release into the cytoplasm. DAG and Ca2+ activate protein kinase C (PKC) that regulates physiological response by phosphorylating various signaling proteins and ion channels [82]. The activation of OctαR by octopamine also stimulates adenylyl cyclase (AC) via the Gs protein, inducing the production of cAMP, which in turn stimulates protein kinase A (PKA) (Fig. 2). However, expression of other octopamine receptors (such as AmOA1, DmOA1, and DmOA1B) in HEK-293 cells increase calcium as well as cAMP levels (Table 3), but increase in cAMP mediated by these receptors is relatively small and can be observed only with nanomolar octopamine concentrations [74, 77]. This supports the notion that under physiological conditions, activation of Oct-α receptors is coupled to PLC pathway. The possibility of OctαR class of octopamine receptors (e.g., octopamine receptors from P. americana, Paoa1; B. mori, BmOAR1) activating more than one signaling pathways when expressed in cells [76, 78] still awaits more detailed pharmacological analyses. The activation of OctβR in response to octopamine increases [cAMP]i levels via AC coupled to Gs protein [2, 61], which stimulates PKA (Fig. 2). This is supported by observations in honeybee A. mellifera where injections of octopamine into the antennal lobes result in activation of PKA [83, 84]. Thus, cAMP modulates sensory response by PKA-mediated phosphorylation of various proteins, ion channels and transcription factors [37, 83, 84]. Active PKA phosphorylates a number of substrates in the cytoplasm involved in encoding and short-term memory formation. PKA migrates from the cytosol to the cell nucleus where it phosphorylates cAMP response element-binding (CREB) protein. The binding of CREB to DNA regulates the transcription of genes required for long-term memory [85]. The activation of TyrR in response to tyramine inhibits AC activity resulting in a decrease in [cAMP]i (Fig. 2). TyrR is activated more potently by tyramine than octopamine for inhibiting AC activity [67–70, 73], whilst it has higher affinity for octopamine than tyramine for induction of calcium release [70, 71, 75] suggesting cAMP reduction through AC coupled to Gi and increase in calcium through PLC coupled to Gq proteins. The dual role of TyrR supports “agonist-receptor trafficking” scheme, where different agonists induce different receptor conformations allowing signaling via different second messenger pathways [86].
Schematic overview of the octopamine receptors and second messenger pathways involved in insect sensory response. The interaction of octopamine (OA) with octopamine receptor (OctαR) sets in motion the stimulation of phospholipse C (PLC) via the Gq protein (G), inducing the generation of DAG and IP3 from PIP2 followed by the release of [Ca2+]i from the endoplasmic reticulum (ER). Calcium and DAG then activate protein kinase C (PKC). A dashed line represents coupling of OctαR with AC activation resulting in relatively small increase in cAMP when cloned octopamine receptor is expressed in HEK-293 cells. The interaction of OA with octopamine receptor (OctβR) activates adenylyl cyclase (AC) activity via stimulatory Gs protein generating cAMP, which stimulates protein kinase A (PKA). Both PKC and PKA influence cellular response by phosphorylating different signaling proteins and are involved in regulation of insect senses. The interaction of tyramine with octopamine/tyramine or tyramine receptor (TyrR) inhibits AC activity via inhibitory Gi protein causing a decrease in cAMP production
Octopamine-modulated senses
Insect sense organs are located on the antenna, leg, palp of the larvae, and other body parts. The sensory organs convert mechanical, chemical, and light energy from the environment into electrical energy of nerve impulses in the sensory neurons. From there, information is carried to the brain or ventral nerve cord resulting in specific behavioral responses [87]. The response of sensory neurons to a neuromodulator depends on the presence of specific receptors in the surface membrane. Octopamine plays a pivotal role as a neurotransmitter, neuromodulator, and neurohormone in influencing variety of physiological processes in insects. Octopamine functions as a neurotransmitter by binding to G protein-coupled receptors on the surface of neurons and other cells affecting diverse behaviors such as arousal, aggression and egg laying [27]. In addition to direct modulation of activities in the nervous system, these responses can be orchestrated through alterations in the metabolism, glycogenolysis, fat metabolism, heart rate, respiration, and muscle performance [27]. Octopamine is mainly released from neurosecretory endings and taken up by the haemolymph circulation (neurohormone). It is transported to relatively distant target organs to modulate many physiological processes. However, when octopamine is secreted from end terminals of a neurosecretory neuron at the synapse to directly affect the post-synaptic sensory receptor neuron, then it acts as a neurotransmitter. When it is involved in altering neurotransmitter-evoked post-synaptic potentiation, which is not sufficient to evoke either an excitatory or inhibitory post-synaptic potentiation, then it acts as a neuromodulator. Thus, octopamine may be considered as neurohormone at one terminal and a neurotransmitter/neuromodulator at another. The following text mainly focuses on modulatory effects of octopamine on five major insect senses (such as hearing, taste, smell, touch, and vision) (Fig. 3). Other insect sensory modalities such as infrared reception, vibration, pressure, thermoreception and magnetoreception [87] will not be discussed in this overview.
Auditory system
Although, octopamine has been reported to modulate response of mechanoreceptors in variety of insects [18, 20, 88–89], but relatively little is known about its influence in auditory system (Table 5). In the peripheral nervous system of insects, auditory receptors transform a sound wave into a train of action potentials. The signal transduction process is carried out by several sequential steps including mechanical coupling, mechanosensory transduction, electrical integration followed by spike generation, which relays information down to the auditory nerve. Insects use auditory system for conspecific communication and discrimination cues. Haemolymph-carried application of chemicals (such as picrotoxin) has been reported to affect firing activity of auditory receptors in noctuid moth Catocala cerogama [90]. However, two independent groups examined noctuid moth auditory organs and observed no efferent fibers in the area of receptor cell bodies suggesting octopamine modulation may not be involved at the peripheral sensory site [91, 92]. MacDermid and Fullard [93] recorded tracings of auditory nerve after exposing it to sound pulses at different frequencies in presence and absence of octopamine in moth C. cerogama notodontid tympanum and reported no effect of octopamine on auditory thresholds [93]. In contrast, octopamine injections in the prothoracic ganglion result in an increase in the sensitivity of auditory system through the modulation of an auditory interneuron in the central nervous system of cricket Gryllus bimaculatus [94]. The reason of this discrepancy is not fully understood. However, it may be either due to damage of auditory organ receptor cell bodies and axons from the haemolymph during its isolation or due to partial desensitization of sensory units causing relatively longer latencies in response to octopamine. Desensitization of octopamine receptors has been previously observed in the expression of cloned octopamine receptor in vertebrate cells [69, 70]. Thus, more studies are needed to understand association of octopaminergic neuromodulation of auditory system at the peripheral sensory site in insects. However, in acoustically evoked behaviors, octopamine-mediated neuromodulatory effect may be restricted to the central nervous system.
