Abstract
Introduction
Effective glioblastoma (GBM) treatment is limited by high invasiveness and heterogeneity. Current therapies target proliferating Glioma Stem Cell (GSC) subpopulations while sparing invading GSCs, which eventually engender tumor recurrence after treatment. Surface receptor CD97/ADRGE5 is associated with invasion and metastasis regulation in non-CNS cancers. Although CD97 expression level positively correlates with poor GBM patient prognosis, its role in this tumor is unclear.
Methods
Here, we examined CD97 function in primary patient-derived GSCs (pdGSCs) obtained from five GBM tumors, belonging to three major genetic subtypes. We compared endogenous CD97 levels in pdGSCs to the corresponding patient MRI’s radiographic invasion pattern aggressiveness. We manipulated CD97 levels in these pdGSCs by knockdown and overexpression and analyzed: (i) stem and subtype marker expression, (ii) in vitro invasive properties, and (iii) cell proliferation.
Results
Endogenous CD97 levels in pdGSCs positively correlated with radiographic invasion pattern aggressiveness on patient MRIs, and in vitro invasion rate. CD97 knockdown decreased pdGSC invasion rates in vitro, most markedly in mesenchymal subtype pdGSCs, as well as classical subtype pdGSCs. Invasion rates in vitro increased after CD97 overexpression predominately in proneural subtype pdGSCs. In the pdGSC line with the lowest endogenous CD97 level, CD97 overexpression increased the proliferation rate almost threefold.
Conclusions
For the first time in pdGSCs, we have shown that CD97 knockdown decreases and overexpression increases invasion rate in vitro. The effect of CD97 on invasion is pdGSC subtype-dependent. Future in vivo and mechanistic studies are needed for validation. Pharmacologic CD97 inhibitors should be identified, as they may potentially therapeutically diminish GBM invasion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Glioblastoma (GBM) is the highest grade and most lethal primary brain tumor with an average 12–15 month survival. The hallmark characteristics of this fatal tumor are its aggressive invasiveness and highly heterogeneous nature [1]. Current standard of care entails surgical resection followed by radiation and chemotherapy with the alkylating agent temozolomide [2], both of which primarily target proliferating tumor cells. During treatment, non-dividing migrating glioma stem cells (GSCs) survive the treatment and eventually populate distal brain areas, giving rise to recurrent therapeutically-resistant tumors. Therefore, having a better understanding of the mechanism regulating GSCs migration will guide the search for alternative therapies targeting not only cell proliferation but also the invasive aspect of GBM.
One group of gene products that play a fundamental role in GCS invasiveness are cell surface receptors, which interact with the extracellular matrix (ECM). The adhesion G-protein coupled receptor (aGPCR) CD97, also known as ADGRE5, has been implicated in the invasive behavior of GBM [3, 4] and metastatic progression of many other tumors [5,6,7,8]. CD97 expression level positively correlates with enhanced GBM invasiveness and shorter survival [3, 4]. CD97 is coupled to the G12/13 family of G proteins [7, 9], which activate signaling pathways involved in cell proliferation, transformation, adhesion, and migration [10, 11]. While CD97’s involvement in invasion and metastasis is well documented for other cancers [12], the role of CD97 in GBM tumorigenesis has not been thoroughly investigated. Specifically, it is not known how this receptor affects the behavior of GCSs of different genetic subtypes. Previous functional studies have used U87, U251 and other commercial GBM cell lines [3, 4, 13] that have been propagated in vitro for many generations and may not represent the properties of original tumors [14].
Here, to understand how CD97 affects GSCs behavior, we identified five GBM patients with varying radiographic invasion patterns, from which we established primary patient-derived GSCs (pdGSCs). We examined the radiographic and clinical data of the original GBM tumors, as well as the in vitro GSC morphology. We correlated these findings with endogenous CD97 levels, as well as the growth and invasion properties of the GSCs. By manipulating the levels of CD97 via over-expression or knockdown, we tested whether this receptor had an effect on cell invasion and proliferation. Our results indicate that CD97 can strongly influence the behavior of primary pdGSCs, and that these effects occur in a GSC subtype-dependent manner.