Tactile system
Many insects make use of their antennae as the main organs for tactile sense. In contrast to other senses that are capable of retrieving spatial information (such as vision), spatial sampling of tactile information requires active movement of the sense organ [95]. Antennal mechanoreception is relevant in several insect behaviors including escape behavior in cockroach P. americana [96], antennal movement pattern during walking in stick insect Carausius morosus [97], pattern recognition in honey bee A. mellifera [98], and communication in cricket Telegryllus oceanicus [99]. This accounts for reasonable information that is available on insect mechanoreceptors and their transduction mechanisms. Octopamine modulates the activity of a single-celled sense organ—stretch receptor—located at the base of each wing projecting into thoracic ganglia influencing flight motor patterns in locust L. migratoria [100]. Octopamine also modulates sensitivity of mechanoreceptors in femoral chordotonal sense organ of legs in stick insects Cuniculina impigra [101], Locusts L. migratoria [88] and S. gregaria [89] in sensing direction, velocity or acceleration of the movement (Table 5). A correlation of tactile associative learning performance and responsiveness to sucrose also has been observed in wild type bees A. mellifera [102, 103] suggesting octopamine’s modulatory role in tactile associated behavior. Mutations in many genes involved in mechanotransduction by tactile bristles in fruit fly D. melanogaster reduce the sensory response of antennal ear [104]. Therefore, it may be presumed that genetically similar transduction mechanisms are involved in tactile and auditory systems.
Visual system
Insects possess a pair of large compound eyes and three single-lense eyes (ocelli), which allow them to receive complex optical signals from the environment. Insect’s eyes absorb photons, which induce transformation of the energized visual pigment molecule, which then triggers the phototransduction process. Photoreceptors, located in the eyes first receive a pattern of image from external environment. The message is then passed on to the brain by projecting neurons, where information is processed and interpreted. Insect eye performs motion detection for navigation as well as for detection of the quality of light and both types of inputs are processed by different photoreceptors in the optic lobe. Octopamine modulates numerous and divergent roles by different photoreceptors [23, 50, 105–109] in insect visual system (Table 5).
Levels of octopamine are affected by constant dark and light conditions in male cabbage looper moth Trichoplusia ni [106]. Octopamine enhances directional specificity of antennal responses to moving visual stimuli when injected into the lobula of honeybee A. mellifera [105]. Serotonin injection in lobula of honeybee A. mellifera inhibits antennal reflex while octopamine injection slightly enhances the reflex [50]. Analogously, serotonin decreases the amplitudes of lobula field potentials evoked by moving stripe patterns while octopamine leads to an increase in these amplitudes in single cell activity recordings suggesting that octopamine and serotonin show opposite effects on visually evoked motion-sensitive antennal reflex [107]. Octopamine participates in arousal of descending contralateral movement detector (DCMD) interneuron within the locust L. migratoria brain [23]. In whole-cell voltage-clamp recordings, cAMP and its analogs (8-bromo-cAMP and dibutyryl-cAMP) slow down the macroscopic light response by increasing quantum bump latency, without any changes in bump amplitude or duration in photoreceptors suggesting its modulatory role in slowing the kinetics of phototransduction [108]. Octopamine closely mimics the effects of cAMP in reversibly slowing down the light-induced current without changing bump shape indicating that it modulates bump latency by AC pathway in phototransduction [108]. Furthermore, null mutants of fruit fly D. melanogaster in “neither inactivation nor afterpotential C” (ninaC) lack photoreceptor-cell specific proteins encoded by this gene. The null mutations in ninaC block all cAMP-induced effects suggesting that phosphorylation of NINAC proteins by PKA results in activation of its own kinase activity. This kinase may change the phosphorylation state of a Gq protein or PLC. These findings are supported by several groups reporting that amplification in fruit fly phototransduction utilizes a phosphoinositide (PI) signaling cascade and PLC pathway [108–110]. Collectively, these studies suggest that octopamine modulates visual system by AC and PLC pathways with a possibility of cross-talk between two signaling cascades.
Gustatory system
For most insects, gustation (sense of taste) is vital for their survival. Sensory gustatory information is vitally important for appropriate selection of food in insects. Gustatory system also plays an important role in other behaviors including host-plant selection and in egg laying behavior in insects. Gustatory receptors are found on mouthparts, body, wings, and legs of various insects (Table 5). Responsiveness to sucrose via proboscis extension reflex (PER) has been evaluated by injections of octopamine into starved adult blowflies Phormia regina [16]. These studies indicate that octopamine positively modulates blowfly feeding behavior. Similarly other investigators have found that either injections of octopamine in the thoraces or feeding of octopamine to honeybee A. mellifera significantly increases PER sucrose responsiveness [8, 103]. Direct injections of octopamine and synephrine (octopaminergic receptor agonist) in nymphs show a significant increase in feeding behavior of cockroach Rhyparobia Madera while phentolamine (octopaminergic receptor antagonist) shows reduction in feeding response without affecting motor activity [111]. Furthermore, injections of octopamine or cAMP in the antennal lobe of honeybee A. mellifera brain evoke a rapid and transient increase in PKA activity after sucrose stimulus implicating that octopamine contributes in processing of chemosensory input [83]. These studies suggest that octopamine plays a regulatory role by modulating AC pathway in insect gustatory system.
Olfactory system
Octopamine plays a modulatory role in insect olfactory system (Table 5). Insects primarily have olfactory receptors located on the olfactory sensory neurons in their antennae. Each sensillum on the antenna has pores through which chemical molecules enter the organ. The axons of the olfactory sensory neurons project to the antennal lobe, where they synapse in neuronal processesing unit called glomeruli (anatomically analogous and functionally similar unit found in the olfactory bulb of vertebrates). Olfactory information is encoded in the antennal lobe and then processed information is relayed to higher order brain centers by the projection neurons. Octopamine-mediated modulation can occur either at the peripheral sensory site or within the central nervous system.
Insects such as cockroach P. Americana have antennal hearts containing neurosecretory endings, which secrete high amounts of octopamine [112]. The evidence for the octopamine receptor expressed in the olfactory sensillae has been provided by in situ hybridization studies performed in moths B. mori and Heliothis virescens [69]. Furthermore, studies showing octopamine-mediated enhancement effect on the sensitivity of pheromone receptor neurons in the male silk worm moth Antherea polyphemus [113] and cabbage looper moth Trichuplusia ni [114] support its direct modulatory effects on the olfactory sensory system.
Octopamine has been reported to modulate various sensory behaviors including olfactory activity in the central nervous system. Application of octopamine in the defined areas of brain increases the sensitivity to olfactory stimuli and learning behavior [6, 9]. Octopamine is required for the foraging behavior of insects [13]. Studies on PKA and PKC have revealed that both enzymes are expressed throughout the brain. A rapid increase in PKA activity after octopamine injection in the antennal lobes has been observed in honeybee A. mellifera [83]. Furthermore, PKA activity in the antennal lobe of honeybee brain is regulated in the same manner by chemosensory stimulation (sucrose) [84] supporting a modulatory action of octopamine in honeybee central nervous system. Induction of olfactory memory results in long-lasting activation of PKC in the antennal lobes of honeybee A. mellifera [115]. Collectively, these studies suggest that octopamine plays a modulatory role by AC and PLC pathways, which results in activation of PKA and PKC, respectively. These enzymes then regulate olfactory system by phosphorylating different target proteins (Fig. 2).
Octopamine-mediated associative learning
Insects have olfactory learning capabilities. In associative learning, an association is made between a particular situation or external stimulus with an event or an unconditioned stimulus. Outdoor honeybees learn to associate sensory cues such as odor, color, visual pattern or landmark with a sucrose reward. However, in the laboratory experiments, honeybee A. mellifera can be trained for appetitive learning, which follows the rules of classical conditioning [116]. In the laboratory, restraind honeybees can be conditioned to respond with feeding movements of the mouthparts using a variety of floral odors as conditioning stimuli. This appetitive reflex is called PER conditioning. Subjects can be conditioned to odor [conditioned stimulus (CS)] paired with sucrose reinforcement [unconditioned stimulus (US)] and can readily discriminate a large number of odors [117]. Odor-evoked learning in honeybees lasts for days after a single trial and for lifetime after three trials. Microinjections of octopamine in the antennal lobes and mushroom bodies of honeybee brain suggest that octopamine is responsible for inducing associative learning in insect brain, and it can substitute for the unconditioned stimulus (sucrose reward) during classical conditioning [7]. Furthermore, octopamine modulates non-associative learning and food-induced arousal [118], responsiveness to conditioned olfactory stimuli [6, 9, 10], and memory consolidation when injected into the mushroom body calyces or antennal lobes (8) in Apis.
Additional evidence that octopamine is involved in mediating associative learning comes from genetic and behavioral studies performed in flies. Schwaerzel et al. [10] have demonstrated that dopamine and octopamine differentiate between aversive (electric shock) and appetitive (sugar) olfactory memories in D. melanogaster [10]. This group tested memory performance in genetic mosaics, exclusively restoring type 1 adenylyl cyclase (rutabaga; rut) function in Kenyon cells of the mushroom bodies. They observed that mutant rut flies have impaired sugar memory and electric shock memory (blockade in retrieval, but not in acquisition or storage) suggesting that both types of memories are rut-dependent and localized within the same subset of mushroom body neurons. Furthermore, these investigators used tyramine-β-hydroxylase (TβH) mutant flies. Tyramine-β-hydroxylase is an enzyme that catalyzes synthesis of octopamine from tyramine. An impairment in appetitive conditioning, but not in aversive conditioning occurs in TβH mutant flies. This blockade can be reversed by feeding flies octopamine prior to but not after conditioning. These findings are analogous to previous studies in honeybees [7] suggesting that octopamine can substitute for US in appetitive conditioning [10]. However, blockade of dopaminergic signaling by using tyrosine hydroxylase-GAL4 driver/UAS-Shi -ts combination results in an inhibition in acquisition but not retrieval of aversive olfactory memory while leaving appetitive learning intact. These findings suggest a differential role of two biogenic amines (octopamine and dopamine) in mediating appetitive and olfactory memories in Drosophila.
Pharmacological approach for the manipulation of octopamine receptor in insect brain
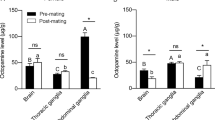
Olfactory memory has three components: (1) encoding, (2) consolidation, and (3) recall. Two of these components (memory consolidation and recall) are facilitated by injections of octopamine or octopaminergic agonists in the defined areas of mushroom bodies in the honeybee A. mellifera brain [7]. The reinforcing function of sucrose can be substituted by injecting octopamine in the two brain sites (mushroom bodies or antennal lobes) of honeybee brain [7]. Octopamine injections in the mushroom bodies do not lead to acquisition but only to a consolidation after conditioning. However, octopamine injections in the antennal lobes facilitate both acquisition and retrieval suggesting that octopamine induces associative learning and memory formation occurring at two independent sites in the honeybee brain [7]. Injections of mianserin, an octopamine receptor antagonist in the antennal lobes of the honeybee brain reduce acquisition (Fig. 4a) and recall (Fig. 4b), but do not affect odor discrimination (Fig 4b). These findings reveal that retrieval process is impaired independent of acquisition suggesting that octopamine mediates consolidation of a component of olfactory memory in the antennal lobe [9]. In addition to its high affinity for octopamine receptor in the central nervous system of honeybee and locust [80], mianserin also has affinity for serotonin receptor in the peripheral nervous system of African migratory locust L. migratoria [119]. Therefore, to rule out any non-specifc effects of this drug through binding to other biogenic amine receptors, it becomes necessary to consider other specific approaches for disrupting octopamine receptors.
Chemical and genetics approach for the manipulation of octopamine receptor in insect brain
Chemical ablation of mushroom bodies in fruit fly D. melanogaster shows an abolishment in odor learning. This suggests an important role of mushroom bodies in learning and memory processes [120]. Further support to the view that mushroom bodies are important neuroanatomical sites for associative learning, and octopamine plays a modulatory role in olfactory learning comes from studies with fruit fly D. melanogaster structural mushroom body mutants [121], and mutants with defects in cAMP, phosphodiesterase, AC, and PKA showing olfactory learning deficits [35]. The cAMP signaling for the formation of appetitive (octopamine-mediated) and aversive (dopamine-mediated) olfactory memories in D. melanogaster takes place within the same set of mushroom body intrinsic neurons. These results show a clear indication that octopamine is required for sugar learning and dopamine for electric shock learning [10]. Injections of mianserin or octopamine receptor dsRNA (AmOA1-dsRNA, synthesized from honeybee A. mellifera brain) in the antennal lobe of honeybee brain disrupt octopamine receptor function or synthesis affecting honeybee’s olfactory response [9]. Receptor disruption by both approaches results in significant reduction in olfactory acquisition (Fig. 4a) and recall (Fig. 4b), but shows no effect on odor discrimination (Fig. 4b). The similarity in the inhibitory patterns of mianserin and AmOA1-dsRNA suggests that both approaches can impair octopaminergic neurotransmission. Further manipulations in the honeybee brain have indicated that immunoblotting with anti-AmOA1 antiserum recognizes 2 protein bands (78 and 72 kDa) in the antennal lobes and four protein bands (78, 72, 60, and 48 kDa) in the remaining parts of the honeybee brain (including mushroom bodies, optic lobes, and suboesophageal ganglion) [28]. These bands are not observed when immunoblotting is performed with pre-immune serum in the antennal lobe or rest of the brain [28]. The intensity of all four bands in the rest of the brain homogenate is significantly reduced (70–90% inhibition) after 48-h of AmOA1-dsRNA treatment (Fig. 5a). The dsRNA-mediated effect is a function of time and distance from a localized site. The rate of dsRNA spread (diffusion) can be quantified from localized injections (in the antennal lobes) to other neuropils (rest of the brain) 48 h post-treatment (Fig. 5b). However, at this point, the molecular nature of bands found in honeybee A. mellifera brain homogenate remains uncertain. It is possible that these bands have been generated by alternative gene splicing or represent different post-translationally modified forms of the receptor. Recent findings from two independent groups have provided evidence for occurrence of alternate splice variants of octopamine receptor in fruit fly D. melanogaster [38, 77]. It is possible that similar to D. melanogaster, splice variants of octopamine receptor occur in honeybee brain, which may be responsible for gene regulation associated with the diversity of octopamine-mediated behaviors.