Materials and methods
Surgical specimens and primary GSC culture
GBM surgical specimens were acquired intraoperatively at our institution from 2 surgeons (MEI, RJK). All research was approved by the University of Miami Institutional Review Board for human research (IRB #20190521). Written and verbal informed preoperative consent were provided from all patients. Clinical data of all patients was collected from the electronic medical records. Primary Glioma Stem Cell (GSC) lines were established as previously described [15] with minor modifications. In short, tissue specimens were minced in Hibernate A media (ThermoFisherScientific, #A1247501) followed by 20 min incubation with 0.25% Trypsin (ThermoFisher, #15090046) and 50ug/ml DNAse (Sigma, #D5025) at 37 °C. Tissue was then washed three times with HibernateA supplemented with B27, triturated gently and filtered through 40 nm sieve. Dissociated cells were centrifuged at 100 g for 5 min, resuspended in red blood cell lysis buffer (Thermo Scientific #00-4333-57) and incubated for 5 min. Cell were plated in flasks coated with PDL (0.1 mg/ml)/Laminin (10ug/ml) for adherent monolayer or into uncoated flasks for spheres formation and cultured at 37 °C 5% CO2 in the following media: Neurocult NC-A media (Stem Cell Technologies #5750) supplemented with B27 (LifeTechnologies, #17504-044), N2 (ThermoFisher, #17502001), 1% Penicillin/Streptomycin (Life Technologies #15140-122), 2 mM l-Glutamine (Life Technologies #A2916801), 1 mM Na Pyruvate (Life Technologies #11360-070), 20 ng/mL EGF (Stem Cell Technologies #78006), 20 ng/ml bFGF (Stem Cell Technologies #78003), and 1 IU/ml Heparin (Stem Cell Technologies #07980).
Plasmid constructs and lentivirus production
shRNA for CD97 and controls were designed using Invivogen siRNA wizard software and cloned into pLKO-Puro-TRC plasmid (Addgene #10878). Of three constructs for CD97 shRNA, we choose the most effective (target sequence 5’ACAAGAAGGTTCGGGAAGAAT) with the knockdown of ~ 60–80%. Flag-tagged CD97 cDNA was PCR-amplified from CD97-Tango (Addgene, #66247) and cloned into pLenti-CMV-Puro (Addgene, # 17448) replacing GFP cDNA. The original plasmid was used as a control. Production of 2nd generation Lentivirus was done according to Addgene protocol using psPAX2 (Addgene, #12260) and pMD2.G (Addgene, #12259) as helper plasmids. In short, all three plasmids were transfected into HEK293T cells (ATCC Cat# CRL-3216) using PEI at 1 mg/ml (764604, Sigma) as a transfection agent. The released virus was harvested twice and filtered through 0.45 um filter. The virus was concentrated with Viro-Peg Concentrator (CHM00B952, Thomas Scientific), aliquoted and stored at − 80 °C. The viral titer was tested by serial dilutions and by cells’ Puromycin resistance.
Invasion assay
pdGSCs were infected with lentiviruses 24 h before the assay. On the day of the assay, the wells of the CIM-plate (cat# 05665825001, ACEA bio) upper chamber were coated with 0.4 mg/ml of Matrigel (Corning #354230) supplemented with 100ug/ml Hyaluronic acid (H7630, Sigma) and incubated at 37 °C for 4 h. 160ul Growth media supplemented with 10% FBS was added to each well of the lower chamber and the CIM plate was assembled. 50 ul growth media was added to each well of the upper chamber and the plate was equilibrated in the xCelligence machine (RTCA-DP, Agilent) at 37 °C 5% CO– incubator. Transduced cells were added to the upper wells at 50,000/well in 100 ul media in quadruplicates, settled for 15 min at room temperature, and the plate was returned to the instrument. Cell index measurements were taken every 15 min for the experimental duration.
Results
Radiographic tumor invasiveness and clinical parameters of GBM patients
For our studies, we chose five GBM patients with a wide range of tumor invasiveness. All five patients had pathologically confirmed W.H.O. Grade IV glioblastomas. Figure 1 shows Magnetic Resonance Images (MRIs) of the tumors before and after the surgery and at the time of tumor recurrence. GBM 22 had the most aggressive recurrence pattern (noncontiguous contralateral hemisphere spread), followed by GBM1 and GBM 18 (contiguous contralateral hemisphere spread), followed by GBM40 (ipsilateral spread to the corpus callosum), and GBM12 had the least aggressive pattern (ipsilateral locoregional spread).