The effect octopamine receptor disruption by mianserin or AmOA1-dsRNA on acquisition and/or recall testing: (a) Acquisition to the conditioned odor (1-octanol). Four nanoliters of saline/mianserin (△/▲) or buffer/AmOA1 dsRNA (○/●) were injected in each antennal lobe 10 min or 24 h before conditioning. (b) Ninety minutes after conditioning, mianserin-treated subjects were tested with odors 1-octanol (C), 1-hexanol (S), and 1-geraniol (D) in a randomized manner. AmOA1-dsRNA-treated subjects were tested 24 h after conditioning with odors C, S, and D in a randomized manner. Sample size (between 30 and 50 honeybees) for each treatment group. Asterisks indicate significant differences of respective points from control group (*P < 0.05; ***P < 0.0001). ns not significantly different. Modified from Farooqui et al. [9]
As stated earlier that octopamine receptors are present in all regions of mushroom bodies while octopamine-like immunoreactivity is restricted exclusively to the calyces and within the γ lobes of mushroom bodies in some insects (such as moth, fruit fly, honeybee, and cockroach) suggesting that calyces and γ lobe play prominent roles in the formation of olfactory memory [39–41]. This interpretation is however speculative and requires further testing using different experimental approaches. Use of RNAi technique to disrupt octopamine receptors in specific mushroom body regions may be helpful in establishing the role of octopamine receptors of specific region in relation to short-term and/or long-term olfactory memory. For these studies, extra precautions are necessary because dsRNA injections should be performed within the specific lobes or calyces of mushroom bodies. Furthermore, optimum post-injection time may be another important factor to avoid any diffusion of dsRNA across these regions. It is possible that reverse genetics approach (such as RNAi) may provide deeper insights into octopamine-mediated modulation of insect sensory behaviors.
Effect of AmOA1-dsRNA on the expression of octopamine receptors in the honeybee brain: (a) Western blot analysis was performed in 48 h injection buffer-treated (-) and AmOA1 dsRNA-treated (+) brain homogenates. Injection buffer containing 5 mM KCl, 10 mM NaH2PO4 pH 7.8 served as control. All injections were made in the antennal lobes. Nitrocellulose membranes were probed with PIS, and anti-AmOA1 antiserum. Four bands (48, 60, 72, and 78 kDa) were recognized by anti-AmOA1 antiserum. The intensity of these bands was significantly reduced with 48 h dsRNA treatment. (b) Intensity of each band in untreated and treated lanes was quantitated and plotted as percent inhibition. Results presented in Fig. 5b represent immunoblot shown in Fig. 5a. These results are supported by our earlier studies [28] except that significant variation has been observed in % reduction of band intensities on day-to-day experimental basis, which could be due to differences in the rate of dsRNA diffusion or dilution of dsRNA with hemolymph in the brains of different subjects. PIS Pre-immune serum, anti-AmOA1 anti-octopamine receptor antiserum, anti-HRP anti-horse raddish peroxidase antibody
Changes in octopamine levels during behavioral development, diapause, and aging
In insect brain, levels of octopamine are significantly increased during metamorphosis. Alterations in octopamine levels may be related to an increase in behavioral activity of insects [122]. Thus, during M. sexta development, tyramine-β-hydroxylase activity is increased 11-fold in the brain and ninefold in the ganglia of moth [123]. This increase in enzyme activity correlates with an increase in octopamine levels in the central nervous system during metamorphosis reflecting that octopamine levels are developmentally regulated either due to: (1) an increase in level of tyramine-β-hydroxylase, (2) an increase in the number of octopaminergic neurons, and/or (3) changes in octopamine metabolism (including octopamine synthesis, release, uptake or degradation). No information is presently available about the levels of octopamine in brain tissue during developmental changes (larvae to adulthood) or due to subtle environmental features in insect species. Since brain size changes during development of insects, therefore total brain protein may vary with age. It will be more appropriate to use a parameter, which remains constant (e.g., expressing octopamine concentration as pg/mg protein). Since change in levels of a neurochemical may also be due to its subsequent metabolism, therefore changes in octopamine concentration alone may not be a good indicator of its specific involvement in any particular function.
Several groups have reported that older honeybee brains (notably foragers) contain higher levels of octopamine compared to brains from younger bees working inside the hive [124–126]. Oral octopamine treatment to honeybees has resulted in a significant increase in brain levels of octopamine and caused a dose-dependent increase in the number of new foragers suggesting that transition to foraging may be due to an increase in octopamine levels in the brain [127]. It is proposed that octopamine may modulate division of labor in honeybee colonies. To determine the association between biogenic amines (such as dopamine, serotonin, and octopamine) and tasks, Schulz et al. [13] have determined levels of biogenic amines using high-performance liquid chromatography analysis in the antennal lobes and mushroom bodies of honeybees A. mellifera from typical, single-cohort, and reversion colonies [13]. These investigators observed that octopamine concentration in the antennal lobes is associated with worker’s behavior regardless of age. In contrast, octopamine concentration in mushroom bodies is associated with age rather than behavior. These observations on behavior-related changes in the octopamine levels in the antennal lobes agree with the notion that octopamine-mediated changes in early olfactory processing regulate behavioral development in the honeybees. However, changes in octopamine levels in mushroom bodies seem to be related to age but not the behavior [13]. The pharmacological properties of octopamine receptors differ in different regions of honeybee brain and these differences may be either due to changes in octopamine receptor density or occurrence of splice variants of octopamine receptors associated with regulation of diverse behaviors. Therefore, more studies and better understanding of neuromodulatory role of octopamine is necessary for division of labor in insect societies.
In insects, adaptation of environmental and genetic changes occurs through diapause. Diapause is a developmental or reproductive arrest that maintains insect’s body in a state of suppressed metabolism during unfavorable conditions for reproduction (such as temperature, time of the day, and time of the year). Insects eventually lift this arrest and can resume successful reproduction. At present no information is available on the neuromodulatory role of octopamine on insect senses during diapause. However, octopamine injections into the female flesh fly, Sarcophaga bullata have been shown to influence expression of pupal diapause in the female’s progeny [128]. Other investigators have also observed increased dopamine levels in hemolymph and brain-subesophageal ganglia from larval to early pupal stages of the silkworm, B. mori [129]. It has been suggested that dopamine enhances expression of diapause hormone gene, which may result in laying of diapause-destined eggs by female adult silkworms. These studies imply that biogenic amines are involved in regulating diapause in insect systems. More information is needed to elucidate the potential role of octopamine on insect senses during induction, maintenance and termination of diapause.