GBM tumors and tumor derived GSCs from five patients. MRIs taken before (top row) and after (middle row) the surgery, and at the time of recurrence (bottom row). Preoperatively, all five of the patients’ tumors were localized to one hemisphere visualized as a brighter white color than the surrounding brain (blue lines). The immediate postoperative MRI shows the resection cavity (orange line). The follow up MRIs at the time of tumor recurrence, with recurrent enhancing tumors are outlined by the red lines. Noncontiguous versus contiguous tumors spread to the contralateral hemisphere are marked by yellow and white arrowheads, respectively. GBM 40 is seen invading the corpus callosum, but GBM12 spread is only within the margins of the primary tumor (bottom right two panels)
Table S1 (Supplemental materials) summarizes clinical parameters for each patient.
Morphology of patient-derived primary GSCs
From each studied GBM tumor, we isolated and established pdGSCs lines that can be maintained in culture in serum free conditions. We observed that the morphology of these GSCs roughly fall into three categories (Supplemental Fig. S1): (i) round loosely adhered cells that tend to form clusters (GBM22 and GBM1), (ii) elongated cells readily growing in a monolayer (GBM18, GBM40) and (iii) GBM12, which is similar to the latter two, but with visibly larger and flatter cell bodies. GBM12,18 and 40 could only be lifted from laminin coated surface using trypsin or accutase. When confluent, they form a dense network of elongated often overlapping cells. In contrast, GBM22 and 1 could easily be detached with non-enzymatic solution and readily form spheres when confluent. This observation suggests that these two GSCs could be more mobile due to their lower affinity to components of ECM such as laminin, correlating with radiographic findings.
The level of CD97
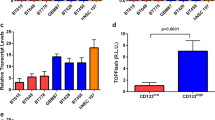
We investigated CD97 expression in our pdGSCs using immunoblot, flow cytometry, immunofluorescence microscopy, and qPCR (Fig. 2). HEK293 cells and normal human astrocytes (NHA) served as negative controls since they do not express CD97. As expected, CD97 was undetectable in either control cells. In contrast, all pdGSCs expressed CD97, albeit at different levels (Fig. 2A). The highest levels were detected in GBM22. GBM12 expressed about 6 times less and had the lowest CD97 among tested GSCs. Notably, these cells were derived from the most invasive and the least invasive tumors (Fig. 1), respectively, consistent with the previous report that CD97 expression correlates with invasive GBM phenotype [4]. We didn’t observe such correlation between the invasive phenotype and CD97 mRNA level which varied only about 2 to 4-fold between our GSCs (Fig. 2E). This data may suggest that CD97 protein levels are regulated post-translationally.
CD97 protein and mRNA expression in primary GSCs. (A) Representative western blot stained with CD97 and pan tubulin (loading control) antibodies shows variable CD97 expression in primary GSCs. CD97 band is not detected in HEK293 cells (HEK) and Normal Human Astrocytes (NHA) used as negative controls (B) Flow cytometry results of non-permeabilized cells stained for PE-conjugated CD97 antibody. The graphs show the distribution of fluorescently labeled CD97 + cells and their percentage of the total single cell population. HEK293 cells are taken as a negative control and Jurkat cells (JRK) as a positive control for CD97 expression. All cell lines included a control without addition of fluorescently labeled antibody to control for autoflorescence (representative distribution for GBM1 control included). (C) Western blots quantitation for CD97 protein normalized to tubulin (N = 3) (D) Flow cytometry data shown on the graph comparing the percentage of cells expressing CD97 receptor on their cell surface. (E) qPCR results for CD97 mRNA fold expression in primary GSCs compare to levels in normal human brain (red dotted line, N = 3). Error bars ± SEM
We then compared CD97 protein expression on the cell surface of GCSs using flow cytometry of non-permeabilized cells with fluorescently-labeled CD97 antibody (Fig. 2B, D). This technique specifically detects cell surface CD97. Among the GSCs with high CD97 expression, GBM1 and GBM22 had more than 80%, and GBM40 had approximately 60% of CD97-positive cells. This proportion was much lower (20–25%) for low CD97-expressing GBM12 and GBM18. Overall, the measurement of total (Fig. 2A, C) and cell surface (Fig. 2B, D) CD97 receptor produced a similar pattern. Immunostaining of permeabilized cells for endogenous CD97 demonstrates diffuse punctate staining of this receptor distributed throughout multiple cellular compartments, including cell surface and cytosol (Suppl. Fig. S2).
Interestingly, confocal imaging revealed a unique pattern of CD97 localization in GBM1 and GBM22. When grown in monolayer these cells showed diffused staining, but in the areas of high cell density, CD97 was concentrated at the border of adjacent cells forming spheres. (Suppl. Fig. S3). This staining pattern was not detected in the other GSCs. This observation may suggest that in GBM22 and GBM1 that have the highest levels of CD97 and the highest radiographic invasiveness, this receptor is upregulated upon cell-to-cell contact during sphere formation in culture.
Stemness of patient-derived GSCs
The notorious heterogeneity of GBM tumors is caused by the presence of cancer stem cells that can proliferate indefinitely and exhibit high degree of plasticity. To test whether the pdGSCs sustain their stem cell characteristics, we measured expression levels of several stem markers (Fig. 3A). All five GSCs express Nestin and Vimentin mRNA more then 50-fold higher than normal brain control. Pluripotency marker Sox2 and the oncogene cMyc are both highly expressed in GBM12, 18 and 40. In contrast, in GBM1 and 22, these stem markers levels are just slightly above those of normal brain. Transcription factors Oct4 and Nanog activate pluripotency genes in complex with Sox2. The expression level of Oct4 was higher in both GBM12 and GBM18, but Nanog was over-expressed only in GBM18.
Endogenous expression of stem and subtype markers in five studied GSCs. (A) mRNA expression of several stem markers tested by qPCR. mRNA levels are normalized to GAPDH and the results on the graphs are shown as fold changes relative to normal brain control (dashed red line). (B) The endogenous mRNA expression of GSC subtype markers EGFR, PDGFRA, Olig2, and cMET, relative to their expression in normal brain (red dashed line). (C) Western blot showing endogenous levels of several marker proteins in studied GSCs. N = 3, error bars ± SEM
GBM1 and 22 have relatively low mRNA levels of these stem markers compared to the other three lines, indicating a potentially higher differentiated status, though still maintaining stemness. These two GSCs also have the highest levels of CD97 (Fig. 2). It is reasonable to suggest that such combination of high expression of this invasion-associated gene and lower stemness predisposes these cells to increased invasiveness. Notably, GBM40 also has high CD97. In this GSC line the expression of Sox2 mRNA and protein is the highest among all five GSC cell lines (Fig. 3A, C). Therefore, in GBM40 the functional effect of Sox2 may dominate over CD97 signaling, reducing its invasive properties while increasing self-proliferation.
Defining GSCs subtypes
Since there is no single molecular marker that clearly defines GBM subtype [1, 16, 17], we tested expression of several additional markers that might suggest the lineage of our pdGSCs (Fig. 3).
Our data indicate that the morphologically similar GBM1 and 22 belong to the Mesenchymal GBM subtype. Indeed, they express high levels of proto-oncogene Mesenchymal Epithelial Transition (cMET) that is associated with Mesenchymal subtype [1].
GBM12 and GBM40 appear to belong to Proneural subtype. They both have high expression of Olig2, a marker of oligodendrocyte progenitors. GBM12 also overexpress PDGFRA, and GBM40 has high levels of Sox2 that is typically expressed in neuronal progenitors. High expression of cMET in GBM12 also suggests the presence of Mesechymal subtype cells.
We define the subtype of GBM18 as Classical on the basis of high EGFR expression [1, 16, 17].