Very little is known about age-related alterations in signal transduction mechanisms of octopaminergic transmission in insects. Octopamine-mediated down regulation of AC occurs during aging in Mediterranean fruit fly Ceratitis Capitata brain [130]. A decrease in octopamine binding to brain plasma membranes can be correlated with a decrease in the affinity and density of octopamine receptors. Age-related changes in octopamine have been reported in worker honeybee A. mellifera [131]. Octopamine-mediated changes in plasticity in brain anatomy and dendritic growth supports the view that octopamine is not only involved in neurotransmission but also in neuromodulation in insect brain [131]. Although octopamine levels do not change significantly with age but the ratio of octopamine/dopamine and octopamine/serotonin is relatively higher in the brains of ant Pheidole denta young workers indicating its modulatory role in behavioral development [132].
Not much is known about the neuromodulatory role of octopamine during senescence in insects. As mentioned earlier, octopamine is required for the foraging behavior of insects [13, 127]. Tofilski [133] has used foraging behavior as a parameter to monitor senescence in honeybee A. mellifera and observed abrupt changes in foraging behavior of foragers during their last flights before death [133]. However, this group has not determined octopamine levels in the brains near senescence. Presently, there are no other studies in insects, which can shed the light on this important issue. Therefore, additional studies are required to determine changes in octopamine levels during senescence in insects. It remains an open question whether decrease in foraging behavior of honeybees is partly due to a decline in levels of octopamine, octopamine receptor expression and/or increase in spliced variants or post-translational modification of octopamine receptor in brain near senescence causing olfactory dysfunction.
Conclusion and future perspectives
Octopamine can act as a neuromodulator, neurotransmitter, and neurohormone in insects. It mediates and regulates diverse physiological functions. Its physiological role in insects is analogous to norepinephrine in vertebrates. Octopamine-mediated diverse effects in insects are mediated by octopamine receptors, which are localized in different regions of insect brain. Octopamine receptors are expressed in multiple forms, which may be generated either through alternative mRNA splicing or by post-translational modification to perform different functions. Octopamine receptors are coupled to G proteins through different second messengers such as cAMP, IP3 and DAG. These second messengers not only modulate levels of intracellular calcium but also regulate phosphorylation of various signaling proteins involved in fine tuning of neuronal signaling affecting different insect sensory behaviors.
Although the occurrence of multiple forms of octopamine receptor has been established in insect brain, but not much is known about their neuromodulatory aspects, interactions with each other and their relationship to various sensory behaviors. The multiplicity of octopamine receptor in insect brain may be responsible for diversity of their function in response to a wide range of external stimuli. It is not known at present whether multiple forms or splice variants of octopamine receptor functionally interchange or act in parallel to transduce behavior related signal transduction processes. Thus, detailed studies are required on the involvement of octopamine receptor multiple forms or splice variants with various octopamine-mediated sensory processes. At present, it is not known how the gene expression of multiple forms of octopamine receptor is controlled and coordinated in insect brain. This would require extensive investigation on the quantification of gene expression profiles of octopamine receptors in different regions of various insects in association with insect senses. Furthermore, protocols should be developed to study the cross-talk between octopamine receptor splice variants, other biogenic amine receptors, and sensory receptors associated with their sensory behaviors.
We have utilized RNAi technology to overcome problems associated with the specificity of antagonists of octopamine receptor in honeybee brain and have clearly shown that in honeybee A. mellifera brain olfactory learning and memory can be impaired by the injection of octopamine receptor dsRNA in a specific brain region—antennal lobe—in time and dose-dependent manner [9]. dsRNA spreads with time from localized region to other brain neuropils [28]. Similarly other investigators have used dsRNA to knock down specific gene functions such as post-mating behavior, alcohol addiction, odor perception and locomotor activity, synaptic target recognition, and gustatory behavior in insects [29–33]. Together these studies suggest that dsRNA can be used to block specific behaviors by silencing specific genes. In future studies, attempts should be made to develop insects over-expressing multiple form or splice variants of octopamine receptors to study insect survival and octopamine-mediated behaviors. These studies may provide more information on modulation of sensory systems. It is hoped that future studies involving targeted disruption of genes encoding octopamine metabolism and its receptors may resolve some of the issues involved in sensory modulation of insects. In addition to previously used techniques (such as immunocytochemical labeling, optical imaging, and electrophysiological methods) for monitoring sensory systems, addition of RNAi tool for disrupting octopamine receptors in the central nervous system along with behavioral pharmacology in the same subjects may extend our understanding of octopamine-mediated molecular mechanisms involved in neuromodulation of insect senses.
References
Erspamer V, Boretti G (1951) Identification and characterization by paper chromatography of enteramine, octopamine, tyramine, histamine, and allied substances in extracts of posterior salivary glands of octopoda and in other tissue extracts of vertebrates and invertebrates. Arch Int Pharmacodyn Ther 88:296–332
Roeder T (1999) Octopamine in invertebrates. Prog Neurobiol 59:533–561
Axelrod J, Saavedra JM (1977) Octopamine. Nature 265:501–504
Evans PD (1985) Octopamine. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry and pharmacology, vol 11. Pergamon Press, Oxford, pp 499–530
Orchard I (1982) Octopamine in insects: neurotransmitter, neurohormone, and neuromodulator. Can J Zool 60:659–669
Mercer AR, Menzel R (1982) The effects of biogenic amines on conditioned and unconditioned responses to olfactory stimuli in the honeybee Apis mellifera. J Comp Physiol 145A:363–368
Hammer M, Menzel R (1998) Multiple sites of associative odor learning as revealed by local brain microinjections of octopamine in honeybees. Learn Mem 5:146–156