Invasion phenotype of primary GSCs in vitro
To test if the invasive properties of our GSCs correlate with CD97 expression in vitro, we assessed the migration of these cells through Matrigel, a commercial preparation of extracellular matrix (ECM) derived from mouse sarcoma. To better mimic the unique composition of brain ECM, we supplemented the Matrigel with hyaluronic acid (HA), the major component of the brain extracellular matrix, that is also known to be upregulated at the brain-tumor interface [18,19,20]. All GSCs migrated through the Matrigel barrier, which confirms their overall invasive nature (Fig. 4). However, the invasion dynamics of the cells were different. Mesenchymal subtype GBM1 and GBM22, which highly express CD97, showed the highest invasiveness. Proneural GBM40 and GBM12 had the lowest invasion rates. The lower in vitro invasiveness of GBM12 correlated with low invasiveness of the original GBM12 tumor (Fig. 1) and the lower expression of CD97. However, there was no such correlation in the case of GBM40 that has high CD97 levels and showed high radiographic tumor migration (Fig. 1). Perhaps the high expression of the stemness marker Sox2 (Fig. 3A, C) negates the invasive properties of this GSC in vitro. Sox2 has a regulatory role promoting the epithelial-to-mesenchymal transition (EMT), a process that promotes proliferation and impedes invasion [21,22,23,24], potentially reducing the in vitro invasion rate of GBM 40.
The in vitro migration rate of Classical subtype GBM18 was intermediate among the tested GSCs which correlated with the intermediate levels of CD97 (Fig. 2A, C).
Overall, the ranking of GSCs invasion rates in vitro corresponds very well to the invasive characteristics of original tumors on MRI. Moreover, for some GSCs, particularly of mesenchymal and classical subtype, we observed a correlation between the level of CD97 and invasive behavior of these cells; this led us to the hypothesis that CD97 can promote GSCs invasion.
Effect of CD97 expression manipulation on GSC invasion in vitro
To test this hypothesis, we performed knockdown and over-expression of CD97 in our pdGSCs using lentivirus vectors carrying either shRNA against CD97 (shCD97) or CMV-driven flag-tagged CD97 (flCD97). As controls, we used GFP cDNA and shRNA against GFP.
The results of in vitro invasion assays showed that CD97 knockdown significantly decreased the invasion rate of GBM1, GBM22, and GBM12 (Fig. 5A). This effect was less noticeable in GBM18 and was not significant in GBM40. Interestingly, the strongest effect of CD97 knockdown was observed in the two GSCs with the highest CD97 expression, GBM1 and GBM22, and the one with the lowest, GBM12. The common feature of these GCSs is the presence of mesenchymal subtype cells (MET positive, Fig. 3A) suggesting that CD97 depletion primarily affects mesenchymal subtype GSCs.
The most striking effect of CD97 over-expression was observed for GBM12 where it strongly increased the invasion rate (Fig. 5B). Since these cells have the lowest endogenous levels of this receptor, it is reasonable to suggest that bringing CD97 expression to higher levels above a threshold induces the invasiveness of these GSCs. CD97 over-expression had no significant effect on invasion rates of GBM1, 22 and 40, and only mildly but significantly increased invasion of GBM18. CD97 over-expression’s lack of effect on invasion in the former three GSCs may be explained by high endogenous levels of this receptor (Fig. 2A, C). The invasive properties of GBM40 did not depend on CD97 expression levels.
Overall, the results from in vitro invasion assay suggest that CD97 selectively affects the invasion properties of several pdGSC subtypes. The only pdGSC resistant to CD97 levels changes was GBM40, the line with the highest Sox2 expression, which as previously mentioned may promote a high proliferation, low invasion state [21,22,23,24]. Future studies will confirm the role of SOX2 in driving the differential phenotype of GBM40 by knocking down SOX2, and determining the effect on invasion rates with and without CD97 manipulation.
Effect of CD97 expression changes on GSC proliferation rate
To investigate whether changes in CD97 expression affects cell proliferation, we stained the virus transduced pdGSCs with antibody against proliferation marker Ki67. We saw no significant changes in cells’ proliferation with either knockdown or over-expression of CD97 in almost all tested GSCs (Suppl. Fig. S4). The only exception was seen in GBM12 in which CD97 over-expression significantly increased proliferation almost threefold. This GSC line was derived from the tumor with the lowest Ki67 proliferation index (see Suppl. Table S1). In GBM18, we saw a trend towards reduced proliferation after CD97 knockdown, however it was not significant. We conclude that CD97 mostly does not contribute to GSCs proliferation, but may affect it in some GSCs.