Menzel R, Heyne A, Kinzel C et al (1999) Pharmacological dissociation between the reinforcing, sensitizing, and response-releasing functions of reward in honeybee classical conditioning. Behav Neurosci 113:744–754
Farooqui T, Robinson K, Vaessin H et al (2003) Modulation of early olfactory processing by an octopaminergic reinforcement pathway in the honeybee. J Neurosci 23:5370–5380
Schwaerzel M, Monastirioti M, Scholz H et al (2003) Dopamine and octopamine differentiate between aversive and appetitive olfactory memories in Drosophila. J Neurosci 23:10495–10502
Sombati S, Hoyle G (1984) Generation of specific behaviours in a locust by local release into neuropils of the natural neuromodulator octopamine. J Neurobiol 15:481–506
Claassen DE, Kammer AE (1986) Effects of octopamine, dopamine, and serotonin on production of flight motor output by thoracic ganglia of Manduca sexta. J Neurobiol 17:1–14
Schulz DJ, Barron AB, Robinson GE (2002) A role for octopamine in honeybee division of labor. Brain Behav Evol 60:350–359
Yellman C, Tao H, He B et al (1997) Conserved and sexually dimorphic behavioral responses to biogenic amines in decapitated Drosophila. Proc Natl Acad Sci USA 94:4131–4136
O’Dell KM (1994) The inactive mutation leads to abnormal experience-dependent courtship modification in male Drosophila melanogaster. Behav Genet 24:381–388
Long TF, Murdock LL (1983) Stimulation of blowfly feeding behaviour by octopaminergic drugs. Proc Natl Acad Sci USA 80:4159–4163
Burrell BD, Smith BH (1995) Modulation of the honeybee (Apis mellifera) sting response by octopamine. J Insect Physiol 41:671–680
Ramirez JM, Orchard I (1990) Octopaminergic modulation of the forewing stretch receptor in the locust Locusta migratoria. J Exp Biol 149:255–279
Sombati S, Hoyle G (1984) Central nervous sensitization and dishabituation of reflex action in an insect by the neuromodulator octopamine. J Neurobiol 15:455–480
Zhang BG, Torkkeli PH, French AS (1992) Octopamine selectively modifies the slow component of sensory adaptation in an insect mechanoreceptor. Brain Res 591:351–355
Fields PE, Woodring JP (1991) Octopamine mobilization of lipids and carbohydrates in the house cricket, Acheta domesticus. J Insect Physiol 37:193–199
David JC, Coulon JF (1985) Octopamine in invertebrates and vertebrates. A review. Prog Neurobiol 24:141–185
Bacon JP, Thompson KS, Stern M (1995) Identified octopaminergic neurons provide an arousal mechanism in the locust brain. J Neurophysiol 74:2739–2743
Erber J, Pribbenow B, Bauer A et al (1993) Antennal reflexes in the honeybee: tools for studying the nervous system. Apidology 24:283–296
Katz PS (1999) What are we talking about? Modes of neuronal communication. In: Katz PS (ed) Beyond neurotransmisson, neuromodulation and its importance for information processing. Oxford University Press, Oxford, pp 1–27
Birmingham JT, Tauck DL (2003) Neuromodulation in invertebrate sensory systems: from biophysics to behavior. J Exp Biol 206:3541–3546
Roeder T (2005) Tyramine and octopamine: ruling behavior and metabolism. Annu Rev Entomol 50:447–477
Farooqui T, Vaessin H, Smith BH (2004) Octopamine receptors in the honeybee (Apis mellifera) brain and their disruption by RNA-mediated interference. J Insect Physiol 50:701–713
Chapman T, Bangham J, Vinti G et al (2003) The sex peptide of Drosophila melanogaster: female post-mating responses analyzed by using RNA interference. Proc Natl Acad Sci USA 100:9923–9928
Dzitoyeva S, Dimitrijevic N, Manev H (2003) γ-Aminobutyric acid B receptor 1 mediates behavior-impairing actions of alcohol in Drosophila: adult RNA interference and pharmacological evidence. Proc Natl Acad Sci USA 100:5485–5490
Winther AM, Acebes A, Ferrus A (2006) Tachykinin-related peptide modulate odor perception and locomotor activity in Drosophila. Mol Cell Neurosci 31:399–406
Marie B, Blagburn JM (2003) Differential roles of engrailed paralogs in determining sensory axon guidance and synaptic target recognition. J Neurosci 23:7854–7862
Amdam GV, Norberg K, Page RE et al (2006) Downregulation of vitellogenin gene activity increases the gustatory responsiveness of honeybee workers (Apis mellifera). Behav Brain Res 169:201–205
Monastirioti M, Linn CE Jr, White K (1996) Characterization of Drosophila tyramine β-hydroxylase gene and isolation of mutant flies lacking octopamine. J Neurosci 16:3900–3911
Davis RL (1996) Physiology and Biochemistry of Drosophila learning mutants. Physiol Rev 76:299–317
McGuire SE, Deshazer M, Davis RL (2005) Thirty years of olfactory learning and memory research in Drosophila melanogaster. Prog Neurobiol 76:328–347
Blenau W, Baumann A (2003) Aminergic signal transduction in invertebrates: focus on tyramine and octopamine receptors. Recent Res Dev Neurochem 6:225–240
Maqueira B, Chatwin H, Evans PD (2005) Identification and characterization of a novel family of Drosophila β-adrenergic-like octopamine G-protein coupled receptors. J Neurochem 94:547–560
Dacks AM, Christensen TA, Agricola HJ et al (2005) Octopamine-immunoreactive neurons in the brain and subesophageal ganglion of the hawkmoth Manduca sexta. J Comp Neurol 488:255–268
Sinakevitch I, Niwa M, Strausfeld NJ (2005) Octopamine-like immunoreactivity in the honey bee and cockroach: comparable organization in the brain and subesophageal ganglion. J Comp Neurol 488:233–254
Sinakevitch I, Strausfeld NJ (2006) Comparison of octopamine-like immunoreactivity in the brains of the fruit fly and blow fly. J Comp Neurol 494:460–475
Monastirioti M, Gorczyca M, Rapus J et al (1995) Octopamine immunoreactivity in the fruit fly Drosophila melanogaster. J Comp Neurol 356:275–287
Stern M (1999) Octopamine in the locust brain: cellular distribution and functional significance in an arousal mechanism. Microsc Res Tech 45:135–141
Kreissl S, Eichmuller S, Bicker G et al (1994) Octopamine-like immunoreactivity in the brain and subesophageal ganglion of the honeybee. J Comp Neurol 348:583–595
Hoyle G (1975) Evidence that insect dorsal unpaired median (DUM) neurons are octopaminergic. J Exp Zool 193:425–431
Bräunig P, Pflüger H-J (2001) The unpaired median neurons of insects. Adv Insect Physiol 28:185–266
Evans PD, O’Shea M (1977) An octopaminergic neurone modulates neuromuscular transmission in the locust. Nature 270:257–259
Nishikawa K, Kidokoro Y (1999) Octopamine inhibits synaptic transmission at the larval neuromuscular junction in Drosophila melanogaster. Brain Res 837:67–74
Hammer M (1993) An identified neuron mediates the unconditioned stimulus in associative learning in honeybees. Nature 366:59–63
Erber J, Kloppenburg P (1995) The modulatory effects of serotonin and octopamine in the visual system of the honeybee (Apis mellifera L.) I. Behavioral analysis of the motion-sensitive antennal reflex. J Comp Physiol 176A:111–118
Lent DD, Kwon HW (2004) Antennal movements reveal associative learning in the American cockroach Periplaneta americana 207:369–375
Bräunig P (1991) Suboesophageal DUM neurons innervate the principal neuropiles of the locust brain. Philos Trans R Soc Biol Sci 332:221–240
Bräunig P, Burrows M (2004) Projection patterns of posterior dorsal unpaired median neurons of the locust subesophageal ganglion. J Comp Neurol 478:164–175
Eckert M, Rapus J, Nurnberger A et al (1992) A new specific antibody reveals octopamine-like immunoreactivity in cockroach ventral nerve cord. J Comp Neurol 322:1–15
Spörhase-Eichmann U, Vullings HG, Buijs RM et al (1992) Octopamine-immunoreactive neurons in the central nervous system of the cricket, Gryllus bimaculatus. Cell Tissue Res 268:287–304
Stevenson P, Meuser S (1997) Octopaminergic innervation and modulation of a locust flight steering muscle. J Exp Biol 200:633–642
Hoyle G, Colquhoun W, Williams M (1980) Fine structure of an octopaminergic neuron and its terminals. J Neurobiol 11:103–126
Watson AHD (1984) The dorsal unpaired median neurons of the locust metathoracic ganglion: neuronal structure and diversity, and synapse distribution. J Neurocytol 13:303–327
Evans PD (1981) Multiple receptor types for octopamine in the locust. J Physiol 318:99–122
Evans PD (1993) Molecular studies on insect octopamine receptors. In: Pichon Y (ed) Comprehensive molecular neurobiology. Birkhauser-Verlag, Basel, pp 286–296
Evans PD, Robb S (1993) Octopamine receptor subtypes and their modes of action. Neurochem Res 18:869–874
Roeder T (1992) A new octopamine receptor class in locust nervous tissue, the octopamine3 (OA3) receptor. Life Sci 50:21–28
Roeder T (1995) Pharmacology of octopamine receptor from locust central nervous tissue (OAR3). Br J Pharmacol 114:210–216
Roeder T, Nathanson JA (1993) Characterization of insect neuronal octopamine receptors (OA3 receptors). Neurochem Res 18:921–925
Evans PD, Maqueira B (2005) Insect octopamine receptors: a new classification scheme based on studies of cloned Drosophila G-protein coupled receptors. Invert Neurosci 5:111–118
Arakawa S, Gocayne JD, McCombie WR et al (1990) Cloning, localization, and permanent expression of a Drosophila octopamine receptor. Neuron 4:343–354
Saudou F, Amlaiky N, Plassat JL et al (1990) Cloning and characterization of a Drosophila tyramine receptor. EMBO J 9:3611–3617
Vanden Broeck J, Vulsteke V, Huybrechts R et al (1995) Characterization of a cloned locust tyramine receptor cDNA by functional expression in permanently transformed Drosophila S2 cells. J Neurochem 64:2387–2395
von Nicksch-Rosenegk E, Krieger J, Kubick S et al (1996) Cloning of biogenic amine receptors from moths (Bombyx mori and Heliothis virescens). Insect Biochem Mol Biol 26:817–827
Robb S, Cheek TR, Hannan FL et al (1994) Agonist-specific coupling of a cloned Drosophila octopamine/tyramine receptor to multiple second messenger systems. EMBO J 13:1325–1330
Reale V, Hannan F, Midgley JM (1997) The expression of a cloned Drosophila octopamine/tyramine receptor in Xenopus oocytes. Brain Res 769:309–320
Han KA, Millar NS, Davis RL (1998) A novel octopamine receptor with preferential expression in Drosophilla mushroom bodies. J Neurosci 18:3650–3658
Blenau W, Balfanz S, Baumann A (2000) Amtyr1: characterization of a gene from honeybee (Apis mellifera) brain encoding a functional tyramine receptor. J Neurochem 74:900–908
Grohmann L, Blenau W, Erber J et al (2003) Molecular and functional characterization of an octopamine receptor from honeybee (Apis mellifera) brain. J Neurochem 86:725–735
Poels J, Suner MM, Needham M et al (2001) Functional expression of a locust tyramine receptor in murine erythroleukemia cells. Insect Mol Biol 10:541–548
Bischof LJ, Enan EE (2004) Cloning, expression and functional analysis of an octopamine receptor from Periplaneta Americana. Insect Biochem Mol Biol 34:511–521
Balfanz S, Strünker T, Frings S et al (2005) A family of octopamine receptors that specifically induce cyclic AMP production or Ca2+ release in Drosophila melanogaster. J Neurochem 93:440–451
Ohtani A, Arai Y, Ozoe F et al (2006) Molecular cloning and heterologous expression of an alpha adrenergic-like octopamine receptor from the silkworm Bombyx mori. Insect Mol Biol 15:763–772
Dacks AM, Dacks JB, Christensen TA et el (2006) The cloning of one putative octopamine receptor and two putative serotonin receptors from the tobacco hawkmoth, Manduca sexta. Insect Biochem Mol Biol 36:741–747
Degen J, Gewecke M, Roeder T (2000) Octopamine receptors in the honeybee and locust nervous system: pharmacological similarities between homologous receptors of distantly related species. Br J Pharmacol 30:587–594
Zars T, Fischer M, Schulz R et al (2000) Localization of a short-term memory in Drosophila. Science 288:672–675
Blenau W, Baumann A (2001) Molecular and pharmacological properties of insect biogenic amine receptors: lessons from Drosophila melanogaster and Apis mellifera. Arch Insect Biochem Physiol 48:13–38
Hildebrandt H, Müller U (1995) Octopamine mediates rapid stimulation of PKA in the antennal lobe of honeybees. J Neurobiol 27:44–50
Hildebrandt H, Müller U (1995) PKA activity in the antennal lobe of honeybees is regulated by chemosensory stimulation in vivo. Brain Res 679:281–288
Eisenhardt D, Friedrich A, Stollhoff N et al (2003) The AmCREB gene is an ortholog of the mammalian CREB/CREM family of transcription factors and encodes several splice variants in the honeybee brain. Insect Mol Biol 12:373–382
Kenakin T (1995) Agonist-receptor efficacy II. Agonist trafficking of receptor signals. Trends Pharmacol Sci 16:232–238
Homberg U (2005) Multisensory processing in the insect brain. In: Christensen TA (ed) Methods in insect sensory neuroscience. CRC Press, Boca Raton, London, New York, Washington, DC, pp 3–25
Bräunig P, Eder M (1998) Locust dorsal unpaired median (DUM) neurones directly innervate and modulate hind leg proprioceptors. J Exp Biol 201:3333–3338
Matheson T (1997) Octopamine modulates the responses and presynaptic inhibition of proprioceptive sensory neurones in the locust Schistocerca gregaria. J Exp Biol 200:1317–1325
Pérez M, Coro F (1986) Effect of picrotoxin on the tympanic organ of a noctuid moth. Naturwissenschaften 73:501–502
Ghiradella H (1971) Fine structure of the noctuid moth ear I. The transducer area and connections to the tympanic membrane in Feltia subgothica Haworth. J Morphol 134:21–45
Surlykke A (1984) Hearing in notodontid moths: a tympanic organ with a single auditory neurone. J Exp Biol 113:323–336
MacDermid V, Fullard J (1998) Not all receptor cells are equal: octopamine exerts no influence on auditory thresholds in the noctuid moth Catocala cergama. Naturwissenschaften 85:505–507
Lühr B, Wiese K (1994) Octopaminergic modulation of the auditory pathway in the cricket Gryllus bimaculatus. Göttingen Neurobiol. Report 329–330