Discussion
CD97/ADGRE5 is an adhesion GPCR that was first found in myeloid cells. CD97 has been implicated in cell adhesion and migration via interactions with the ECM and proteins on other cell surfaces. High CD97 expression has been associated with aggressive metastatic and invasive behavior of gastric, hepatocellular, and many other malignancies [5,6,7, 21, 25]. Recently, correlation between higher CD97 expression and increased invasion was noted in commercially-available glioblastoma cell lines [3, 13, 26] that may be genetically dissimilar to their original tumors [14], but the cause-and-effect relationship of this phenomenon has not been investigated.
In this paper, for the first time, we investigated the role of CD97 in pdGCSs by actively manipulating the levels of CD97 expression. We found that genetic inhibition of CD97 reduced, and overexpression increased, the invasion rates of GCSs in vitro. The reduction in invasion mostly affected the GSCs of mesenchymal and to a lesser extent classical subtype. In contrast, the observed increase in invasion after CD97 overexpression was seen mostly in proneural subtype GSCs. Thus, our results indicate that this effect of CD97 depends on GSC subtype.
Unlike with invasion, the effect on proliferation was observed only in GBM12, which had the lowest endogenous CD97 level. In these cells, overexpression of CD97 increased the proliferation rate almost threefold. Combined, these results support the notion that CD97 signaling varies among GBM subtypes.
In summary, the original results presented here show that CD97 can control functional invasion of pdGSCs. We found that inhibition of CD97 in GSCs of mesenchymal and classical subtype reduces their invasion rate in vitro. A strong stimulatory effect of CD97 on proliferation was observed only in one GSC with the lowest endogenous CD97 levels. Considering that in vitro invasiveness of patient derived GSCs corresponds to invasive characteristics of the original tumors, these findings support the regulatory role of CD97 in GBM invasion in vivo. Adhesion GPCRs recently emerged as potential drug targets in cancer [27]. Therefore, finding new strategies utilizing CD97 and associated mechanisms as new pharmacological targets may be promising for glioblastoma.
Data availability
The data generated and analyzed in this study are provided in the text and in the figure, tables, and supplementary tables. Any additional data needed can be requested from the corresponding author.
References
Behnan J, Finocchiaro G, Hanna G (2019) The landscape of the mesenchymal signature in brain tumours. Brain 142:847–866. https://doi.org/10.1093/brain/awz044
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, Organisation European, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. New Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
Safaee M, Clark AJ, Oh MC, Ivan ME, Bloch O, Kaur G, Sun MZ, Kim JM, Oh T, Berger MS, Parsa AT (2013) Overexpression of CD97 confers an invasive phenotype in glioblastoma cells and is associated with decreased survival of glioblastoma patients. PLoS ONE 8: https://doi.org/10.1371/journal.pone.0062765
Safaee M, Fakurnejad S, Bloch O, Clark AJ, Ivan ME, Sun MZ, Oh T, Phillips JJ, Parsa AT (2015) Proportional upregulation of CD97 isoforms in glioblastoma and glioblastoma-derived brain tumor initiating cells. PLoS ONE 10: https://doi.org/10.1371/journal.pone.0111532
He Z, Wu H, Jiao Y, Zheng J (2015) Expression and prognostic value of CD97 and its ligand CD55 in pancreatic cancer. Oncol Lett 9:793–797. https://doi.org/10.3892/ol.2014.2751
Ward Y, Lake R, Yin JJ, Heger CD, Raffeld M, Goldsmith PK, Merino M, Kelly K (2011) LPA receptor heterodimerizes with CD97 to amplify LPA-initiated RHO-dependent signaling and invasion in prostate cancer cells. Cancer Res 71:7301–7311. https://doi.org/10.1158/0008-5472.CAN-11-2381