Krause AF, Durr V (2004) Tactile efficiency of insect antennae with two hinge joints. Biol Cybern 91:168–181
Ye S, Leung V, Khan A et al (2003) The antennal system and cockroach evasive behavior I. Roles for visual and mechanosensory cues in the response. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 189:89–96
Durr V, Konig Y, Kittmann R (2001) The antennal motor system of the stick insect Carausius morosus: anatomy and antennal movement pattern during walking. J Comp Physiol 187A:131–144
Kevan PG, Lane MA (1985) Flower petal microtexture is a tactile cue for bees. Proc Natl Acad Sci USA 82:4750–4752
Balakrishnan R, Pollack G (1997) The role of antennal sensory cues in female responses to courting males in the cricket Teleogryllus oceanicus. J Exp Biol 200:511–522
Altman JS, Tyer NM (1977) The locust wing hinge stretch receptors I. Primary sensory neurons with enormous central arborizations. J Comp Neurol 172:409–430
Ramirez JM, Buschges A, Kittmann R (1993) Octopaminergic modulation of the femoral chordotonal organ in the stick insect. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 173:209–219
Scheiner R, Erber J, Page RE Jr (1999) Tactile learning and the individual evaluation of the reward in honeybees (Apis mellifera L.). J Comp Physiol 185A:1–10
Scheiner R, Page RE Jr, Erber J (2001) The effects of genotype, foraging role, and sucrose responsiveness on the tactile learning and performance of honeybees (Apis mellifera. L.). Neurobiol Learn Mem 76:138–150
Eberl DF, Hardy RW, Kernan MJ (2000) Genetically similar transduction mechanisms for touch and hearing in Drosophila. J Neurosci 20:5981–5988
Mercer AR, Erber J (1983) The effects of amines on evoked potentials recorded in the mushroom bodies of the bee brain. J Comp Physiol 151A:469–476
Linn CE, Campbell MG, Poole KR et al (1996) Effects of photoperiod on the circadian timing of pheromone response in male Trichoplusia ni: relationship to the modulatory action of octopamine. J Insect Physiol 42:881–891
Hamdorf K, Hoglund G, Juse A et al (1989) Effect of neurotransmitters on movement of screening pigment in insect superposition eyes. Z Naturforschung 44C:992–998
Kloppenburg P, Erber J (1995) The modulatory effects of serotonin and octopamine in the visual system of the honeybee (Apis mellifera L.) II. Electrophysiological analysis of motion sensitive neurons in the lobula. J Comp Physiol 176A:119–129
Chyb S, Hevers W, Forte M et al (1999) Modulation of light response by cAMP in Drosophila photoreceptors. J Neurosci 19:8799–8807
Hardie RC, Martin F, Cochrane GW et al (2002) Molecular basis of amplification on Drodophila phototransduction: roles for G protein, phospholipase C, and diacylglycerol kinase. Neuron 36:689–701
Cohen RW, Mahoney DA, Can HD (2002) Possible regulation of feeding behavior in cockroach nymphs by the neurotransmitter octopamine. J Insect Behav 15:37–50
Pass G, Sperk G, Agricola H et al (1988) Octopamine in a neurohaemal area within the antennal heart of the american cockroach. J Exp Biol 135:495–498
Pophof B (2000) Octopamine modulates the sensitivity of silkmoth pheromone receptor neurons. J Comp Physiol 186A:307–313
Linn CE, Roelofs WL (1986) Modulatory effects of octopamine and serotonin on male sensitivity and periodicity of response to sex pheromone in the cabbage looper moth, Trichuplusia ni. Arch Insect Biochem Physiol 3:161–171
Grünbaum L, Müller U (1998) Induction of a specific olfactory memory leads to a long-lasting activation of protein kinase C in the antennal lobes of the honeybee. J Neurosci 18:4384–4392
Rescorla RA (1988) Behavioral studies of Pavlovian conditioning. Annu Rev Neurosci 11:329–352
Menzel R (1990) Learning, memory, and “cognition” in honeybees. In: Kesner RP, Olten DS (eds) Neurobiology of comparative cognition. Erlbaum, Hillsdale, NJ, pp 237–292
Braun G, Bicker G (1992) Habituation of an appetitive reflex in the honeybee. J Neurophysiol 67:588–598
Molaei G, Lange AB (2003) The association of serotonin with the alimentary canal of the African migratory locust, Locusta migratoria: distribution, physiology and pharmacological profile. J Insect Physiol 49:1073–1082
de Belle JS, Heisenberg M (1994) Associative odor learning in Drosophila abolished by chemical ablation of mushroom bodies. Science 263:692–695
Heisenberg M, Borst A, Wagner S et al (1985) Drosophila mushroom body mutants are deficient in olfactory learning. J Neurogenet 2:1–30
Bodnaryk RP (1980) Changes in brain octopamine levels during metamorphosis of the moth Mamestra configurata Wlk. Insect Biochem 10:169–173
Lehman HK, Murgiuc CM, Hildebrand JG (2000) Characterization and developmental regulation of tyramine-β-hydroxylase in the CNS of the moth, Manduca sexta. Insect Biochem Mol Biol 30:377–386
Harris JW, Woodring J (1992) Effect of stress, age, season, and source colony on levels of octopamine, dopamine and serotonin in the honeybee (Apis mellifera L.) brain. J Insect Physiol 38:29–35
Taylor DJ, Robinson GE, Logan BJ et al (1992) Changes in brain amine levels associated with the morphological and behavioral development of the worker honeybee. J Comp Physiol 170A:715–721
Wagener-Hulme C, Kuehn JC, Schulz DJ et al (1999) Biogenic amines and division of labor in honey bee colonies. J Comp Physiol 184A:471–479
Schulz DJ, Robinson GE (2001) Octopamine influences division of labor in honeybee colonies. J Comp Physiol 187A:53–61
Webb M-LZ, Denlinger D (1998) GABA and picrotoxin alter expression of a maternal effect that influences pupal diapause in the flesh fly, Sarcophaga bullata. Physiol Entomol 23:184–191
Noguchi H, Hayakawa Y (2001) Dopamine is a key factor for the induction of egg diapause of the silkworm, Bombyx mori. Eur J Biochem 268:774–780
Pérez-Baun JC, Galve I, Ruiz-Verdú A et al (1994) Octopamine-sensitive adenylyl cyclase and G proteins in Ceratitis capitata brain during aging. Neuropharmacol 33:641–646
Humphries MA, Müller U, Fondrk MK et al (2003) PKA and PKC content in the honeybee central brain differs in genotypic strains with distinct foraging behavior. J Comp Physiol 189A:555–562
Seid MA, Traniello JF (2005) Age-related changes in biogenic amines in individual brains of the ant Pheidole dentate. Naturwissenschaften 92:198–201
Tofilski A (2000) Senescence and learning in honeybee (Apis mellifera) workers. Acta Neurobiol Exp (Wars) 60:35–39
Acknowledgments
Supported by NIH award 747127 from National Center for Research Resources to Brian H. Smith and NIH award 60005617 to Terman D. Hillel. The author thanks anonymous reviewers for their valuable suggestions and constructive criticisms on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farooqui, T. Octopamine-Mediated Neuromodulation of Insect Senses. Neurochem Res 32, 1511–1529 (2007). https://doi.org/10.1007/s11064-007-9344-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9344-7