Ward Y, Lake R, Martin PL, Killian K, Salerno P, Wang T, Meltzer P, Merino M, Cheng SY, Santoro M, Garcia-Rostan G, Kelly K (2013) CD97 amplifies LPA receptor signaling and promotes thyroid cancer progression in a mouse model. Oncogene 32:2726–2738. https://doi.org/10.1038/onc.2012.301
Liu D, Li C, Trojanowicz B, Li X, Shi D, Zhan C, Wang Z, Chen L (2016) CD97 promotion of gastric carcinoma lymphatic metastasis is exosome dependent. Gastric Cancer 19:754–766. https://doi.org/10.1007/s10120-015-0523-y
Yu OM, Benitez JA, Plouffe SW, Ryback D, Klein A, Smith J, Greenbaum J, Delatte B, Rao A, Guan KL, Furnari FB, Chaim OM, Miyamoto S, Brown JH (2018) YAP and MRTF-A, transcriptional co-activators of RhoA-mediated gene expression, are critical for glioblastoma tumorigenicity. Oncogene 37:5492–5507. https://doi.org/10.1038/s41388-018-0301-5
Wu V, Yeerna H, Nohata N, Chiou J, Harismendy O, Raimondi F, Inoue A, Russell RB, Tamayo P, Gutkind JS (2019) Illuminating the Onco-GPCRome: novel G protein-coupled receptor-driven oncocrine networks and targets for cancer immunotherapy. J Biol Chem 294:11062–11086. https://doi.org/10.1074/jbc.REV119.005601
Vizurraga A, Adhikari R, Yeung J, Yu M, Tall GG (2020) Mechanisms of adhesion G protein-coupled receptor activation. J Biol Chem 295:14065–14083. https://doi.org/10.1074/jbc.REV120.007423
Juneja J, Casey PJ (2009) Role of G12 proteins in oncogenesis and metastasis. Br J Pharmacol 158:32–40. https://doi.org/10.1111/j.1476-5381.2009.00180.x
Chidambaram A, Fillmore HL, Van Meter TE, Dumur CI, Broaddus WC (2012) Novel report of expression and function of CD97 in malignant gliomas: correlation with Wilms tumor 1 expression and glioma cell invasiveness. J Neurosurg 116:843–853. https://doi.org/10.3171/2011.11.JNS111455
Allen M, Bjerke M, Edlund H, Nelander S, Westermark B (2016) Origin of the U87MG glioma cell line: Good news and bad news. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aaf6853
Pollard SM, Yoshikawa K, Clarke ID, Danovi D, Stricker S, Russell R, Bayani J, Head R, Lee M, Bernstein M, Squire JA, Smith A, Dirks P (2009) Glioma stem cell lines expanded in adherent culture have tumor-specific phenotypes and are suitable for chemical and genetic screens. Cell Stem Cell 4:568–580. https://doi.org/10.1016/j.stem.2009.03.014
Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP, Alexe G, Lawrence M, O’Kelly M, Tamayo P, Weir BA, Gabriel S, Winckler W, Gupta S, Jakkula L, Feiler HS, Hodgson JG, James CD, Sarkaria JN, Brennan C, Kahn A, Spellman PT, Wilson RK, Speed TP, Gray JW, Meyerson M, Getz G, Perou CM, Hayes DN, Cancer Genome Atlas Research N (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17:98–110. https://doi.org/10.1016/j.ccr.2009.12.020
Neftel C, Laffy J, Filbin MG, Hara T, Shore ME, Rahme GJ, Richman AR, Silverbush D, Shaw ML, Hebert CM, Dewitt J, Gritsch S, Perez EM, Gonzalez Castro LN, Lan X, Druck N, Rodman C, Dionne D, Kaplan A, Bertalan MS, Small J, Pelton K, Becker S, Bonal D, Nguyen QD, Servis RL, Fung JM, Mylvaganam R, Mayr L, Gojo J, Haberler C, Geyeregger R, Czech T, Slavc I, Nahed BV, Curry WT, Carter BS, Wakimoto H, Brastianos PK, Batchelor TT, Stemmer-Rachamimov A, Martinez-Lage M, Frosch MP, Stamenkovic I, Riggi N, Rheinbay E, Monje M, Rozenblatt-Rosen O, Cahill DP, Patel AP, Hunter T, Verma IM, Ligon KL, Louis DN, Regev A, Bernstein BE, Tirosh I, Suva ML (2019) An integrative model of cellular states, plasticity, and genetics for glioblastoma. Cell 178(835–849): https://doi.org/10.1016/j.cell.2019.06.024
Wolf KJ, Shukla P, Springer K, Lee S, Coombes JD, Choy CJ, Kenny SJ, Xu K, Kumar S (2020) A mode of cell adhesion and migration facilitated by CD44-dependent microtentacles. Proc Natl Acad Sci USA 117:11432–11443. https://doi.org/10.1073/pnas.1914294117
Bellail AC, Hunter SB, Brat DJ, Tan C, Van Meir EG (2004) Microregional extracellular matrix heterogeneity in brain modulates glioma cell invasion. Int J Biochem Cell Biol 36:1046–1069. https://doi.org/10.1016/j.biocel.2004.01.013
Novak U, Kaye AH (2000) Extracellular matrix and the brain: components and function. J Clin Neurosci 7:280–290. https://doi.org/10.1054/jocn.1999.0212
Yin Y, Xu X, Tang J, Zhang W, Zhangyuan G, Ji J, Deng L, Lu S, Zhuo H, Sun B (2018) CD97 promotes tumor aggressiveness through the traditional G Protein-coupled receptor-mediated signaling in hepatocellular carcinoma. Hepatology 68:1865–1878. https://doi.org/10.1002/hep.30068
Iglesias JM, Gumuzio J, Martin AG (2017) Linking pluripotency reprogramming and cancer. Stem Cells Transl Med 6:335–339. https://doi.org/10.5966/sctm.2015-0225
Han X, Fang X, Lou X, Hua D, Ding W, Foltz G, Hood L, Yuan Y, Lin B (2012) Silencing SOX2 induced mesenchymal-epithelial transition and its expression predicts liver and lymph node metastasis of CRC patients. PLoS ONE 7: https://doi.org/10.1371/journal.pone.0041335
Zhang S, Cui W (2014) Sox2, a key factor in the regulation of pluripotency and neural differentiation. World J Stem Cells 6:305–311. https://doi.org/10.4252/wjsc.v6.i3.305
Hilbig D, Dietrich N, Wandel E, Gonsior S, Sittig D, Hamann J, Aust G (2018) The interaction of CD97/ADGRE5 with beta-catenin in adherens junctions is lost during colorectal carcinogenesis. Front Oncol 8:182. https://doi.org/10.3389/fonc.2018.00182
Liu JK, Lubelski D, Schonberg DL, Wu Q, Hale JS, Flavahan WA, Mulkearns-Hubert EE, Man J, Hjelmeland AB, Yu J, Lathia JD, Rich JN (2014) Phage display discovery of novel molecular targets in glioblastoma-initiating cells. Cell Death Differ 21:1325–1339. https://doi.org/10.1038/cdd.2014.65
Gad AA, Balenga N (2020) The emerging role of adhesion GPCRs in cancer. ACS Pharmacol Transl Sci 3:29–42. https://doi.org/10.1021/acsptsci.9b00093
Funding
Dr. Daniel Eichberg and this work are supported by the Neurosurgery Research & Education Foundation (2021-22 NREF & StacheStrong Research Fellowship Grant on behalf of the AANS/CNS Section on Tumors). Dr. Daniel Eichberg is supported by training grant T32-CA-211034 from the National Cancer Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Dr. Michael Ivan is funded by the Elsa U. Pardee Foundation Cancer Grant and the American Cancer Society Institutional Grant for this work.
Author information
Authors and Affiliations
Contributions
Conception and design: DGE, TIS, ALP, RJK, MEI. Experimentation, collection and assembly of data: DGE, TIS, ALP. Data analysis/interpretation DGE, TIS, ALP, RJK, MEI. Cytological examination of tumor tissues: DGE, TIS. Contributions to manuscript writing: DGE, TIS, MEI. Final approval of manuscript: All authors.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Michael Ivan reports being a consultant to and receiving research funding from Medtronic and the NX Development Corporation.
Consent for publication
No individual patient data is reported in this study.
Ethical approval
University of Miami Institutional Review Board for human research (IRB #20190521). Both written and verbal informed preoperative consent were provided from all patients.
Informed consent
Informed consent was obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Eichberg, D.G., Slepak, T.I., Pascoini, A.L. et al. Genetic manipulation of adhesion GPCR CD97/ADGRE5 modulates invasion in patient-derived glioma stem cells. J Neurooncol 153, 383–391 (2021). https://doi.org/10.1007/s11060-021-03778-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-021-03778-8